Abstract
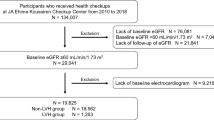
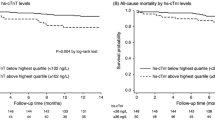
The prognostic value of electrocardiograms (ECGs) has been reported in predialysis patients but not in incident hemodialysis patients with overhydration and electrolyte disturbances, both of which potentially affect ECG results. We performed a retrospective multicenter cohort study involving incident hemodialysis patients and examined whether ECG parameters immediately before hemodialysis initiation can predict subsequent cardiovascular disease (CVD) using Cox proportional hazards models. We explored potential effect modifications by several electrolytes on the predictive power of ECG abnormalities. Among the 618 enrolled patients, 16%, 10%, 46%, and 22% showed a PR interval ≥ 200 ms, QRS interval ≥120 ms, QTc interval ≥ 450/460 ms (male/female), and left ventricular hypertrophy (LVH) by voltage criteria, respectively. Over a median 3-year follow-up, 19% and 16% of the patients developed atherosclerotic and nonatherosclerotic CVD, respectively. The Cox regression model results revealed that the sum of the number of abnormalities in PR, QRS, and QT intervals was a significant risk factor for nonatherosclerotic CVD (hazard ratios (HRs) [95% confidence interval (CI)]: 1.58 [1.24–2.01] per number of abnormalities). The predictive value of LVH for atherosclerotic CVD was attenuated over time. At up to 36 months, although the proportional hazards assumption was met, LVH was significantly associated with atherosclerotic CVD (HR [95% CI]: 1.89 [1.15–3.11]). The adjusted HR was particularly high (HR [95% CI]: 4.02 [1.68–9.60]) among patients who were in the lowest tertile of serum magnesium levels (P for interaction = 0.04). PR, QRS, and QT prolongation additively predicted nonatherosclerotic CVD, while LVH predicted atherosclerotic CVD in the short term.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Deo R, Shou H, Soliman EZ, Yang W, Arkin JM, Zhang X, et al. Electrocardiographic measures and prediction of cardiovascular and noncardiovascular death in CKD. J Am Soc Nephrol. 2016;27:559–69.
Kestenbaum B, Rudser KD, Shlipak MG, Fried LF, Newman AB, Katz R, et al. Kidney function, electrocardiographic findings, and cardiovascular events among older adults. Clin J Am Soc Nephrol. 2007;2:501–8.
Dobre M, Brateanu A, Rashidi A, Rahman M. Electrocardiogram abnormalities and cardiovascular mortality in elderly patients with CKD. Clin J Am Soc Nephrol. 2012;7:949–56.
Gouda P, Brown P, Rowe BH, McAlister FA, Ezekowitz JA. Insights into the importance of the electrocardiogram in patients with acute heart failure. Eur J Heart Fail. 2016;18:1032–40.
Park SJ, On YK, Byeon K, Kim JS, Choi JO, Choi DJ, et al. Short- and long-term outcomes depending on electrical dyssynchrony markers in patients presenting with acute heart failure: clinical implication of the first-degree atrioventricular block and QRS prolongation from the Korean Heart Failure registry. Am Heart J. 2013;165:57–64.
Kannel WB, Abbott RD. A prognostic comparison of asymptomatic left ventricular hypertrophy and unrecognized myocardial infarction: the Framingham Study. Am Heart J. 1986;111:391–7.
Rautaharju PM, Kooperberg C, Larson JC, LaCroix A. Electrocardiographic abnormalities that predict coronary heart disease events and mortality in postmenopausal women: the Women’s Health Initiative. Circulation. 2006;31:473–80.
Cheng S, Keyes MJ, Larson MG, McCabe EL, Newton-Cheh C, Levy D, et al. Long-term outcomes in individuals with prolonged PR interval or first-degree atrioventricular block. JAMA. 2009;301:2571–7.
Genovesi S, Rossi E, Nava M, Riva H, De Franceschi S, Fabbrini P, et al. A case series of chronic haemodialysis patients: mortality, sudden death, and QT interval. Europace. 2013;15:1025–33.
Silva RT, Martinelli Filho M, Peixoto Gde L, Lima JJ, Siqueira SF, Costa R, et al. Predictors of Arrhythmic events detected by implantable loop recorders in renal transplant candidates. Arq Bras Cardiol. 2015;105:493–502.
Krane V, Heinrich F, Meesmann M, Olschewski M, Lilienthal J, Angermann C, et al. German diabetes and dialysis study investigators: electrocardiography and outcome in patients with diabetes mellitus on maintenance hemodialysis. Clin J Am Soc Nephrol. 2009;4:394–400.
Poulikakos D, Malik M. Challenges of ECG monitoring and ECG interpretation in dialysis units. J Electrocardiol. 2016;49:855–9.
