Abstract
Background
To assess multimodal imaging findings of focal scleral nodule (FSN) to evaluate its origin and natural course.
Methods
This was a retrospective observational case series and included 14 patients with FSN who underwent multimodal imaging. Clinical information was gathered from patients’ medical records. Primary outcome measures were standardized grading of imaging features.
Results
The mean follow-up duration was 68.8 ± 43.6 months (range, 6-139 months). Most lesions were solitary (92.6%), but one patient had two adjacent lesions (7.1%). Optical coherence tomography revealed that all lesions were confined to the sclera. Lesions showed mostly outer retinal abnormality, with external limiting membrane thinning or absence in 41.6% of lesions and ellipsoid layer absence in 84.6% of lesions. Most lesions showed an absence (69.2%) or thinning (23.1%) of the choroid above the lesion, and the mean choroidal thickness above the lesion for choroids with measurable thickness was 36 ± 75 μm (median, 0; range, 0–265 μm). Of 13 lesions with available follow-up data, only three lesions showed minimal growth over time.
Conclusions
This study demonstrates for the first time that bifocal lesions of FSN in the same eye are possible and reaffirms the relative stability of this entity.
Similar content being viewed by others
Introduction
Focal scleral nodule (FSN) is a newly coined term for the entity previously known as solitary idiopathic choroiditis (SIC) or unifocal helioid choroiditis. It was first described by Hong PH, Jampol LM, Dodwell DG, Hrisomalos NF and Lyon AT [1] as a solitary, elevated, yellow-white active focus of choroiditis with overlying subretinal fluid and in some cases subretinal haemorrhage. As the lesion can resemble some of the more detrimental entities such as amelanotic melanoma, metastasis, osteoma and retinoblastoma, it was commonly misdiagnosed and overtreated before its clinical description. Later, Shields JA, Shields CL, Demirci H and Hanovar S. [2] reported 60 consecutive patients with such clinical findings and described their clinical features and natural course. Patients were mostly young with a mean age of 35 years, and 33% of them showed signs of active inflammation even though 60% of the lesions remained stable on long-term follow-up. With the widespread use of optical coherence tomography (OCT), Fung AT, Kaliki S, Shields CL, Mashayekhi A and Shields JA [3] observed 10 eyes with SIC using OCT with enhanced depth imaging (EDI) and suggested the possibility of the lesions arising “from [the] sclera, outer choroid, or both.”
Recently, Fung AT, Waldstein SM, Gal-Or O, Pellegrini M, Preziosa C, Shields JA, et al. [4] performed swept-source OCT scans on 63 patients with SIC and revealed that the lesion was located solely at the sclera, not at the choroid as previously understood. They proposed a new name—focal scleral nodule—for SIC to represent the newly identified location of the nodule and the hypothesis that the lesion need not be solitary, although no patients with more than one FSN lesion had been identified. In this retrospective case series, we present 14 cases of FSN—including a case with two lesions—assessed by various imaging modalities.
Methods
This study was a single-centre, retrospective observational case series. It adhered to the tenets of the Declaration of Helsinki and received approval from the institutional review board of Severance Hospital to waive informed consent (IRB no. 4-2021-0606). Due to retrospective nature of this study, informed consent was not required.
This study included patients with a diagnosis of FSN who were first evaluated between August 2009 and March 2020 at Severance Hospital. Clinical information of patients with multimodal imaging results was gathered from medical records. Demographic and clinical data including age, gender, presenting symptoms, referral diagnosis, and best-corrected visual acuity were collected for all patients. Medical histories were inspected for any systemic conditions related to their condition, such as a history of tuberculosis, syphilis, sarcoidosis, histoplasmosis, toxoplasmosis, cat-scratch disease, blastomycosis, or Aspergillus species infection.
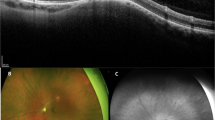
An FSN lesion was defined as a circular yellow-white subretinal fundus lesion without associated systemic disease, based on an article by Fung AT, Waldstein SM, Gal-Or O, Pellegrini M, Preziosa C, Shields JA, et al. [4], which used the definition of SIC by Shields JA, Shields CL, Demirci H and Hanovar S. [2] Choroidal or retinal lesions mimicking FSN, such as amelanotic nevus, amelanotic melanoma, metastasis, osteoma, granuloma, retinoblastoma, or astrocytic hamartoma, were excluded. Characteristic multimodal imaging findings of FSN are demonstrated in Fig. 1.
The lesions typically appear (A, C) yellow-white or (B) yellow. (A, B, C) An orange halo is present. Fundus autofluorescence shows hyperfluorescence (C), stippled appearance or hypofluorescence (A). OCT demonstrates scleral dome-shaped (A, B, C) or nodular lesions. Lesions are typically located at the posterior pole near the optic disc (A, B, C). Lesions can sometimes occur with minimal intraretinal cysts (A) or subretinal fluid (C). B-scan ultrasonography can show either high echogenicity or low echogenicity.
