Abstract
Chrysanthemum is frequently attacked by aphids, which greatly hinders the growth and ornamental value of this plant species. WRKY transcription factors play an important role in the response to biotic stresses such as pathogen and insect stresses. Here, chrysanthemum CmWRKY53 was cloned, and its expression was induced by aphid infestation. To verify the role of CmWRKY53 in resistance to aphids, CmWRKY53 transgenic chrysanthemum was generated. CmWRKY53 was found to mediate the susceptibility of chrysanthemum to aphids. The expression levels of secondary metabolite biosynthesis genes, such as peroxidase- and polyphenol oxidase-encoding genes, decreased in CmWRKY53-overexpressing (CmWRKY53-Oe) plants but dramatically increased in chimeric dominant repressor (CmWRKY53-SRDX) plants, suggesting that CmWRKY53 contributes to the susceptibility of chrysanthemum to aphids, possibly due to its role in the regulation of secondary metabolites.
Similar content being viewed by others
Introduction
Aphids are a major group of crop pests that cause severe damage to plants by consuming nutrients from phloem sap1. In addition to obtaining nutrients from the phloem, they are also vectors for numerous viruses2. Aphids have complex life cycles and produce offspring via cyclical parthenogenesis (CP), making it difficult to control aphids in plants3. Plants can employ direct or indirect defenses against herbivore attack. With respect to direct defense responses, plants produce different chemical components, such as phenolics, alkaloids and lignin, to deter herbivores or hinder the growth, development and reproduction of insects4. Phenolics, alkaloids and lignin are plant secondary metabolites that confer insect defense to plants5. In narrow-leafed lupin, quinolizidine alkaloid biosynthesis is altered in response to aphid predation6. Nicotine, an abundant secondary metabolite in tobacco, is a highly toxic chemical to the green peach aphid Myzus persicae7. Pyrrolizidine alkaloids have been used to deter general insect herbivores, but their relative effects differ between insect species8. Lignin is an aromatic heteropolymer synthesized via phenylpropanoid metabolism. Peroxidase (POD) enzymes catalyze the polymerization of monolignols, yielding lignin9. Lignin in turn contributes to the defense against aphids10. Overexpression of CmMYB19, a MYB transcription factor, promotes aphid resistance in chrysanthemum by regulating lignin biosynthesis11.
WRKY proteins are mainly characterized by one or two highly conserved WRKY domains (WRKYGQK) and a zinc finger motif in the C-terminal region12. The domains can specifically bind to W-box sequences ((T/C)TGAC(T/C)) in the promoter regions of target genes to regulate the expression of related genes13. Evidence indicates that PAMP-triggered immunity (PTI) and effector-triggered immunity (ETI) defense networks are positively or negatively regulated by WRKY transcription factors at the transcriptional level in response to pathogens14,15. In Arabidopsis, MPK3/MPK6 phosphorylate WRKY33 and activate the biosynthesis of phytoalexins in response to Botrytis cinerea infection16. AtWRKY33 can also regulate redox homeostasis, salicylic acid (SA) signaling, and ET/JA-mediated defense networks in response to Botrytis cinerea infection17. In addition, several WRKYs are involved in the resistance to herbivores. In Nicotiana attenuata, NaWRKY3 and NaWRKY6 mediate the resistance to Manduca sexta larvae in a JA-dependent manner18. Similarly, the tomato genes SlWRKY72a and SlWRKY72b mediate basal defense against potato aphids19. The expression of WRKY53 and WRKY DNA-binding activities are regulated by SA20, which provides a link to the subsequent pathogen response. WRKY53 plays roles in the biotic stress response and senescence. Arabidopsis WRKY53 mainly acts as a node in the multilayer regulation of the networks that control senescence and pathogen defense21. Furthermore, AtWRKY53 works in conjunction with WRKY46 and WRKY70 by mediating basal resistance against Pseudomonas syringae pathogens22. However, to our knowledge, there is no information available on the involvement of WRKY53 in the response to aphids in chrysanthemum.
Chrysanthemum (Chrysanthemum morifolium Ramat.) is one of four cut flower species throughout the world and is widely grown for ornamental, tea, and medicinal uses23,24,25. Chrysanthemum plants are frequently attacked by aphids (Macrosiphoniella sanborni) during their growth and development26, which causes significant economic losses. Transcriptomic changes in response to aphid infestation have been analyzed; where the expression of CmWRKY53 was significantly upregulated in chrysanthemum fed aphids (unpublished data), inferring that CmWRKY53 might be associated with the resistance of chrysanthemum to aphids. To test this hypothesis, we cloned the CmWRKY53 gene in chrysanthemum and studied its function by generating CmWRKY53 transgenic plants. We showed that CmWRKY53 mediates the sensitivity of chrysanthemum to aphids by regulating the synthesis of secondary metabolites, highlighting a novel chrysanthemum defense mechanism against aphids.
