Abstract
Soil pH regulates soil biogeochemical processes and has cascading effects on terrestrial ecosystem structure and functions. Afforestation has been widely adopted to increase terrestrial carbon sequestration and enhance water and soil preservation. However, the effect of afforestation on soil pH is still poorly understood and inconclusive. Here we investigate the afforestation-caused soil pH changes with pairwise samplings from 549 afforested and 148 control plots in northern China. We find significant soil pH neutralization by afforestation—afforestation lowers pH in relatively alkaline soil but raises pH in relatively acid soil. The soil pH thresholds (TpH), the point when afforestation changes from increasing to decreasing soil pH, are species-specific, ranging from 5.5 (Pinus koraiensis) to 7.3 (Populus spp.) with a mean of 6.3. These findings indicate that afforestation can modify soil pH if tree species and initial pH are properly matched, which may potentially improve soil fertility and promote ecosystem productivity.
Similar content being viewed by others
Introduction
Soil pH, which measures the acidity or alkalinity of soils, is associated with many soil properties such as hydrolysis equilibrium of ions1, microbial communities2,3, and organic matter contents4. Recent climate and anthropogenic changes have significantly modified soil properties including soil pH5,6. In particular, soil acidification has been widely reported across a variety of ecosystem types and regions5,6,7. Change in soil pH may alter soil biogeochemical processes, and has cascading effects on terrestrial ecosystem structure and functions8,9,10,11,12. For instance, soil acidification caused by nitrogen deposition has led to diversity loss throughout the world8,9. Further soil acidification could increase leaching loss of cation nutrients, intensifying the scarcity of some nutrient elements essential for plant growth, thus reducing plant productivity10. Accordingly, the impacts of soil acidification have attracted increasing concerns from both the scientific community and the public and raised discussions on possible mitigation measures13, and it is generally agreed that the potential impact on soil pH needs to be included in the design and evaluation of many land use change projects14,15.
Afforestation is one increasingly popular type of land use change projects primarily designated for wood production, soil and water conservation, increasing carbon storage and mitigating climate change16. However, it could also change soil pH14,15,17,18. Soil pH is determined by the balance between production and consumption of soil hydrogen ions19,20, which is closely associated with nutrient (e.g. carbon, nitrogen, phosphorus, sulfur, calcium) cycles19,20,21,22,23. Afforestation could affect the nutrient cycles through plant uptake of exchangeable cations17,23, capture of acid deposition19,20, modification in the quality, and quantity of litter input and rhizosphere processes19,20, which consequently impact the generation and consumption of soil hydrogen ions and soil pH. It is commonly reported that afforestation decreases soil pH, although results widely varied among different studies and different regions17,18,23,24. Furthermore, limited by research scale, we still know little on the spatial heterogeneity of afforestation-induced soil pH changes, and on how factors like background soil physical and chemical properties, and species choices may influence soil pH. Large-scale comparative studies between afforested and non-afforested control sites may provide critical information to address these questions.
The primary objective of this study is therefore to investigate the effect of afforestation on soil pH across different tree species and soil pH gradient. To achieve this goal, we conducted a comparative study using samples from northern China, a broad geographical region spanning 2000 km from east to west and known for ambitious state-sponsored large-scale afforestation efforts. China is the world’s largest cultivator of forest plantations, and afforestation has contributed to approximately 90% of its forest area expansion and 49.3% of its forest carbon sink since the 1970s25. The Three-North Shelterbelt Development Program (TNSDP), lying in North, Northwest, and Northeast China and covering an area of 4,069,000 km2 since 197826, is one of China’s earliest large-scale afforestation projects. The TNSDP program has generated important environmental and socioeconomic benefits, including reduced soil erosion and sand storms, and increased carbon sequestration27,28. This large-scale afforestation project also provides a rare opportunity to investigate the effects of afforestation on soil properties. In 2012−2013, we sampled across 148 sites within the TNSDP area to conduct pairwise comparisons on soil pH between planted forests and non-afforested control plots. For each site, we sampled one non-afforested control plot and several afforestation plots (see the section “Methods”) of different stand ages, resulting in a sum of 697 sampled plots (Fig. 1, 549 afforested and 148 non-afforested control plots) that made 549 afforestation−control pairs (note most of the control plots (119 of 148) corresponded to more than one afforestation plots). Five tree species, Pinus (P.) koraiensis, Larix (L.) gmelinii, Pinus (P.) sylvestris var. mongolica, Pinus (P.) tabuliformis and Populus spp., were used in these afforestation plots; and all plots were monocultures. Our results show significant soil pH neutralization by afforestation—afforestation decreases soil pH in alkaline soils but increases soil pH in acidic soils. These findings provide improved understandings on how afforestation impacts soil pH across a broad range of soil types and afforestation tree species, which is critical for developing climate change mitigation strategies and ecological sustainability plans.
