Abstract
Structural determination of inorganic clusters relies heavily on mass spectrometry because of, in most cases, their poor responsivities toward nuclear magnetic resonance, ultraviolet/visible, and infrared analyses. Nevertheless, mass spectrometry analysis of oligosulfurs (S n ), which are unique clusters with copious allotropic forms, usually displays their fragment peaks. Here we report that a polyaromatic capsule acts as a supramolecular matrix for the mass determination of the neutral sulfur clusters. Upon encapsulation, molecular ion peaks derived from the host–guest complexes including cyclic S6 and S8 clusters are exclusively detected by common electrospray ionization time-of-flight mass spectrometry analysis. Furthermore, mass spectrometry analysis of a cyclic S12 cluster, which is in situ prepared from two S6 clusters within the matrix upon light irradiation, is achieved by the same way. The present matrix can remarkably stabilize the otherwise labile S6 and S12 clusters in the polyaromatic shell not only under mass spectrometry conditions but also in an ambient solution state.
Similar content being viewed by others
Introduction
Mass spectrometry (MS) is of particular importance for the structural characterization of inorganic clusters, because more precise information is not easily obtained by other analytical techniques such as nuclear magnetic resonance (NMR), ultraviolet-visible (UV/Vis), and infrared (IR) spectroscopies1, 2. However, most of the neutral clusters fully or partially decompose within MS3, which prevents us from determining the molecular weight of the target structures. Sulfur clusters continue to attract attention in the fields of physical and synthetic chemistry due to their unique structures and reactivities4,5,6,7. More than 30 allotropes of sulfur (e.g., S6-15) are known to date, yet mass determination of the uncharged structures has been virtually impossible by previous MS methods. Owing to the instability of oligosulfur ions generated under usual electron ionization conditions, immediate and substantial fragmentation reactions are typically observed (Fig. 1a, left). Therefore, no reliable method to obtain the structural information of such metastable clusters in solution has been reported. We anticipated that if the neutral sulfur clusters (Fig. 1b) were fully wrapped with an ionic capsular matrix, the exact molecular weights could be readily available on the MS analysis (Fig. 1a, right), because of the protection of the encapsulated clusters and the promotion of the efficient ionization. In addition, development of a new class of supramolecular matrixes for common mass spectrometry would assist in the creation of not only unknown sulfur clusters but also novel metalloclusters. There are many reports on the successful stabilization and observation of highly reactive organic and organometallic compounds within supramolecular cages and capsules8,9,10,11,12,13,14,15,16,17,18. Nevertheless, those of labile inorganic clusters are limited to white phosphorus (Fig. 1c)19 and yellow arsenic20, 21.
MS analysis of an inorganic cluster and structure of sulfur clusters and polyaromatic capsule 1. a Schematic representation of the MS analysis of a neutral inorganic cluster within/without a capsular matrix and their typical ion peaks. b Sulfur clusters S6, S8, and S12 and their crystal or optimized structures. c White phosphorus and its crystal structure37. d Polyaromatic capsule 1 and the space-filling representation of the crystal structure (substituents and counterions are omitted for clarity)
Here we report the MS analysis of cyclic sulfur clusters S n (n = 6, 8, and 12; Fig. 1b) upon encapsulation. As an analytical matrix, we employ supramolecular capsule 1 (Fig. 1d)22 bearing a positively charged (4+) polyaromatic shell suitable for electrospray ionization time-of-flight (ESI-TOF) MS analysis2. The composition of the supramolecular matrix is simple, i.e., M2L4 with two Pd(II) ions and four bent bispyridine ligands with two anthracene panels23. The spherical cavity surrounded by the multiple polyaromatic panels can efficiently accommodate various organic molecules with dimensions of up to 1 nm (e.g., BODIPY, corannulene, and fullerene C60) in aqueous solutions through the hydrophobic effect and π-stacking interactions18, 24,25,26. However, solution-state host–guest interactions between the inorganic sulfur clusters and our capsules as well as other supramolecular cages remained uncertain so far27, 28. In the present work, we demonstrate the exact mass determination of cyclic S6 and S8 clusters by common ESI-TOF MS analysis upon encapsulation within capsular matrix 1. In addition, we accomplish selective preparation of a cyclic S12 cluster from two S6 clusters within the matrix upon light irradiation, as revealed by subsequent MS analysis of the host–guest complex.