Material and methods
Plant materials and growth conditions
The chrysanthemum cultivar Jinba was obtained from the Chrysanthemum Germplasm Resource Preserving Center, Nanjing Agricultural University, Nanjing, China. Cuttings were transplanted into pots filled with a 1:2 (v/v) mixture of nutrient-enriched soil:vermiculite. The plants were grown in a greenhouse with a relative humidity of 70% and a 16 h/8 h (light/dark) photoperiod. The day/night temperature was 23 °C/18 °C, respectively, and the light intensity was 100 μmol m−2 s−1. Roots, stems and leaves along with tubular florets and ray florets were sampled for RNA extraction.
Isolation and sequence analysis of CmWRKY53
Total RNA was extracted from leaves using RNAiso reagent (TaKaRa, Tokyo Japan), and reverse transcription was performed using M-MLV reverse transcriptase (TaKaRa) according to the two-step protocol. WRKY53-F/R primers were used to clone the WRKY53 open reading frame by PCR. The PCR product was purified and cloned into pMD19-T (TaKaRa) for sequencing. Multiple sequence alignments of CmWRKY53 and its homologs were conducted by DNAMAN 6 software27. A phylogenetic tree was then constructed by MEGA 5 software using the neighbor-joining method. The polypeptide sequences of WRKY53 homologs were retrieved from the NCBI website (https://www.ncbi.nlm.nih.gov).
Expression profile of CmWRKY53 in response to aphid infestation
Wild-type plants at the 6-8-fully expanded leaf stage were used, and the uppermost leaves and stems of the top three nodes of the shoots were infested with twenty five-instar aphids. Aphid-infested leaves and stems were sampled at different time points after aphid infestation26, and non-aphid-infested plants (controls) were sampled at the same time points, with each time point including three individual plants.
Transactivation assays
The transactivation activity and transcriptional activation domains were analyzed as described previously28. The coding region and truncated sequences of CmWRKY53 were cloned into a pDEST-GBKT7 vector. The resulting pDEST-GBKT7-CmWRKY53 fusion plasmids were then transformed into cells of yeast strain Y2H Gold (Clontech, Mountain View, CA, USA). Strains introduced with plasmids of pCL1 and/or pDEST-GBKT7 served as positive and negative controls, respectively. The transformants were plated on SD/-Trp media, while the pCL1-harboring strain was grown on SD/-Leu media. Several colonies were then transferred to SD/-His/-Ade media and incubated at 30 °C for three days to determine the activation activity.
Quantitative real-time PCR (qRT-PCR)
The total RNA of different tissues of aphid-infested samples and control samples was extracted with RNAiso reagent (TaKaRa). cDNA was synthesized using a PrimeScript RT Reagent Kit (TaKaRa). Gene-specific primers were designed using Primer 5 (Supplementary Table S1), and CmEF1a was used as a reference gene29. The expression of CmWRKY53 was quantified using SYBR® Premix Ex TaqTM II (Tli RNaseH Plus) (TaKaRa). Three independent biological replicates were used, and the qRT-PCR data were calculated using the 2−ΔΔCt method30.
Subcellular localization of CmWRKY53
The ORF of CmWRKY53 was cloned into a pMDC43 overexpression vector, generating a construct with the ORF of CmWRKY53 fused to GFP in the N-terminal region. The plasmids were individually introduced into the Agrobacterium tumefaciens strain GV3101, 35S::GFP-CmWRKY53 and 35S::D53-RFP were transiently co-transformed into Nicotiana benthamiana leaves, and 35S::D53-RFP acted as a nuclear marker31. Expression of GFP and RFP was observed using a Zeiss LSM800 (Germany) laser scanning confocal microscope.