Results
The overall effect of afforestation on soil pH
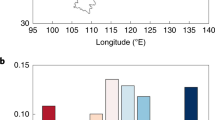
We found a rather mixed result on the post-afforestation change in soil pH (CIP), although on average the change of soil pH across the 549 afforestation−control pairs was not significant (Fig. 2a; mean pH was 6.51 and 6.45 for the control group and the afforestation group, respectively, p = 0.076 from a paired t-test). Afforested plots had a lower soil pH than corresponding control plots in 53.7% (295/549) of the afforestation−control pairs. In 295 of these plots, the mean and median of H+ generation were 44.32 and 6.98 mole ha−1, respectively (Supplementary Fig. 1). In contrast, in the remaining 254 (46.3% of the total pairs) pairs, afforested plots had a higher pH value, with mean and median of H+ consumption rate of 52.52 and 17.98 mole ha−1, respectively (Supplementary Fig. 1). On average, across all the 549 pairs, H+ reduced by 3.26 mole ha−1 (Supplementary Fig. 1), although insignificant (p = 0.69). However, the frequency distribution of soil pH values was significantly altered by afforestation (p = 0.02, Siegel−Tukey test). Afforestation reduced the frequencies of both the low (pH < 5) and the high (pH > 7) soil pH values, but increased the frequency of intermediate (5 < pH < 7) pH values (Fig. 2a).
Comparison of soil pH in control and afforested groups. a The frequency distribution of soil pH in control and afforested groups. Siegel−Tukey test was used to compare the frequency distributions of soil pH between the control and afforested groups. b Soil pH for the control and afforested groups across five tree species. Error bars indicate standard errors. * indicates 0.01 < p < 0.05, ** indicates 0.001 < p < 0.01, and *** indicates p < 0.001 from paired t-test. The inset shows the results of two-way ANOVA on the effect of afforestation (control vs. afforested) and species on soil pH. Interaction indicates afforestation × species
In the light of these results, we then divided the 549 pairs based on the pH value of control plots, named initial soil pH hereafter, into three groups—pH > 7, 5 < pH < 7, and pH < 5 (Fig. 3, one-way ANOVA, p < 0.05)—and investigated the influence of afforestation for each pH group. For the pH > 7 group, afforestation reduced soil pH in 74.1% of the pairs (Supplementary Fig. 2), with an average pH reduction of 0.57 (Fig. 3, p < 0.001). This group was further divided into three sub-groups according to initial soil pH, i.e. 7–8, 8–9, and >9. Significant reduction in soil pH was found in all the three sub-groups (p < 0.001, p < 0.001, and p < 0.05, respectively; Fig. 3), and the largest soil pH reduction was found in the sub-group of initial soil pH = 7−8, whose mean value of CIPs was −0.71 (Fig. 3). In contrast, for the pH < 5 group, afforestation increased soil pH in all cases (Supplementary Fig. 2). The mean of CIPs in this group was +0.96 (Fig. 3, p < 0.001). For the group 5 < pH < 7, afforestation increased soil pH in about a half (51.7%) of the pairs. The increase in the sub-group 5–6 was modest (mean CIPs is +0.18) but significant (p < 0.001). Furthermore, for the group with initial soil pH = 6–7, afforestation-induced soil pH change was not significant (p = 0.74).
Comparison of CIP across groups of different initial pH. In box-whisker plots, the central mark indicates the median, and the bottom and top edges of the box indicate the 25th and 75th percentiles, respectively. The maximum whisker lengths are specified as 1.5 times the interquartile range and outliers are marked using +. Independent sample t-tests with false discovery rate (FDR) correction were conducted to compare data of each group with 0. Symbols *, **, and *** indicate that the null hypothesis could be rejected at a significance level of 0.05, 0.01, and 0.001, respectively. Different letters mean significant differences between different pH groups (p < 0.05, one-way ANOVA, post-hoc LSD test)
Impacts of different tree species on soil pH changes
The impact of afforestation on soil pH was further complicated by the choice of different afforestation species (Fig. 2b). In particular, afforestation with P. koraiensis and P. tabuliformis significantly reduced soil pH from 5.84 to 5.64 (p < 0.05) and from 7.39 to 7.21 (p < 0.05), respectively. In contrast, afforestation with P. sylvestris var. mongolica, L. gmelinii and Populus spp. had no statistically detectable effect on soil pH (Fig. 2b). Further analyses showed that afforestation species had significant effects on soil pH, but afforestation (control vs. afforested) and the interaction of afforestation and species did not have a significant impact on soil pH (Fig. 2b). Furthermore, the vertical patterns of CIPs also varied among different species. For instance, P. koraiensis and P. sylvestris var.mongolica significantly acidified the deep layers, while P. tabuliformis afforestation mainly affected top soil layers (0–20 cm, Supplementary Table 1).