Results
MS analysis of a cyclic S8 cluster
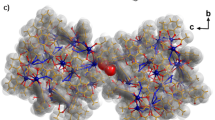
We firstly carried out the quantitative encapsulation and mass determination of cyclo-octasulfur (S8) using capsular matrix 1 in water. Stirring excess hydrophobic S8 (3.9 μmol) in a D2O solution (0.5 ml) of 1 (0.40 μmol) at room temperature for 30 min led to the exclusive formation of a 1⊃(S8)2 complex (Fig. 2a). After removal of suspended free S8 by filtration, the 1:2 host–guest structure was confirmed by NMR, UV-visible, and X-ray crystallographic analyses. In the 1H NMR spectrum, the appearance of new aromatic signals and the disappearance of the original matrix signals are indicative of the quantitative encapsulation of the sulfur cluster (Figs. 2b, c and see Supplementary Fig. 1). Due to the inclusion, the internal H f signal of 1 was largely shifted upfield (Δδ = −0.40 p.p.m.), whereas the external H h signal remained almost unchanged. UV-visible spectrum of the product showed slight red-shifts (Δλ max = +6 nm) of the absorption bands derived from the anthracene moieties of 1 (Fig. 2d), indicating S-π interactions29. Direct evidence of the 1⊃(S8)2 structure was obtained from the X-ray crystallographic analysis. Pale-yellow crystals suitable for X-ray analysis grew by the slow evaporation of a 25:1 H2O/CH3CN solution of the product at room temperature for 1 month. The molecular structure revealed that two molecules of S8 fully occupy the spherical cavity of 1 in a stacked fashion (Figs. 2e, f and see Supplementary Fig. 3). Each of the crown-shaped rings is in close proximity with the closest intermolecular S•••S distance of 3.1 Å. With regard to the host–guest interactions, the closest distances between the sulfur atoms and the eight anthracene panels are less than 3.6 Å (see Supplementary Fig. 4), whereas those between the sulfur atoms and the Pd(II) centers are more than 4.8 Å. The S8 dimer can be isolated from the bulk phase by the multiple anthracene panels of 1 (Fig. 2g).
Formation and characterization of 1⊃(S8)2. a Schematic representation of the encapsulation of two S8 clusters within matrix 1. 1H NMR spectra (500 MHz, D2O, room temperature) of b 1 and c 1⊃(S8)2. d UV-visible spectra (room temperature) of 1 and 1⊃(S8)2 in H2O, and S8 in CH3OH. X-ray crystal structure of 1⊃(S8)2 (host and guest parts): e cylinder and space-filling representation, f ball-and-stick, and g space-filling representations (the peripheral substituents are replaced by hydrogen atoms). h MALDI-TOF MS spectrum of S8. i ESI-TOF MS spectrum (H2O) of 1⊃(S8)2 and the expansion of the [1⊃(S8)2−4•NO3 −]4+ peak
Although the S8 cluster is the most stable sulfur allotrope6, the matrix-assisted laser desorption ionization (MALDI)-TOF MS spectrum displays several ion peaks derived from the fragments such as S3 +, S4 +, and S5 + species (m/z = 96, 128, and 160, respectively; Fig. 2h) even under various conditions30. ESI-TOF MS analysis of S8 also shows no target peak (see Supplementary Fig. 5). In contrast, ESI-TOF MS spectrum of the product showed prominent peaks at m/z = 988.0 and 1338.1, corresponding to 1:2 host–guest [1⊃(S8)2 – n•NO3 −]n+ species (n = 4 and 3, respectively; Fig. 2i and see Supplementary Fig. 6). No MS peaks assignable to the empty matrix and host–guest complexes including decomposed S8 clusters were detected so that the significant stabilization of S8 under MS conditions was demonstrated upon encapsulation within 1. The non-covalent host–guest structure remains intact in water even under highly diluted conditions (<5.0 μM; see Supplementary Fig. 7), suggesting the potential application for microgram-scale MS analysis. Interestingly, the binding is stronger than that of hydrophobic cyclooctane. 1H NMR competitive binding experiments revealed that the matrix encapsulates S8 clusters with >90% selectivity from a 1:1 mixture of the cyclic sulfur and alkane (20 equiv. each) under ambient aqueous conditions (see Supplementary Fig. 8).
