Abstract
Hybrids between divergent populations commonly show hybrid sterility; this reproductive barrier hinders hybrid breeding of the japonica and indica rice (Oryza sativa L.) subspecies. Here we show that structural changes and copy number variation at the Sc locus confer japonica–indica hybrid male sterility. The japonica allele, Sc-j, contains a pollen-essential gene encoding a DUF1618-domain protein; the indica allele, Sc-i, contains two or three tandem-duplicated ~ 28-kb segments, each carrying an Sc-j-homolog with a distinct promoter. In Sc-j/Sc-i hybrids, the high-expression of Sc-i in sporophytic cells causes suppression of Sc-j expression in pollen and selective abortion of Sc-j-pollen, leading to transmission ratio distortion. Knocking out one or two of the three Sc-i copies by CRISPR/Cas9 rescues Sc-j expression and male fertility. Our results reveal the gene dosage-dependent allelic suppression as a mechanism of hybrid incompatibility, and provide an effective approach to overcome the reproductive barrier for hybrid breeding.
Similar content being viewed by others
Introduction
Hybrid sterility (HS, including male and female sterilities) reflects the genetic incompatibility between evolutionarily divergent populations in the same or different species. This major form of postzygotic reproductive isolation restricts gene flow and maintains the separation of species (subspecies) during speciation1,2,3. The classical Bateson–Dobzhansky–Muller (BDM) model proposes that the hybrid incompatibility results from deleterious genetic interactions between divergent alleles4,5,6. Asian cultivated rice (Oryza sativa L.) was domesticated from the wild rice (Oryza rufipogon Griff.) and differentiated into two subspecies, japonica and indica 7. Rice HS, as first reported in 1928, occurs extensively in the inter-subspecific (japonica–indica) hybrids, and is one of the major factors determining the genetic differentiation of these two subspecies8.
The development and application of hybrid rice technology has contributed greatly to increase rice yield9,10,11. In the past decades, hybrid rice, which boosted grain yield by about 20% compared to inbred varieties, has been planted ca. 17 million hectares annually in China alone (accounting for about 60% of its total rice planting area)10. However, current hybrid rice varieties were bred mainly from crosses between lines from the same subspecies, mostly indica lines, and hybrid yields have reached a plateau due to the narrow genetic diversity of the parental lines, which results in lower degrees of hybrid vigor (heterosis). By contrast, the hybrids between japonica and indica cultivars have much stronger heterosis, thus holding great promise for further increasing yield potential12.
In japonica-indica hybrids, the accumulated genetic effects of multiple HS loci cause very low pollen and spikelet fertilities, which prevents seed formation. Therefore, HS is a major barrier preventing the utilization of the stronger japonica–indica heterosis for achieving higher grain yield, and overcoming HS remains a major challenge in high-yield rice breeding. Genetic studies have identified approximately 50 loci involved in HS in rice3, but the allelic relationships of some loci remain unclear. Two general models have been proposed to explain genetic HS in rice: the one-locus sporo–gametophytic interaction model and the duplicate gametic–lethal model13,14,15. The former model proposes that genetic interaction between divergent alleles of a single locus in the sporophytic cells causes abortion of gametes carrying a specific allele. In the later model, epistatic interactions between divergent alleles of two loci cause HS.
So far, the HS-causal genes have been cloned from five loci (Sa, S5, HSA1, S7, and the locus pair DPL1/DPL2) in japonica–indica hybrids16,17,18,19,20,21, from a locus pair (S27/S28) in Asian rice–wild rice (Oryza glumaepatula) hybrids22, and from a locus (S1) in hybrids of Asian rice and African rice23. Sa confers hybrid male sterility (HMS), and was identified as the first HS complex locus consisting of two adjacent genes SaF and SaM, which encode an F-box protein and a small ubiquitin-like modifier E3 ligase, respectively. In Sa-heterozygotes, detrimental interactions among proteins from three alleles (indica SaF and SaM, and japonica SaM) cause selective abortion of pollen grains carrying the japonica SaM allele, thus establishing the two-gene/three component interaction model at the molecular level16. S5 also is a complex locus containing three tightly linked genes, ORF3, ORF4, and ORF5. During female sporogenesis, co-action of the killer/partner genes ORF4 and ORF5 of the indica allele causes endoplasmic reticulum stress and abortion of embryo sacs that contain the non-functional protector gene ORF3 in the japonica allele; the functional ORF3 of the indica allele rescues these effects18. Similarly, the HSA1 locus contains two closely linked genes, HSA1a and HSA1b; epistatic interactions among the japonica and indica alleles of the genes in japonica–indica hybrids cause female gamete abortion19. At the S7 locus, a gene encoding a tetratricopeptide repeat domain-containing protein confers the Aus–japonica–indica hybrid female sterility20. The locus pairs DPL1/DPL2 and S27/S28 were generated from duplication of the pollen-essential genes on different chromosomes, followed by reciprocal loss of one of the duplicated genes in the divergent species; chromosomal recombination in male meiosis of the hybrids carrying DPL1/DPL2 or S27/S28 produces ~ 25% sterile pollen grains that lack both of the functional loci21,22. At the S1 locus, a gene OgTPR1 encoding a protein containing two trypsin-like peptidase domains and a ribosome biogenesis regulatory domain is required for the interspecific hybrid male and female sterilities23. Even though much has been learned about the genetic basis of HS in rice, we still lack a complete picture of its complexity and efficient methods to overcome HS for hybrid rice breeding.
Natural hybrid-compatible (neutral) alleles, which do not cause HS in hybrids, have been found for some HS loci (such as S5-n at S5 and Sa-n at Sa) in rice3,14,15,16,17. However, previous efforts using neutral alleles to develop the inter-subspecific hybrids have had only limited success. In addition, breeding of hybrid wide-compatible lines with natural neutral alleles by successive backcrossing is time consuming, and often not feasible because no neutral alleles have been detected for many HS loci.
Genomic variations are the basis for genetic diversity, genome evolution, and natural selection in organisms. Genomic structural variations, including segmental insertion, deletion, inversion, translocation, duplication, and copy number variation (CNV), have significant genetic effects on various biological processes, agronomic traits, and human diseases24,25. For example, genomic CNVs increase nematode resistance in soybean26 and contribute to grain-size diversity in rice27. However, how genomic structural variations generate speciation genes that contribute to reproductive isolation and regulate allele-specific gene expression and function remains largely unclear.