Note that different tree species were planted on different soils, we further used ordinary least squares (OLS) curve fitting to depict the relationship between CIP and initial soil pH for each of the five afforestation species. The results suggested that afforestation decreased soil pH in alkaline soils but increased it in acidic soils consistently across all the five species (Fig. 4). The decrease of CIP with increasing initial soil pH was highest for L. gmelinii plantations (slope −0.72), followed by that of P. koraiensis (slope −0.67), Populus spp. (slope −0.47), P. tabuliformis (slope −0.25), and P. sylvestris var. mongolica (slope −0.24); the slope was −0.31 when all the five species were combined (Fig. 4a). We then defined a threshold soil pH (TpH), which is the point when afforestation-induced CIP changes from positive to negative, shown as the horizontal axis intercept of the OLS curve in Fig. 4a. We found that the overall TpH was about 6.3 when all the species were included, with considerably species-specific variations. Specifically, Populus spp. plantations had the highest TpH (7.3), and the P. koraiensis plantations had the lowest one (TpH = 5.5). Afforestation with L. gmelinii, P. sylvestris var. mongolica, and P. tabuliformis acidified soils when the initial soil pH was >5.8, 5.9, and 6.7, respectively. In general, Populus spp. caused the largest increase of soil pH when the initial soil pH < 5.5; but L. gmelinii and P. koraiensis resulted in the largest decrease of pH value when the initial pH > 7. Furthermore, result of two-way ANOVA showed that initial soil pH, species, and their interaction all had significant effects on CIP (Fig. 4b).
Relationships between change in soil pH and initial pH across five plantation tree species. a Plotting of change in soil pH (CIP) against initial soil pH for each of the five plantation species. Solid lines indicate the ordinary least squares (OLS) fit of the linear equation (CIP = a*pH+b, where pH indicates the pH of the control group) for each of the five plantation species (the colors are the same as the legend in b) and all the species pooled together. b The dependence of mean CIP on initial soil pH across the five plantation species. Mean CIP is averaged for each unit of initial soil pH. Error bars indicate standard errors. Independent sample t-tests with false discovery rate (FDR) correction were conducted to compare data of each group with 0. Symbols *, **, and *** indicate that the null hypothesis could be rejected at a significance level of 0.05, 0.01, and 0.001, respectively. The inset shows the results of two-way ANOVA on the effect of initial soil pH and species on CIP. Interaction indicates initial soil pH × species
Robustness tests of soil pH neutralization by afforestation
The observed neutralization effect of afforestation on soil pH may have also been confounded by some other factors such as original vegetation types, stand age, soil types, climate and net primary productivity (NPP). To test the robustness of our finding, we used generalized linear models (GLM) to evaluate the effects of all the factors (i.e. initial soil pH, afforestation tree species, original vegetation types, stand age, soil types, precipitation, temperature, and NPP) on CIP (Supplementary Table 2). Results confirmed that initial soil pH was the most dominant factor controlling the variation in CIP, followed by afforestation species choices. It is noteworthy that stand age has little impact on CIP variations (Supplementary Fig. 3, Supplementary Table 2), likely due to the wide distribution of sampled sites over a very large region. The spatial heterogeneity of those sites could be large enough to cover up possible effect of stand age on CIP.
Moreover, we investigated the relationship between CIP and initial soil pH across different soil depths (Supplementary Fig. 4), and the results also confirmed the robustness of afforestation-induced soil pH neutralization. For example, similar patterns of CIP were observed at different soil depths, except for P. tabuliformis soils below 30 cm (p = 0.41 at 30–60 cm and 0.64 at 60–100 cm, Supplementary Fig. 4).
Discussion
Collectively, our results showed that afforestation tended to neutralize soil pH. An earlier meta-analysis study of site-scale observations suggested that afforestation acidified soil globally14. Our results, however, suggested that post-afforestation soil pH could change toward either direction (i.e. increased or decreased soil pH), dependent on the initial soil pH value. Afforestation acidified alkaline soils and alkalinized acid soils for all the five afforestation tree species. For a given tree species, there existed a tipping point of soil pH (TpH), where afforestation changed from increasing to decreasing soil pH. Measuring pre-afforestation initial soil pH and identifying species-specific TpH are thus essential for accurately predicting and dealing with post-afforestation soil pH changes.