Generation of CmWRKY53 transgenic chrysanthemum
The ORF sequence of CmWRKY53 was first cloned into a pENTR1A gateway vector and then cloned into a pMDC43 overexpression vector, with CmWRKY53 driven by a 2 × 35S promoter. It has been demonstrated that the ERF-associated amphiphilic repression (EAR) motif functions as a repression domain in plants32, and fusion of the EAR repression domain (SRDX) to transcriptional activators is sufficient to convert them into strong repressors33,34. Plants expressing a chimeric repressor mimic plants with the corresponding loss-of-function alleles35,36. In the present study, CmWRKY53 presented transcriptional activity. To reduce the activities of endogenous and functionally redundant factors, CmWRKY53 was fused to SRDX to generate a dominant repressor, i.e., CmWRKY53-SRDX. The plasmids were introduced into the A. tumefaciens EHA105 strain, which was subsequently transformed into chrysanthemum via Agrobacterium-mediated transformation37. Overexpression of CmWRKY53 and the genotype of SRDX transgenic plants were verified by PCR analysis using vector primers and gene-specific primers, and the expression levels of CmWRKY53 were measured by qRT-PCR using CmWRKY53-RT-F/R primers. All the primers used are listed in Table S1.
Analysis of aphid resistance in CmWRKY53 transgenic chrysanthemum
Wild-type and transgenic plants at the 6-8-leaf stage were infested with 5 recently hatched aphid nymphs, and the total number of aphids on the plants was counted at 14 days after infestation. The multiplication rate (MR) and inhibition ratio (IR) parameters were used to quantify the plant resistance. The MR is defined as Ni/5, where Ni represents the total number of aphids on the plants, and the IR is defined 100(NW-NT)/NW, where NW and NT represent the mean numbers of aphids counted at 14 days after infestation on WT and transgenic plants, respectively37. Every infestation assay involved 10 plants of each line, and the assay included three biological replicates.
Transcriptome analysis of CmWRKY53 transgenic plants
CmWRKY53-overexpressing plants (CmWRKY53-Oe2), CmWRKY53-SRDX transgenic plants (CmWRKY53-SRDX2), and wild-type plants at the 6-8-leaf stage were used, the third leaf counted from the apex was collected, and nine individual plants were included in each replicate. The experiment included two biological replicates. Total RNA was extracted as mentioned above. A total of 1.5 μg of RNA of CmWRKY53-Oe2, CmWRKY53-SRDX2 or WT plants was used for RNA-seq (Novogene, China). The library preparation and generation for RNA sequencing followed the methods described by Qi38. The DESeq method39 was applied to analyze differential gene expression, and the screening threshold was padj <0.05.
Statistical analysis
SPSS 20.0 software was used to determine statistical significance, and the means and results of the WT and transgenic plants were expressed as the means ± standard errors. The least significant difference (LSD) multiple range test was used to analyze the results after one-way analysis of variance was performed.
Results
CmWRKY53 sequence characteristics
The sequence of CmWRKY53 (KM359566), which is 1,114 bp in length and contains an 816 bp open reading frame encoding a polypeptide of 271 amino acid residues, was isolated from Jinba chrysanthemum. Amino acid sequence comparisons showed that the typical WRKY domain is not present in CmWRKY53 (Fig. 1a). Phylogenetic analysis showed that the sequence of CmWRKY53 is most similar to that of Artemisia apiacea AaWRKY53 (Fig. 1b).
Subcellular localization of CmWRKY53
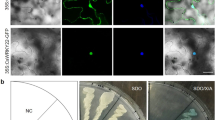
A. tumefaciens transformed with pMDC43-GFP-CmWRKY53 or a pMDC43-GFP empty vector was infiltrated into the leaves of N. benthamiana. GFP fluorescence was detected only in the nucleus of pMDC43-GFP-CmWRKY53 fusion protein-infiltrated tobacco cells, while GFP fluorescence was evenly distributed throughout the observed tobacco cells that were infiltrated with pMDC43-GFP (Fig. 2). Taken together, these results indicated that CmWRKY53 localized to the nucleus in vivo.
Transcriptional activity of CmWRKY53 and analysis of its transcriptional activation domain
The pDEST-GBKT7-CmWRKY53 plasmid was transformed into the yeast strain Y2H to determine the transcriptional activity of CmWRKY53. The Y2H Gold yeast transformed with pDEST-GBKT7-CmWRKY53 grew normally in double-deficient media, the negative control yeast transformed with pDEST-GBKT7 did not grow, and the positive control yeast transformed with pCL1 grew normally (Fig. 3). The results indicated that the whole CmWRKY53 protein is transcriptionally active. To identify the specific transactivation region of the CmWRKY53 protein, different truncated segments of CmWRKY53 from the C- and N-termini were cloned into pDEST-GBKT7. The results showed that the yeast strains containing the recombinant plasmids of the pDEST-GBKT7-CmWRKY53 (1-168 aa) and pDEST-GBKT7-CmWRKY53 (169-225 aa) fragments did not grow on the double-deficient media, but fragments of 1-225 aa and the remaining segments all grew normally (Fig. 3). Together, these results suggest that the activation domain is located within the 229-271 aa region at the C-terminus and that the intact protein of approximately 168 aa is important for activation activity.