Madias JE, Narayan V. Augmentation of the amplitude of electrocardiographic QRS complexes immediately after hemodialysis: a study of 26 hemodialysis sessions of a single patient, aided by measurements of resistance, reactance, and impedance. J Electrocardiol. 2003;36:263–71.
Hancock EW, Deal BJ, Mirvis DM, Okin P, Kligfield P, Gettes LS, et al. American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; American College of Cardiology Foundation; Heart Rhythm Society: AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part V: electrocardiogram changes associated with cardiac chamber hypertrophy: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol. 2009;53:992–1002.
Oka T, Hamano T, Sakaguchi Y, Yamaguchi S, Kubota K, Senda M, et al. Proteinuria-associated renal magnesium wasting leads to hypomagnesemia: a common electrolyte abnormality in chronic kidney disease. Nephrol Dial Transpl. 2019;34:1154–62.
Surawicz B. Relationship between electrocardiogram and electrolytes. Am Heart J. 1967;73:814–34.
Yamaguchi S, Hamano T, Doi Y, Oka T, Kajimoto S, Kubota K, et al. Hidden hypocalcemia as a risk factor for cardiovascular events and all-cause mortality among patients undergoing incident hemodialysis. Sci Rep. 2020;10:4418.
Heemskerk CPM, Pereboom M, van Stralen K, Berger FA, van den Bemt PMLA, Kuijper AFM, et al. Risk factors for QTc interval prolongation. Eur J Clin Pharm. 2018;74:183–91.
Noordam R, Young JW, Salman R, Kanters KJ, van den Berg ME, van Heemst D, et al. Effects of Calcium. Magnes, Potassium Concentrations Ventricular Repolarization Unselected Individ J Am Coll Caridol. 2019;73:3118–31.
Shimaoka T, Wang Y, Morishima M, Miyamoto S, Ono K. Magnesium deficiency causes transcriptional downregulation of Kir2.1 and Kv4.2 channels in cardiomyocytes resulting in QT interval prolongation. Circ J. 2020;84:1244–53.
Tagawa M, Hamano T, Nishi H, Tsuchida K, Hanafusa N, Fukatsu A, et al. Mineral metabolism markers are associated with myocardial infarction and hemorrhagic stroke but not ischemic stroke in hemodialysis patients: a longitudinal study. PLoS One. 2014;9:e11468.
Liao F, Folsom AR, Brancati FL. Is low magnesium concentration a risk factor for coronary heart disease? The atherosclerosis risk in communities (ARIC) Study. Am Heart J. 1998;136:480–90.
Bazett HC. An analysis of time relations of electrocardiogram. Heart. 1920;7:353–70.
Sokolow M, Lyon TP. The ventricular complex in left ventricular hypertrophy as obtained by unipolar precordial and limb leads. Am Heart J. 1949;37:161–86.
Wheeler DC, London GM, Parfrey PS, Block GA, Correa-Rotter R, Dehmel B, et al. Effects of cinacalcet on atherosclerotic and nonatherosclerotic cardiovascular events in patients receiving hemodialysis: the evaluation of cinacalcet HCL therapy to lower cardiovascular events (EVOLVE) Trial. J Am Heart Assoc. 2014;3:e001363.
United States Renal Data System. 2018 USRDS annual data report: Epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD, 2018.
Schumacher K, Dagres N, Hindricks G, Husser D, Bollmann A, Kornej J. Characteristics of PR interval as predictor for atrial fibrillation: association with biomarkers and outcomes. Clin Res Cardiol. 2017;106:767–75.
Iwasaki YK, Nishida K, Kato T, Nattel S. Atrial fibrillation pathophysiology: implications for management. Circulation. 2011;124:2264–74.
Salden FCWM, Kutyifa V, Stockburger M, Prinzen FW, Vernooy K. Atrioventricular dromotropathy: evidence for a distinctive entity in heart failure with prolonged PR interval? Europace. 2018;20:1067–77.
Namdar M, Biaggi P, Stähli B, Bütler B, Casado-Arroyo R, Ricciardi D, et al. A novel electrocardiographic index for the diagnosis of diastolic dysfunction. PLoS One. 2013;8:e79152.
Kane GC, Karon BL, Mahoney DW, Redfield MM, Roger VL, Burnett JC Jr, et al. Progression of left ventricular diastolic dysfunction and risk of heart failure. JAMA. 2011;306:856–63.
Sandhu R, Bahler RC. Prevalence of QRS prolongation in a community hospital cohort of patients with heart failure and its relation to left ventricular systolic dysfunction. Am J Cardiol. 2004;93:244–6.
Shamim W, Yousufuddin M, Cicoria M, Gibson DG, Coats AJ, Henein MY. Incremental changes in QRS duration in serial ECGs over time identify high risk elderly patients with heart failure. Heart. 2002;88:47–51.
