Abstract
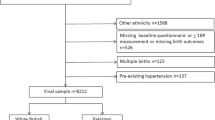
This study aimed to explore the association of systolic blood pressure (SBP) trajectories of pregnant women with the risk of adverse outcomes of pregnant women and their fetuses. A register-based cohort of 63,724 pregnant women and their fetuses from January 2013 to December 2017 was investigated. Demographic characteristics, history of disease and family history of disease for pregnant women and perinatal outcomes were recorded, and blood pressure was measured during the whole pregnancy. SBP trajectories were estimated with latent mixture modeling by Proc Traj in SAS using SBP data from the first antenatal care appointment (8–14 weeks), the highest SBP before admission, the admission SBP and the SBP at 2 h postpartum. A censored normal model (CNORM) was considered appropriate, and model fit was assessed using the Bayesian information criterion (BIC). A logistic regression model was used to examine the association between SBP trajectories and the risk of adverse perinatal outcomes. Four distinct SBP trajectory patterns over the pregnancy period were identified and were labeled as low-stable, moderate-stable, high-decreasing and moderate-increasing. Three maternal and three fetal adverse outcomes were selected as the main outcome measures. After adjusting for confounding factors, compared with pregnant women with the low-stable pattern, those with the high-decreasing pattern had a higher risk of developing poor growth outcomes of fetuses, while those with the moderate-increasing pattern had higher risks of developing both adverse maternal and fetal outcomes. Our study results suggest that pregnant women should pay attention to the control of blood pressure throughout pregnancy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Lo JO, Mission JF, Caughey AB. Hypertensive disease of pregnancy and maternal mortality. Curr Opin Obstet Gynecol. 2013;25:124–32.
Gulmezoglu AM, Say L, Betran AP, Villar J, Piaggio G. WHO systematic review of maternal mortality and morbidity: methodological issues and challenges. BMC Med Res Methodol. 2004;4:16.
Nzelu D, Dumitrascu-Biris D, Hunt KF, Cordina M, Kametas NA. Pregnancy outcomes in women with previous gestational hypertension: a cohort study to guide counselling and management. Pregnancy Hypertens. 2018;12:194–200.
Say L, Chou D, Gemmill A, Tunçalp Ö, Moller A-B, Daniels J, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2:e323–33.
Ananth CV, Hansen AV, Williams MA, Nybo Andersen AM. Cardiovascular disease in relation to placental abruption: a population-based cohort study from Denmark. Paediatr Perinat Epidemiol. 2017;31:209–18.
Soh MC, Dib F, Nelson-Piercy C, Westgren M, McCowan L, Pasupathy D. Maternal-placental syndrome and future risk of accelerated cardiovascular events in Parous Swedish women with systemic lupus erythematosus—a population-based retrospective cohort study with time-to-event analysis. Rheumatology. 2016;55:1235–42.
Moussa HN, Hosseini Nasab S, Amro FH, Hoayek J, Haidar ZA, Blackwell SC, et al. Adverse pregnancy outcomes in deliveries prior to, at and beyond 39 weeks; low- and high-risk women. J Matern Fetal Neonatal Med. 2018;31:2545–9.
Boriboonhirunsarn D, Pradyachaipimol A, Viriyapak B. Incidence of superimposed preeclampsia among pregnant Asian women with chronic hypertension. Hypertens Pregnancy. 2017;36:226–31.
Shahul S, Ramadan H, Mueller A, Nizamuddin J, Nasim R, Lopes Perdigao J, et al. Abnormal mid-trimester cardiac strain in women with chronic hypertension predates superimposed preeclampsia. Pregnancy Hypertens. 2017;10:251–5.
Ruiter L, Ravelli AC, de Graaf IM, Mol BW, Pajkrt E. Incidence and recurrence rate of placental abruption: a longitudinal linked national cohort study in the Netherlands. Am J Obstet Gynecol. 2015;213:573 e571–578.
Downes KL, Shenassa ED, Grantz KL. Neonatal outcomes associated with placental abruption. Am J Epidemiol. 2017;186:1319–28.
Starr MC, Hingorani SR. Prematurity and future kidney health: the growing risk of chronic kidney disease. Curr Opin Pediatr. 2018;30:228–35.
Huckstep OJ, Williamson W, Telles F, Burchert H, Bertagnolli M, Herdman C, et al. Physiological stress elicits impaired left ventricular function in preterm-born adults. J Am Coll Cardiol. 2018;71:1347–56.
