Abstract
Rare-earth nickelates form an intriguing series of correlated perovskite oxides. Apart from LaNiO3, they exhibit on cooling a sharp metal-insulator electronic phase transition, a concurrent structural phase transition, and a magnetic phase transition toward an unusual antiferromagnetic spin order. Appealing for various applications, full exploitation of these compounds is still hampered by the lack of global understanding of the interplay between their electronic, structural, and magnetic properties. Here we show from first-principles calculations that the metal-insulator transition of nickelates arises from the softening of an oxygen-breathing distortion, structurally triggered by oxygen-octahedra rotation motions. The origin of such a rare triggered mechanism is traced back in their electronic and magnetic properties, providing a united picture. We further develop a Landau model accounting for the metal-insulator transition evolution in terms of the rare-earth cations and rationalizing how to tune this transition by acting on oxygen rotation motions.
Similar content being viewed by others
Introduction
First synthesized in 19711, rare-earth nickelates (RNiO3, with R = rare earth) are appealing for various applications2, 3, and the possibility to tune their properties in epitaxial films and heterostructures4 has recently fueled an even larger interest5,6,7. RNiO3 compounds belong to the family of perovskite oxides with a reference \(Pm\bar 3m\) cubic structure (Fig. 1a), nevertheless not directly observed. Apart for LaNiO3, which always keeps a metallic \(R\bar 3c\) phase and will not be further discussed here, all RNiO3 adopt at reasonably high temperature a metallic Pbnm orthorhombic phase8. This phase, rather ubiquitous9 amongst perovskites with small Goldschmidt tolerance factor8, t, corresponds to a distortion of the cubic structure arising from the appearance of combined anti-phase rotations of the oxygen octahedra along the x and y directions, R xy (Fig. 1b), with the same amplitude Q R and in-phase rotations of the oxygen octahedra along z, M z (Fig. 1c), with amplitude Q M.
Reference cubic perovskite structure and most relevant atomic distortions. a Sketch of the reference \(Pm\bar 3m\) cubic perovskite cell of RNiO3 compound with R at the corner, Ni at the center, and O atoms at the middle of the faces, forming corner-shared octahedra surrounding the B atoms. Starting from this reference, three main atomic distortions drive the system successively to the Pbnm and then P21/n phases: b anti-phase rotations of oxygen octahedra of same amplitudes about x- and y-axis (R xy ), c in-phase rotations of oxygen octahedra about z-axis (M z ), d breathing of the oxygen octahedra (B OC). The energy wells associated to the freezing of individual distortion of increasing amplitude in the cubic cell are shown for different R cations, associated to distinct tolerance factor t, and either a ferromagnetic (full line) or E′-type antiferromagnetic (dashed line, only for R = Y) spin arrangement: YNiO3 (t = 0.920, blue); GdNiO3 (t = 0.938, orange) and SmNiO3 (t = 0.947, green). The atomic distortions are normalized to their amplitude in the P21/n AFM-E′ ground state of YNiO3. Calculations are done for each compound in a cubic cell that has the same volume as the ground state
In this phase, all Ni atoms are equivalent and formally in a Jahn-Teller active d 7 (likely \(t_{{\mathrm{2g}}}^6e_{\mathrm{g}}^1\) low spin) configuration. Surprisingly, although compatible with the Pbnm symmetry, cooperative Jahn-Teller distortions do not appear. Instead, at a temperature T MI, which strongly evolves with the R cation (i.e., with t)10, RNiO3 compounds exhibit an electronic metal-insulator transition (MIT), which was shown to be concurrent with a structural transition from Pbnm to monoclinic P21/n symmetry11. This symmetry lowering is accompanied with the appearance of a breathing distortion of the oxygen octahedra, B OC (Fig. 1d), which alternatively expand and contract with amplitude Q B, according to a rock-salt pattern. This gives rise to two types of Ni sites with long and short Ni–O bonds, respectively.
