Abstract
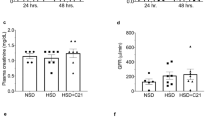
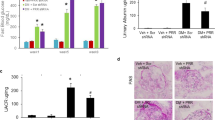
The progress in the research field of diabetic kidney disease (DKD) has been disturbed by the lack of reliable animal models. Angiotensin II (Ang II) type 1 receptor (AT1R)-associated protein (ATRAP) promotes internalization of AT1R and selectively inhibits pathological AT1R signaling. In this study, we investigated whether overactivation of the renin-angiotensin system (RAS) through a combination of ATRAP deletion with Ang II stimulation developed a progressive DKD model in C57BL/6 mice, which are resistant to the development of kidney injury. Eight-week-old male systemic ATRAP-knockout mice on the C57BL/6 strain (KO) and their littermate wild-type mice (Ctrl) were divided into five groups: 1) Ctrl, 2) Ctrl-streptozotocin (STZ), 3) KO-STZ, 4) Ctrl-STZ-Ang II, and 5) KO-STZ-Ang II. Ang II was administered for 6 weeks from 4 weeks after STZ administration. At 10 weeks after STZ administration, mice were euthanized to evaluate kidney injuries. Neither ATRAP deletion alone nor Ang II stimulation alone developed a progressive DKD model in STZ-induced diabetic C57BL/6 mice. However, a combination of ATRAP deletion with Ang II stimulation accelerated the development of DKD as manifested by overt albuminuria, glomerular hypertrophy, podocyte loss, mesangial expansion, kidney interstitial fibrosis and functional insufficiency, concomitant with increased angiotensinogen and AT1R expression in the kidneys. In STZ-induced diabetic C57BL/6 mice that are resistant to the development of kidney injury, the combination of ATRAP deletion and Ang II stimulation accelerates the development of DKD, which may be associated with intrarenal RAS overactivation.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All relevant data are included within the paper. The datasets are available from the corresponding authors upon reasonable request.
References
Tuttle KR, Bakris GL, Bilous RW, Chiang JL, de Boer IH, Goldstein-Fuchs J, et al. Diabetic kidney disease: a report from an ADA Consensus Conference. Diabetes Care. 2014;37:2864–83.
Heerspink HJL, Kosiborod M, Inzucchi SE, Cherney DZI. Renoprotective effects of sodium-glucose cotransporter-2 inhibitors. Kidney Int. 2018;94:26–39.
Lo KB, Rangaswami J, Vaduganathan M. Nonsteroidal mineralocorticoid receptor antagonists and cardiorenal outcomes in chronic kidney disease. Nephrol Dial Transplant. 2022;38:845–54.
Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383:1436–46.
Herrington WG, Staplin N, Wanner C, Green JB, Hauske SJ, Emberson JR, et al. Empagliflozin in patients with chronic kidney disease. N Engl J Med. 2023;388:117–27.
Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, et al. Effect of Finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med. 2020;383:2219–29.
Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, et al. Cardiovascular events with finerenone in kidney disease and type 2 diabetes. N Engl J Med. 2021;385:2252–63.
Rayego-Mateos S, Rodrigues-Diez RR, Fernandez-Fernandez B, Mora-Fernández C, Marchant V, Donate-Correa J, et al. Targeting inflammation to treat diabetic kidney disease: the road to 2030. Kidney Int. 2023;103:282–96.
Azushima K, Gurley SB, Coffman TM. Modelling diabetic nephropathy in mice. Nat Rev Nephrol. 2018;14:48–56.
Breyer MD, Böttinger E, Brosius FC 3rd, Coffman TM, Harris RC, Heilig CW, et al. Mouse models of diabetic nephropathy. J Am Soc Nephrol. 2005;16:27–45.
Brosius FC 3rd, Alpers CE, Bottinger EP, Breyer MD, Coffman TM, Gurley SB, et al. Mouse models of diabetic nephropathy. J Am Soc Nephrol. 2009;20:2503–12.
Thomas MC, Brownlee M, Susztak K, Sharma K, Jandeleit-Dahm KA, Zoungas S, et al. Diabetic kidney disease. Nat Rev Dis Primers. 2015;1:15018.
Daviet L, Lehtonen JY, Tamura K, Griese DP, Horiuchi M, Dzau VJ. Cloning and characterization of ATRAP, a novel protein that interacts with the angiotensin II type 1 receptor. J Biol Chem. 1999;274:17058–62.
Tamura K, Tanaka Y, Tsurumi Y, Azuma K, Shigenaga A, Wakui H, et al. The role of angiotensin AT1 receptor-associated protein in renin-angiotensin system regulation and function. Curr Hypertens Rep. 2007;9:121–7.