Sc is a major locus conferring HMS in japonica–indica hybrids and was identified using a cross between the japonica cultivar Taichung 65 (T65, carrying a japonica allele, Sc-j) and its near-isogenic line E5 (which has the T65 nuclear background but contains an indica allele, Sc-i introgressed from the indica cultivar Peh-ku)13,28. We previously mapped Sc to a region of chromosome 3 (ref. 29). Here we report the isolation and characterization of the gene for Sc, and reveal that genomic structural changes and CNV at Sc result in allelic suppression that causes the hybrid incompatibility. Based on these findings, we also provide an effective approach that uses genome editing to break down the reproductive barrier for hybrid breeding.
Results
Identification of a DUF1618 protein gene at the Sc locus
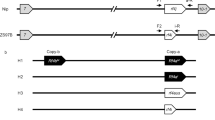
The F1 hybrid (Sc-j/Sc-i) of the T65/E5 cross is partially (semi-) sterile due to the abortion of pollen grains carrying Sc-j, resulting in a severe, male-dependent transmission ratio distortion (mTRD) of the Sc alleles (Fig. 1a). Therefore, the Sc-j and Sc-i alleles are genetically incompatible in the inter-subspecific hybrids. So far, no neutral allele at the Sc locus has been identified in rice germplasm. By fine-mapping, using the mapping population derived from the T65/E5 cross, we delimited Sc to an 8.6-kb region (Fig. 1b). In japonica cultivars, this allele contains an annotated gene Os03g0247300, here named Sc-j (Fig. 1c). Sc-j encodes a DUF1618 domain-containing protein (446 amino acids, aa), belonging to a monocot-specific gene family30.
Cloning of Sc reveals variations in genomic structure and copy number. a Pollen phenotypes of T65, E5, and their F1. Arrows indicate smaller, sterile pollen grains (scale bars, 50 μm). The self-fertilized F2 family shows a severely distorted segregation of the alleles (P < 0.001 for the chi-square test). b Fine-mapping of Sc using F2 plants of the T65/E5 cross. The markers indicate the positions (kb) in the BAC of the japonica cultivar (cv) Nipponbare (Nip). c Genomic configurations of the Sc alleles in japonica lines (from GenBank except for T65) and indica lines (determined in this study). The Sc-i allele variants have three or two tandemly duplicated segments, each containing the gene homologous to Os03g0247300. T1 (2016 bp) and T2 (9087 bp) are transposon insertions (a DDE-type and a Rim2/Hipa-type, respectively). The Sc-i paralogous genes have recombinant promoter/upstream sequences different from that of Sc-j. d Fiber-FISH images of the MH63 BAC (~ 180 kb) and the E5 chromatin (genomic) DNA (gDNA). For the BAC fiber-FISH, Probes I and II were labeled for red fluorescence, and the whole BAC DNA was labeled for green fluorescence. The labeled Probe I (or Probe II) was mixed with the labeled BAC for the hybridization. For fiber-FISH of E5 gDNA, Probes I and II were labeled for red and green fluorescence, respectively, and mixed for hybridization. Scale bars, 2 μm. Note that the BAC and chromatin DNA fibers for the same segments had different extension levels, similar to the previous observations34. FF full fertility, SS semi-sterility
Complex genomic structural variations in the Sc-i alleles
Analysis of the genome sequence at the Sc locus in the indica cultivar 93-11 (GenBank accession numbers: AAAA02008136.1, AAAA02008130.1, AAAA02008132.1, AAAA02008135.1) suggested complex structural variations at Sc, but the available sequence in this region was largely incomplete. We therefore sequenced a Sc-i-containing bacterial artificial chromosome (BAC) of the indica cultivar MH63, and PCR-amplified and sequenced the genomic and cDNA sequences of the Sc-i region in 93-11 and E5. The results showed that the complete genomic sequence of the Sc-i region in E5 contains four copies of the Os03g0247300 sequence (Fig. 1c and Supplementary Figs. 1, 2). One copy has a similar promoter sequence to Sc-j but is a pseudogene (named Sc-is) due to a 2-bp deletion and two transposon insertions (a DDE-type31 and a Rim2/Hipa-type32) in this gene (Supplementary Fig. 2). The other three copies, named Sc-ia, Sc-ib1, and Sc-ib2, each contain the complete Os03g0247300-homologous sequence, and are located in three tandemly duplicated segments (28.2 or 28.4 kb in length) (Fig. 1c and Supplementary Figs. 1, 2). MH63 and 93-11 also have Sc-is and two duplications of the segment (28.4 kb) with Sc-ib1 and Sc-ib2, but lack the Sc-ia-containing segment (Fig. 1c). Notably, the 23.9-kb upstream/promoter sequence of Sc-ia, Sc-ib1, and Sc-ib2 differ completely from that of Sc-j (Fig. 1c and Supplementary Figs. 1, 2). The coding regions of Sc-j and these paralogous Sc-i genes include nineteen single-nucleotide polymorphisms (SNPs) and three 3-bp insertion/deletions, which result in several aa substitutions, but the coding sequences of Sc-ib1 and Sc-ib2 are identical (Supplementary Figs. 3 and 4). The Sc-i allele variants probably arose from genomic rearrangement between the ancestral DUF1618 gene and the 23.9-kb genomic sequence to produce a new recombinant gene, followed by inactivation of Sc-is and CNV of the ~ 28-kb segment.
Using PCR assays, we genotyped 14 additional japonica cultivars and 21 indica cultivars, and found that all tested japonica cultivars have the Sc-j allele and all indica cultivars have the Sc-i allele with at least two duplications of the segment (Supplementary Fig. 5a–c). We further genotyped this locus in O. rufipogon and found that, of the nine accessions analyzed, five have the Sc-i-type structure (including Sc-is and at least two duplications of the ~ 28-kb segment) and four have the Sc-j-type structure (Supplementary Fig. 5d). This suggests that the divergence of Sc occurred before the domestication of O. sativa from O. rufipogon and the differentiation of the japonica and indica subspecies.
Since the PCR assay could not determine the actual copy number of the ~ 28-kb segment at the Sc-i allele variants, we further carried out DNA fiber-fluorescence in situ hybridization (fiber-FISH)33,34 to examine the Sc-i structure of several indica cultivars. We found that five cultivars (MH63, 93-11, HHZ, Dular, and 02428) contain two duplicates (not including Sc-is) of the segment, whereas three other cultivars (E5, ZS97B, and GLA4) contain three such duplicates (Fig. 1d and Supplementary Fig. 6), consistent with the sequencing results of E5, MH63, and 93-11. These results indicate that the genomic structural change and the segment CNV at Sc is a common feature in indica cultivars.