Okin PM, Devereux RB, Kjeldsen SE, Edelman JM, Dahlöf B. Incidence of heart failure in relation to QRS duration during antihypertensive therapy: the LIFE study. J Hypertens. 2009;27:2221–7.
El-Chami MF, Brancato C, Langberg J, Delurgio DB, Bush H, Brosius L, et al. QRS duration is associated with atrial fibrillation in patients with left ventricular dysfunction. Clin Cardiol. 2010;33:132–83.
Quinn SJ, Thomsen ARB, Pang JL, Kantham L, Bräuner-Osborne H, Pollak M, et al. Interactions between calcium and phosphorus in the regulation of the production of fibroblast growth factor 23 in vivo. Am J Physiol Endocrinol Metab. 2013;304:E310–320.
Huang SY, Chen YC, Kao YH, Hsieh MH, Lin YK, Chung CC, et al. Fibroblast growth factor 23 dysregulates late sodium current and calcium homeostasis with enhanced arrhythmogenesis in pulmonary vein cardiomyocytes. Oncotarget. 2016;7:69231–42.
Mathew JS, Sachs MC, Katz R, Patton KK, Heckbert SR, Hoofnagle AN, et al. Fibroblast growth factor-23 and incident atrial fibrillation: the Multi-Ethnic Study of Atherosclerosis (MESA) and the Cardiovascular Health Study (CHS). Circulation. 2014;130:298–307.
Covic AC, Buimistriuc LD, Green D, Stefan A, Badarau S, Kalra PA. The prognostic value of electrocardiographic estimation of left ventricular hypertrophy in dialysis patients. Ann Noninvasive Electrocardiol. 2013;18:188–98.
Cice G, Di Benedetto A, D’Andrea A, D’Isa S, De Gregorio P, Marcelli D, et al. Heart rate as independent prognostic factor for mortality in normotensive hemodialysed patients. J Nephrol. 2008;21:704–12.
Kim SJ, Oh HJ, Yoo DE, Shin DH, Lee MJ, Kim HR, et al. Electrocardiographic left ventricular hypertrophy and outcome in hemodialysis patients. PLoS One. 2012;7:e35534.
Sakaguchi Y, Hamano T, Obi Y, Monden C, Oka T, Yamaguchi S, Matsui I, et al. A randomized trial of magnesium oxide and oral carbon adsorbent for coronary artery calcification in predialysis CKD. J Am Soc Nephrol. 2019;30:1073–85.
Pearson PJ, Evora PR, Seccombe JF, Schaff HV. Hypomagnesemia inhibits nitric oxide release from coronary endothelium: protective role of magnesium infusion after cardiac operations. Ann Thorac Surg. 1998;65:967–72.
Shechter M, Merz CN, Paul-Labrador M, Meisel SR, Rude RK, Molloy MD, et al. Oral magnesium supplementation inhibits platelet-dependent thrombosis in patients with coronary artery disease. Am J Cardiol. 1999;84:152–6.
Tereshchenko LG, Kim ED, Oehler A, Meoni LA, Ghafoori E, Rami T, et al. Electrophysiologic substrate and risk of mortality in incident hemodialysis. J Am Soc Nephrol. 2016;27:3413–20.
Acknowledgements
We thank Dr. Shihomi Maeda, Dr. Fumiyuki Morioka, Dr. Syou Matsui, Dr. Sayaka Katou, Dr. Akane Ito, Dr. Eriko Iwahashi, (JCHO Osaka Hospital), Dr. Kiyoshi Shozu (Aino Hospital), Dr. Yutaro Azuma (Azuma Dialysis Clinic), Dr. Akira Wada (Azuma Renal Clinic), Dr. Jong Il Kim (Chibune Kidney and Dialysis Clinic), Dr. Sachiko Yamada (Saiseikai Ibaraki Hospital), Dr. Yasuhiro Enomoto (Enomoto Clinic), Dr. Ryoichi Miyazaki (Fujita Memorial Hospital), Dr. Yuji Murata (Harbor-Town Clinic), Dr. Toshiaki Hirabayashi (Heimeikai Clinic), Dr. Fukuhara Isao (Heisei Urologic Clinic), Dr. Shigeru Yamaguchi (Hokusetsu General Hospital), Dr. Takashi Tomokuni (Ibaraki Iseikai Hospital), Dr. Toyohumi Fukuda (Ibuki Clinic), Dr. Kazuyuki Hayashi (Ikeda Municipal Hospital), Dr. Nobuyuki