Abstract
Background
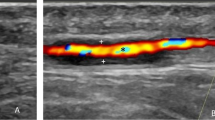
Increasing number of centres are establishing sequential fast track pathways (FTP) for management of giant cell arteritis (GCA), with temporal artery ultrasound (US) replacing temporal artery biopsy (TAB) as the first investigational method. Biopsy is performed as second investigation, when US is negative/inconclusive. This study investigates the role of TAB in a sequential GCA-FTP and its utility in those with negative/inconclusive US.
Methods
Prospective study of patients referred for TAB as part of Coventry sequential GCA-FTP May 2014-June 2019. Analysis included sensitivity and specificity of TAB, impact of arterial specimen length and duration of treatment with corticosteroids on sensitivity of TAB and the clinical predictors for a positive biopsy.
Results
A total of 1149 patients with suspected GCA were referred to this GCA-FTP, with 109 (9.5%) referred for TAB. Overall sensitivity of TAB was 47% (specificity: 100%) and in patients with negative/inconclusive US sensitivity was 39% (specificity:100%). Post-fixation arterial specimen length <15 mm showed lower sensitivity (14%), which increased to 52% when specimen length was ≥15 mm. Sensitivity of TAB was highest in first 7 (60%) to 10 days (59%) from starting corticosteroids. Predictors of positive biopsy using univariate logistic regression analysis were jaw claudication (OR = 5.40; p = 0.0057), elevated erythrocyte sedimentation rate (OR = 5.50; p = 0.013) and elevated C-reactive protein (OR = 23.7; p = 0.0043).
Conclusion
This is the first study to look at the role of TAB in a sequential GCA-FTP. Biopsy plays an important role in GCA-FTP, when US is negative/inconclusive. Sensitivity of TAB improved when specimen length was ≥15 mm and performed within 10 days of commencing corticosteroids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
We would be happy to provide de-identified participant data if needed, at the time of publication, including the statistical analysis plan.
References
Luqmani R, Lee E, Singh S, Gillett M, Schmidt WA, Bradburn M, et al. The role of ultrasound compared to biopsy of temporal arteries in the diagnosis and treatment of giant cell arteritis (TABUL): a diagnostic accuracy and cost-effectiveness study. Health Technol Assess. 2016;20:1–270.
Dasgupta B, Borg FA, Hassan N, Alexander L, Barraclough K, Bourke B, et al. BSR and BHPR Guidelines for the management of giant cell arteritis. Rheumatol (Oxf). 2010;49:1594–7.
Ješe R, Rotar Z, Tomšič M, Hočevar A. The role of colour doppler ultrasonography of facial and occipital arteries in patients with giant cell arteritis: A prospective study. Eur J Radio. 2017;95:9–12.
Roncato C, Allix-Béguec C, Brottier-Mancini E, Gombert B, Denis G. Diagnostic performance of colour duplex ultrasonography along with temporal artery biopsy in suspicion of giant cell arteritis. Clin Exp Rheumatol. 2017;35:119–22.
Aranda-Valera IC, García Carazo S, Monjo, Henry I. de Miguel Mendieta E. Diagnostic validity of Doppler ultrasound in giant cell arteritis. Clin Exp Rheumatol. 2017;103:123–7.
Diamantopoulos A, Haugeberg G, Hetland H, Soldal DM, Bie R, Myklebust G. Diagnostic value of color Doppler ultrasonography of temporal arteries and large vessels in giant cell arteritis: a consecutive case series. Arthritis Care Res. 2014;66:113–9.
Schmidt WA. Ultrasound in the diagnosis and management of giant cell arteritis. Rheumatology. 2018;57:22–31.
Dubey S, Pinnell J, Tiivas C, Mehta P. Coventry fast-track pathway for managing giant cell arteritis. Int J Clin Rheumtol. 2020;15:21–25.
Monti S, Floris A, Ponte CB, Schmidt WA, Diamantopoulos AP, Pereira C, et al. The proposed role of ultrasound in the management of giant cell arteritis in routine clinical practice. Rheumatology. 2018;57:112–9.