Expression profiles of CmWRKY53 in different tissues in response to aphid infestation
The relative expression levels of CmWRKY53 were monitored in the root, stem, leaf and flower tissues of chrysanthemum. The results demonstrated that the relative expression levels were highest in the stems, followed by those in roots and leaves, while the disk florets presented the lowest levels of expression (Fig. 4a). The relative expression of CmWRKY53 increased by 1.28-fold at 9 h after aphid infestation and by 25.11-fold at 12 h after aphid infestation compared with that in the non-infested plants (Fig. 4b).
CmWRKY53 contributes to the aphids susceptibility of chrysanthemum
To determine the function of CmWRKY53, overexpression and gene-silenced transgenic chrysanthemum lines were obtained. Transgenic overexpression lines were verified by PCR amplification using a 35S forward primer and a reverse CmWRKY53 gene-specific primer, and CmWRKY53-SRDX lines were verified using a forward gene primer and an SRDX reverse primer. The expected bands were present for the transgenic lines and the positive control samples but not for WT or negative control samples (Fig. 5a). The transgenic plants were further verified using qRT-PCR (Fig. 5b). Aphid infestation assays showed that the number of aphids on the WT plants was lower than that on CmWRKY53-overexpressing plants but higher than that on CmWRKY53-SRDX gene-silenced transgenic plants (Fig. 6). The aphid MR on the WT plants was 28.92, while on the CmWRKY53-overexpressing lines, the rates were 34.06 and 33.48, and those in CmWRKY53-SRDX gene-silenced lines were 20.86 and 22.34. The IRs for CmWRKY53-overexpressing lines were −17.77% and −15.77%, whereas they were 27.87% and 22.75% for the SRDX lines (Table 1), suggesting that CmWRKY53 contributed to the sensitivity of chrysanthemum to aphids.
a PCR-based identification of pMDC43-CmWRKY53 and pSRDX-CmWRKY53 transgenic lines using vector- and gene-specific primers. For the positive control, the pMDC43-CmWRKY53 and pSRDX-CmWRKY53 plasmid were used as template, and for the negative control, no template was added. b Relative expression levels of CmWRKY53 in the transgenic plants
Expression profiles of genes involved in secondary metabolism in CmWRKY53 transgenic plants
Transcriptome analysis showed that there are 675 differentially expressed genes (DEGs) between CmWRKY53-overexpressing lines and wild-type plants and 404 DEGs between SRDX lines and wild-type plants. Compared with the wild-type plants, the CmWRKY53-overexpressing lines and gene-silenced lines shared 183 DEGs (Supplementary Fig. S1a). DEG_GO enrichment analysis showed that the expression levels of metabolism-related genes decreased in the overexpression plants compared to the wild-type plants, while they increased in the SRDX plants (Supplementary Fig. S1b). KEGG pathway enrichment analysis suggested that the DEGs whose expression changed in the transgenic plants are mainly related to the biosynthesis of isoquinoline alkaloids and phenylpropanoids (Table 2). The transcript levels of three polyphenol oxidase genes (CmPPO1, CmPPO2, CmPPO3) involved in the biosynthesis of isoquinoline alkaloids increased in pSRDX-CmWRKY53 plants but decreased in the overexpression plants compared with the wild-type plants (Table 2). KEGG pathway enrichment analysis demonstrated that the peroxidase 66 gene, which is involved in phenylpropanoid biosynthesis, increased in the SRDX line but decreased in CmWRKY53-overexpressing plants compared to WT plants (Table 2). The expression levels of the abovementioned DEGs were verified by qRT-PCR (Fig. 7).