Different expression patterns of the Sc-j and Sc-i alleles
We speculated that the structural variations might alter the expression pattern and/or level of the diverged Sc alleles; therefore, we examined their expression by semi-quantitative and quantitative reverse transcription PCR (qRT-PCR). Sc-j in T65 was expressed specifically in anthers, peaking but having a low level at the early-bicellular pollen stage (Fig. 2a and Supplementary Fig. 7a), which is consistent with the microarray-based transcriptome data from the japonica cultivar Nipponbare (Nip, with Sc-j) (Supplementary Fig. 7b). The repeated Sc-i paralogs in E5, however, were expressed broadly in vegetative organs and anthers of different stages, and the total expression level of all paralogs was much higher than that of Sc-j (Fig. 2a and Supplementary Fig. 7a). In the early-bicellular stage anthers, the Sc-j expression level (0.020, expressed as the ratio to OsActin 1 expression) in T65 was only ~ 13% that of Sc-i (0.151) in E5 (Fig. 2a).
Expression of the Sc alleles. a Expression of Sc-i and Sc-j in the developing anthers of E5, T65, and their F1 by qRT-PCR using a common primer pair (P3/P8, Supplementary Fig. 1) for all the alleles/Sc-i paralogs (except for Sc-is). The values (means of three biological replicates with s.e.m.) were normalized to OsActin 1 transcript levels. b qRT-PCR analysis of Sc-j (using P3/P8) in separated pollen and anther wall cells (at early-bicellular stage) of T65. Data are shown as means ± s.e.m. (n = 3). c Different Sc protein abundances in anthers (at stages of uninucleate late-microspore to early-bicellular pollen) of the parents and the F1, detected with anti-Sc antibodies
To verify if Sc-j is expressed in pollen, we carried out mRNA in situ hybridization to Sc-j in anthers of T65, but failed to obtain a clear signal, probably due to its very low-level expression. Alternatively, we separated pollen grains from the anther wall (epidermis) cells (at the early-bicellular stage) of T65, The results showed that Sc-j was expressed mainly in pollen and showed much less expression in the anther wall (Fig. 2b), consistent with its proposed function in pollen development (see below). The differential expression of the Sc-j and Sc-i alleles was also reflected by their different protein abundances (Fig. 2c and Supplementary Fig. 8). We speculated that the high-level and broad expression patterns of Sc-i could result from the repeated gene copies and the distinct promoter sequence.
The expression of Sc-j is largely suppressed in the hybrid
To explore the possible causal relationship between the differential expression of the Sc alleles and HMS in the hybrid, we examined Sc-j expression levels in the hybrid and T65 using Sc-j-specific primers. In the early-bicellular stage anthers, the expression level of Sc-j per copy in the hybrid was about 1/5 of that in T65 (Fig. 3a). Similar results were obtained in isolated early-bicellular stage pollen grains (Fig. 3a), suggesting that Sc-j expression is largely repressed in the hybrid pollen grains. We further analyzed the relative transcript abundances of Sc-j and the Sc-i paralogs in the hybrid by genotyping cDNA clones based on the SNPs in Sc-j and the Sc-i paralogs (Supplementary Fig. 3). In anthers of the meiocyte (microspore mother cell and meiosis I) and uninucleate microspore stages, no or very few Sc-j-type cDNA clones were detected; in the early-bicellular stage anthers, Sc-j type cDNAs accounted for only 4.7% of the total clones (Fig. 3b and Supplementary Table 1), much lower than the expression level of Sc-j in T65 (13%) compared to E5 (Fig. 2a). Together, these results suggest that normal pollen development requires a threshold level of Sc-j expression in the early-bicellular pollen, and that the suppression of expression Sc-j in the hybrid, a phenomenon that we term “allelic suppression”, might be the cause of Sc-j-pollen abortion.
Functional analysis of Sc-j. a qRT-PCR analysis of relative Sc-j expression levels (using the Sc-j-specific primers P1/P2) in the anthers and isolated pollen (at early-bicellular stage) of the F1 (T65/E5) and T65, calculated as the ratios of the transcript per copy Sc-j to the transcript of OsActin 1 (two copies), because there is only one Sc-j copy in the F1. The data are shown as means ± s.e.m. (n = 3), and * and ** indicate significance at P < 0.05 and P < 0.01, respectively, by two-tailed Student’s t test (the same for Fig. 3c). b Percentages of cDNA clones from Sc-j, Sc-ia, and Sc-ib (Sc-ib1/ib2) of the F1 anthers of two stages indicated. c Expression of Sc-j in the early-bicellular stage anthers of two T1 (T65 background) plants carrying the hemizygous antisense-transgene of Sc-j. The detected transcript levels in Anti-1 and Anti-2 could be mainly from the pollen grains (~ 50%) without the antisense-transgene. d Semi-sterility of three T1 plants carrying the hemizygous antisense-transgene. WT (T-), a T1 segregant without the antisense-transgene. Scale bars, 50 μm
Previous cytological observation of sectioning pollen of the T65/E5 hybrid showed that at the bicellular stage, the cell wall (callose) of the mitosis I-generated generative cell (nucleus) within Sc-j-pollen failed to degenerate at the proper time and the generative nucleus could not move with the vegetative nucleus as occurs in normal bicellular pollen. This leads to the generative nucleus being unable to complete mitosis II35. Therefore, this allelic suppression of Sc-j in early-bicellular pollen may be related to the cytological mechanism of impaired Sc-j-pollen development.
Sc is a gametophytic factor for pollen development
To further test if Sc is essential for pollen development, we introduced an Sc-antisense construct into T65 plants. As expected, the hemizygous transgenic plants with reduced Sc-j expression in their anthers showed partial male-sterility, similar to the Sc-mediated HMS (Fig. 3c, d). Furthermore, the transgene segregation ratio in five T1 families (from self-fertilization) fitted the 1:1 ratio expected for a hemizygous gametophytic sterility gene (Supplementary Table 2). Therefore, we concluded that this DUF1618 gene has a primary function as a gametophytic factor, and normal pollen development requires a threshold level of Sc expression.
To test whether the abnormal development of Sc-j-pollen involves changes in the expression of genes known to affect male fertility36,37, we analyzed a microarray-based transcriptome data set from anthers of T65 and the T65/E5 F1. Of 14 genes with known functions in microspore/pollen development, four (OsADF, OsGSL5, OsUgp2, and OsCAP1) showed differential microarray hybridization signals (Supplementary Fig. 9a). However, further qRT-PCR analysis did not find significant difference in expression of these four genes in developing anthers between T65 and the F1 (Supplementary Fig. 9b).