Stabilization and MS analysis of a cyclic S6 cluster
To investigate how capsular matrix 1 impacts the MS analysis of a metastable sulfur allotrope, we next synthesized pure cyclo-hexasulfur (S6) at low temperatures and prepared the host–guest complex31. In a manner similar to the encapsulation of S8, simple mixing hydrophobic S6 (13 equiv.) with 1 in D2O gave rise to a 1⊃(S6)2 complex predominantly (Fig. 3a). A 1H NMR spectrum of the resultant solution displayed new prominent signals in the aromatic region derived from the desired product (Fig. 3b and see Supplementary Fig. 10). One set of minor signals was also found at, e.g., 9.27, 8.07, and 8.01 p.p.m., which suggests the formation of a 1⊃(S6•S8) complex (<10%) due to the decomposition of S6 during the mixing. UV-visible absorption bands of 1 around 390 nm were again slightly red-shifted (Δλ max = +3 nm) after treatment with the S6 solid (see Supplementary Fig. 11). The encapsulation of two cyclic S6 clusters within the matrix was preliminarily confirmed by the X-ray crystallographic analysis (Fig. 3f and see Supplementary Figs 12 and 13) and yet definitely proved by the ESI-TOF MS analysis. Prominent molecular ion peaks were observed in the spectrum at m/z = 956.0, 1295.4, and 1974.6, derived from [1⊃(S6)2−n•NO3 −]n+ species (n = 4, 3, and 2, respectively; Fig. 3c). The isotope patterns of the peaks fully coincide with the calculated ones (see Supplementary Fig. 14). As expected, minor MS peaks found at m/z = 972.0 and 1317.0 were assignable to a 1⊃(S6•S8) species. In contrast to the host–guest complex, without the matrix, the MALDI-TOF MS analysis of free S6 showed only the fragment peaks (see Supplementary Fig. 15).
Formation and characterization of 1⊃(S6)2 and its stability. a Schematic representation of the encapsulation of two S6 clusters within matrix 1 in D2O (right) and the decomposition of S6 in CS2 (left). 1H NMR (500 MHz, D2O, room temperature; left) and ESI-TOF MS spectra (H2O; right) of 1⊃(S6)2 b, c before and d, e after 8 days at room temperature (asterisks indicate signals derived from 1⊃(S6•S8)). f X-ray crystal structure of 1⊃(S6)2: cylindrical and space-filling representation (solvents and counterions are omitted for clarity). g Decomposition of S6 (%) within matrix 1 in water and without matrix 1 in CS2
It is worth noting that the otherwise labile S6 cluster, which possesses a highly strained ring structure4, 6, 32, is remarkably stabilized within matrix 1 not only under MS conditions but also in an ambient solution state33. Free S6 dissolved in CS2 was completely decomposed into S8 and other oligosulfurs within ~1 h at room temperature (Fig. 3g and see Supplementary Fig. 17). In sharp contrast, time-dependent 1H NMR analysis revealed that 75% of the proton signals of 1⊃(S6)2 remain unchanged after 8 days (Fig. 3d and see Supplementary Fig. 18) and the ESI-TOF MS analysis of the resultant solution indicated the generation of 1⊃(S6•S8) and 1⊃(S6•S7) species (Fig. 3e). The τ 1/2 of the S6 clusters within 1 was estimated to be 770 h by 1H NMR studies (Fig. 3g). The decomposition is further suppressed at lower temperatures (e.g., τ 1/2 = 1733 h at 4 °C). The observed, unusual stabilization of encapsulated S6 clusters in solution as well as under the MS conditions presumably arises from the isolation effect of the polyaromatic shell of matrix 1.