All study eyes underwent colour fundus photography or ultra-widefield fundus photography using an Optos fundus camera (Optos PLC, Dunfermine, United Kingdom). Eyes were evaluated to obtain the following clinical data: colour, basal diameter, location, distance relative to the optic nerve head and fovea, and presence of associated clinical features, such as orange halo, orange pigment, drusen, and surrounding retinal pigment epithelium (RPE) changes. When available, spectral-domain OCT with EDI was performed using Spectralis OCT (Heidelberg Engineering, Heidelberg, Germany). Using the OCT image, the greatest horizontal diameter of the lesion was evaluated, and associated features, such as drusen, pigment epithelial detachment, subretinal fluid, and intraretinal fluid, were assessed. When the posterior margin of the lesion was visible, the thickness of the lesion was measured. When possible, with adequate image quality, anatomic alterations of neurosensory retinal layers overlying the lesion were evaluated. The thickness of the choroid overlying the lesion was measured at the thinnest point using a calliper tool.
A subset of patients (n = 5) underwent OCT angiography using Spectralis HRA + OCT (Heidelberg Engineering), and retinal and choroidal circulation status was evaluated using reconstructed images. Fundus autofluorescence images were acquired using a fundus camera, and their fluorescence pattern was reviewed and categorized into hypoautofluorescent, hyperautofluorescent, or stippled autofluorescent groups. B-scan ultrasonography was performed in 10 lesions, and the echogenicity and size of the lesion were checked. In addition, related clinical features such as calcification, extraocular extension, and choroidal excavation were evaluated. Ten lesions underwent fluorescein angiography with indocyanine green angiography. The presence of early and late hyperfluorescence and hypofluorescence at the location of the lesions was noted.
Using descriptive and frequency analysis, the means, medians, and standard deviations of the measured clinical features of the FSN lesions were demonstrated. Because this study was an observational series with a descriptive nature, statistical meaning was not evaluated.
Results
A total of 14 eyes from 14 patients with a diagnosis of FSN were included. The mean follow-up duration was 68.8 ± 43.6 months (range, 6-139 months). Demographic characteristics of the patients are summarized in Table 1.
The mean age at presentation was 37.71 ± 16.92 years (range, 3–60 years), and 8 out of 14(57%) patients were female. The median best-corrected visual acuity at presentation was 20/20 (range, 25/20–16/20). Most patients (64.3%) were referred for subretinal or choroidal mass, some patients (28.6%) were referred for choroidal melanoma, and one patient (7.1%) was referred for other retinal disease. None of the patients showed any ocular symptoms at presentation. In addition, no patient had a history of diseases that can cause retinal or choroidal lesions similar to FSN, such as tuberculosis, syphilis, sarcoidosis, histoplasmosis, toxoplasmosis, cat-scratch disease, blastomycosis, or Aspergillus species infection.
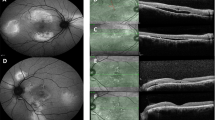
The clinical features of the patients are summarized in Table 1. Most of the lesions were solitary (92.6%), but one patient had two adjacent lesions (7.1%). Imaging characteristics of this patient are presented in Fig. 2. All lesions had clearly demarcated borders without any active signs of inflammation, such as blurry margins, haemorrhage, or exudation. The lesions were mostly yellow (40.0%) or yellow-white (46.7%), but some were orange (13.3%). Many of the lesions had an orange halo (66.7%), and none showed coexisting drusen (0%). Some of the lesions (40%) showed orange pigment. Seven lesions (46.7%) showed overlying RPE hypopigmentation, and 6 (40.4%) showed RPE hyperpigmentation. The lesions were typically located directly superior or superonasally/inferonasally to the disc with a mean distance of 4.3 mm (range, 0–12.2 mm) from the optic nerve. All of the lesions were at the postequatorial location with a mean distance to the fovea of 7.2 mm (range, 2.1–13.2 mm). The mean maximal linear basal diameter was 2.6 mm (median, 2.3 mm; range, 1.2–5.4 mm).
This patient had two FSN lesions in her left eye, which is shown in wide-field colour fundus photo(top left) and autofluorescence fundus photo(top middle). Two lesions were adjacent to each other but not directly connected. OCT(top right) shows two distinct FSN lesions with a dome shape. Raster line for OCT is drawn on wide-field fundus photo. OCT angiography shows decreased choriocapillary, choroid flow at each level, and marked signal void at the intralesional level.