At low temperature (100–200 K), RNiO3 compounds finally show a magnetic phase transition toward an unusual E′-type antiferromagnetic (AFM) spin order identified by a Bragg vector q = (1/4, 1/4, 1/4) in pseudocubic notation11,12,13. For large cations (R = Nd and Pr), the Néel temperature T N = T MI and the system goes directly from paramagnetic metal (PM-M) to AFM insulator (AFM-I). For smaller R cations, T N is much lower than T MI; the two phase transitions are decoupled and the system goes through an intermediate paramagnetic insulating phase (PM-I).
The origin of the MIT has been widely debated in the literature14,15,16,17,18,19. It was sometimes interpreted as a charge disproportionation at Ni sites20: 2(d 7) → d 8 + d 6. However, the importance of Ni 3d-O 2p hybridization—i.e., transfer of electrons from O to Ni and formation of oxygen holes \(\left( {\underline L } \right)\) keeping Ni in a d 8 configuration (i.e., \(d^{8 - n} \approx d^8\underline L ^n\))—was evoked early on15. Nowadays, the MIT is usually viewed as a charge ordering of the type \(2\left( {d^8\underline L ^1} \right) \to \left( {d^8} \right) + \left( {d^8\underline L ^2} \right)\) 17, 21, 22. In this scenario, B OC appears important to stabilize the charge ordering and open the gap. As suggested in ref. 23 and confirmed from statistical correlation techniques24, R xy and M z are also expected to play an active role. However, a complete picture linking electronic, structural and magnetic properties is yet to emerge.
Unlike recent theoretical studies, which were focusing specifically on the electronic properties17,18,19, 21, we investigate here self-consistently the electronic and structural degrees of freedom of RNiO3 compounds from density functional theory calculations (DFT, see Methods). Specific care was given to the validation of our approach, which turns out to provide an unprecedented agreement with experimental data. Focusing on YNiO3, we show (Supplementary Notes 1–4) that not only the atomic structure but also the AFM-E′ ground state, the estimated T N and the electronic bandgap of the insulating phase are very accurately reproduced, making therefore our approach a method of choice to shed light on the interlink between these different features.
Results
Energetics of individual lattice distortions
We start from the reference \(Pm\bar 3m\) cubic structure. Inspection of the phonon dispersion curves (Supplementary Note 5) reveals dominant structural instabilities at R (ω R = 310i cm−1) and M (ω M = 278i cm−1) points of the Brillouin zone (BZ), which are associated, respectively, to the R xy and M z distortions responsible for the Pbnm phase. These imaginary frequencies ω i are linked to a negative energy curvature α i at the origin \(\left( {\alpha _{\mathrm{i}} \propto \omega _{\mathrm{i}}^2 < 0} \right)\) and to a typical double-well (DW) shape of the energy when freezing R xy and M z distortions of increasing amplitude within the cubic structure, as illustrated in Fig. 1. These wells are nearly independent of the spin order but strongly evolve with the R cation: they become shallower when R size increases, consistently with a reduction of the related distortion amplitudes in the Pbnm phase.
In contrast, the B OC motion, corresponding to another phonon at R, is stable and extremely stiff (in fact the stiffest mode with ω B = 700 cm−1), in line with the single-well (SW) shape illustrated in Fig. 1. Decreasing R cation size tends to stabilize slightly further B OC, in apparent contradiction with the observed increase of T MI. As illustrated for YNiO3, switching from ferromagnetic (FM) to AFM-E′ spin order reduces slightly the curvature but does not reverse it; instead it shifts the SW to lower energy13, yielding a finite Q B at the minimum. Although B OC tends to make the system insulating, this amplitude (25% of ground state’s value) is however not large enough to open a gap (more than 75% would be required). This shows that B OC and the magnetic order alone cannot explain the MIT by themselves.