In situ preparation and MS analysis of a cyclic S12 cluster
It is known that sulfur clusters are photosensitive and, in most cases, rapidly decomposed into complex mixtures in solutions upon light irradiation4,5,6. Notably, selective photochemical transformation from two S6 clusters into a cyclic S12 cluster and subsequent MS analysis of the product were attained using matrix 1. When a frozen aqueous glass of 1⊃(S6)2 cooled at −196 °C was irradiated by LED lamps (3 W × 4, λ irrd = 425 ± 15 nm) for 30 min, distinct shifts of the matrix signals were observed in the NMR spectrum (Figs. 4a, c and see Supplementary Fig. 19). The signal pattern is quite different from that of empty 1 and host–guest complex 1⊃(S8)2, elucidating the formation of a new photoproduct within 1. In addition, a huge upfield shift of inner protons H f is indicative of the encapsulation of a bulky cluster. The product could not be extracted with CS2 solutions, because of most probably its steric demand. This photoreaction occurred very slowly under UV light irradiation (λ irrd = 360 nm), probably due to the strong absorption bands (~380 nm) of the polyaromatic capsule (see Supplementary Fig. 20). A Raman spectrum of the product supported the formation of the cyclic S12 cluster (Figs. 4d, f and see Supplementary Fig. 21). The observed new peaks at 445 and 457 cm−1 are overlapped with the characteristic peaks of free S12 (i.e., 445 and 456 cm−1, respectively)34. On the other hand, Raman peaks derived from clusters S6 (e.g., 200 and 264 cm−1; Fig. 4e) and S8 (e.g., 149 and 218 cm−1) within 1 were virtually undetected in the spectrum. These peaks are slightly shifted (up to −4 cm−1) as compared with those of free S6 and S8 (see Supplementary Figs 16 and 9). Finally, ESI-TOF MS spectrum of the product exhibited intense peaks at m/z = 956.0 and 1295.7, assigned to 1:1 host–guest [1⊃S12−n•NO3 −]n+ species (n = 4 and 3, respectively; Fig. 4g and see Supplementary Fig. 22). The MS peaks for 1 and 1⊃(S8)2 were not observed in the spectrum. The optimized structure of 1ʹ⊃S12 (R = −OCH3) by force-field calculations displays that the spherical S12 cluster with a diameter of 0.66 nm is fully insulated by the spherical polyaromatic framework of 1ʹ (Fig. 4h and see Supplementary Fig. 23). The otherwise reactive S12 cluster in the capsular shell remains in more than 60% for 8 days at room temperature (see Supplementary Figs 24 and 25). Concerning the unusual clusterization in the isolated nanospace, the S6 cluster provides a highly strained, small ring structure, whereas the product is the next stable sulfur allotrope after S8. Thus, the formation of the S12 cluster through the generation of a biradical species (Fig. 4b), by homolytic photocleavage of the S–S bond of the encapsulated S6, might be a thermodynamically favorable process32, 35.
Preparation and characterization of 1⊃S12. a Schematic representation of the transformation from two S6 clusters into a S12 cluster within matrix 1 upon light irradiation and b the proposed intermediate. c 1H NMR spectra (500 MHz, D2O, room temperature) of 1⊃S12 (red line) and 1⊃(S6)2 (gray line). Raman spectra (He−Ne laser, λ ex = 632.8 nm, room temperature) of d 1, e 1⊃(S6)2, and f 1⊃S12. g ESI-TOF MS spectrum (H2O) of 1⊃S12. h Optimized structure of 1ʹ⊃S12 (R = −OCH3): cylindrical and space-filling representation (left) and ball-and-stick representation (right)
Discussion
We have succeeded in the facile MS characterization of inorganic sulfur clusters S6, S8, and S12 by using a polyaromatic capsule as a new supramolecular matrix, whereas the clusters themselves fragment under usual MS conditions. Simple mixing cyclic S6 or S8 clusters with the capsular matrix provides stable 1:2 host–guest complexes for common ESI-TOF MS analysis. The cationic and closed polyaromatic shell of the matrix facilitates the efficient ionization of the uncharged clusters without troublesome fragmentation. In addition, the capsular matrix can be used for not only in situ synthesis but also MS analysis of a cyclic S12 cluster. The otherwise labile S6 and S12 clusters are also significantly stabilized within the matrix in an ambient solution state. The present functions as an analytical tool as well as a reaction vessel for sulfur clusters prompt us to discover unknown inorganic clusters by using the polyaromatic capsular matrix and its potential derivatives36.