CNV of Sc-i is associated with the degree of HMS
To investigate the relationship between the Sc-i CNV and HMS, we crossed the japonica cultivar Nip with indica cultivars that have three copies (E5 and ZS97B) or two copies (93-11, HHZ, and 02428) of the ~ 28-kb segments (Supplementary Fig. 6). The two F2 families with three Sc-i copies had more severe mTRD (with 0.4% and 3.3% of Sc-j homozygotes) than those of the three F2 families with two Sc-i copies (with 4.7–9.7% of Sc-j homozygotes) (Table 1). Particularly, the recombinant inbred lines (RILs) derived from crossing Nip with the indica cultivar GLA4 (carrying three Sc-i copies) produced only 0.9% Sc-j homozygotes, whereas the frequency was 21.1% in the RILs derived from crossing Nip with 93-11. The more severe mTRD reflects greater abortion of Sc-j-pollen in the Sc-heterozygotes. These results suggest that the severity of HMS in the hybrids is associated with the CNV at Sc-i. Notably, all the indica cultivars analyzed have at least two Sc-i copies (Supplementary Figs 5 and 6) and these Sc-i allele types produced different degrees of genetic incompatibility to japonica cultivars (Table 1).
Reduction of the Sc-i dosage rescues the HMS
Based on the relationship of the Sc-i CNV with the HMS, we tested whether decreasing the number of functional Sc-i copies by genome editing could generate artificial neutral alleles. Using our CRISPR/Cas9 plant genome editing system38, we made two binary constructs, one to delete the ~ 28-kb segment(s) and another to specifically edit a unique target site in Sc-ib1 and Sc-ib2 (but not in Sc-ia) based on a SNP that forms the protospacer adjacent motif (PAM) required for CRISPR/Cas9 genome editing (Fig. 4a). By transformation of E5 with these constructs, we identified a plant (E5-d1) with one of the segmental duplications deleted (Fig. 4a and Supplementary Fig. 6), and two plants (E5-ed1 and E5-ed2) with nucleotide mutations in Sc-ib1 and Sc-ib2, but with their Sc-ia intact due to the lack of a PAM (Fig. 4a). We crossed these edited plants with T65 to obtain F1 plants (F1-d1, F1-ed1, and F1-ed2) and confirmed the edited sites by sequencing (Supplementary Fig. 10). The pollen fertility of these hybrids was greatly improved (Fig. 4b) and the F2 families showed less mTRD; two F2 families from F1-ed1 and F1-ed2 had segregation ratios fitting the 1:2:1 ratio (Table 2), thus confirming the restoration of Sc-j-pollen fertility. Therefore, reduction of the gene dosage by the targeted mutation of two of the three Sc-i paralogs can convert the hybrid-incompatible Sc-i allele into completely compatible alleles while maintaining the basic function of Sc in gametogenesis. We name these edited Sc-i alleles Sc-n, as they produce no significant HMS.
Genomic editing of the Sc-i paralogs rescues Sc-j expression and male fertility in the hybrids. a Targeted editing of the sites (arrowed) in E5 produced a plant (E5-d1) with deletion of one of the ~ 28-kb segments (Supplementary Fig. 6), and editing another site (underlined) specific to Sc-ib1 and Sc-ib2 of E5 identified two plants (E5-ed1 and E5-ed2) with mutated Sc-ib1 and Sc-ib2 and intact Sc-ia (Supplementary Fig. 10). The SNP (blue) in Sc-ib1 and Sc-ib2 formed the PAM (NGG) required for genome editing. Primers P9a-P9e combined with the Sc-i-specific P10 were used for qRT-PCR of the intact and mutated Sc-i transcripts show in (c). b Improvement of pollen fertility (sterile pollen grains arrowed) in the edited F1 plants from crossing E5-d1, E5-ed1, and E5-ed2 with T65. Scale bars, 50 μm. c Expression levels (in the meiocyte stage anthers) of the Sc-i paralogs in the F1 (T65/E5), and the intact Sc-ia and mutated Sc-ib1/Sc-ib2 in the edited F1 plants. Data are shown as means ± s.e.m. (n = 3). d Expression levels of per copy Sc-j (qRT-PCR using P1/P2) in the early-bicellular stage anthers of the intact and edited F1 plants and T65. The data are shown as means ± s.e.m. (n = 3), and * and ** indicate significance at P < 0.05 and P < 0.01, respectively, by two-tailed Student’s t test
To verify the causal relationship between expression of Sc-i and Sc-j and male fertility in the edited hybrids, we analyzed the mRNA levels of Sc-ia, the mutated Sc-ib1 and Sc-ib2, and Sc-j in anthers of the edited F1 plants using the intact and edited allele-specific primers (Fig. 4a). The mutated mRNA levels were greatly reduced relative to the Sc-ia transcript (Fig. 4c), probably due to the common nonsense-mediated mRNA decay mechanism for abnormal mRNAs39. In contrast, the Sc-j transcript levels (at the early-bicellular stage) of these edited F1 plants significantly increased (Fig. 4d). Thus, the Sc-j expression level and the pollen fertility in the Sc-heterozygotes are negatively associated with the Sc-i gene dosage and its expression level.
Discussion
In this study, we isolated and characterized the gene at the HMS locus Sc. Based on the results, we propose a molecular genetic model for the Sc-HMS system. In the wild rice, the pollen-essential Sc gene underwent structural changes and CNV, and the divergent Sc-j-type and Sc-i-type alleles were inherited into the japonica and indica subspecies, respectively, during domestication. In the japonica–indica hybrids, the Sc-i paralogs are highly expressed in the sporophytic cells and the product may interact, directly or indirectly, with the promoter region of Sc-j by a yet-unknown mechanism to mediate the suppression of Sc-j expression. This interaction may involve transcriptional-repressive epigenetic modification(s) of the Sc-j promoter region, and the modification state (epiallele) may be retained, through meiosis, in the pollen grains, thus leading to the suppression of Sc-j transcription in pollen and the allele-specific male-gamete elimination (Fig. 5). This model of gene dosage-dependent HMS is consistent with the partial, sometimes almost complete, restoration of hybrid male fertility produced by reducing the number of copies in the Sc-i locus in the natural allele variants and the genome-edited alleles of Sc-i.