Pinnell J, Tiivas C, Mehta P, Dubey S. Corticosteroids reduce vascular ultrasound sensitivity in fast-track pathways: results from Coventry Multidisciplinary FTP for cranial Giant cell Arteritis. Accepted Scandinavian J Rheumatol. https://doi.org/10.1080/03009742.2022.2051279.
Mackie SL, Dejaco C, Appenzeller S, Camellino D, Duftner C, Gonzalez-Chiappe S, et al. British Society for Rheumatology guideline on diagnosis and treatment of giant cell arteritis. Rheumatol (Oxf). 2020;59:e1–23.
Monti S, Águeda AF, Luqmani RA, Buttgereit F, Cid M, Dejaco C, et al. Systematic literature review informing the 2018 update of the EULAR recommendation for the management of large vessel vasculitis: focus on giant cell arteritis. RMD Open. 2019;5:e001003.
Oh L, Wong EH, Gill AJ, McCluskey P, Smith JEH. Value of temporal artery biopsy length in diagnosing giant cell arteritis. ANZ J Surg. 2018;88:191–5.
Chu R, Foster C, Ali M, Chaba T, Clifford AH, Mahr A, et al. Optimal length and usefulness of temporal artery biopsies in the diagnosis of giant cell arteritis: a 10-year retrospective review of medical records. Lancet Rheumatol. 2020. https://doi.org/10.1016/S2665-9913(20)30222-8.
Ray-Chaudhuri N, Kiné DA, Tijani SO, Parums D, Cartliddge N, Strong NP, et al. Effect of prior steroid treatment of temporal artery biopsy findings in giant cell arteritis. Br J Ophthalmol. 2002;86:530–2.
Allison MC, Gallagher PJ. Temporal artery biopsy and corticosteroid treatment. Ann Rheum Dis. 1984;43:416–17.
Jakobsson K, Jacobsson L, Mohammad AJ, Nilsson JA, Warrington K, Matteson EL, et al. The effect of clinical features and glucocorticoids on biopsy findings in giant cell arteritis. BMC Musculoskelet Disord. 2016;17:363.
Toren A, Ezekiel W, Patel V, Monteith B, Gilberg S, Jordan D. Clinical predictors of positive temporal artery biopsy. Can J Ophthalmol. 2016;51:476–81.
Aghdam KA, Sanjari MS, Manafi N, Khorramdel S, Alemzadeh SA, Navahi RAA. Temporal artery biopsy for diagnosing giant cell arteritis: a ten-year review. J Ophthalmic Vis Res. 2020;15:201–9.
Fernandez E, Monjo-Henry I, Bonilla G, Plasencia C, Carus M, Balsa A, et al. False positives in the ultrasound diagnosis of giant cell arteritis: some diseases can also show the halo sign. Rheumatology. 2020;59:2443–7.
Acknowledgements
The authors would like to acknowledge Carl Tiivas for his contribution. There are no funding sources to declare for any author. This data was presented at: 1. European League Against Rheumatism (EULAR) Annual European Congress of Rheumatology, 3–5th June 2020, 2. 10th Kettering Eye Meeting, 11th September 2020, Kettering (won first prize for oral presentation) 3. Midlands Ophthalmological Society annual meeting, 2nd October 2020, Birmingham.
Author information
Authors and Affiliations
Contributions
All listed authors provided substantial contributions to this work. PM, AS, and MG were involved with the conception and study design. SD contributed to study design and ethical approval. The literature review was led by AS with help from PM. RC provided detailed statistical analysis. All authors have contributed to the critical revision of the manuscript and agreed on the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sachdev, A., Dubey, S., George, M. et al. Role of Temporal artery biopsy in a sequential Giant Cell Arteritis fast-track pathway: a 5-year prospective study. Eye 37, 1614–1618 (2023). https://doi.org/10.1038/s41433-022-02132-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-02132-0
This article is cited by
-
Diagnostic validity of ultrasound including extra-cranial arteries in giant cell arteritis
Clinical Rheumatology (2023)