Methods
General
NMR: Bruker ASCEND-500 (500 MHz), ESI-TOF MS: Bruker micrOTOF II, FT-IR: JASCO FT/IR-4200, X-ray: Bruker AXS D8 VENTURE/PHOTON 100 diffractometer Raman: JASCO NRS-4100 or HORIBA, Ltd LabRAM HR Evolution. Optimized structure: Materials Studio (ver. 5.5.3). Solvents and reagents: TCI Co., Ltd, Wako Pure Chemical Industries Ltd, Kanto Chemical Co., Inc., Sigma-Aldrich Co., and Cambridge Isotope Laboratories, Inc. Polyaromatic capsule 1 and sulfur cluster S6 (see Supplementary Methods) were synthesized according to previously reported procedures18, 31.
Raman analysis
The excitation wavelength from an He-Ne laser was 632.8 nm with a power on the sample typically 22 μW. A long-working distance objective (× 50 magnification, 10 mm focal length) was used to focus the laser onto the sample. The Raman signal was collected in a back-scattering geometry.
Synthesis of 1⊃(S8)2
Capsular matrix 1 (1.5 mg, 0.40 μmol), S8 (1.0 mg, 3.9 μmol), and D2O (0.5 ml) were added to a microtube (2 ml) and the resultant mixture was stirred at room temperature for 30 min. The quantitative formation of a 1⊃(S8)2 complex was confirmed by NMR (see Supplementary Figs 1 and 2), X-ray crystallographic (see Supplementary Figs 3 and 4 and Supplementary Table 1), ESI-TOF MS (see Supplementary Fig. 6), and Raman (see Supplementary Fig. 9) analyses. The host–guest structure of 1⊃(S8)2 is stable enough in water at room temperature even under high dilution conditions (5.0 μM), as confirmed by 1H NMR analysis (see Supplementary Fig. 7).
1H NMR (500 MHz, D2O, room temperature): δ 2.40 (s, 24H), 3.00 (m, 16H), 3.44 (s, 12H), 3.90 (m, 16H), 4.02 (m, 8H), 4.42 (m, 4H), 4.60 (m, 4H), 6.27 (s, 4H), 6.38 (d, J = 8.5 Hz, 8H), 6.80 (dd, J = 8.5, 7.5 Hz, 8H), 6.96 (d, J = 8.5 Hz, 8H), 7.16 (dd, J = 8.5, 7.5 Hz, 8H), 7.52 (dd, J = 8.5, 7.5 Hz, 8H), 7.68 (s, 8H), 7.72 (d, J = 8.5 Hz, 8H), 7.78 (dd, J = 8.5, 7.5 Hz, 8H), 8.05 (d, J = 8.5 Hz, 8H), 8.33 (dd, J = 8.0, 5.5 Hz, 8H), 8.56 (d, J = 8.0 Hz, 8H), 9.10 (d, J = 5.5 Hz, 8H). 13C NMR (125 MHz, D2O, room temperature): δ 57.0 (CH3), 58.1 (CH3), 70.6 (CH2), 71.7 (CH2), 72.6 (CH2), 72.7 (CH2), 124.2 (CH), 124.8 (CH), 126.2 (CH), 126.5 (CH), 127.0 (CH), 127.2 (CH), 127.3 (CH), 127.7 (C q ), 128.1 (C q ), 128.4 (CH), 129.3 (C q ), 129.4 (C q ), 129.5 (C q ), 129.8 (CH), 134.9 (C q ), 138.4 (C q ), 145.3 (CH), 145.6 (C q ), 151.5 (C q ), 151.7 (CH), 152.1 (CH). Raman (λ ex = 632.8 nm, 22 μW, cm−1): 149, 218, 238, 400, 478. ESI-TOF MS (H2O): m/z 2038.6 [1⊃(S8)2−2•NO3 −]2+, 1338.1 [1⊃(S8)2−3•NO3 −]3+, 988.0 [1⊃(S8)2−4•NO3 −]4+.
Synthesis of 1⊃(S6)2
Capsular matrix 1 (1.5 mg, 0.40 μmol), S6 (1.0 mg, 5.1 μmol), and D2O (0.5 ml) were added to a microtube (2 ml). The mixture was stirred at room temperature for 15 min. The selective formation of a 1⊃(S6)2 complex (>90% yield) was confirmed by NMR (see Supplementary Fig. 10), UV-visible (see Supplementary Fig. 11), X-ray crystallographic (see Supplementary Figs 12 and 13 and Supplementary Table 2), ESI-TOF MS (see Supplementary Fig. 14), and Raman (see Supplementary Fig. 16) analyses.