Macdonald-Wallis C, Tilling K, Fraser A, Nelson SM, Lawlor DA. Associations of blood pressure change in pregnancy with fetal growth and gestational age at delivery: findings from a prospective cohort. Hypertension. 2014;64:36–44.
Gaillard R, Bakker R, Willemsen SP, Hofman A, Steegers EA, Jaddoe VW. Blood pressure tracking during pregnancy and the risk of gestational hypertensive disorders: the Generation R Study. Eur Heart J. 2011;32:3088–97.
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr., et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 2003;42:1206–52.
Macdonald-Wallis C, Silverwood RJ, Fraser A, Nelson SM, Tilling K, Lawlor DA, et al. Gestational-age-specific reference ranges for blood pressure in pregnancy: findings from a prospective cohort. J Hypertens. 2015;33:96–105.
Steer PJ, Little MP, Kold-Jensen T, Chapple J, Elliott P. Maternal blood pressure in pregnancy, birth weight, and perinatal mortality in first births: prospective study. BMJ. 2004;329:1312.
Pressure NHBPEPWGoHB, Pregnancy i. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000;183:s1–s22.
Ananth CV, Keyes KM, Wapner RJ. Pre-eclampsia rates in the United States, 1980-2010: age-period-cohort analysis. BMJ. 2013;347:f6564.
Youngstrom M, Tita A, Grant J, Szychowski JM, Harper LM. Perinatal outcomes in women with a history of chronic hypertension but normal blood pressures before 20 weeks of gestation. Obstet Gynecol. 2018;131:827–34.
Viteri OA, England JA, Alrais MA, Lash KA, Villegas MI, Ashimi Balogun OA, et al. Association of nonsteroidal antiinflammatory drugs and postpartum hypertension in women with preeclampsia with severe features. Obstet Gynecol. 2017;130:830–5.
Chappell LC, Enye S, Seed P, Briley AL, Poston L, Shennan AH. Adverse perinatal outcomes and risk factors for preeclampsia in women with chronic hypertension: a prospective study. Hypertension.2008;51:1002–9.
pregnancy ACoOaGHi. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122:1122–31.
Bramham K, Parnell B, Nelson-Piercy C, Seed PT, Poston L, Chappell LC. Chronic hypertension and pregnancy outcomes: systematic review and meta-analysis. BMJ. 2014;348:g2301.
Sibai BM, Koch MA, Freire S, Pinto e Silva JL, MVC Rudge, Martins-Costa S, et al. The impact of prior preeclampsia on the risk of superimposed preeclampsia and other adverse pregnancy outcomes in patients with chronic hypertension. Am J Obstet Gynecol. 2011;204:345.e341–345.e346.
Lane-Cordova AD, Gunderson EP, Carnethon MR, Catov JM, Reiner AP, Lewis CE, et al. Pre-pregnancy endothelial dysfunction and birth outcomes: The Coronary Artery Risk Development in Young Adults (CARDIA) Study. Hypertens Res. 2018;41:282–9.
Tay J, Foo L, Masini G, Bennett PR, McEniery CM, Wilkinson IB, et al. Early and late preeclampsia are characterized by high cardiac output, but in the presence of fetal growth restriction, cardiac output is low: insights from a prospective study. Am J Obstet Gynecol. 2018;218:517.e511–517.e512.
Ohkuchi A, Iwasaki R, Ojima T, Matsubara S, Sato I, Suzuki M, et al. Increase in systolic blood pressure of > or = 30 mm Hg and/or diastolic blood pressure of > or = 15 mm Hg during pregnancy: is it pathologic? Hypertens Pregnancy. 2003;22:275–85.
Acknowledgements
We thank all members of our study in the Maternal and Child Health Bureau of Kunshan and School of Public Health, Medical College of Soochow University.
Funding
This work was supported by the Natural Science Foundation of Jiangsu Province (Project Number: BK20181438).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Guo, Q., Feng, P., Yu, Q. et al. Associations of systolic blood pressure trajectories during pregnancy and risk of adverse perinatal outcomes. Hypertens Res 43, 227–234 (2020). https://doi.org/10.1038/s41440-019-0350-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-019-0350-3
Keywords
This article is cited by
-
Hypertensive disorders of pregnancy: definition, management, and out-of-office blood pressure measurement
Hypertension Research (2022)
-
Gestational systolic blood pressure trajectories and risk of adverse maternal and perinatal outcomes in Chinese women
BMC Pregnancy and Childbirth (2021)
-
Placental abruption in each hypertensive disorders of pregnancy phenotype: a retrospective cohort study using a national inpatient database in Japan
Hypertension Research (2021)