Wakui H. The pathophysiological role of angiotensin receptor-binding protein in hypertension and kidney diseases: Oshima Award Address 2019. Clin Exp Nephrol. 2020;24:289–94.
Tamura K, Azushima K, Kinguchi S, Wakui H, Yamaji T. ATRAP, a receptor-interacting modulator of kidney physiology, as a novel player in blood pressure and beyond. Hypertens Res. 2022;45:32–9.
Haruhara K, Suzuki T, Wakui H, Azushima K, Kurotaki D, Kawase W, et al. Deficiency of the kidney tubular angiotensin II type1 receptor-associated protein ATRAP exacerbates streptozotocin-induced diabetic glomerular injury via reducing protective macrophage polarization. Kidney Int. 2022;101:912–28.
Maeda A, Tamura K, Wakui H, Dejima T, Ohsawa M, Azushima K, et al. Angiotensin receptor-binding protein ATRAP/Agtrap inhibits metabolic dysfunction with visceral obesity. J Am Heart Assoc. 2013;2:e000312.
Wakui H, Tamura K, Tanaka Y, Matsuda M, Bai Y, Dejima T, et al. Cardiac-specific activation of angiotensin II type 1 receptor-associated protein completely suppresses cardiac hypertrophy in chronic angiotensin II-infused mice. Hypertension. 2010;55:1157–64.
Ohsawa M, Tamura K, Wakui H, Maeda A, Dejima T, Kanaoka T, et al. Deletion of the angiotensin II type 1 receptor-associated protein enhances renal sodium reabsorption and exacerbates angiotensin II-mediated hypertension. Kidney Int. 2014;86:570–81.
Azushima K, Ohki K, Wakui H, Uneda K, Haku S, Kobayashi R, et al. Adipocyte-specific enhancement of Angiotensin II Type 1 receptor-associated protein Ameliorates diet-induced visceral obesity and insulin resistance. J Am Heart Assoc. 2017;6:e004488.
Tsurumi Y, Tamura K, Tanaka Y, Koide Y, Sakai M, Yabana M, et al. Interacting molecule of AT1 receptor, ATRAP, is colocalized with AT1 receptor in the mouse renal tubules. Kidney Int. 2006;69:488–94.
Wakui H, Tamura K, Matsuda M, Bai Y, Dejima T, Shigenaga A, et al. Intrarenal suppression of angiotensin II type 1 receptor binding molecule in angiotensin II-infused mice. Am J Physiol Renal Physiol. 2010;299:F991–1003.
Taguchi S, Azushima K, Yamaji T, Urate S, Suzuki T, Abe E, et al. Effects of tumor necrosis factor-α inhibition on kidney fibrosis and inflammation in a mouse model of aristolochic acid nephropathy. Sci Rep. 2021;11:23587.
Zhao HJ, Wang S, Cheng H, Zhang MZ, Takahashi T, Fogo AB, et al. Endothelial nitric oxide synthase deficiency produces accelerated nephropathy in diabetic mice. J Am Soc Nephrol. 2006;17:2664–9.
Wakui H, Uneda K, Tamura K, Ohsawa M, Azushima K, Kobayashi R, et al. Renal tubule angiotensin II type 1 receptor-associated protein promotes natriuresis and inhibits salt-sensitive blood pressure elevation. J Am Heart Assoc. 2015;4:e001594.
Kinguchi S, Wakui H, Azushima K, Haruhara K, Koguchi T, Ohki K, et al. Effects of ATRAP in renal proximal tubules on Angiotensin-dependent hypertension. J Am Heart Assoc. 2019;8:e012395.
Song S, Meyer M, Türk TR, Wilde B, Feldkamp T, Assert R, et al. Serum cystatin C in mouse models: a reliable and precise marker for renal function and superior to serum creatinine. Nephrol Dial Transplant. 2009;24:1157–61.
Nakagawa T, Sato W, Glushakova O, Heinig M, Clarke T, Campbell-Thompson M, et al. Diabetic endothelial nitric oxide synthase knockout mice develop advanced diabetic nephropathy. J Am Soc Nephrol. 2007;18:539–50.
Kanetsuna Y, Takahashi K, Nagata M, Gannon MA, Breyer MD, Harris RC, et al. Deficiency of endothelial nitric-oxide synthase confers susceptibility to diabetic nephropathy in nephropathy-resistant inbred mice. Am J Pathol. 2007;170:1473–84.
Mohan S, Reddick RL, Musi N, Horn DA, Yan B, Prihoda TJ, et al. Diabetic eNOS knockout mice develop distinct macro- and microvascular complications. Lab Invest. 2008;88:515–28.
Forbes MS, Thornhill BA, Park MH, Chevalier RL. Lack of endothelial nitric-oxide synthase leads to progressive focal renal injury. Am J Pathol. 2007;170:87–99.