Afforestation could lead to soil pH neutralization over the long term by altering the balance between soil hydrogen ion generation and consumption during nutrient cycle. Firstly, afforestation-caused changes in litter decomposition and rhizospheric processes29,30 may play a major role in soil pH neutralization. The addition of plant residues can increase, decrease or have little effect on soil pH31,32,33,34,35, depending on initial soil pH, nitrogen in plant materials, and the proportion of cations and organic anions. The input of plant residues decreases soil pH at high initial soil pH through nitrogen nitrification in the residue19,20,30. However, at low initial soil pH, the activity of nitrification bacteria will be suppressed and mineralized nitrogen tends to be ammonified and thus increase soil pH19,20,31. In addition, the organic anion-to-acid ratio of plant residues also has a significant role in determining whether litter decomposition would increase or decrease soil pH, subject to the value of initial soil pH as well31,32,33,34,35. Moreover, root exudates (e.g. HCO3−, OH−, and H+) can modify rhizospheric pH to enhance nutrient uptake by plant roots36. Roots often secrete HCO3− when plants take up more anions than cations from acid soils, leading to a higher rhizospheric pH than that of bulk soils. By contrast, in calcareous soils, plants take up more cations than anions, which reduces the rhizospheric pH by releasing H+ from their roots to maintain charge balance36. The differences in the composition of plant residues and rhizospheric processes thus lead to different CIPs across different initial soil pH and species. Furthermore, these processes also vary with soil depths and hence generate the vertical differentiations of CIP.
Secondly, root exudates may also indirectly influence soil pH by solubilizing unavailable soil aluminum (Al), which provides major buffering capacity in acidic soils37,38,39,40. The soil−root interface of forested soils contains more water-extractable Al than bulk soils, and afforestation may increase soil Al concentrations41,42 through rhizospheric processes43,44. The ability of hydrated aluminum ions to donate and accept H+ would make it eligible to be an acid or a base, depending on pH of soil solution. Under the domination of the reaction Al3+ + 2H2O <=> AlO2− + 4 H+, soil pH reaches an equilibrium at about 5−5.5 (the accurate value depends on the total Al concentration37,38,39). When soil pH is below the equilibrium value, Al(OH) n 3−n condenses to Al3+ through the reaction Al(OH)3 (s) + 3H+ → Al3+ + 3H2O, thus reducing [H+] and increasing soil pH. Here, Al3+ plays the same role as base cations (e.g. Ca2+, Mg2+) do. When soil pH is above the equilibrium value, Al3+ is hydrolyzed to Al(OH)3 (s), which releases more H+ and thus decreases soil pH. At that time, aluminum ion is an acid cation. However, these equilibrium (or threshold) pH values have only been empirically estimated by few local-scale studies in places such as Sweden39 and northeastern United States45, which may limit its applications in broader geographical regions without further verifications. In particular, the semi-arid climate in northern China’s forests and their soil properties may differ from that of Skyllberg39 and Ross et al.45. Therefore, it still remains open questions whether and how Al buffers soil pH in temperate forest of northern China.
Thirdly, afforestation affects soil pH through influencing the base cation cycle19,20,46,47,48. It has been demonstrated that afforestation can decrease soil pH through plant uptake of base cations19,20. However, in acid soils, base cations are relatively scanty. Plants therefore need to get cations from deeper soil through hydrological processes20. Moreover, the increases of evapotranspiration48 caused by afforestation will reduce the leaching loss of base cations46,47, and thus increase soil pH. However, we did not observe a consistent relationship between CIP and change in soil moisture content (SMC) (Supplementary Table 3), which may result from the high sensitivity of SMC to weather conditions, for which one-time sampling may not well represent long-term soil hydrological characteristics.
Terrestrial ecosystems are under the threat of soil acidification caused by the deposition of atmospheric nitrogen and sulfur on local5,6,7 and global scales49. For instance, bulk nitrogen deposition has increased by approximately 60% in China during the past three decades50 and will probably continue to increase in the foreseeable future due to elevated levels of anthropogenic nitrogen inputs to ecosystems51. Our study indicates that afforestation has the potential to alleviate soil acidification caused by enhanced acidic deposition with the appropriate selection of tree species and thus could further increase ecosystem productivity and carbon sequestration. Admittedly, further field studies are still needed to determine best tree species according to soil properties, water availability and climate suitability, and designated ecosystem and socioeconomic goals. Possible mechanisms for the observed effect of soil pH neutralization by afforestation and its potential in mitigating soil acidification caused by increased acidic deposition also remain to be investigated, both qualitatively and quantitatively. Nonetheless, our finding challenges the conventional notion that afforestation usually acidifies soils. Instead, our comparative study along a 2000 km transection in northern China finds that afforestation neutralizes soil pH. This can be another benefit of afforestation: when appropriate tree species are selected based on initial soil pH, afforestation may have the potential to modify soil pH, which will promote soil health and increase ecosystem productivity12.
