Abstract
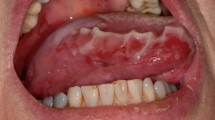
Fixed Drug Eruptions (FDE) represent a distinctive type of adverse drug reaction, typically characterized by recurring, sharply demarcated skin lesions occurring at identical sites with each administration of the causative drug. A less frequent, albeit significant manifestation of FDE, is balanoposthitis, an inflammatory condition affecting the glans penis and prepuce. This rare case report explores the clinical presentation, diagnosis, and therapeutic management of FDE-induced balanoposthitis in a 34-year-old male patient who developed this condition following azithromycin administration to treat a pulmonary infection. The patient’s distinctive symptoms, coupled with a medical history of similar antibiotic-induced reactions, pointed strongly towards an FDE diagnosis. Management entailed immediate discontinuation of the offending drug and initiation of symptomatic treatment, culminating in a positive therapeutic outcome. This case illuminates the potential of commonly prescribed medications, such as antibiotics, to incite balanoposthitis via FDE. It underscores the critical need for healthcare professionals to include FDE in their differential diagnosis for balanoposthitis, especially when patient exposure to high-risk medications is evident. Furthermore, the report emphasizes the pressing requirement for additional research to elucidate the pathogenesis of FDE-induced balanoposthitis and to devise effective therapeutic and preventive measures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The data generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Flowers H, Brodell R, Brents M, Wyatt JP. Fixed drug eruptions: presentation, diagnosis, and management. South Med J. 2014;107:724–7.
Brahimi N, Routier E, Raison-Peyron N, Tronquoy AF, Pouget-Jasson C, Amarger S, et al. A three-year-analysis of fixed drug eruptions in hospital settings in France. Eur J Dermatol. 2010;20:461–4.
Zaouak A, Chabchoub I, Essid D, Ben Jennet S, Hammami H, Fenniche S. Genital involvement in bullous fixed drug eruption. Skinmed. 2019;17:306–9.
Fazeli SA, Abbasi M, Jalali H, Eskandari S, Shamshirgaran F, Dehghani Z, et al. Bullous fixed drug eruption following ibuprofen ingestion. J Res Pharm Pract. 2018;7:51–6.
Shiohara T, Mizukawa Y. Fixed drug eruption: a disease mediated by self-inflicted responses of intraepidermal T cells. Eur J Dermatol. 2007;17:201–8.
Mizukawa Y, Yamazaki Y, Shiohara T. In vivo dynamics of intraepidermal CD8+ T cells and CD4+ T cells during the evolution of fixed drug eruption. Br J Dermatol. 2008;158:1230–8.
Mizukawa Y, Shiohara T. Fixed drug eruption: a prototypic disorder mediated by effector memory T cells. Curr Allergy Asthma Rep. 2009;9:71–7.
Shiohara T. Fixed drug eruption: pathogenesis and diagnostic tests. Curr Opin Allergy Clin Immunol. 2009;9:316–21.
Sehgal VN, Srivastava G. Fixed drug eruption (FDE): changing scenario of incriminating drugs. Int J Dermatol. 2006;45:897–908.
Mahboob A, Haroon TS. Drugs causing fixed eruptions: a study of 450 cases. Int J Dermatol. 1998;37:833–8.
Anderson HJ, Lee JB. A review of fixed drug eruption with a special focus on generalized bullous fixed drug eruption. Medicina (Kaunas). 2021;57:925.
Lammintausta K, Kortekangas-Savolainen O. The usefulness of skin tests to prove drug hypersensitivity. Br J Dermatol. 2005;152:968–74.
Oezkaya E. Fixed drug eruption: state of the art. J Dtsch Dermatol Ges. 2008;6:181–8.
Bos JD, Zonneveld I, Das PK, Krieg SR, Vanderloos CM, Kapsenberg ML. The skin immune system (SIS): distribution and immunophenotype of lymphocyte subpopulations in normal human skin. J Invest Dermatol. 1987;88:569–73.
Edwards SK, Bunker CB, Ziller F, van der Meijden WI. 2013 European guideline for the management of balanoposthitis. Int J STD AIDS. 2014;25:615–26.
Barbaud A. Skin testing and patch testing in non-IgE-mediated drug allergy. Curr Allergy Asthma Rep. 2014;14:442.
Barbaud A, Collet E, Milpied B, Assier H, Staumont D, Avenel-Audran M, et al. A multicentre study to determine the value and safety of drug patch tests for the three main classes of severe cutaneous adverse drug reactions. Br J Dermatol. 2013;168:555–62.
Barbaud A, Gonçalo M, Bruynzeel D, Bircher A. Guidelines for performing skin tests with drugs in the investigation of cutaneous adverse drug reactions. Contact Dermat. 2001;45:321–8.
Rawlings AV, Harding CR. Moisturization and skin barrier function. Dermatol Ther. 2004;17:43–8.
Gelincik A, Cavkaytar O, Kuyucu S. An update on the management of severe cutaneous drug hypersensitivity reactions. Curr Pharm Des. 2019;25:3881–901.
Barbaud A. Drug patch testing in systemic cutaneous drug allergy. Toxicology. 2005;209:209–16.
Zang X, Du W, Geng F, Meng P, Lingling E, Zhang H, et al. Clinical observation of allergic contact stomatitis treated with Kangfuxin solution as adjuvant: case report. Ann Transl Med. 2022;10:721.
Verma AA, Khuu W, Tadrous M, Gomes T, Mamdani MM. Fixed-dose combination antihypertensive medications, adherence, and clinical outcomes: a population-based retrospective cohort study. PLoS Med. 2018;15:e1002584.
Thong BYH, Tan TC. Epidemiology and risk factors for drug allergy. Br J Clin Pharm. 2011;71:684–700.
Funding
This study was supported by the Sichuan Science and Technology Program (Award Number: 2023NSFSC1802, Recipient: FY).
Author information
Authors and Affiliations
Contributions
JYZ and FY conceptualized the research study. YX, MJW, and ALY conducted the investigation. FY provided the necessary resources for the study. The original draft of the manuscript was written by JYZ. The manuscript was reviewed and edited by FY, MJW, FZ, and XJY. The project was supervised by XJY, who also administered the project alongside YXG and FZ.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Per the guidelines of our institution, no formal ethical approval or participant consent is necessary for a single case report if it contains no patient-identifying information. However, we have obtained the patient’s consent, and it can be requested from the corresponding author if needed. The methodologies employed in this study involving human participants align with the ethical norms of our institutional and national research committees and adhere to the principles outlined in the 1964 Helsinki Declaration, its subsequent amendments, or equivalent ethical standards.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, J., Yang, F., Wang, M. et al. Fixed drug eruption-induced balanoposthitis: a case report. Int J Impot Res (2023). https://doi.org/10.1038/s41443-023-00817-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41443-023-00817-y