Discussion
Structural characteristics and transcriptional activation activity of CmWRKY53
WRKY transcription factors are mainly characterized by the presence of the WRKYGQK domain in the N-terminus and a zinc finger structure in the C-terminus12. Despite WRKYGQK being a highly conserved region in the structural domain of WRKYs, it has been found that there are also variations in this sequence. These variations mainly occur for R, G, Q, and K, where the change of Q to E and K is the most common; for example, there are 19 variations in the rice WRKY domain structure: there are 7 of WRKYGEKs, 5 WRKYGKKs, and 1 each of WRICGQK, WRMCGQK, WKKYGQK, WIKYGQK, WKRYGQK, WSKYEQK and WRKYSEK. This genetic variation in the WRKY family in rice may due to the process of evolution40. AtWRKY53 and WRKY53 in wheat have conserved WRKYGQK domains41; however, a conserved WRKY domain was not observed in the WRKY53 protein from Artemisia annua (Fig. 1). Similarly, a conserved WRKY domain was not observed in chrysanthemum CmWRKY53, suggesting that CmWRKY53 might be evolutionally different from WRKY proteins in other species.
Our transactivation assay showed that the whole protein segment of CmWRKY53 is transcriptionally active. An N-terminal truncation assay showed that a protein segment ranging from 229 to 271 aa has transcriptional activity. Furthermore, a C-terminal truncation assay showed that an intact fragment of approximately 168 aa is important for transactivation activity (Fig. 3).
CmWRKY53 negatively regulates the resistance of chrysanthemum to aphids
Previous studies have shown that a few members of the WRKY family play important roles in aphid resistance. Silencing of SlWRKY70 attenuated the resistance of tomato to potato aphids (Macrosiphum euphorbiae), and SlWRKY70 was reported to be required for Mi-1-enhanced resistance to aphids42. In Arabidopsis, AtWRKY22 increases susceptibility to green peach aphids (Myzus persicae) via the suppression of salicylic acid signaling43. However, in wheat, silencing WRKY53 increased the susceptibility to aphids while decreasing the expression level of PAL, and PAL-silenced plants are also susceptible to aphids, which implies that these genes operate via the same defense mechanism41. In the present study, CmWRKY53 contributed to the susceptibility of chrysanthemum to aphids, which suggests that CmWRKY53 might regulate aphid resistance in a way different from that of WRKY53 in wheat.
CmWRKY53-altered aphid resistance is potentially related to secondary metabolism
WRKYs affect a number of secondary metabolites, including phenylpropanoids, alkaloids, and terpenes, by regulating genes involved in metabolite biosynthesis43,44,45,46. Brassica napus WRKY41-1 regulates the production of anthocyanin, and WRKY23 regulates the biosynthesis of flavonols in Arabidopsis44,47. In potato, StWRKY8 regulates resistance to late blight by regulating the isoquinoline alkaloid pathway48. WsWRKY1 positively regulates the biosynthesis of phytosterol and triterpenoid withanolide accumulation and defense against biotic stress in Withania somnifera49. Here, transcriptome analysis showed that the identified differentially expressed genes between wild-type plants and CmWRKY53 transgenic plants are mainly involved in secondary metabolites (Supplementary Fig. S1). Plants are able to overproduce reactive oxygen species (ROS) when they are challenged with insect feeding, and POD and polyphenol oxidase (PPO), which are involved in plant defense against insects and pathogens50,51. Increased activities of peroxidase and polyphenol oxidase enhance the resistance of cassava to the spider mite Tetranychus urticae52. Our previous study showed that peroxidase and polyphenol oxidase activity were enhanced by aphids, and the activities of polyphenol oxidase enzymes were increased in the resistant cultivars of chrysanthemum after aphid inoculation, suggesting that polyphenol oxidase might contribute to aphid resistance in chrysanthemum53. Transcripts of the PPO family encode PPO enzymes, which are characterized by a common central domain of tyrosinase, that can catalyze the hydroxylation of tyrosine, thereby forming DOPA, which is thought to contribute to the biosynthesis of benzylisoquinoline alkaloids54,55. PPO also contributes to lignification and produces polyphenols that reduce infestation rates of green peach aphids56,57. PRX66 encodes a peroxidase involved in the lignification of tracheary elements in Arabidopsis thaliana58. In the present study, we found that genes encoding peroxidase and polyphenol oxidase decreased in CmWRKY53-overexpressing plants but increased in SRDX lines. Thus, we propose that CmWRKY53 mediates sensitivity to aphids and that the mechanism could be related to plant secondary metabolism. However, more data need to be obtained before a definitive conclusion can be made.
Conclusions
In conclusion, CmWRKY53 was cloned from chrysanthemum, and its expression was induced by aphid infestation. The results showed that CmWRKY53 mediates chrysanthemum susceptibility to aphids. The expression levels of secondary metabolite biosynthesis genes, such as peroxidase- and polyphenol oxidase-encoding genes, decreased in CmWRKY53-overexpressing plants, while they dramatically increased in CmWRKY53-SRDX plants. This suggests that the decreased levels of secondary metabolites in CmWRKY53 contributed to the susceptibility of chrysanthemum to aphids.