Mode coupling and triggered mechanism
Our central result is presented in Fig. 2 where we report in panel a the evolution of the B OC energy well of YNiO3 at various fixed amplitudes of oxygen rotation motions. It highlights that although initially stable (SW), B OC is progressively destabilized (DW) by the appearance of R xy and M z . As illustrated in panel b, α B is renormalized into \(\tilde \alpha _{\mathrm{B}}\), which evolves linearly with \(Q_{\mathrm{R}}^2\) and \(Q_{\mathrm{M}}^2\). The slope associated to Q R is twice as large as that related to Q M, attesting that each of the three individual rotations similarly affects B OC. This behavior arises from the presence in the energy expansion of cooperative (λ < 0) bi-quadratic coupling terms between B OC and oxygen rotations (\(E \approx \lambda _{{\mathrm{Bi}}}Q_{\mathrm{B}}^{\mathrm{2}}Q_{\mathrm{i}}^{\mathrm{2}}\), i = M,R), which, being the lowest-order couplings allowed by symmetry, should give rise to the appearance of B OC through a triggered phase transition according to Holakovsky25. The same behavior is observed independently of the magnetic order (Supplementary Note 8). From now we focus on representative FM results while coming back to the role of magnetism later.
Triggered mechanism. a Evolution of the energy E in terms of the amplitude of the breathing distortion (Q B) for fixed amplitudes of oxygen rotations (Q R = Q M from 0 to 110%) in the FM cubic cell of YNiO3 (same volume as the ground state). It highlights the softening of the energy well associated to B OC, triggered by the oxygen rotations R xy and M z . b Linear evolution of the energy curvature at the origin, \(\tilde \alpha _B\) along Q B, in terms of the square of the amplitude of the individual distortions Q R (orange) and Q M (blue)
Landau model and phase diagram
To further assess the relevance of such a triggered mechanism in nickelates, we built a Landau model, including R xy , M z , and B OC degrees of freedom24, restricting ourselves to lowest coupling terms and assuming temperature dependence of the oxygen rotations only:
Within this model, R xy and M z appear at T 0R and T 0M, respectively. On cooling, they progressively develop within the Pbnm phase and renormalize the energy curvature α B of B OC as made clear when grouping the \(Q_{\mathrm{B}}^2\) terms in Eq. (1):
When reaching a critical amplitude at which \(\tilde \alpha _{\mathrm{B}} = 0\), they trigger the appearance of B OC and produce concurrent structural and metal-insulator phase transitions. The phase transition appears to be second order within this model, which is however too simple to be conclusive on this point (Supplementary Note 10).
All parameters and their evolution with R were directly fitted from first principles; only Curie temperatures were uniformly scaled to reproduce the experimental T MI of YNiO3 (Supplementary Note 8). The phase diagram of nickelates as predicted within this model is reported in Fig. 3. This figure demonstrates that the cooperative coupling of B OC with R xy and M z is a key mechanism that, by itself, accounts for the experimentally observed evolution of T MI with the tolerance factor.
Nickelate phase diagram. Phase diagram of RNiO3 compounds in terms of their tolerance factor t and the temperature T. It includes three phases: a metallic Pbnm paramagnetic phase (PM-M, yellow area); an insulating P21/n paramagnetic phase (PM-I, green area); and an insulating P21/n E′-type AFM phase (AFM-I, magenta area). The yellow line and dots show the experimental evolution of T MI with the tolerance factor t. The blue line and squares is the prediction of the simple Landau model fitted on the first-principles data (FM order). The dashed blue line is the fit of the Landau expression of T MI (t) on experimental data. The blue star is the magnetic phase transition predicted for YNiO3 from first principles. The blue arrows indicate the correction to be applied on T MI for large cations when properly incorporating the change of magnetic order
Hence, the MIT in nickelates turns out to be a concrete example of triggered phase transition26, 27, a kind of transition never identified before in simple perovskites. Indeed, although bi-quadratic interactions are generic in this class of compounds, different distortions usually compete and exclude each other9. The cooperative coupling of B OC with oxygen rotations pointed out here is therefore an unusual and intriguing feature, whose origin is now traced back in the electronic band structure.