A proposed model for the molecular genetic mechanism of Sc-mediated HMS. In a japonica–indica hybrid, the Sc-i paralogs are highly expressed in sporophytic cells and the product may interact with the Sc-j promoter region. This might cause transcriptional-repressive epigenetic modification(s) (red asterisk) in the Sc-j promoter region. This putative modification state (Sc-j*) could be retained, through meiosis, in the male gametophytes, thus causing allelic suppression of Sc-j expression and the consequent Sc-j-specific pollen abortion (HMS) and mTRD. Only two of the four pollen grains from a meiocyte, and one Sc-j copy and one set of Sc-i copies in the bicellular and tricellular pollen grains, are shown
The change of the promoter sequence of Sc-i via structural variations not only alters its gene expression pattern and levels, producing the putative allelic interaction in the sporophytic cells, but might also allow the Sc-i paralogs to escape repression by their own products. Thus, the Sc-i allele variants have evolved into selfish speciation genes in this HMS system, and can cause specific abortion of pollen with Sc-j but not pollen carrying the Sc-i variants in the hybrids. Such a scenario exemplifies the molecular genetic basis of the classical one-locus sporo-gametophytic interaction model for plant HS13,14,15. The BDM-type genetic incompatibility results from the divergence and interaction of genetic loci during evolution. It would be interesting to know if other intermediate types of Sc-i-like and/or Sc-j-like allele variants that are likely hybrid-neutral or hybrid-incompatible exist in wild and cultivated rice populations, for example, the primarily diverged Sc-i-like allele having a novel, recombinant promoter but without the segmental duplication. Therefore, the Sc-HMS system may be an ideal model for study of BDM-type or a novel type of postzygotic hybrid incompatibility caused by a single locus. Further investigation will be required to analyze the allelic interaction and the molecular mechanism of Sc, as well as its evolution.
The development of anthers and pollen involves a large number of genes, and many of them are highly conserved between monocot and dicot plant species36,37. However, this study revealed that the Sc gene is a member of the monocot-specific DUF1618 gene family (121 members in japonica rice). To date, only two genes of this family in rice, HSA1a 19 and Sc, have been functionally studied. These studies showed that HSA1a and Sc have important roles in reproductive development and participate in hybrid female and male sterilities, respectively. These findings demonstrate that rice has evolved genetic networks distinct from those of dicots for male and female gametogenesis.
The findings from studies of a number of cloned HS loci in plants suggest that hybrid incompatibility involves diverse molecular genetic mechanisms40; the only common feature among some hybrid incompatibility systems is that they are caused by gene duplication/reciprocal loss-of-function mutations, as shown in the DPL1/DPL2 and S27/28 locus pairs21,22. In this study we revealed a new type of combinatorial genomic variation involving structural changes and CNV, and demonstrate that this type has an important genetic effect on hybrid incompatibility. Our results show that the Sc-j and Sc-i alleles are associated with japonica and indica cultivars, suggesting that this hybrid incompatibility might have contributed to the differentiation of the subspecies, although extensive analysis will be required to resolve this issue. The allelic suppression described here suggests a regulatory mode for differential allelic expression, which is characterized by allele-specific (Sc-j) repression and is driven by the opposite allele (Sc-i) of the same gene. This phenomenon has features distinct from the previously described paramutations41 and monoallelic expression forms (parent-of-origin imprinting, random monoallelic expression of autosomal genes, and X-inactivation genes)42,43,44.
In this study, we used CRISPR/Cas9 genome-editing technology to create neutral alleles of Sc by precisely reducing the gene dosage of Sc-i but maintaining its basic function in pollen development. By contrast, precise down-regulation of target genes remains difficult to achieve with other methods, such as antisense and RNA interference. The genome editing-based approach can rapidly generate neutral alleles at Sc and other HS loci that require loss-of-function mutations to generate neutral alleles, such as Sa, S5, and S1 16,17,23,45. This may enable breeding of wide-compatible lines to break down the reproductive barrier and overcome HS in the inter-subspecific and inter-specific hybrid breeding in crops, and thus may have significant implications for agriculture.
Methods
Rice materials
Rice materials for the gene cloning and functional analysis included the japonica line T65 and its near-isogenic line E513,28 and their hybrid progeny prepared in this study. The other indica cultivars MH63, ZS97B, HHZ, and GLA4 are Minghui 63, Zhengshan 97B, Huanghuazhan, and Guangluai 4, respectively. The line 02428 with a majority japonica background was derived from a japonica–indica cross46. The wild and cultivated rice species used for the haplotype survey and crosses were maintained in our laboratory. The rice plants were planted in experimental fields or in a phytotron (for some seedlings) at the South China Agricultural University in Guangzhou.
Map-based cloning
F2 plants of the T65/E5 cross were used for mapping. The pollen phenotypes (male fertile or male sterile) of rice were observed by placing pollen in a 1% I2-KI (potassium iodide) solution and observing the pollen under a microscope (Leica DMLB, Zeiss Axio Observer D1). Plants homozygous for Sc produced fully fertile pollen (>95% pollen fertile); plants heterozygous for Sc produced semi-fertile pollen (ca. 50% of pollen grains sterile). A number of insertion/deletion markers (Supplementary Table 3) were used for the fine-mapping. The key recombinant plants (Fig. 1b) were further confirmed by analyzing the phenotypic and genotypic segregation in their F3 progenies. Genetic segregation of the genotypes in various populations was analyzed by χ 2 test.
Sequence analysis
The BAC clone (OSIMNBa0008K19) was from an MH63 genomic library47. With the incomplete genomic sequences of the indica 93-11 at the Sc region (GenBank accession numbers: AAAA02008136.1, AAAA02008130.1, AAAA02008132.1, AAAA02008135.1) as references, a number of primer pairs were prepared and long-length PCR with KOD FX DNA polymerase (TOYOBO, Japan) was used to amplify the Sc-i region sequences from the BAC and from gDNAs of E5 and 93-11 in multiple fragments. The PCR products were sub-cloned into a plasmid vector and multiple clones for each fragment were sequenced. The sequences were assembled using SeqMan of the Lasergene package, and analyzed by BLAST (http://www.ncbi.nlm.nih.gov/BLAST/). The DDE-type and Rim2/Hipa-type transposon insertions in Sc-is were analyzed by BLAST (http://www.ncbi.nlm.nih.gov/BLAST/).