1H NMR (500 MHz, D2O, room temperature): δ 2.39 (s, 24H), 3.00 (m, 16H), 3.43 (s, 12H), 3.82 (m, 8H), 3.89 (m, 8H), 3.96 (m, 8H), 4.39 (m, 4H), 4.59 (m, 4H), 6.14 (d, J = 8.5 Hz, 8H), 6.21 (dd, J = 8.5, 7.5 Hz, 8H), 6.25 (s, 4H), 6.67 (dd, J = 8.5, 7.5 Hz, 8H), 7.11 (d, J = 8.5 Hz, 8H), 7.54 (dd, J = 8.5, 7.5 Hz, 8H), 7.59 (d, J = 8.5 Hz, 8H), 7.66 (dd, J = 8.5, 7.5 Hz, 8H), 7.92 (d, J = 8.5 Hz, 8H), 8.32 (s, 8H), 8.43 (dd, J = 8.0, 6.0 Hz, 8H), 8.53 (d, J = 8.0 Hz, 8H), 9.35 (d, J = 6.0 Hz, 8H). Raman (λ ex = 632.8 nm, 22 μW, cm−1): 200, 238, 264, 398, 478. ESI-TOF MS (H2O): m/z 1974.6 [1⊃(S6)2−2•NO3 −]2+, 1295.4 [1⊃(S6)2−3•NO3 –]3+, 956.0 [1⊃(S6)2−4•NO3 –]4+.
Formation of 1⊃S12
An aqueous solution of 1⊃(S6)2 (0.8 mM, 0.5 ml) was irradiated with Relyon LED lamps (3 W × 4: λ irrd = 425 ± 15 nm) for 30 min at −196 °C (under liquid N2). The selective formation of a 1⊃S12 complex was confirmed by 1H NMR (see Supplementary Fig. 19), UV-visible (see Supplementary Fig. 20), Raman (see Supplementary Fig. 21), and ESI-TOF MS (see Supplementary Fig. 22) analyses. On the other hand, light irradiation (3 W × 2) of 1⊃(S6)2 in D2O for 2 h at room temperature gave rise to complex mixtures.
1H NMR (500 MHz, D2O, room temperature): δ 2.39 (s, 24H), 3.02 (m, 16H), 3.45 (s, 12H), 3.92 (m, 16H), 3.99 (m, 8H), 4.44 (m, 4H), 4.61 (m, 4H), 6.15 (d, J = 8.5 Hz, 8H), 6.36 (dd, J = 8.5, 7.0 Hz, 8H), 6.49 (s, 4H), 6.92 (dd, J = 8.5, 7.0 Hz, 8H), 7.01 (d, J = 8.5 Hz, 8H), 7.49 (dd, J = 8.5, 7.0 Hz, 8H), 7.72-7.69 (m, 16H), 7.82 (s, 8H), 8.00 (d, J = 8.5 Hz, 8H), 8.36 (dd, J = 7.5, 5.5 Hz, 8H), 8.49 (d, J = 7.5 Hz, 8H), 9.22 (d, J = 5.5 Hz, 8H). Raman (λ ex = 632.8 nm, 22 μW, cm−1): 180, 216, 238, 263, 288, 398, 410, 445, 457, 475, 490. ESI-TOF MS (H2O): m/z 1974.0 [1⊃S12 – 2•NO3 −]2+, 1295.7 [1⊃S12 – 3•NO3 −]3+, 956.0 [1⊃S12 – 4•NO3 −]4+.
Data availability
The authors declare that the data supporting the findings of this study are available within the Supplementary Information files and from the corresponding author upon reasonable request. CCDC-1509515 and CCDC-1525707 contain the supplementary crystallographic data for the structures reported in this article. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre (CCDC) via www.ccdc.cam.ac.uk/data_request/cif.
References
Gauglitz, G. & Moore, D. S. Handbook of Spectroscopy 2nd enlarged edn (Wiley-VCH, 2014).