Methods
Study region
Soils samples were collected from northern China, in the provinces of Heilongjiang, Jilin, Liaoning, Hebei, Shanxi, Shaanxi, and the Inner Mongolia Autonomous Region. The sampling region covered most of the TNSDP area, which extends from 34.20 to 51.80°N and 106.81 to 133.31°E (Fig. 1). Mean annual temperature and precipitation range from −3 to 15 °C and 355 to 1068 mm year−1, respectively. Dominant soil types in this region include black soil, bog soil, brown coniferous forest soil, brown earths, brown pedocals, castanozems, chernozems, cold brown calcic soil, yellow earths and yellow-brown earths52, roughly corresponding to phaeozems, gleysols, humic cambisols, haplic/albic luvisols or eutric/dystric cambisols, haplic calcisols, kastanozems, chernozems, cambisols, haplic alisols, and ferric/haplic luvisols of the United Nations Food and Agriculture Organization (FAO)53, respectively.
Sampling design
We established a paired afforestation-control system to evaluate the effects of afforestation on soil pH. For each site, we chose one non-afforested plot as a control plot and several afforested plots with different stand ages (in some sites with only one tree species and one stand age, we sampled only one afforested plot; in other sites, we chose at least two afforested plots to cover different tree species and stand ages (maximum = 30 afforested plots)). Within a site, the distance between any afforested plot to its corresponding non-afforested control plot was less than 2.5 km to minimize the variation in soil and climatic properties between the pair; and that between any two afforested plots was more than 50 m but less than 5 km. It is noteworthy that the pre-afforestation vegetation types and soil types of afforested plots were the same as that of the corresponding control plots, according to the records provided by local forestry administrations. Specifically, the original vegetation types in this study include cropland, barren land, grassland, natural forest, and desert. Data of stand afforestation age were also obtained from local forestry administrations. For each plot, we dug three replicate soil profiles to a depth of 1 m. For each profile, soils were sampled from six layers (0–5, 5–10, 10–20, 20–30, 30–60, and 60–100 cm) using a cutting ring. Therefore, except for a few plots that we could not reach to 1 m in depth, we collected 18 soil samples in each plot; and 11,118 soil samples for the whole project. We also recorded the planted tree species for each afforested plot; and as a result, five major afforestation tree species, including P. koraiensis, L. gmelinii, P. sylvestris var. mongolica, P. tabuliformis, and Populus spp. (including Populus simonii, Populus × beijingensis, and Populus × xiaohei), were documented in this study.
Laboratory measurement of soil pH
All soil samples were air-dried to constant weights in a ventilated room, and roots and stones were removed. Samples were then gently grinded in a mortar and passed through 2-mm sieves. The pH of each sample was measured in 1:2.5 mixtures of soil and deionized water with a pH meter (PHS-3C, Lei-ci). Soil solutions were shaken for 30 min and then kept static for 5 min before pH measurement.
Data analysis
Because each plot included three replicate profiles and each profile included samples from different soil depths, we needed to appropriately derive the value of soil pH for each plot so that it could represent the mean concentration of hydrogen ions of soil in the whole plot. The mean concentration of hydrogen ions (Hp) for the entire soil profile was calculated from hydrogen ion concentration, [H+], of each layer weighted by its thickness:
where w j and H j are the thickness and the concentration of hydrogen ions of the jth layer, respectively. Similarly, the mean concentration of hydrogen ions in a plot (\(\overline {H_{\rm p}}\)) was calculated by averaging Hp of its three replicate profiles, then we calculated the total hydrogen ions of a plot and transformed it into the hydrogen ions in one-hectare soil (mole ha−1). However, the hydrogen ion content varies across some orders of magnitude and it is not approximately normal distribution, so logarithmic transformation is needed. Therefore, we got the average pH for each plot from a log transformation of \(\overline {H_{\rm p}}\):
For each afforestation−control pair, we calculated the change in pH (CIP) as:
Note that pH is the negative logarithm of [H+] and thus CIP represents the effected changes in the ratio of [H+] caused by afforestation. Given that each afforested plot corresponded to one control plot, we got 549 pairs of control-afforested data to conduct the following analyses. Firstly, we applied a Siegel−Tukey test to compare the frequency distributions of soil pH between the control and afforested groups. Secondly, we used a paired t-test to evaluate the difference of soil pH between the paired plots. In the same time, we also conducted independent sample t-tests to find if CIPs were significantly different from 0. False discovery rate (FDR) correction54 was used to control potential error rates in multiple comparisons. FDR correction sorts all the p values of t-tests in ascending order: p (1) <= p (2)⋯ <= p (m), and compares each p value (p(i)) with q*i/m (where i is the order, m is the number of groups, q is pre-defined significance level, e.g., 0.05, 0.01, and 0.001). If p(i) <= q*i/m, the null hypothesis is rejected. Thirdly, we used one-way and two-way analysis of variance (ANOVA) to test the effect of different factors on soil pH and CIP. Those factors used in this analysis included afforestation (control vs. afforested), tree species, and initial soil pH. Fourthly, we performed ordinary least square (OLS) to examine the correlation between CIP and initial pH. Finally, we conducted GLM to synthetically evaluate the effects of all the factors (i.e. initial soil pH, tree species, original vegetation types, stand age, soil types, precipitation, temperature, and NPP) on CIP (Supplementary Table 2). Data of mean annual precipitation and temperature were acquired from the China Meteorological Forcing Dataset55,56. This data set was created by merging a variety of data sources and included 3-h data at a resolution of 0.1° x 0.1°. For NPP, we used the Moderate Resolution Imaging Spectroradiometer (MODIS) data, MOD17A3 data set57. Data of soil type were obtained from the Harmonized World Soil Database (HWSD) v.1.258. Longitude and latitude of each plot recorded by global positioning system were used to extract the data of climate and NPP for each plot. Furthermore, partial regressions were used to evaluate the effects of stand age on CIP after controlling for initial soil pH (Supplementary Fig. 3). Changes in soil moisture content (SMC) were calculated from SMC in afforested plots minus SMC in control plots (Supplementary Table 3). Independent t-tests were used to exam whether the changes were significantly different from 0. All statistical analyses were conducted using MATLAB R2012b (MathWorks, Natick, MA, USA).
Data availability
The authors declare that the source data supporting the finding of this study are provided with the paper.
References
Tyler, G. & Olsson, T. Concentrations of 60 elements in the soil solution as related to the soil acidity. Eur. J. Soil Sci. 52, 151–165 (2001).
Bååth, E., Frostegård, A., Pennanen, T. & Fritze, H. Microbial community structure and pH response in relation to soil organic-matter quality in wood-ash fertilized, clear-cut or burned coniferous forest soils. Soil Biol. Biochem. 27, 229–240 (1995).
Kooijman, A. M. & Cammeraat, E. Biological control of beech and hornbeam affects species richness via changes in the organic layer, pH and soil moisture characteristics. Funct. Ecol. 24, 469–477 (2010).
Lambkin, D. C. et al. Soil pH governs production rate of calcium carbonate secreted by the earthworm Lumbricus terrestris. Appl. Geochem. 26, S64–S66 (2011).
Guo, J. H. et al. Significant acidification in major Chinese croplands. Science 327, 1008–1010 (2010).
Yang, Y. H. et al. Significant soil acidification across northern China’s grasslands during 1980s–2000s. Glob. Change Biol. 18, 2292–2300 (2012).
Yang, Y. H. et al. Long-term changes in soil pH across major forest ecosystems in China. Geophys. Res. Lett. 42, 933–940 (2015).
Stevens, C. J., Dise, N. B., Mountford, J. O. & Gowing, D. J. Impact of nitrogen deposition on the species richness of grasslands. Science 303, 1876–1879 (2004).
Stevens, C. J. et al. Nitrogen deposition threatens species richness of grasslands across Europe. Environ. Pollut. 158, 2940–2945 (2010).
Bowman, W. D., Cleveland, C. C., Halada, L., Hreško, J. & Baron, J. S. Negative impact of nitrogen deposition on soil buffering capacity. Nat. Geosci. 1, 767–770 (2008).
Janssens, I. A. et al. Reduction of forest soil respiration in response to nitrogen deposition. Nat. Geosci. 3, 315–322 (2010).
Kunito, T. et al. Aluminum and acidity suppress microbial activity and biomass in acidic forest soils. Soil Biol. Biochem. 97, 23–30 (2016).
Zhao, Y. et al. Soil acidification in China: is controlling SO2 emissions enough? Environ. Sci. Technol. 43, 8021–8026 (2009).
Berthrong, S. T., Jobbagy, E. G. & Jackson, R. B. A global meta-analysis of soil exchangeable cations, pH, carbon, and nitrogen with afforestation. Ecol. Appl. 19, 2228–2241 (2009).
Jackson, R. B. et al. Trading water for carbon with biological sequestration. Science 310, 1944–1947 (2005).
Bonan, G. B. Forests and climate change: forcings, feedbacks, and the climate benefits of forests. Science 320, 1444–1449 (2008).
Rhoades, C. & Binkley, D. Factors influencing decline in soil pH in Hawaiian Eucalyptus and Albizia plantations. For. Ecol. Manag. 80, 47–56 (1996).