DNA fiber-FISH
For BAC fiber-FISH, the BAC plasmid was isolated from 200 ml LB culture using the alkaline lysis method. The BAC DNA was diluted to about 10 ng/μl in AF solution (100% glycerin:1 M sodium chloride:double-distilled water, 180:3:17, v/v, pH 7.5) with cut pipet tips. The diluted BAC DNA (2 µL) was added to Poly-Prep slides (Sigma-Aldrich, USA), then the slides were slowly covered with coverslips (22 × 22 mm) and set vertically to stretch the BAC DNA. The slides were air-dried for 10 min, then carefully uncovered by removing the coverslips. The slides were fixed in fresh Carnoy’s solution (ethanol:glacial acetic acid, 3:1, v/v). Probe I (16.3 kb) and Probe 2 (11.9 kb) DNAs were amplified by PCR in three and two segments, respectively (primer sequences are provided in Supplementary Table 3), and purified on a 1% agarose gel with a gel-extraction kit. According to the DNA fiber-FISH protocol33, the probe sequences (Probe I and Probe II) were labeled by a nick translation reaction with digoxigenin-11-dUTP (Roche, Switzerland), and mixed with the whole BAC DNA labeled with biotin-16-dUTP (Roche, Switzerland) for two separate hybridizations. For hybridization, the probes were firstly added to the slides and covered with the cover slips. Then the slides were denatured at 80 °C in for 5 min in an oven. Then the slides were immediately transferred to another oven and incubated at 37 °C overnight. Immunodetection of the probes were performed by sequentially adding various antibodies to gain amplifying the fluorescence signals. The biotin-labeled probes were detected successively with fluorescein-avidin D (Vector, USA), biotinylated anti-avidin antibody (Vector, USA), and fluorescein-avidin D (Vector, USA). The digoxigenin-labeled probes were firstly detected by anti-digoxigenin-rhodamine (Roche, Switzerland), then by texas red® goat anti-rabbit IgG antibody (Vector, USA). The slides were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) in vectashield mounting medium (Vector, USA).
For genomic DNA fiber-FISH, nuclei were prepared from young rice seedling leaves, using the plant nuclei isolation method48. The seedling leaves were grounded to powder in liquid nitrogen, and the powder was suspended in buffer (10 mM Tris–HCl (pH 9.5), 10 mM EDTA (pH 8.0), 100 mM KCl, 0.5 M sucrose, 4 mM spermidine, 1 mM spermine, 0.1% mercaptoethanol). After filtering sequentially through 149, 74 and 37 μm nylon meshes, the nuclei were collected by centrifuging at 1500 g for 10 min. The genomic DNA fiber-FISH was performed following the protocol34. Briefly, the DNA fibers were extended by dragging with a cover slip upon the Poly-L-lysine (Sigma-Aldrich, USA) slides to get long and clean DNA fibers. The slides were hybridized using mixed Probe I labeled with digoxigenin-11-dUTP and Probe II labeled with biotin-16-UTP. The following procedures of hybridization and probes detection were carried as the same as the above description for BAC fiber-FISH. The fiber-FISH images of the BAC and genomic DNAs were captured digitally using a CCD camera (QIMAGING, USA) attached to an Olympus BX61 epifluorescence microscope. Gray-scale images were captured for each color channel and then merged. The final image optimization was performed using Adobe Photoshop CS3 (Adobe, USA).
Immunoblot analysis
A peptide antigen (MVEGNEGEESTKRMKVK) corresponding to amino acid residues 427–443 of Sc-i (430–446 of Sc-j) was synthesized and used for immunizing rabbits to produce the anti-Sc antibodies by Invitrogen (USA) as a customary service.
Detection of Sc-j and Sc-i with the antiserum was carried out as described with some modifications49. About 50 mg anthers (at stages of late-microspore to early-bicellular pollen) of T65, E5, and their hybrid F1 were grinded to fine powder in liquid nitrogen and lysed in 500 μl 2 × protein extraction buffer (100 mM Tris-HCl (pH 7.5), 300 mM NaCl, 2 mM EDTA, 10% glycerol, 0.5% TritonX-100 and protease inhibitor cocktail from Roche, PMSF from AMRESCO). After centrifugation at 12,000 g for 15 min, the supernatants were collected and the total proteins were quantified with the 2-D Quant Kit (GE Healthcare, USA). Extracted proteins were mixed with an equal volume of the 2 × loading buffer (100 mM Tris-HCl, 5 mM DTT, 4% SDS, 0.01% bromophenol blue and 30% glycerol, pH 6.8) and boiled for 5 min, then separated by 12% SDS-PAGE and transferred onto an Immobilon-PPSQP transfer membrane (polyvinylidene fluoride (PVDF) type, Millipore) using a Bio-Rad mini-transfer cell. The membranes were incubated in blocking buffer (5% milk, 0.1% Tween-20, 0.1% Triton X-100, 100 mM Tris-HCl and 150 mM NaCl, pH 7.5) for 2 h at room temperature, washed twice with 2 × TBST buffer (0.1% Tween-20, 100 mM Tris-HCl and 150 mM NaCl, pH 7.5). Then the membranes were incubated with anti-Sc polyclonal antibodies (1:2000 dilution) or the anti-Hsp90 monoclonal antibody (Beijing Protein Innovation, China; AbM51099-31-PU) diluted (1:10000) in blocking buffer for about 20 h at 4 °C. After washing three times with TBST for 5 min each, the membranes were incubated in the secondary antibodies, HRP-conjugated goat anti-rabbit IgG or HRP-conjugated goat anti-mouse IgG (TransGen Biotech, China), with 1:10000 dilution for 1 h at room temperature and washed three times with TBST for 5 min each. The membrane blots were incubated in the ECL substrate (Bio-rad, USA) for 1 min. The photos were captured using Gel Logic 6000 Pro (Kodak, USA) and processed with Adobe Photoshop CS3 (Adobe, USA).
Expression analysis
Separation of pollen grains from anther wall cells was carried out by filtering broken anthers in 2% sucrose through a nylon mesh (48 μm in diameter), followed by centrifugation at 250 g for 10 min to pellet pollen. The separated pollen grains and anther wall cells were collected for RNA preparation. Total RNA was extracted from rice tissues (anthers, isolated pollen, anther wall cells, young panicles, leaves, stems, and roots) of T65, E5, and their hybrid using TRIzol reagent (Invitrogen, USA). The RNA samples were treated with DNase I (Promega, USA). First-strand cDNA was synthesized from 2 ~ 3 μg total RNA using Superscript III reverse transcriptase (Invitrogen, USA) with oligo dT or a Sc-specific primer. The expressions of Sc and the pollen-essential marker genes were investigated by qRT-PCR analysis with OsActin 1 (Os03g0718100) for normalization by three biological replicates, and the significance was analyzed by two-tailed Student’s t test. SYBR Green QPCR mix (Bio-Rad) was used for qRT-PCR. The reactions were carried out using the Bio-rad CFX Connect Real-Time PCR System with the following profile: 95 °C for 3 min; 42 cycles of 95 °C for 10 s, 60 °C for 15 s, 72 °C for 20 s. The expression pattern of Sc-j (Os03g0247300) in Nip was from the Rice Expression Profile Database (http://ricexpro.dna.affrc.go.jp/)50.
