Abstract
We report a 1-year-old girl with congenital stromal corneal dystrophy confirmed by genetic analysis. The ocular phenotype included diffuse opacity over the corneal stroma bilaterally. We performed a genetic analysis to provide counseling to the parents regarding the recurrence rate. Whole exome sequencing was performed on her and her parents, and a novel de novo variant, NM_001920.5: c.953del, p.(Asn318Thrfs*10), in the DCN gene was identified in the patient.
Similar content being viewed by others
Congenital stromal corneal dystrophy (CSCD) is an exceedingly rare disease1,2,3. It is an autosomal dominant trait4 and occurs bilaterally5. The primary corneal alterations are not associated with previous inflammation or secondary to a systemic disease1.
CSCD is caused by mutations in the decorin (DCN) gene on the long arm of chromosome 123,5,6. Decorin is a member of the small leucine-rich proteoglycan gene family with a variety of binding properties with matrix structural components, including collagens and growth factors6,7,8,9. Decorin is distributed throughout the body, including the cornea, respiratory system, pancreas, muscle, skin, and ovary. However, no reports have described systemic diseases caused by DCN mutations. The only phenotype associated with a mutation in the DCN gene is CSCD9.
Decorin has a role in the assembly of corneal collagen fibers and supports corneal transparency3,10,11,12. In patients with CSCD with DCN mutations, transmission electron microscopy has confirmed disruption of collagen fibers, i.e., separation of the normal lamella of the collagen fibrils by abnormal collagen filaments1,2,5,13.
Five families with CSCD have been reported2,5, and we recently identified a Japanese child with CSCD as the sixth CSCD pedigree in the world. We also identified a novel mutation in the DCN gene. We present the detailed clinical profile of the case with CSCD confirmed by genetic analysis.
The proband was a 1-year 7-month-old girl referred to the Division of Ophthalmology, National Center for Child Health and Development for further examinations and treatment for bilateral corneal opacities. She had no medical history, and her parents were not consanguineous.
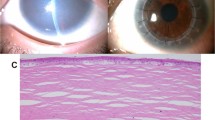
At the first visit, she showed no nystagmus and maintained orthophoria without limitations of extraocular movements. Fixation and following behavior of each eye were observed; the visual acuity (VA) using the Teller Acuity Cards II was 20/380 bilaterally. Slit-lamp microscopy showed diffuse opacity bilaterally. The anterior chamber depth was normal, and the corneal diameters were equally normal in both eyes (10.5 mm vertically and 11.0 mm horizontally). The central corneal thickness was 620 μm in the right eye and 640 μm in the left eye (Fig. 1A). The respective intraocular pressures (Eye Care Hand-held Tonometer; Icare TA01i, Helsinki, Finland) were 18 mmHg and 14 mmHg. We conducted a detailed ophthalmic examination with the child under general anesthesia; swept-source optical coherence tomography (SS-OCT) (Topcon, Tokyo, Japan) showed hyperreflective zones consistent with the corneal opacity involving the entire stroma in both eyes (Fig. 1B). Ophthalmoscopy showed no apparent abnormalities in either fundus but poor translucency due to corneal opacity (Fig. 1C). Electroretinography (ERG) (Neuropack, Nihon Koden, Tokyo, Japan) based on the International Society for Clinical Electrophysiology of Vision protocol14 showed normal responses (Fig. 1D).
Both eyes had similar findings. A The anterior segment has diffuse corneal opacity. B An OCT image shows hyperreflective zones consistent with corneal opacity involving the entire stroma in both eyes. C A fundus photograph shows poor translucency due to corneal opacity, but no abnormalities are seen. D The full-field ERG in the left eye shows normal responses of the combined rod-cone, rods, and cones. The calibrations are shown for each stimulation.
We performed a systemic investigation to eliminate other diseases that cause diffuse corneal opacity in infants, e.g., congenital metabolic disorders and congenital infection. Laboratory analyses of the serum antibodies, treponema pallidum, and TORCH antibodies (IgG, IgM) were negative. No abnormal findings were detected in a lysosomal enzyme activity assay or urinary mucopolysaccharide analysis. Imaging, i.e., electrocardiography, echocardiography, abdominal ultrasonography, and bone X-rays, were negative. We clinically diagnosed congenital corneal dystrophy, although the type was not determined.
Because the parents inquired about potential recurrence in a second child, we performed a genetic analysis. Since our patient was born to healthy parents, the inheritance was suspected to be autosomal recessive or dominant inheritance of a de novo mutation.
To investigate the genetic background, we performed whole exome sequencing (WES) in three family members and analyzed them via autosomal recessive and dominant models. Genomic DNA was extracted from peripheral blood using standard procedures. Exome data processing, variant calling, and variant annotation were performed as previously described15 using human GRCh38 as the reference genome. To identify disease-causing variants, we focused on nonsynonymous variants and splice-site variants, which were within 10 base pairs (bp) of the exon‒intron boundaries (±10 bp), and excluded synonymous and noncoding exonic variants from the analysis. We treated common genetic variants (allele frequency >0.01 for recessive variants; >0.001 for dominant variants) in any of the ethnic subgroups found in the following single nucleotide polymorphism databases and in-house exome data (n = 218) as putative nonpathogenic sequence alterations using the Genome Aggregation Database (https://gnomad.broadinstitute.org/) and Tohoku Medical Megabank Organization database (4.7KJPN, https://jmorp.megabank.tohoku.ac.jp/).