Ohki K, Wakui H, Azushima K, Uneda K, Haku S, Kobayashi R, et al. ATRAP expression in brown adipose tissue does not influence the development of diet-induced metabolic disorders in mice. Int J Mol Sci. 2017;18:676.
Wakui H, Tamura K, Masuda S, Tsurumi-Ikeya Y, Fujita M, Maeda A, et al. Enhanced angiotensin receptor-associated protein in renal tubule suppresses angiotensin-dependent hypertension. Hypertension. 2013;61:1203–10.
Wakui H, Dejima T, Tamura K, Uneda K, Azuma K, Maeda A, et al. Activation of angiotensin II type 1 receptor-associated protein exerts an inhibitory effect on vascular hypertrophy and oxidative stress in angiotensin II-mediated hypertension. Cardiovasc Res. 2013;100:511–9.
Wesseling S, Ishola DA Jr, Joles JA, Bluyssen HA, Koomans HA, Braam B. Resistance to oxidative stress by chronic infusion of angiotensin II in mouse kidney is not mediated by the AT2 receptor. Am J Physiol Renal Physiol. 2005;288:F1191–200.
Kirchhoff F, Krebs C, Abdulhag UN, Meyer-Schwesinger C, Maas R, Helmchen U, et al. Rapid development of severe end-organ damage in C57BL/6 mice by combining DOCA salt and angiotensin II. Kidney Int. 2008;73:643–50.
Kamiyama M, Farragut KM, Garner MK, Navar LG, Kobori H. Divergent localization of angiotensinogen mRNA and protein in proximal tubule segments of normal rat kidney. J Hypertens. 2012;30:2365–72.
Uneda K, Wakui H, Maeda A, Azushima K, Kobayashi R, Haku S, et al. Angiotensin II type 1 receptor-associated protein regulates kidney aging and lifespan independent of Angiotensin. J Am Heart Assoc. 2017;6:e006120.
Russo GT, De Cosmo S, Viazzi F, Mirijello A, Ceriello A, Guida P, et al. Diabetic kidney disease in the elderly: prevalence and clinical correlates. BMC Geriatr. 2018;18:38.
Jiang W, Wang J, Shen X, Lu W, Wang Y, Li W, et al. Establishment and validation of a risk prediction model for early diabetic kidney disease based on a systematic review and meta-analysis of 20 cohorts. Diabetes Care. 2020;43:925–33.
Ruggenenti P, Cravedi P, Remuzzi G. The RAAS in the pathogenesis and treatment of diabetic nephropathy. Nat Rev Nephrol. 2010;6:319–30.
Alicic RZ, Rooney MT, Tuttle KR. Diabetic kidney disease: challenges, progress, and possibilities. Clin J Am Soc Nephrol. 2017;12:2032–45.
DeFronzo RA, Reeves WB, Awad AS. Pathophysiology of diabetic kidney disease: impact of SGLT2 inhibitors. Nat Rev Nephrol. 2021;17:319–34.
Acknowledgements
The authors are grateful to Prof. Thomas M. Coffman (Duke-NUS Medical School, Duke University School of Medicine) for useful discussions. We also thank Editage for editing and reviewing the English in this manuscript.
Funding
This work was supported by grants from the Yokohama Foundation for Advancement of Medical Science; Uehara Memorial Foundation; Japan Society for the Promotion of Science; Japan Kidney Association-Nippon Boehringer Ingelheim Joint Research Program; Japanese Association of Dialysis Physicians; Salt Science Research Foundation; Strategic Research Project of Yokohama City University; Japan Agency for Medical Research and Development (AMED); Translational Research program; Strategic Promotion for Practical Application of Innovative Medical Technology (TR-SPRINT) from AMED; Moriya Scholarship Foundation; Bayer Scholarship for Cardiovascular Research; and Mochida Memorial Foundation for Medical and Pharmaceutical Research.
Author information
Authors and Affiliations
Contributions
KA designed and conducted the research. STaguchi, KA, TY, AY, HW, and KT wrote the manuscript. STaguchi, KA, TS, EA, ST, KH, STsukamoto, RM, RK, SK, and HW performed the research. STaguchi and KA analyzed the data. KT supervised the research. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Taguchi, S., Azushima, K., Yamaji, T. et al. Angiotensin II type 1 receptor-associated protein deletion combined with angiotensin II stimulation accelerates the development of diabetic kidney disease in mice on a C57BL/6 strain. Hypertens Res 47, 55–66 (2024). https://doi.org/10.1038/s41440-023-01496-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-023-01496-4
Keywords
This article is cited by
-
PGC1-α in diabetic kidney disease: unraveling renoprotection and molecular mechanisms
Molecular Biology Reports (2024)