For precise quantification of the ratio of relative Sc-j and Sc-i copy numbers and transcript levels in the F1, the gDNA and cDNA segments that included all the alleles/paralogs in different stage anthers of the F1 plants were amplified using primers P5/P7. The PCR products were cloned into a plasmid vector, and multiple clones (196–380) for each sample were genotyped. The insertion/deletion marker 3-N1/3-N2 (Supplementary Table 3) was used to differentiate Sc-j from Sc-ia/Sc-ib1/Sc-ib2, and the SNP marker HRM1F/HRM1R to differentiate Sc-ia from Sc-ib1/Sc-ib2 (Supplementary Fig. 8). SNP genotype was detected by high-resolution melting analysis (LightScanner, USA) using EvaGreen (Biotium, USA) as a fluorescent dye51.
Constructs for rice transformation
A 771-bp fragment covering the 3’ coding region of Sc-j (from +514 to +1284 bp) was amplified and inserted in reverse orientation into an overexpression vector with the maize Ubiquitin promoter to prepare an antisense expression construct. Two genome-targeting constructs for fragmental deletion (the target site: 5′-GGCACAATGAGCGGGCTGGATGG-3′, PAM underlined) and Sc-ib1/ib2 mutation (Fig. 4a) were prepared using the CRISPR/Cas9 vector system38. These target site sequences were searched against the rice genome sequence with the web-based tool CRISPR-GE/targetDesign (http://skl.scau.edu.cn/)52 to confirm its specificity in the genome. The target-adapter primers are listed in Supplementary Table 3. All constructs were introduced into Agrobacterium tumefaciens strain EHA105 by electroporation and used for rice transformation. The PCR amplicons containing the target sites were directly sequenced and cloned in a plasmid for sequencing. The superimposed chromatograms of the direct sequencing were decoded with CRISPR-GE/DSDecodeM52,53, and the PCR products also were cloned in a plasmid vector and multiple clones were sequenced.
Data availability
The nucleotide sequence data for Sc-j of T65 and the Sc-i variants of E5 and MH63 have been deposited in GenBank nucleotide database under accession numbers MF370526, KX495643 and KX495644, respectively. All the relevant data are available from the authors upon request.
References
Bomblies, K. Doomed lovers: mechanisms of isolation and incompatibility in plants. Annu. Rev. Plant. Biol. 61, 109–124 (2010).
Nosil, P. & Schluter, D. The genes underlying the process of speciation. Trends Ecol. Evol. 26, 161–167 (2011).
Ouyang, Y. & Zhang, Q. Understanding reproductive isolation based on the rice model. Annu. Rev. Plant. Biol. 64, 111–135 (2013).
Bateson, W. in Darwin and modern science (ed. Seward, A. C.) 85–101 (Cambridge Univ. Press, Cambridge, 1909).
Dobzhansky, T. Genetics and the origin of species (Columbia University Press, New York, 1937).
Muller, H. J. Isolating mechanisms, evolution and temperature. Biol. Symp. 6, 71–125 (1942).
Huang, X. et al. A map of rice genome variation reveals the origin of cultivated rice. Nature 490, 497–501 (2012).
Kato, S., Kosaka, H. & Hara, S. On the affinity of rice varieties as shown by fertility of hybrid plants. Bull. Sci. Fac. Agric. Kyushu Univ. 3, 132–147 (1928).
Yuan, L. Hybrid Rice Technology: New Developments and Future Prospects. 1–6 (International Rice Research Institute, Philippines, 1994).
Cheng, S., Zhuang, J., Fan, Y., Du, J. & Cao, L. Progress in research and development on hybrid rice: a super-domesticate in China. Ann. Bot. 100, 959–966 (2007).
Yuan, L. Development of hybrid rice to ensure food security. Rice Sci. 21, 1–2 (2014).
Qian, Q., Guo, L., Smith, S. & Li, J. Breeding high-yield superior-quality hybrid super-rice by rational design. Natl Sci. Rev. 3, 283–294 (2016).
Oka, H. Analysis of genes controlling F1 sterility in rice by the use of isogenic lines. Genetics 77, 521–534 (1974).
Ikehashi, H. & Araki, H. Genetics of F1 sterility in remote crosses of rice. Rice Genet. 119–130 (1986).
Sano, Y. The genic nature of gamete eliminator in rice. Genetics. 125, 183–191 (1990).
Long, Y. et al. Hybrid male sterility in rice controlled by interaction between divergent alleles of two adjacent genes. Proc Natl Acad. Sci. USA 105, 18871–18876 (2008).
Chen, J. et al. A triallelic system of S5 is a major regulator of the reproductive barrier and compatibility of japonica-indica hybrids in rice. Proc. Natl Acad. Sci. USA 105, 11436–11441 (2008).
Yang, J. et al. A Killer-Protector system regulates both hybrid sterility and segregation distortion in rice. Science 337, 1336–1340 (2012).
Kubo, T., Takashi, T., Ashikari, M., Yoshimura, A. & Kurata, N. Two tightly linked genes at the hsa1 locus cause both F1 and F2 hybrid sterility in rice. Mol. Plant 9, 221–232 (2016).
Yu, Y. et al. Hybrid sterility in rice (Oryza sativa L.) involves the tetratricopeptide repeat domain containing protein. Genetics 203, 1439–1451 (2016).
Mizuta, Y., Harushima, Y. & Kurata, N. Rice pollen hybrid incompatibility caused by reciprocal gene loss of duplicated genes. Proc Natl Acad Sci USA 107, 20417–20422 (2010).
Yamagata, Y. et al. Mitochondrial gene in the nuclear genome induces reproductive barrier in rice. Proc Natl Acad Sci USA 107, 1494–1499 (2010).
Xie, Y. et al. Interspecific hybrid sterility in rice is mediated by OgTPR1 at the S1 locus encoding a peptidase-like protein. Mol. Plant 10, 1137–1140 (2017).
Weischenfeldt, J., Symmons, O., Spitz, F. & Korbel, J. O. Phenotypic impact of genomic structural variation: insights from and for human disease. Nat. Rev. Genet. 14, 125–138 (2013).
Alkan, C., Coe, B. P. & Eichle, E. E. Genome structural variation discovery and genotyping. Nat. Rev. Genet. 12, 363–375 (2011).
Cook, D. E. et al. Copy number variation of multiple genes at Rhg1 mediates nematode resistance in soybean. Science 338, 1206–1209 (2012).
Wang, Y. et al. Copy number variation at the GL7 locus contributes to grain size diversity in rice. Nat. Genet. 47, 944–948 (2015).