Potential pathogenic variants detected by WES were validated using Sanger sequencing according to the standard protocol16. Sanger sequencing segregation analyses were performed in three family members to investigate cosegregation of the potentially pathogenic variants. The following primer set for the DCN gene was used: forward primer 5’-AAGGGCCTCAACATATTTAGAGAAT-3’ and reverse primer 5’-TGCAGTTAGGTTTCCAGTATCTAGC-3’.
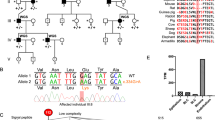
Based on the WES data of the affected child (II-1) and parents (I-1 and I-2), we identified a novel heterozygous variant in the patient, NM_001920.5: c.953del, p.(Asn318Thrfs*10), in the DCN gene that was de novo in origin.
Since DCN was reported as the causative gene for CSCD1,2,3,5,6,9, we further validated the DCN variant by Sanger sequencing and cosegregation (Fig. 2). Sanger sequencing indicated that the variant was not in any database described in the methods, indicating that the variant was extremely rare. As a result of the mutation, the asparagine at position 318 becomes threonine. Subsequently, the amino acid sequence changed, and the following 10th codon became a termination codon. This suggests that p.(Asn318Thrfs*10) is deleterious to the helical structure of the protein. This variant was classified as pathogenic17 (Supplementary Table 1).
After the definitive diagnosis was made, we performed genetic counseling for the parents. We explained that the recurrence rate in a second child would be generally considered to be low because we identified a de novo variant of the DCN gene. However, this could vary depending on gonadal mosaicism and postzygotic DNA18. Following genetic counseling, they gave birth to their second child.
The patient was followed up to 3 years of age. The corneal opacity remained unchanged, and the decimal VA was 0.03 bilaterally.
CSCD is a rare hereditary stromal corneal dystrophy caused by DCN mutations1,2,3,5,6 that has been reported in only five genetically confirmed pedigrees2,5. Detailed descriptions of genetic mutations and clinical characteristics of previous patients and the current patient are shown in Supplementary Table 2.
We documented a de novo novel mutation of the DCN gene, NM_001920.5: c.953del, p.(Asn318Thrfs*10), in a Japanese child with CSCD. Interestingly, in the previously reported families with CSCD, the DCN mutations were located only in exon 8 of the gene1,2,3,4,5, as in the current patient. The structural/functional effects of the mutation on the DCN protein classify it as a truncation mutation of the decorin protein, and the EAR repeat at the C-terminus region was shortened2,12. This might affect the collagen-decorin interaction. In a knockout mouse model of CSCD at the C-terminus region, separation of collagen lamellae was seen, and this was seen in CSCD patients9. In the model, the truncated DCN possibly had a dominant negative effect on wild-type decorin protein that interacted with keratocytes through the signaling receptor on the surface, which resulted in the downregulation of endogenous decorin, biglycan, lumican, and keratocan. Based on the American College of Medical Genetics and Genomics guidelines, the current frameshift mutation was considered pathogenic. This could be because the mutation was located between those of the Norwegian and Belgian families1,4, which induced truncation of decorin in the penultimate and longest leucine-rich repeats, known as the EAR repeat region.
The corneal abnormality appeared in infancy, with diffuse corneal opacity and corneal thickening. Although no nystagmus was observed, the measurable VA was 0.03 bilaterally. In a Norwegian report, corneal changes were seen in affected individuals during the first months after birth based on a family interview. The corneal thickness increased, and the VA varied from light perception to 0.6. In the Belgian families, diffuse corneal opacity was observed during early childhood, and the corneal thickness was normal13. Compared to that of the Norwegian and Belgian families1,13, the age of onset of the current case was comparable. As discussed previously, the current mutation was localized between those of the Norwegian and Belgian cases in the EAR region of decorin, and the phenotype of the current case might be consistent with those cases.
Most previous patients underwent penetrating keratoplasty1,2,3,5,13 between 1 and 44 years of age. The postoperative VA improved or was maintained in many cases, and a few required regrafting because of keratitis, perforation, and corneal opacities. Therefore, penetrating keratoplasty may also have provided good postoperative VA in the current case.
We performed prompt genetic studies that were useful to determine the diagnosis, causative gene, and hereditary form for genetic counseling regarding the birth of a second child and may contribute to treatment decisions.