Gross, J. H. Mass Spectrometry: A Textbook 2nd edn (Springer, 2011).
Alonso, J. A. Structure and Properties of Atomic Nanoclusters 2nd edn (Imperial College Press, 2011).
Meyer, B. Elemental sulfur. Chem. Rev. 76, 367–388 (1976).
Steudel, R. Homocyclic sulfur molecules. Top. Curr. Chem. 102, 149–176 (1982).
Steudel, R. & Eckert, B. Solid sulfur allotropes. Top. Curr. Chem. 230, 1–79 (2003).
Liu, H. & Jiang, X. Transfer of sulfur: from simple to diverse. Chem. Asian J. 8, 2546–2563 (2013).
Cram, D. J., Tanner, M. E. & Thomas, R. The taming of cyclobutadiene. Angew. Chem. Int. Ed. 30, 1024–1027 (1991).
Warmuth, R. o-Benzyne: strained alkyne or cumulene ?—NMR characterization in a molecular container. Angew. Chem. Int. Ed. 36, 1347–1350 (1997).
Warmuth, R. & Marvel, M. A. 1,2,4,6-Cycloheptatetraene: room-temperature stabilization inside a hemicarcerand. Angew. Chem. Int. Ed. 39, 1117–1119 (2000).
Ziegler, M., Brumaghim, J. L. & Raymond, K. N. Stabilization of a reactive cationic species by supramolecular encapsulation. Angew. Chem. Int. Ed. 39, 4119–4121 (2000).
Yoshizawa, M., Kusukawa, T., Fujita, M., Sakamoto, S. & Yamaguchi, K. Cavity-directed synthesis of labile silanol oligomers within self-assembled coordination cages. J. Am. Chem. Soc. 123, 10454–10459 (2001).
Kawano, M., Kobayashi, Y., Ozeki, T. & Fujita, M. Direct crystallographic observation of a coordinatively unsaturated transition-metal complex in situ generated within a self-assembled cage. J. Am. Chem. Soc. 128, 6558–6559 (2006).
Dong, V. M., Fiedler, D., Carl, B., Bergman, R. G. & Raymond, K. N. Molecular recognition and stabilization of iminium ions in water. J. Am. Chem. Soc. 128, 14464–14465 (2006).
Iwasawa, T., Hooley, R. J. & Rebek, J. Jr. Stabilization of labile carbonyl addition intermediates by a synthetic receptor. Science 317, 493–496 (2007).
Nishimura, N. & Kobayashi, K. Self-assembled boronic ester cavitand capsule as a photosensitizer and a guard nanocontainer against photochemical reactions of 2,6-diacetoxyanthracene. J. Org. Chem. 75, 6079–6085 (2010).
Horiuchi, S., Murase, T. & Fujita, M. Noncovalent trapping and stabilization of dinuclear ruthenium complexes within a coordination cage. J. Am. Chem. Soc. 133, 12445–12447 (2011).
Yamashina, M., Sei, Y., Akita, M. & Yoshizawa, M. Safe storage of radical initiators within a polyaromatic nanocapsule. Nat. Commun. 5, 4662 (2014).
Mal, P., Breiner, B., Rissanen, K. & Nitschke, J. R. White phosphorus is air-stable within a self-assembled tetrahedral capsule. Science 324, 1697–1699 (2009).
Yang, D. et al. Air- and light-stable P4 and As4 within an anion-coordination-based tetrahedral cage. J. Am. Chem. Soc. 139, 5946–5951 (2017).
Schwarzmaier, C. et al. Stabilization of tetrahedral P4 and As4 molecules as guests in polymeric and spherical environments. Angew. Chem. Int. Ed. 52, 10896–10899 (2013).
Kishi, N., Li, Z., Yoza, K., Akita, M. & Yoshizawa, M. An M2L4 molecular capsule with an anthracene shell: encapsulation of large guests up to 1 nm. J. Am. Chem. Soc. 133, 11438–11441 (2011).
Debata, N. B., Tripathy, D. & Chand, D. K. Self-assembled coordination complexes from various palladium(II) components and bidentate or polydentate ligands. Coord. Chem. Rev. 256, 1831–1945 (2012).