Imai (Imai Naika Clinic), Dr. Yasuji Kuroda (Inadera Dialysis Clinic), Dr. Yuko Fujiwara, Dr. Yoshihiro Tsujimoto (Inoue Hospital), Dr. Takahisa Hotta (Iseikai Fuzoku Clinic), Dr. Takahiro Fujii (Itami Bara Clinic), Dr. Tomoko Yokokawa (Itami Gardens Clinic), Dr. Kazuo Yasuda (Jinyuukai Yasuda Clinic), Dr. Masanori Joko (Joko Clinic), Dr. Yukiomi Namba (Jouto Iseikai Clinic), Dr. Jyunya Takahara (Takahara Clinic), Dr. Mayumi Sakurai (Jyurakukai Clinic), Dr. Masato Baden (Kaiseikai Takarazuka Hospital), Dr. Hisato Nakamori (Katano Hospital), Dr. Hironori Kawamura (Kawamura Clinic), Dr. Chikako Monden (Kisei Hospital), Dr. Ken Kishida (Kishida Clinic), Dr. Eikun Maruyama (Kitagawa Clinic), Dr. Fumi Sato (KKR Otemae Hospital), Dr. Shigeru Hanada (Koseikai Taisei Clinic), Dr. Akihisa Kuki (Kuki Clinic), Dr. Takayuki Okamoto (Kyoritsu Hospital), Dr. Mitsuru Kubo (Kyowakai Hospital), Dr. Haruki Fuse (Maizuru Kyosai Hospital), Dr. Yutaka Matsui (Matsui Dietary & Dementia Clinic), Dr. Noriko Mikami (Mikami Clinic), Dr. Hiroshi Miyao (Miyo Urology Clinic), Dr. Mutsuko Mori (Mori Clinic), Dr. Rumi Sakai (Motoyama Sakai Rumi Clinic), Dr. Shunro Agata (Nagahara Hospital), Dr. Takanori Nagai (Nagai Clinic), Dr. Hideyuki Nakagawa (Nakagawa Clinic), Dr. Masashi Shinozaki (Nakano Clinic), Dr. Yasuo Ohyama (Neyagawa Keijinkai Clinic), Dr. Kanji Nozaki (Nozaki Clinic), Dr. Tetsuya Kaneko (NTT West Osaka Hospital), Dr. Takeshi Omichi (Ohmichi Clinic), Dr. Motohiro Okamura (Okamuraiin Jin Clinic), Dr. Akira Okasho (Okasho Urology Clinic), Dr. Kiyoshi Goto (Ono-Naika Clinic), Dr. Hidekazu Shimizu (Onoyama Clinic), Dr. Masahiro Kyo (Osaka Umeda Iseikai Dialysis Clinic), Dr. Nobuhiko Narukawa (Sakuragaoka Hospital), Dr. Kei Hirai (Sanko Clinic), Dr. Kazuhisa Yamamoto (Sanko Hospital), Dr. Satoshi Sasaki (Sasaki Medical Clinic), Dr. Masugi Satoh (Satoh Clinic Kuzuha), Dr. Daisuke Nakagawa (Second Rokushima Clinic), Dr. Hiroyuki Araki (Settsu Iseikai Hospital), Dr. Shintaro Yamanaka (Shingu Municipal Medical Center), Dr. Atsushi Yokota (Shin-Osaka Iseikai Clinic), Dr. Ken Takahara (Sone Hospital), Dr. Zenzo Fujii (St. Hill Hospital), Dr. Jun Matsuda (Suita City Hospital), Dr. Yoshinori Sai (Suminodo Clinic), Dr. Kanji Yoshida (Suzawa Clinic), Dr. Tadashi Hatayama (Neyagawaikunobyouinn), Dr. Yukito Kokado, Dr. Tohru Takahashi (Takahashi Clinic), Dr. Kazuyuki Takahashi (Takahashi Kazuyuki Clinic), Dr. Yoshiaki Hirosue (Takasago Municipal Hospital), Dr. K.Y. (Tanaka Clinic), Dr. Masatou Tanaka (Tanaka Urology Clinic Dialysis Center Furuedai), Dr. Hiroko Tokizane (Tokizane Clinic), Dr. Keiji Isshiki (Tomita Clinic), Dr. Jun Tomita (Toyonaka Watanabe Clinic, Kansai Medical Hospital), Dr. Yumiko Honda (Toyonakamidorigaoka Hospital), Dr. Takahide Sugiyama (Tsukuno Fujii Clinic), Dr. Akira Moriguchi (Umeda-Higashi Blood Purification Clinic), Dr. Takatoshi Yamamoto (Yamamoto Clinic), Dr. Masaru Shin (Yao Tokushukai General Hospital), and Dr. Toru Yoshie (Yoshie Hospital) for their assistance in collecting data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Yamaguchi, S., Hamano, T., Oka, T. et al. Electrocardiogram findings at the initiation of hemodialysis and types of subsequent cardiovascular events. Hypertens Res 44, 571–580 (2021). https://doi.org/10.1038/s41440-020-00592-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-020-00592-z