Parfitt, R. & Ross, D. Long-term effects of afforestation with Pinus radiata on soil carbon, nitrogen, and pH: a case study. Soil Res. 49, 494–503 (2011).
Binkley D. & Fisher R. Ecology and Management of Forest Soils, 4th edn (Wiley-Blackwell, 2013).
Binkley, D. & Richter, D. Nutrient cycles and H+budgets of forest ecosystems. Adv. Ecol. Res 16, 1–15 (1987).
Paul, K. I. et al. Change in soil carbon following afforestation. For. Ecol. Manag. 168, 241–257 (2002).
Laganiere, J., Angers, D. A. & Pare, D. Carbon accumulation in agricultural soils after afforestation: a meta-analysis. Glob. Change Biol. 16, 439–453 (2010).
Binkley, D., Valentine, D., Wells, C. & Valentine, U. An empirical-analysis of the factors contributing to 20-year decrease in soil-pH in an old-field plantation of loblolly-pine. Biogeochemistry 8, 39–54 (1989).
Rigueiro-Rodríguez, A., Mosquera-Losada, M. R. & Fernández-Núñez, E. Afforestation of agricultural land with Pinus radiata D. don and Betula alba L. in NW Spain: effects on soil PH, understorey production and floristic diversity eleven years after establishment. Land Degrad. Dev. 23, 227–241 (2012).
Piao, S. L. et al. The carbon balance of terrestrial ecosystems in China. Nature 458, 1009–1013 (2009).
Duan, H. C. et al. Assessing vegetation dynamics in the Three-North Shelter Forest region of China using AVHRR NDVI data. Environ. Earth Sci. 64, 1011–1020 (2011).
He, B., Chen, A. F., Wang, H. L. & Wang, Q. F. Dynamic response of satellite-derived vegetation growth to climate change in the Three North Shelter Forest Region in China. Remote Sens. 7, 9998–10016 (2015).
Zhang, Y. et al. Multiple afforestation programs accelerate the greenness in the ‘Three North’ region of China from 1982 to 2013. Ecol. Indic. 61, 404–412 (2016).
Ande Schrijver et al. Tree species traits cause divergence in soil acidification during four decades of post agricultural forest development. Glob. Change Biol. 18, 1127–1140 (2012).
Rukshana, F., Butterly, C. R., Xu, J. M., Baldock, J. A. & Tang, C. X. Organic anion-to-acid ratio influences pH change of soils differing in initial pH. J. Soils Sediment. 14, 407–414 (2014).
Xu, J. M., Tang, C. & Chen, Z. L. The role of plant residues in pH change of acid soils differing in initial pH. Soil Biol. Biochem. 38, 709–719 (2006).
Marschner, B. & Noble, A. D. Chemical and biological processes leading to the neutralisation of acidity in soil incubated with litter materials. Soil Biol. Biochem. 32, 805–813 (2000).
Rukshana, F., Butterly, C. R., Baldock, J. A. & Tang, C. X. Model organic compounds differ in their effects on pH changes of two soils differing in initial pH. Biol. Fertil. Soils 47, 51–62 (2011).
Tang, C. & Yu, Q. Impact of chemical composition of legume residues and initial soil pH on pH change of a soil after residue incorporation. Plant. Soil 215, 29–38 (1999).
Xu, J. M., Tang, C. & Chen, Z. L. Chemical composition controls residue decomposition in soils differing in initial pH. Soil Biol. Biochem. 38, 544–552 (2006).
Dakora, F. D. & Phillips, D. A. Root exudates as mediators of mineral acquisition in low-nutrient environments. Plant. Soil 245, 35–47 (2002).
Clarholm, M. & Skyllberg, U. Translocation of metals by trees and fungi regulates pH, soil organic matter turnover and nitrogen availability in acidic forest soils. Soil Biol. Biochem. 63, 142–153 (2013).
Skyllberg, U. Aluminum associated with a pH-increase in the humus layer of a boreal haplic podzol. Interciencia 19, 356–365 (1994).
Skyllberg, U. Small scale pH buffering in organic horizons of two boreal coniferous forest stands. Plant. Soil 179, 99–107 (1996).
Skyllberg, U., Raulund-Rasmussen, K. & Borggaard, O. K. pH buffering in acidic soils developed under Picea abies and Quercus robur—effects of soil organic matter, adsorbed cations and soil solution ionic strength. Biogeochemistry 56, 51–74 (2001).
Adams, M. L., Davis, M. R. & Powell, K. J. Effects of grassland afforestation on exchangeable soil and soil solution aluminium. Aust. J. Soil Res. 39, 1003–1014 (2001).