Electronic origin of the triggered mechanism
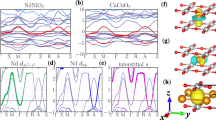
In the cubic phase, as expected from the formal Ni 3d 7 \(\left( {t_{{\mathrm{2g}}}^6e_{\mathrm{g}}^1} \right)\) occupancy, the Fermi energy E f crosses levels of dominant Ni 3d-e g character (i.e., anti-bonding Ni 3d-O 2p states); such levels form an isolated and rather dispersive set of two e g bands, shifted above the t 2g levels by the crystal field (Fig. 4). Forcing into this cubic structure a B OC distortion, associated to a phonon at q R = (1/2, 1/2, 1/2), can open a gap at q c = (1/4, 1/4, 1/4) within the e g bands but well above E f and without any direct impact on the metallic character and the occupied states. Nevertheless, the oxygen rotation motions substantially affect the e g bands (Fig. 4), reducing their bandwidth and yielding a progressive down shift of the e g levels at q c. As the rotations gradually increase and the bandwidth decreases, B OC more substantially pushes down the electronic states around E f, providing a progressive gain of electronic energy, which, in turn, results in the softening of ω B. The critical rotation amplitude at which B OC becomes unstable \(\left( {\tilde \alpha_{\mathrm{B}} = 0} \right)\) is precisely that at which the e g levels at q c cross E f. At these amplitudes, the electronic system itself becomes unstable; the appearance of B OC is favored and opens a gap at E f, making the system insulating. As such, the MIT can therefore be interpreted as a Peierls instability but one that is not initially present and has been triggered by oxygen rotations.
Electronic properties. a Electronic dispersion curves of YNiO3 along different high-symmetry line of the Brillouin zone of the \(Pm\bar 3m\) phase (FM case, majority spins): Γ = (0, 0, 0), X = (1/2, 0, 0), M = (1/2, 1/2, 0), and R = (1/2, 1/2, 1/2). The Ni 3d e g bands are highlighted in green. The Fermi energy corresponds to the horizontal blue line. The point q c = (1/4, 1/4, 1/4) is located by vertical dashed blue lines. b–g Electronic dispersion curves around the Fermi energy E f (FM order, majority spins) along high-symmetry points (coordinates in pseudocubic notations) in the Brillouin zone of the Pbnm or P21/n 20-atom cell, in which bands have been folded respect to a. Consecutive panels show the evolution of the dispersion curves when freezing into the \(Pm\bar 3m\) phase increasing amplitudes of oxygen rotations (Q R = Q M = b 0%, d 50%, e 75%, or f 90%, lines) and eventually adding the breathing distortion (Q B = 100% and Q R = Q M = c 0% or g 90%, dashed lines)
Discussion
For compounds with small R cations, Q R and Q M are large and able to produce the MIT at relatively high temperatures, well above T N. For large cations (R = Nd, Pr), oxygen rotations are reduced and, from our Landau model (built on FM results), no more sufficient to trigger the MIT (Fig. 3). However, as previously mentioned, the AFM-E′ spin order is compatible by symmetry with B OC and induces its appearance as an improper order (linear shift of B OC SW, Fig. 1d). Hence, although not opening a gap in the cubic phase, the onset of the AFM-E′ order in the Pbnm phase of NdNiO3 and PrNiO3 promotes the occurrence of the MIT almost triggered by the rotations. In these compounds, we have therefore T MI = T N; the transition is more abrupt and first-order8. Such active role of magnetism for large cations is supported by the experimental results and discussion in ref. 28. It is also confirmed by our first-principles calculations on NdNiO3, showing that the predicted T MI is rescaled when including the change of magnetic order: while the system prefers to stay in the Pbnm metallic phase when imposing a FM order, it switches to the P21/n phase when adopting a AFM-E′ order. The cooperative effect of the magnetic order remains true for small cations but without any impact on T MI (>T N).
In conclusion, the concurrent electronic and structural transitions at T MI in RNiO3 compounds take the form of a Peierls instability, which, primarily, is structurally triggered by the oxygen rotation motions R xy and M z and, eventually, is further assisted by the appearance of the E′-type AFM magnetic ordering. Our Landau model, and its possible extension to incorporate explicitly strain degrees of freedom neglected here for simplicity, provides a simple and useful quantitative tool to estimate and interpret how T MI can be tuned toward the monitoring of oxygen rotation motions R xy and M z when making solid solutions10, applying pressure8, or playing with the epitaxial strain and the orientation of the substrate in thin films29. Our findings are relevant to other families of perovskites like A2+ Fe4+ O3 compounds30. For instance, they can explain why CaFeO3, which exhibits oxygen rotations, undergoes a MIT while SrFeO3 and BaFeO3, which stay cubic, remain metallic. In addition, the same physics is also inherent to manganites like LaMnO3, suggesting a close competition between charge and orbital orderings in this family compounds. However, the situation is slightly different in a bismutate like BaBiO3, in which B OC is intrinsically unstable in the cubic phase31.
