Abstract
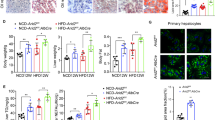
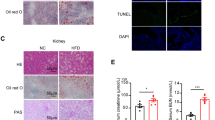
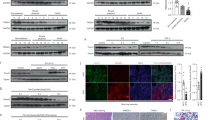
In recent years, receptor for advanced glycation end-products (RAGE) has been documented to induce liver fibrosis and inflammatory reaction. Further, microarray data analysis of this study predicted high expression of RAGE in non-alcoholic fatty liver disease (NAFLD). However, its specific mechanisms remain to be elucidated. Hence, this study is aimed at investigating the mechanistic insights of RAGE in chronic intermittent hypoxia (CIH)-induced NAFLD. ApoE knockout (ApoE−/−) mice were exposed to CIH to induce NAFLD, and primary hepatocytes were also exposed to CIH to mimic in vitro setting. Accordingly, we found that RAGE and NF-κB were upregulated in the liver tissues of CIH-induced NAFLD mice and CIH-exposed hepatocytes. Depleted RAGE attenuated CIH-induced hepatocyte injury, lipid deposition, and inflammation. The relationship between RAGE and NF-κB was analyzed by in silico analysis and correlation analysis. It was demonstrated that knockdown of RAGE inhibited the NF-кB pathway, thus alleviating CIH-induced disorders in hepatocytes. Moreover, in vivo experiments also verified that depletion of RAGE alleviated CIH-induced NAFLD by inhibiting NF-кB pathway. Collectively, loss of RAGE blocked the NF-кB pathway to alleviate CIH-induced NAFLD, therefore, highlighting a potential hepatoprotective target for treating NAFLD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article (and/or) its supplemental material file. Further enquiries can be directed to the corresponding author.
References
Wang XJ, Malhi H. Nonalcoholic fatty liver disease. Ann Intern Med. 2018;169:ITC65–ITC80.
Marjot T, Moolla A, Cobbold JF, Hodson L, Tomlinson JW. Nonalcoholic fatty liver disease in adults: current concepts in etiology, outcomes, and management. Endocr Rev. 2020;41:bnz009.
Lemoine M, Serfaty L. Chronic intermittent hypoxia: a breath of fresh air in the understanding of NAFLD pathogenesis. J Hepatol. 2012;56:20–2.
Isaza SC, Del Pozo-Maroto E, Dominguez-Alcon L, Elbouayadi L, Gonzalez-Rodriguez A, Garcia-Monzon C. Hypoxia and non-alcoholic fatty liver disease. Front Med. 2020;7:578001.
Wu Q, Li S, Li X, Sui Y, Yang Y, Dong L, et al. Inhibition of advanced glycation endproduct formation by lotus seedpod oligomeric procyanidins through RAGE-MAPK Signaling and NF-kappaB activation in high-fat-diet rats. J Agric Food Chem. 2015;63:6989–98.
Santilli F, Blardi P, Scapellato C, Bocchia M, Guazzi G, Terzuoli L, et al. Decreased plasma endogenous soluble RAGE, and enhanced adipokine secretion, oxidative stress and platelet/coagulative activation identify non-alcoholic fatty liver disease among patients with familial combined hyperlipidemia and/or metabolic syndrome. Vascul Pharmacol. 2015;72:16–24.
Wan J, Wu X, Chen H, Xia X, Song X, Chen S, et al. Aging-induced aberrant RAGE/PPARalpha axis promotes hepatic steatosis via dysfunctional mitochondrial beta oxidation. Aging Cell. 2020;19:e13238.
Asadipooya K, Lankarani KB, Raj R, Kalantarhormozi M. RAGE is a potential cause of onset and progression of nonalcoholic fatty liver disease. Int J Endocrinol. 2019;2019:2151302.
Chandrashekaran V, Seth RK, Dattaroy D, Alhasson F, Ziolenka J, Carson J, et al. HMGB1-RAGE pathway drives peroxynitrite signaling-induced IBD-like inflammation in murine nonalcoholic fatty liver disease. Redox Biol. 2017;13:8–19.
Zhou J, Bai W, Liu Q, Cui J, Zhang W. Intermittent hypoxia enhances THP-1 monocyte adhesion and chemotaxis and promotes M1 macrophage polarization via RAGE. Biomed Res Int. 2018;2018:1650456.
Panday A, Inda ME, Bagam P, Sahoo MK, Osorio D, Batra S. Transcription factor NF-kappaB: an update on intervention strategies. Arch Immunol Ther Exp. 2016;64:463–83.
Chen Z, Yu R, Xiong Y, Du F, Zhu S. A vicious circle between insulin resistance and inflammation in nonalcoholic fatty liver disease. Lipids Health Dis. 2017;16:203.
Liu J, Li W, Zhu W, He W, Zhao H, Xiang Y, et al. Chronic intermittent hypoxia promotes the development of experimental non-alcoholic steatohepatitis by modulating Treg/Th17 differentiation. Acta Biochim Biophys Sin. 2018;50:1200–10.
Khan SA, Tyagi M, Sharma AK, Barreto SG, Sirohi B, Ramadwar M, et al. Cell-type specificity of beta-actin expression and its clinicopathological correlation in gastric adenocarcinoma. World J Gastroenterol. 2014;20:12202–11.