Li, Y. Q. & Mathews, B. W. Effect of conversion of sugarcane plantation to forest and pasture on soil carbon in Hawaii. Plant. Soil 335, 245–253 (2010).
Gottlein, A., Heim, A. & Matzner, E. Mobilization of aluminium in the rhizosphere soil solution of growing tree roots in an acidic soil. Plant. Soil 211, 41–49 (1999).
Seguin, V., Gagnon, C. & Courchesne, F. Changes in water extractable metals, pH and organic carbon concentrations at the soil−root interface of forested soils. Plant. Soil 260, 1–17 (2004).
Ross, D. S., David, M. B., Lawrence, G. B. & Bartlett, R. J. Exchangeable hydrogen explains the pH of Spodosol Oa horizons. Soil Sci. Soc. Am. J. 60, 1926–1932 (1996).
Slessarev, E. W. et al. Water balance creates a threshold in soil pH at the global scale. Nature 540, 567–569 (2016).
Chytrý, M. et al. Plant species richness in continental southern Siberia: effects of pH and climate in the context of the species pool hypothesis. Glob. Ecol. Biogeogr. 16, 668–678 (2007).
Yao, Y. Y. et al. The effect of afforestaion on soil moisture content in northeastern China. PLoS ONE 11, e016776 (2016).
Lu, M. et al. Responses of ecosystem nitrogen cycle to nitrogen addition: a meta-analysis. New Phyto 189, 1040–1050 (2011).
Liu, X. J. et al. Enhanced nitrogen deposition over China. Nature 494, 459–462 (2013).
Gu, B. J. et al Integrated reactive nitrogen budgets and future trends in China. Proc. Natl. Acad. Sci. USA 112, 8792–8797 (2015).
Xiong, Y., & Li, Q. K.. Soils in China (Press of Sciences: Beijing, 1987).
Xie, Z. B. et al. Soil organic carbon stocks in China and changes from 1980s to 2000s. Glob. Change Biol. 13, 1989–2007 (2007).
Benjamini, Y. & Yekutieli, D. The control of false discovery rate in multiple testing under dependency. Ann. Stat. 4, 1165–1188 (2001).
Yang, K., He, J., Tang, W. J., Qin, J. & Cheng, C. C. On downward shortwave and longwave radiations over high altitude regions: observation and modeling in the Tibetan Plateau. Agric. Meteorol. 150, 38–46 (2010).
Chen, Y. Y. et al. Improving land surface temperature modeling for dry land of China. J. Geophys. Res. Atmos. 116, 999–1010 (2011).
Running, S. Q. M. MOD17A3H MODIS/TerraNet Primary Production Yearly L4 Global 500m SIN Grid V006. NASA EOSDIS Land Processes DAAC. https://doi.org/10.5067/modis/mod17a3h.006 (2015).
FAO, IIASA, ISRIC, ISSCAS & JRC. Harmonized World Soil Database (version 1.2). (FAO and IIASA: Rome, Italy, and Laxenburg, Austria, 2012).
Acknowledgements
This study was supported by the National Key R&D Program of China (2017YFA0604702), the National Natural Science Foundation of China (41530528 and 31621091), the 111 Project (B14001). Funding for this study was also provided by undergraduate student research training program of the Ministry of Education. A.C. acknowledges the support of a research fellowship provided by the Purdue University Department of Forestry and Natural Resources.
Author information
Authors and Affiliations
Contributions
S. Piao designed the research; S.H. performed the analysis; and S. Piao, S.H., A.C., Y.L., L.L., S. Peng, J.S., Y.S., J.P., H.Z. contributed to the interpretation of the results and to the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hong, S., Piao, S., Chen, A. et al. Afforestation neutralizes soil pH. Nat Commun 9, 520 (2018). https://doi.org/10.1038/s41467-018-02970-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-02970-1
This article is cited by
-
Positive effects of plant species diversity on organic carbon accumulation in soil aggregates driven by mineral protection in a subtropical forest in southwest China
Journal of Soils and Sediments (2024)
-
Natural forests exhibit higher organic carbon concentrations and recalcitrant carbon proportions in soil than plantations: a global data synthesis
Journal of Forestry Research (2024)
-
Comparative Analysis of the Drivers of Soil Organic Carbon, Total Nitrogen, and Phosphorus Stocks in Different Coniferous Plantations on the Eastern Tibetan Plateau
Journal of Soil Science and Plant Nutrition (2024)
-
Analysis of heavy metal content and microbial characteristics in the pioneer plant soil system of typical manganese tailing ponds in Guangxi
Plant and Soil (2024)
-
Asymmetry of carbon sequestrations by plant and soil after forestation regulated by soil nitrogen
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.