Methods
First-principles calculations
First-principles calculations were performed in the framework of DFT32, 33 using a Projected Augmented Wave (PAW) approach34 as implemented within the ABINIT package35,36,37,38. The calculations relied on the Generalized Gradient Approximation using the PBEsol39 exchange-correlation functional. We worked within a collinear spin approximation. We included a Hubbard correction U = 1.5 eV40 on the 3d orbitals of Ni atoms. A special care was devoted to the determination of the appropriate U parameter (Supplementary Notes 1– 4).
We made use of JTH atomic potentials41. For the wavefunctions, we used an energy cutoff of 24 Ha (38 Ha for the second grid in the PAW spheres), which guarantees a convergence better than 1 meV on the total energy. The BZ was sampled with k-point meshes equivalent to a 12 × 12 × 12 grid in the five-atom unit cell. During structural relaxations, thresholds of 10−5 Ha/bohr on the maximum force and of 10−7 Ha/bohr3 on the maximum stress have been considered.
Structural analysis
The Goldschmidt tolerance factor42, t = d R−O/\(\sqrt 2\) d Ni−O, of RNiO3 compounds were determined using Nicole Benedek’s tool43 relying on a bond valence model44 to calculate d R−O and d Ni−O, respectively, the ideal R–O and Ni–O bond lengths in the cubic perovskite structure.
Symmetry-adapted mode analysis have been performed with AMPLIMODE45, 46. The modes are normalized to their amplitude in the P21/n AFM-E′ ground state. This normalization is such that in cubic phase (volume of the ground state) Q R = 100% corresponds to rotation angles ϕ x = ϕ y = 11.33° (Ni–O–Ni angle of 157.33°), Q R = 100% corresponds to a rotation angle ϕ z = 12.12° (Ni–O–Ni angle of 155.75°) and Q B = 100% corresponds to oxygen displacements d O = 0.0358.
Landau model
The Landau model parameters have been fitted for YNiO3, GdNiO3, and SmNiO3 on first-principles data using in each case a FM cubic phase (volume of the ground state) and interpolated for the other compounds. T MI was determined analytically (Supplementary Notes 6–9).
Data availability
All relevant data are available from the authors.
References
Demazeau, G., Marbeuf, A., Pouchard, M. & Hagenmuller, P. Sur une série de composés oxygènes du nickel trivalent derivés de la perovskite. J. Solid State Chem. 3, 582 (1971).
Shi, J., Ha, S. D., Zhou, Y., Schoofs, F. & Ramanathan, S. A correlated nickelate synaptic transistor. Nat. Commun. 4, 2676 (2013).
Zhou, Y. et al. Strongly correlated perovskite fuel cells. Nature 534, 231–234 (2016).
Middey, S. et al. Physics of ultrathin films and heterostructures of rare-earth nickelates. Annu. Rev. Mater. Res. 46, 305–334 (2016).
Först, M. et al. Spatially resolved ultrafast magnetic dynamics initiated at a complex oxide heterointerface. Nat. Mater. 14, 883–888 (2015).
Kim, T. H. et al. Polar metals by geometric design. Nature 533, 68–72 (2016).
Grisolia, M. N. et al. Hybridization-controlled charge transfer and induced magnetism at correlated oxide interfaces. Nat. Phys. 12, 484–492 (2016).
Catalan, G. Progress in perovskite nickelate research. Phase Transit. 81, 729–749 (2008).
Benedek, N. A. & Fennie, C. J. Why are there so few perovskite ferroelectrics? J. Phys. Chem. C 117, 13339–13349 (2013).
Medarde, M. L. Structural, magnetic and electronic properties of RniO3 perovskites (R = rare earth). J. Phys. Condens. Matter 9, 1679 (1997).