HGV Database
The relevant data from this Data Report are hosted at the Human Genome Variation Database at https://doi.org/10.6084/m9.figshare.hgv.3283
References
Bredrup, C., Knappskog, P. M., Majewski, J., Rodahl, E. & Boman, H. Congenital stromal dystrophy of the cornea caused by a mutation in the decorin gene. Invest Ophthalmol. Vis. Sci. 46, 420–426 (2005).
Kim, J. H. et al. A novel mutation of the decorin gene identified in a Korean family with congenital hereditary stromal dystrophy. Cornea 30, 1473–1477 (2011).
Lee, J. H., Ki, C. S., Chung, E. S. & Chung, T. Y. A novel decorin gene mutation in congenital hereditary stromal dystrophy: a Korean family. Korean J. Ophthalmol. 26, 301–305 (2012).
Rodahl, E., Van Ginderdeuren, R., Knappskog, P. M., Bredrup, C. & Boman, H. A second decorin frame shift mutation in a family with congenital stromal corneal dystrophy. Am. J. Ophthalmol. 142, 520–521 (2006).
Jing, Y. et al. Novel decorin mutation in a Chinese family with congenital stromal corneal dystrophy. Cornea 33, 288–293 (2014).
Danielson, K. G. et al. The human decorin gene: intron-exon organization, discovery of two alternatively spliced exons in the 5’ untranslated region, and mapping of the gene to chromosome 12q23. Genomics 15, 146–160 (1993).
Chen, S. et al. Pathophysiological mechanisms of autosomal dominant congenital stromal corneal dystrophy: C-terminal-truncated decorin results in abnormal matrix assembly and altered expression of small leucine-rich proteoglycans. Am. J. Pathol. 179, 2409–2419 (2011).
Gubbiotti, M. A., Vallet, S. D., Ricard-Blum, S. & Lozzo, R. V. Decorin interacting network: a comprehensive analysis of decorin-binding partners and their versatile functions. Matrix Biol. 55, 7–21 (2016).
Seidler, D. G. The galactosaminoglycan-containing decorin and its impact on diseases. Curr. Opin. Struct. Biol. 22, 578–582 (2012).
Iozzo, R. V. The biology of the small leucine-rich proteoglycans. Functional network of interactive proteins. J. Biol. Chem. 274, 18843–18846 (1999).
Michelacci, Y. M. Collagens and proteoglycans of the corneal extracellular matrix. Braz. J. Med Biol. Res. 36, 1037–1046 (2003).
Chen, S., Sun, M., Iozzo, R. V., Kao, W. W. & Birk, D. E. Intracellularly-retained decorin lacking the C-terminal ear repeat causes ER stress: a cell-based etiological mechanism for congenital stromal corneal dystrophy. Am. J. Pathol. 183, 247–256 (2013).
Van Ginderdeuren, R., De Vos, R., Casteels, I. & Foets, B. Report of a new family with dominant congenital heredity stromal dystrophy of the cornea. Cornea 21, 118–120 (2002).
McCulloch, D. L. et al. ISCEV Standard for full-field clinical electroretinography (2015 update). Doc. Ophthalmol. 130, 1–12 (2015).
Tachibana, N. et al. Maternal uniparental isodisomy of chromosome 4 and 8 in patients with retinal dystrophy: SRD5A3-congenital disorders of glycosylation and RP1-related retinitis pigmentosa. Genes (Basel). 16, 13 (2022).
Hosono, K. et al. Two novel mutations in the EYS gene are possible major causes of autosomal recessive retinitis pigmentosa in the Japanese population. PLoS One 7, e31036 (2012).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 17, 405–424 (2015).
Bernkopf, M. et al. Personalized recurrence risk assessment following the birth of a child with a pathogenic de novo mutation. Nat. Commun. 14, 853 (2023).
Acknowledgements
The authors thank the patient and parents who participated in this study. Part of this work was performed at the Advanced Research Facilities & Services, Preeminent Medical Photonics Education & Research Center, Hamamatsu University School of Medicine. This work was supported by the Health and Labor Sciences Research Grant of Research on Intractable Diseases (20FC1055 and 20FC1046 to S.N.) from the Ministry of Health, Labor and Welfare, Tokyo, Japan, a grant for the Initiative on Rare and Undiagnosed Diseases (JP22ek0109549 to Y.H.) from the Japan Agency for Medical Research and Development (AMED), and the Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research (JP20K09825 to Y.H., JP16K11284 to K.H.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statements
The Institutional Review Board of the National Center for Child Health and Development and Hamamatsu University School of Medicine approved this study, which adhered to the tenets of the Declaration of Helsinki. The parents of the current patient provided written informed consent for ophthalmic examinations under general anesthesia, genetic analysis including WES, and publication of this report.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morikawa, H., Nishina, S., Torii, K. et al. A pediatric case of congenital stromal corneal dystrophy caused by the novel variant c.953del of the DCN gene. Hum Genome Var 10, 9 (2023). https://doi.org/10.1038/s41439-023-00239-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41439-023-00239-8
This article is cited by
-
Autosomal dominant stromal corneal dystrophy associated with a SPARCL1 missense variant
European Journal of Human Genetics (2024)