Kang HH, Kim IK, Lee HI, Joo H, Lim JU, Lee J, et al. Chronic intermittent hypoxia induces liver fibrosis in mice with diet-induced obesity via TLR4/MyD88/MAPK/NF-kB signaling pathways. Biochem Biophys Res Commun. 2017;490:349–55.
De Vincentis A, Rahmani Z, Muley M, Vespasiani-Gentilucci U, Ruggiero S, Zamani P, et al. Long noncoding RNAs in nonalcoholic fatty liver disease and liver fibrosis: state-of-the-art and perspectives in diagnosis and treatment. Drug Discov Today. 2020;25:1277–86.
Chen X, Tan XR, Li SJ, Zhang XX. LncRNA NEAT1 promotes hepatic lipid accumulation via regulating miR-146a-5p/ROCK1 in nonalcoholic fatty liver disease. Life Sci. 2019;235:116829.
Lin ZP, Lin HL, Yu XP, Zheng YJ, Cheng SY. TLR4 mediates inflammation and hepatic fibrosis induced by chronic intermittent hypoxia in rats. Mol Med Rep. 2020;22:651–60.
Sundaram SS, Halbower AC, Klawitter J, Pan Z, Robbins K, Capocelli KE, et al. Treating obstructive sleep apnea and chronic intermittent hypoxia improves the severity of nonalcoholic fatty liver disease in children. J Pediatr. 2018;198:67–75 e1.
Bai W, Zhou J, Zhou N, Liu Q, Cui J, Zou W, et al. Hypoxia-increased RAGE expression regulates chemotaxis and pro-inflammatory cytokines release through nuclear translocation of NF-kappa B and HIF1alpha in THP-1cells. Biochem Biophys Res Commun. 2018;495:2282–8.
Palma-Duran SA, Kontogianni MD, Vlassopoulos A, Zhao S, Margariti A, Georgoulis M, et al. Serum levels of advanced glycation end-products (AGEs) and the decoy soluble receptor for AGEs (sRAGE) can identify non-alcoholic fatty liver disease in age-, sex- and BMI-matched normo-glycemic adults. Metabolism. 2018;83:120–7.
Pang Q, Sun Z, Shao C, Cai H, Bao Z, Wang L, et al. CML/RAGE signal bridges a common pathogenesis between atherosclerosis and non-alcoholic fatty liver. Front Med. 2020;7:583943.
Chen RC, Yi PP, Zhou RR, Xiao MF, Huang ZB, Tang DL, et al. The role of HMGB1-RAGE axis in migration and invasion of hepatocellular carcinoma cell lines. Mol Cell Biochem. 2014;390:271–80.
Yang JM, Sun Y, Wang M, Zhang XL, Zhang SJ, Gao YS, et al. Regulatory effect of a Chinese herbal medicine formula on non-alcoholic fatty liver disease. World J Gastroenterol. 2019;25:5105–19.
Yuan S, Liu H, Yuan D, Xu J, Chen Y, Xu X, et al. PNPLA3 I148M mediates the regulatory effect of NF-kB on inflammation in PA-treated HepG2 cells. J Cell Mol Med. 2020;24:1541–52.
Lu S, Zhang T, Gu W, Yang X, Lu J, Zhao R, et al. Volatile oil of amomum villosum inhibits nonalcoholic fatty liver disease via the gut-liver axis. Biomed Res Int. 2018;2018:3589874.
Amen Y, Sherif AE, Shawky NM, Abdelrahman RS, Wink M, Sobeh M. Grape-leaf extract attenuates alcohol-induced liver injury via interference with NF-kappaB signaling pathway. Biomolecules. 2020;10:558.
Li CX, Gao JG, Wan XY, Chen Y, Xu CF, Feng ZM, et al. Allyl isothiocyanate ameliorates lipid accumulation and inflammation in nonalcoholic fatty liver disease via the Sirt1/AMPK and NF-kappaB signaling pathways. World J Gastroenterol. 2019;25:5120–33.
Leung C, Herath CB, Jia Z, Andrikopoulos S, Brown BE, Davies MJ, et al. Dietary advanced glycation end-products aggravate non-alcoholic fatty liver disease. World J Gastroenterol. 2016;22:8026–40.
Yano T, Hagiwara Y, Ando A, Kanazawa K, Koide M, Sekiguchi T, et al. RAGE-dependent NF-kB inflammation processes in the capsule of frozen shoulders. J Shoulder Elbow Surg. 2020;29:1884–91.
Acknowledgements
This study was supported by Key Project of Jiangxi Provincial Education Department (No. GJJ190026), the National Natural Science Foundation of China (No. 82060025), and the Natural Science Foundation of Jiangxi Province (No. 2019BAB205002).
Author information
Authors and Affiliations
Contributions
HT: Conceptualization, Data Curation, Formal analysis, Writing—Original Draft, Writing—Review & Editing; JH: Conceptualization, Investigation, Methodology, Writing—Review & Editing; WZ: Resources, Supervision, Writing—Original Draft; YH: Software, Writing—Original Draft; JC: Visualization, Writing—Original Draft; FG: Validation, Writing—Original Draft; WB: Conceptualization, Project administration, Writing—Review & Editing
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Tan, H., Hu, J., Zuo, W. et al. Loss of RAGE prevents chronic intermittent hypoxia-induced nonalcoholic fatty liver disease via blockade of NF-кB pathway. Gene Ther 30, 278–287 (2023). https://doi.org/10.1038/s41434-022-00351-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-022-00351-4