References
Nalam, V., Louis, J. & Shah, J. Plant defense against aphids, the pest extraordinaire. Plant Sci.279, 96–107 (2019).
Ng, J. C. & Perry, K. L. Transmission of plant viruses by aphid vectors. Mol. Plant Pathol.5, 505–511 (2004).
Figueroa, C. C., Fuentes-Contreras, E., Molina-Montenegro, M. A. & Ramírez, C. C. Biological and genetic features of introduced aphid populations in agroecosystems. Curr. Opin. Insect Sci.26, 63–68 (2018).
Jamieson, M. A. et al. Global change effects on plant–insect interactions: the role of phytochemistry. Curr. Opin. Insect Sci.23, 70–80 (2017).
Hartmann, T. Plant-derived secondary metabolites as defensive chemicals in herbivorous insects: a case study in chemical ecology. Planta219, 1–4 (2004).
Frick, K. M., Foley, R. C., Siddique, K. H., Singh, K. B. & Kamphuis, L. G. The role of jasmonate signalling in quinolizidine alkaloid biosynthesis, wounding and aphid predation response in narrow-leafed lupin. Funct. Plant Biol.46, 443–454 (2019).
Pan, Y., Xu, P., Zeng, X., Liu, X. & Shang, Q. Characterization of UDP-glucuronosyltransferases and the potential contribution to nicotine tolerance in Myzus persicae. Int. J. Mol. Sci.20, 3637 (2019).
Macel, M. et al. Differences in effects of pyrrolizidine alkaloids on five generalist insect herbivore species. J. Chem. Ecol.31, 1493–1508 (2005).
Lin, C.-C., Chen, L.-M. & Liu, Z.-H. Rapid effect of copper on lignin biosynthesis in soybean roots. Plant Sci.168, 855–861 (2005).
Liu, Q., Luo, L. & Zheng, L. Lignins: biosynthesis and biological functions in plants. Int. J. Mol. Sci.19, 335 (2018).
Wang, Y. et al. CmMYB19 over-expression improves aphid tolerance in chrysanthemum by promoting lignin synthesis. Int. J. Mol. Sci.18, 619 (2017).
Eulgem, T., Rushton, P. J., Robatzek, S. & Somssich, I. E. The WRKY superfamily of plant transcription factors. Trends Plant Sci.5, 199–206 (2000).
Ülker, B. & Somssich, I. E. WRKY transcription factors: from DNA binding towards biological function. Curr. Opin. Plant Biol.7, 491–498 (2004).
Bakshi, M. & Oelmüller, R. WRKY transcription factors: jack of many trades in plants. Plant Signal Behav.9, e27700 (2014).
Pandey, S. P. & Somssich, I. E. The role of WRKY transcription factors in plant immunity. Plant Physiol.150, 1648–1655 (2009).
Mao, G. et al. Phosphorylation of a WRKY transcription factor by two pathogen-responsive MAPKs drives phytoalexin biosynthesis in Arabidopsis. Plant Cell23, 1639–1653 (2011).
Birkenbihl, R. P., Diezel, C. & Somssich, I. E. Arabidopsis WRKY33 is a key transcriptional regulator of hormonal and metabolic responses toward Botrytis cinerea infection. Plant Physiol.159, 266–285 (2012).
Skibbe, M., Qu, N., Galis, I. & Baldwin, I. T. Induced plant defenses in the natural environment: Nicotiana attenuata WRKY3 and WRKY6 coordinate responses to herbivory. Plant Cell20, 1984–2000 (2008).
Bhattarai, K. K., Atamian, H. S., Kaloshian, I. & Eulgem, T. WRKY72‐type transcription factors contribute to basal immunity in tomato and Arabidopsis as well as gene‐for‐gene resistance mediated by the tomato R gene Mi‐1. Plant J.63, 229–240 (2010).
Dong, J., Chen, C. & Chen, Z. Expression profiles of the Arabidopsis WRKY gene superfamily during plant defense response. Plant Mol. Biol.51, 21–37 (2003).
Zentgraf, U. & Doll, J. Arabidopsis WRKY53, a node of multi-layer regulation in the network of senescence. Plants8, 578 (2019).
Hu, Y., Dong, Q. & Yu, D. Arabidopsis WRKY46 coordinates with WRKY70 and WRKY53 in basal resistance against pathogen Pseudomonas syringae. Plant Sci.185, 288–297 (2012).