Zhang, G., Lu, Y., Zhang, H., Yang, J. & Liu, G. Genetic studies on the hybrid sterility in cultivated rice (Oryza sativa L.) IV. Genotypes for F1 pollen sterility. Acta Genet. Sinica 21, 34–41 (1994).
Yang, C., Chen, Z., Zhuang, C., Mei, M. & Liu, Y.-G. Genetic and physical fine-mapping of the Sc locus conferring indica-japonica hybrid sterility in rice (Oryza sativa L.). Chinese Sci. Bull. 49, 1718–1721 (2004).
Wang, L., Shen, R., Chen, L. & Liu, Y.-G. Characterization of a novel DUF1618 gene family in rice. J. Integr. Plant Biol. 56, 151–158 (2014).
Marchler-Bauer, A. et al. CDD: NCBI’s conserved domain database. Nucleic Acids Res. 43, D222–D226 (2015).
Wang, G. D. et al. Genomic characterization of Rim2/Hipa elements reveals a CACTA-like transposon superfamily with unique features in the rice genome. Mol. Genet. Genomics 270, 234–242 (2003).
Jackson, S. A. et al. Application of fiber-FISH in physical mapping of Arabidopsis thaliana. Genome 41, 566–572 (1998).
Cheng, Z. et al. Functional rice centromeres are marked by a satellite repeat and a centromere-specific retrotransposon. Plant Cell 14, 1691–1704 (2002).
Zhang, Z., Lu, Y., Liu, X., Feng, J. & Zhang, G. Cytological mechanism of pollen abortion resulting from allelic interaction of F1 pollen sterility locus in rice (Oryza sativa L.). Genetica 127, 295–302 (2006).
Guo, J. & Liu, Y.-G. Molecular control of male reproductive development and pollen fertility in rice. J. Integr. Plant Biol. 54, 967–978 (2012).
Shi, J., Cui, M., Yang, L., Kim, Y.-J. & Zhang, D. Genetic and Biochemical Mechanisms of Pollen. Trends Plant. Sci. 20, 741–753 (2015).
Ma, X. et al. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol. Plant 8, 1274–1284 (2015).
Maquat, L. E. Nonsense-mediated mRNA decay: splicing, translation and mRNP dynamics. Nat. Rev. Mol. Cell Biol. 5, 89–99 (2004).
Rieseberg, L. H. & Blackman, B. K. Speciation genes in plants. Ann. Bot 106, 439–455 (2010).
Chandler, V. & Stam, M. Chromatin conversations: Mechanisms and implications of paramutation. Nat. Rev. Genet. 5, 532–544 (2004).
Lawson, H. A., Cheverud, J. M. & Wolf, J. B. Genomic imprinting and parent-of-origin effects on complex traits. Nat. Rev. Genet. 14, 609–617 (2013).
Reinius, B. & Sandberg, R. Random monoallelic expression of autosomal genes: stochastic transcription and allele-level regulation. Nat. Rev. Genet. 16, 653–664 (2015).
Chess, A. Random and non-random monoallelic expression. Neuropsychopharmacology 38, 55–61 (2013).
Xie, Y. et al. Suppression or knockout of SaF/SaM overcomes the Sa-mediated hybrid male sterility in rice. J. Integr. Plant Biol. 59, 669–679 (2017).
Xie, W. et al. Breeding signatures of rice improvement revealed by a genomic variation map from a large germplasm collection. Proc. Natl Acad. Sci. USA 112, E5411–E5419 (2015).
Wang, X. et al. Global genomic diversity of Oryza sativa varieties revealed by comparative physical mapping. Genetics 196, 937–949 (2014).
Liu, Y.-G. & Whittier, R. F. Rapid preparation of megabase plant DNA from nuclei in agarose plugs and microbeads. Nucleic Acids Res. 22, 2168–2169 (1994).
Luo, D. et al. A detrimental mitochondrial-nuclear interaction causes cytoplasmic male sterility in rice. Nat. Genet. 45, 573–577 (2013).
Sato, Y. et al. RiceXPro version 3.0: Expanding the informatics resource for rice transcriptome. Nucleic Acids Res. 41, D1206–D1213 (2013).
Li, Y. D. et al. A cost-effective high-resolution melting approach using the EvaGreen dye for DNA polymorphism detection and genotyping in plants. J. Integr. Plant Biol. 52, 1036–1042 (2010).
Xie, X. et al. CRISPR-GE: A convenient software toolkit for CRISPR-based genome editing. Mol. Plant. 10, 1246–1249 (2017).
Liu, W. et al. DSDecode: a web-based tool for decoding of sequencing chromatograms for genotyping of targeted mutations. Mol. Plant 8, 1431–1433 (2015).
Acknowledgements
We thank G. Zhang, C. Zhuang and X.D. Liu (South China Agricultural University, SCAU) for providing T65, E5, and some other rice materials. We also thank H. Wang (SCAU), D. Charlesworth (University of Edinburgh), K. Tsunewaki (Kyoto University), H. Ma (Fudan University), Q. Zhang and Y. Ouyang (Huazhong Agricultural University) for comments on the manuscript. This work was supported by grants from the Ministry of Science and Technology of China (2013CBA01401, 2013CB126904 and 2012AA10A303), the National Nature Science Foundation of China (31401449), and the Nature Science Foundation of Guangdong Province, China (S2012040007477).
Author information
Authors and Affiliations
Contributions
R.S. performed most of the experiments; L.W., X.L., J.W., W.J., X.Z., X.X., Q.Z., H.T., Q.L., and L.C. performed some of the experiments. R.S. and Y.-G.L. analyzed tne data. Y.-G.L. conceived and supervised the project, and Y.-G.L. and R.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shen, R., Wang, L., Liu, X. et al. Genomic structural variation-mediated allelic suppression causes hybrid male sterility in rice. Nat Commun 8, 1310 (2017). https://doi.org/10.1038/s41467-017-01400-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-017-01400-y
This article is cited by
-
Cleave and Rescue gamete killers create conditions for gene drive in plants
Nature Plants (2024)
-
Overriding Mendelian inheritance in Arabidopsis with a CRISPR toxin–antidote gene drive that impairs pollen germination
Nature Plants (2024)
-
Haplotype-resolved gapless genome and chromosome segment substitution lines facilitate gene identification in wild rice
Nature Communications (2024)
-
Rapid generation of fragrant thermo-sensitive genic male sterile rice with enhanced disease resistance via CRISPR/Cas9
Planta (2024)
-
Domain of unknown function (DUF) proteins in plants: function and perspective
Protoplasma (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.