Kishi, N. et al. Wide-ranging host capability of a Pd(II)-linked M2L4 molecular capsule with an anthracene shell. Chem. Eur. J. 19, 6313–6320 (2013).
Yamashina, M. et al. Preparation of highly fluorescent host-guest complexes with tunable color upon encapsulation. J. Am. Chem. Soc. 137, 9266–9269 (2015).
Yamashina, M., Matsuno, S., Sei, Y., Akita, M. & Yoshizawa, M. Recognition of multiple methyl groups on aromatic rings by a polyaromatic cavity. Chem. Eur. J. 22, 14147–14150 (2016).
Ohtsu, H., Choi, W., Islam, N., Matsushita, Y. & Kawano, M. Selective trapping of labile S3 in a porous coordination network and the direct X‐ray observation. J. Am. Chem. Soc. 135, 11449–11452 (2013).
Duan, P.-C., Wang, Z.-Y., Chen, J.-H., Yang, G. & Raptis, R. G. Trigonal prismatic Cu(I) and Ag(I) pyrazolato nanocage hosts: encapsulation of S8 and hydrocarbon guests. Dalton. Trans. 42, 14951–14954 (2013).
Salonen, L. M., Ellermann, M. & Diederich, F. Aromatic rings in chemical and biological recognition: energetics and structures. Angew. Chem. Int. Ed. 50, 4808–4842 (2011).
Zheng, D. & Qu, D. Chromatographic separation of polysulfide species in non-aqueous electrolytes—revisited. J. Electrochem. Soc. 161, A1164–A1166 (2014).
Mäusle, H.-J. & Steudel, R. Einfache darstellung von cyclohexaschwefel (S6) aus dichlordisulfan (S2Cl2) und ionischen iodidnen. Z. Anorg. Allg. Chem. 463, 27–31 (1980).
Steudel, R., Strauss, R. & Koch, L. Quantitative HPLC analysis and thermodynamics of sulfur melts. Angew. Chem. Int. Ed. 24, 59–60 (1985).
Sugimoto, K., Uemachi, H., Maekawa, M. & Fujiwara, A. Air-stable cyclohexasulfur as cocrystal. Cryst. Growth Des. 13, 433–436 (2013).
Steudel, R. & Rebsch, M. Infrared and raman spectra of cyclo dodecasulphur. J. Mol. Spectrosc. 51, 189–193 (1974).
Jones, R. O. & Ballone, P. Density functional and Monte Carlo studies of sulfur. I. Structure and bonding in S n rings and chains (n = 2–18). J. Chem. Phys. 118, 9257–9265 (2003).
Yoshizawa, M. & Yamashina, M. Coordination-driven nanostructures with polyaromatic shells. Chem. Lett. 46, 163–171 (2017).
Simon, A., Borrmann, H. & Craubner, H. Crystal structure of ordered white phosphorus (β-P). Phosphorus Sulfur Relat. Elem. 30, 507–510 (1987).
Acknowledgements
This work was supported by JSPS KAKENHI (Grant No. JP25104011/JP26288033/JP17H05359/JP15H05757) and “Support for Tokyotech Advanced Researchers (STAR)”. M.Ya. thanks the JSPS for an Overseas Research Fellowship. A.K. and K.Y. acknowledge the financial support from JST ERATO (Grant No. JPMJER1503).
Author information
Authors and Affiliations
Contributions
S.M., M.Ya., and M.Yo. designed the work, carried out research, analyzed data, and wrote the paper. Y.S., M.A., A.K., and K.Y. were involved in the work discussion. Y.S. and A.K. contributed to X-ray crystallographic and Raman analyses, respectively. M.Yo. is the principal investigator. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Matsuno, S., Yamashina, M., Sei, Y. et al. Exact mass analysis of sulfur clusters upon encapsulation by a polyaromatic capsular matrix. Nat Commun 8, 749 (2017). https://doi.org/10.1038/s41467-017-00605-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-017-00605-5
This article is cited by
-
Modern cluster design based on experiment and theory
Nature Reviews Chemistry (2021)
-
Strategies for binding multiple guests in metal–organic cages
Nature Reviews Chemistry (2019)
-
Cramming versus threading of long amphiphilic oligomers into a polyaromatic capsule
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.