Ohmiya, A. Molecular mechanisms underlying the diverse array of petal colors in chrysanthemum flowers. Breed. Sci.68, 119–127 (2018).
Zhou, X., Chen, X., Wu, X., Cao, G. & Zhang, J. Characterization of the chemical composition of white chrysanthemum flowers of Hangzhou by using high‐performance ion trap mass spectrometry. J. Sep. Sci.39, 1218–1222 (2016).
Kuang, C.-l et al. Chemical composition and antimicrobial activities of volatile oil extracted from Chrysanthemum morifolium Ramat. J. Food Sci. Technol.55, 2786–2794 (2018).
Xia, X. et al. Gene expression profiles responses to aphid feeding in chrysanthemum (Chrysanthemum morifolium). BMC Genomics15, 1050 (2014).
Larkin, M. A. et al. Clustal W and Clustal X version 2.0. Bioinformatics23, 2947–2948 (2007).
Gao, H. et al. The heterologous expression in Arabidopsis of a chrysanthemum Cys2/His2 zinc finger protein gene confers salinity and drought tolerance. Planta235, 979–993 (2012).
Gu, C. et al. Reference gene selection for quantitative real-time PCR in Chrysanthemum subjected to biotic and abiotic stress. Mol. Biotechnol.49, 192 (2011).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res.29, e45 (2001).
Zhou, F. et al. D14–SCF D3-dependent degradation of D53 regulates strigolactone signalling. Nature504, 406–410 (2013).
Ohta, M., Matsui, K., Hiratsu, K., Shinshi, H. & Ohmetakagi, M. Repression domains of class II ERF transcriptional repressors share an essential motif for active repression. Plant Cell13, 1959–1968 (2001).
Hiratsu, K., Matsui, K., Koyama, T. & Ohmetakagi, M. Dominant repression of target genes by chimeric repressors that include the EAR motif, a repression domain, in Arabidopsis. Plant J.34, 733–739 (2003).
Hiratsu, K., Mitsuda, N., Matsui, K. & Ohmetakagi, M. Identification of the minimal repression domain of SUPERMAN shows that the DLELRL hexapeptide is both necessary and sufficient for repression of transcription in Arabidopsis. Biochem. Biophys. Res. Commun.321, 172–178 (2004).
Huang, Y., Niu, C., Yang, C. & Jinn, T. The heat stress factor HSFA6b connects ABA signaling and ABA-mediated heat responses. Plant Physiol.172, 1182–1199 (2016).
Narumi, T. et al. Arabidopsis chimeric TCP3 repressor produces novel floral traits in Torenia fournieri and Chrysanthemum morifolium. Plant Biotechnol.28, 131–140 (2011).
Li, P. et al. The over-expression of a chrysanthemum WRKY transcription factor enhances aphid resistance. Plant Physiol. Biochem.95, 26–34 (2015).
Qi, X., Wang, H. & Song, A. et al. Genomic and transcriptomic alterations following intergeneric hybridization and polyploidization in the Chrysanthemum nankingense×Tanacetum vulgare hybrid and allopolyploid (Asteraceae). Hortic. Res.5, 5 (2018).
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Genome Biol.11, 1–12 (2010).
Xie, Z. et al. Annotations and functional analyses of the rice WRKY gene superfamily reveal positive and negative regulators of abscisic acid signaling in aleurone cells. Plant Physiol.137, 176–189 (2005).
Van Eck, L. et al. Virus-induced gene silencing of WRKY53 and an inducible phenylalanine ammonia-lyase in wheat reduces aphid resistance. Plant Biotechnol. J.8, 1023–1032 (2010).
Atamian, H. S., Eulgem, T. & Kaloshian, I. SlWRKY70 is required for Mi-1-mediated resistance to aphids and nematodes in tomato. Planta235, 299–309 (2012).
Kloth, K. J. et al. AtWRKY22 promotes susceptibility to aphids and modulates salicylic acid and jasmonic acid signalling. J. Exp. Bot.67, 3383–3396 (2016).
Duan, S. et al. Functional characterization of a heterologously expressed Brassica napus WRKY41-1 transcription factor in regulating anthocyanin biosynthesis in Arabidopsis thaliana. Plant Sci.268, 47–53 (2018).
Schluttenhofer, C., Pattanaik, S., Patra, B. & Yuan, L. Analyses of Catharanthus roseus and Arabidopsis thaliana WRKY transcription factors reveal involvement in jasmonate signaling. BMC Genomics15, 502 (2014).