Alonso, J. A. et al. Charge disproportionation in RniO 3 perovskites: simultaneous metal-insulator and structural transition in YNiO 3 . Phys. Rev. Lett. 82, 3871–3874 (1999).
García-Muñoz, J. L., Rodríguez-Carvajal, J. & Lacorre, P. Neutron-diffraction study of the magnetic ordering in the insulating regime of the perovskites RniO3 R = Pr and Nd). Phys. Rev. B 50, 978–992 (1994).
Lee, S., Chen, R. & Balents, L. Landau theory of charge and spin ordering in the nickelates. Phys. Rev. Lett. 106, 016405 (2011).
Torrance, J. B., Lacorre, P., Nazzal, A. I., Ansaldo, E. J. & Niedermayer, C. Systematic study of insulator-metal transitions in perovskites RniO3 R = Pr, Nd, Sm, Eu) due to closing of charge-transfer gap. Phys. Rev. B 45, 8209–8212 (1992).
Mizokawa, T., Khomskii, D. I. & Sawatzky, G. A. Spin and charge ordering in self-doped Mott insulators. Phys. Rev. B 61, 11263–11266 (2000).
Raebiger, H., Lany, S. & Zunger, A. Charge self-regulation upon changing the oxidation state of transition metals in insulators. Nature 453, 763–766 (2008).
Park, H., Millis, A. J. & Marianetti, C. A. Site-selective mott transition in rare-earth-element nickelates. Phys. Rev. Lett. 109, 156402 (2012).
Ruppen, J. et al. Optical spectroscopy and the nature of the insulating state of rare-earth nickelates. Phys. Rev. B 92, 155145 (2015).
Varignon, J., Grisolia, M. N., Iniguez, J., Barthélémy, A. & Bibes, M. Complete phase diagram of rare-earth nickelates from first principles. npj Quant. Mater. 2, 21 (2017).
Mazin, I. I. et al. Charge ordering as alternative to jahn-teller distortion. Phys. Rev. Lett. 98, 176406 (2007).
Johnston, S., Mukherjee, A., Elfimov, I., Berciu, M. & Sawatzky, G. A. Charge disproportionation without charge transfer in the rare-earth-element nickelates as a possible mechanism for the metal-insulator transition. Phys. Rev. Lett. 112, 106404 (2014).
Bisogni, V. et al. Ground state oxygen holes and the metal-insulator transition in the negative charge transfer rare-earth nickelates. Nat. Commun. 7, 13017 (2016).
de la Cruz, F. P. et al. Possible common ground for the metal-insulator phase transition in the rare-earth nickelates RniO3 (R = Eu, Ho, Y). Phys. Rev. B 66, 153104 (2002).
Balachandran, P. V. & Rondinelli, J. M. Interplay of octahedral rotations and breathing distortions in charge-ordering perovskite oxides. Phys. Rev. B 88, 054101 (2013).
Holakovsky, J. A new type of ferroelectric phase transition. Phys. Status Solidi B 56, 615–619 (1973).
Flerov, I. N., Gorev, M. V., Voronov, V. N., Tressaud, A. & Grannec, J. Triggered phase transitions in Rb2KB3+F6(B3+: Er, ho) elpasolites. J. Solid State Chem. 3, 582 (1971).
Iwata, M., Zhao, C. H., Aoyagi, R., Maeda, M. & Ishibashi, Y. Splitting of triggered phase transition in Bi4−x La x Ti3O12 mixed crystals. Japonese. J. Appl. Phys. 46, 5894 (2007).
Vobornik, I. et al. Electronic-structure evolution through the metal-insulator transition in RniO3. Phys. Rev. B 60, R8426–R8429 (1999).
Catalano, S. et al. Tailoring the electronic transitions of NdNiO3 films through (111)pc oriented interfaces. APL Mater. 3, 062506 (2015).
Akao, T. et al. Charge-ordered state in single-crystalline CaFeO3 thin film studied by x-ray anomalous diffraction. Phys. Rev. Lett. 91, 156405 (2003).
Liechtenstein, A. I. et al. Structural phase diagram and electron-phonon interaction in Ba1−x K x BiO3. Phys. Rev. B 44, 5388–5391 (1991).