Suttipanta, N. et al. The transcription factor CrWRKY1 positively regulates the terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Plant Physiol.157, 2081–2093 (2011).
Grunewald, W. et al. Transcription factor WRKY23 assists auxin distribution patterns during Arabidopsis root development through local control on flavonol biosynthesis. Proc. Natl. Acad. Sci. USA109, 1554–1559 (2012).
Yogendra, K. N. et al. StWRKY8 transcription factor regulates benzylisoquinoline alkaloid pathway in potato conferring resistance to late blight. Plant Sci.256, 208–216 (2017).
Singh, A. K. et al. A WRKY transcription factor from Withania somnifera regulates triterpenoid withanolide accumulation and biotic stress tolerance through modulation of phytosterol and defense pathways. N. Phytol.215, 1115–1131 (2017).
Mittler, R., Vanderauwera, S., Gollery, M. & Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci.9, 490–498 (2004).
Jaiti, F., Verdeil, J. L. & El Hadrami, I. Effect of jasmonic acid on the induction of polyphenoloxidase and peroxidase activities in relation to date palm resistance against Fusarium oxysporum f. sp. albedinis. Physiol. Mol. Plant Pathol.74, 84–90 (2009).
Liang, X. et al. Increased activities of peroxidase and polyphenol oxidase enhance cassava resistance to Tetranychus urticae. Exp. Appl. Acarol.71, 195–209 (2017).
He, J. et al. Chrysanthemum leaf epidermal surface morphology and antioxidant and defense enzyme activity in response to aphid infestation. J. Plant Physiol.168, 687–693 (2011).
Song, X., Pan, Y., Li, L., Wu, X. & Wang, Y. Composition and diversity of rhizosphere fungal community in Coptis chinensis Franch. continuous cropping fields. PLoS ONE13, 3 (2018).
Lovkova, M. Y., Buzuk, G. & Sokolova, S. Regulatory role of elements in the formation and accumulation of alkaloids in Papaver somniferum L. seedlings. Appl. Biochem. Microbiol.42, 420–423 (2006).
Jordan, M. O., Sauge, M. H. & Vercambre, G. Chemical and growth traits of the peach tree may induce higher infestation rates of the green peach aphid, Myzus persicae (Sulzer) Pest Manag. Sci.76, 797–806 (2020).
Ralph, J. et al. in Recent advances in polyphenol research, Vol. 1 (eds Fouad Daayf & Vincenzo Lattanzio) Ch. 2 (Wiley-Blackwell, 2008).
Tokunaga, N., Kaneta, T., Sato, S. & Sato, Y. Analysis of expression profiles of three peroxidase genes associated with lignification in Arabidopsis thaliana. Physiol. Plant.136, 237–249 (2009).
Acknowledgements
This study was financially supported by the National Natural Science Foundation of China (31672192), the Agricultural Science and Technology Innovation Fund Project of Jiangsu Province (CX (18) 2020), the National Key Research and Development Project (2018YFD1000402), and the Foundation of Key Laboratory of Landscaping (KF201801).
Author information
Authors and Affiliations
Contributions
F.C. and S.C. conceived and designed the project. T.G. and P.L. generated the CmWRKY53 transgenic chrysanthemum plants. W.Z. performed the aphid infestation and RNA-seq experiments and analyzed the data. W.Z. and S.C. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, W., Gao, T., Li, P. et al. Chrysanthemum CmWRKY53 negatively regulates the resistance of chrysanthemum to the aphid Macrosiphoniella sanborni. Hortic Res 7, 109 (2020). https://doi.org/10.1038/s41438-020-0334-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41438-020-0334-0
This article is cited by
-
Chrysanthemum (Chrysanthemum morifolium) CmHRE2-like negatively regulates the resistance of chrysanthemum to the aphid (Macrosiphoniella sanborni)
BMC Plant Biology (2024)
-
Evaluation of aphid resistance on different rose cultivars and transcriptome analysis in response to aphid infestation
BMC Genomics (2024)
-
Potential pathways and genes expressed in Chrysanthemum in response to early fusarium oxysporum infection
BMC Plant Biology (2023)
-
A sorghum genome-wide association study (GWAS) identifies a WRKY transcription factor as a candidate gene underlying sugarcane aphid (Melanaphis sacchari) resistance
Planta (2022)
-
A functional genomics approach to dissect spotted alfalfa aphid resistance in Medicago truncatula
Scientific Reports (2020)