Hohenberg, P. & Kohn, W. Inhomogeneous electron gas. Phys. Rev. 136, B864–B871 (1964).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Gonze, X. et al. ABINIT: First-principles approach to material and nanosystem properties. Comput. Phys. Commun. 180, 2582–2615 (2009).
Gonze, X. et al. First-principles computation of material properties: the ABINIT software project. Computat. Mater. Sci. 25, 478–492 (2002).
Gonze, X. et al. A brief introduction to the abinit software package. Z. Kristallogr. Cryst. Mater. 220, 558–562 (2005).
Torrent, M., Jollet, F., Bottin, F., Zérah, G. & Gonze, X. Implementation of the projector augmented-wave method in the ABINIT code: application to the study of iron under pressure. Comput. Mater. Sci. 42, 337–351 (2008).
Perdew, J. P. et al. Restoring the density-gradient expansion for exchange in solids and surfaces. Phys. Rev. Lett. 100, 136406 (2008).
Liechtenstein, A. I., Anisimov, V. I. & Zaanen, J. Density-functional theory and strong interactions: orbital ordering in mott-hubbard insulators. Phys. Rev. B 52, R5467–R5470 (1995).
Jollet, F., Torrent, M. & Holzwarth, N. Generation of Projector Augmented-Wave atomic data: a 71 element validated table in the XML format. Comput. Phys. Commun. 185, 1246–1254 (2014).
Goldschmidt, V. M. Die gesetze der krystallochemie. Naturwissenschaften 14, 477–485 (1926).
Benedek, N. Tolerance factor calculator v0.2.1.
Lufaso, M. W. & Woodward, P. M. Prediction of the crystal structures of perovskites using the software program spuds. Acta. Crystallogr. B. 57, 725 (2001).
Orobengoa, D., Capillas, C., Aroyo, M. I. & Perez-Mato, J. M. AMPLIMODES: symmetry-mode analysis on the Bilbao Crystallographic Server. J. Appl. Crystallogr. 42, 820–833 (2009).
Perez-Mato, J. M., Orobengoa, D. & Aroyo, M. I. Mode crystallography of distorted structures. Acta Crystallogr. A 66, 558–590 (2010).
Acknowledgements
We thank Y. Zhang, E. Bousquet, F. Ricci, M. Verstraete, J.-Y. Raty, S. Catalano, J.-M. Triscone, A. Georges and J. Varignon for fruitful discussions. This work was supported by FRS-FNRS project HiT4FiT and ARC project AIMED. J.I. acknowledges support form FNR Luxembourg Grant P12/4853155 “COFERMAT”. Calculations have been performed on the Céci facilities funded by F.R.S-FNRS (Grant No 2.5020.1) and Tier-1 supercomputer of the Fédération Wallonie-Bruxelles funded by the Walloon Region (Grant No 1117545). They also took advantage of HPC resources from the French Research and Technology Computing Center (CCRT) and from the PRACE project Megapasta.
Author information
Authors and Affiliations
Contributions
P.G. conceived the study with A.M. and J.I. and directed the work. A.M. and J.B. did the first-principles simulations. A.M. and P.G. did the Landau modeling. P.G. wrote the manuscript with A.M. and J.B. All authors discussed the results at different stages of the study and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mercy, A., Bieder, J., Íñiguez, J. et al. Structurally triggered metal-insulator transition in rare-earth nickelates. Nat Commun 8, 1677 (2017). https://doi.org/10.1038/s41467-017-01811-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-017-01811-x
This article is cited by
-
Dataset of theoretical multinary perovskite oxides
Scientific Data (2023)
-
Origin of superconductivity in hole doped SrBiO3 bismuth oxide perovskite from parameter-free first-principles simulations
npj Computational Materials (2023)
-
Quantifying the role of the lattice in metal–insulator phase transitions
Communications Physics (2022)
-
Persistent half-metallic ferromagnetism in a (111)-oriented manganite superlattice
npj Computational Materials (2022)
-
Hole doping in a negative charge transfer insulator
Communications Physics (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.