Multimodal imaging characteristics of the lesions are summarized in Table 2. OCT data were available for 13 out of 15 lesions and revealed that all of the lesions were confined to the sclera, with most lesions (85%) being dome shaped and some (15%) having a nodular shape. The mean maximal horizontal length for the lesions was 2.6 ± 1.2 mm. For lesions with visible posterior margins, the mean thickness was 697 ± 174 μm. OCT also revealed that the eyes had mostly outer retinal abnormalities, with external limiting membrane thinning or absence in 38.5% and ellipsoid layer absence in 84.6%. Retinal pigment epithelium irregularity was observed in 23.1% of lesions, with thickened RPE in 53.8%, normal RPE in 38.5%, and thinning of RPE in 7.7%. Most lesions showed absent choroid (69.2%) or thinning (23.1%) of the choroid above the lesion, and the mean choroidal thickness above the lesion for choroids with measurable thickness was 36 ± 75 μm (median, 0; range, 0-265 μm). Mild subretinal fluid was observed in four lesions (30.8%).
OCT angiography of five lesions showed normal retinal circulation in all eyes, but choroidal flow void was eminent in all five lesions. Fundus autofluorescence was available for 12 lesions. Four lesions (33.3%) showed hyperautofluorescence while another four (33.3%) showed hypoautofluorescence and two (16.6%) showed a stippled appearance. The halo of the lesions showed mostly normal autofluorescence (50.0) with some hyperautofluorescence (25.0%) and hypoautofluorescence (25.0%). B-scan ultrasonography was performed in 10 lesions: 70.0% showed low echogenicity and 30.0% showed high echogenicity. The mean lesion thickness with B-scan was 1.8 ± 0.7 mm. None of the lesions revealed signs of calcification, extraocular extension, or choroidal excavation.
Ten lesions underwent fluorescein angiography and indocyanine green angiography. In fluorescein angiography, seven lesions (70.0%) showed early hypofluorescence, and six lesions (60.0%) showed late hypofluorescence. Indocyanine green angiography demonstrated early and late hyperfluorescence in all eyes.
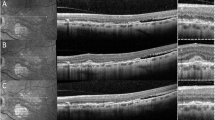
Of the 13 lesions with available follow-up data for progression evaluation, three lesions showed minimal growth over time. The change of FSN lesion size over time is demonstrated in Fig. 3. No lesion showed aggravation to the point where deterioration of vision or symptom appearance would occur. Most of the lesions (76.9%) showed no change, and no lesion showed regression.
Discussion
Earlier reports on FSN regarded it as a form of idiopathic focal choroidal inflammation and assumed that the lesion was primarily singularly located, thus the term “unifocal” (as in unifocal helioid choroiditis) or “solitary” (as in SIC) [1, 2]. What stands out in our study is that one patient had two adjacent lesions in the same eye. Although theoretically suggested, multiple lesions of FSN have never, to our knowledge, been reported before, and our result elucidates the possibility of multiple lesions in a single eye, which may help elucidating the nature of this poorly understood entity. This result was consistent with Fung AT, Waldstein SM, Gal-Or O, Pellegrini M, Preziosa C, Shields JA, et al. [4], who assumed the future possibility of multifocality despite all of their patients showing unifocal disease and therefore suggested the term “focal” rather than “solitary,” which excludes the chance of multiple lesions. Our finding reaffirms that this entity should not be described as “unifocal” or “solitary”. All lesions were confined to the sclera and displayed a dome or nodular shape on OCT, which is in agreement with recent studies that suggested the scleral origin of FSN, rather than choroid. Nearly all of the lesions showed thinning of the overlying choroidal layer with anatomical changes involving mostly the outer retina.
As the two lesions of the bifocal FSN case were adjacent to each other, it is possible that this is a single bilobed lesion, which also to our knowledge has never been described in FSN. As shown in colour fundus photo and autofluorescence photos (Fig. 2), each white area of the lesion seems to correspond mainly to RPE changes, and these manifest as two lesions. Due to limitations in available depth of OCT imaging, whether these two lesions are merged within deep sclera cannot be confirmed, with available OCT angiography image at its deepest setting possible showing two adjacent but distinct lesions. A retinal vessel does exist between two lesions, but this is likely to be coincidental as many cases of FSN lie directly underneath retinal vessels.
The pathophysiology of FSN is still poorly understood, and disputes are still ongoing about the essential nature of this entity. Some reports have suggested that FSN is an inflammatory lesion after infection, and its association with Coxsackie virus [5] or Bartonella [6] infection has been hypothesized. However, as Coxsackie virus and Bartonella infection are associated with chorioretinitis, an association with FSN, which is essentially a scleral lesion, is hardly likely. Others have suggested that these lesions are choroiditis of unknown origin, as can be speculated from the names “unifocal helioid choroiditis” or “solitary idiopathic choroiditis.” [1, 2] Shields JA, Shields CL, Demirci H and Hanovar S [2]. assumed that the lesion has active and inactive phases and showed a spectrum of cases that adheres to the requisites of active diseases, but natural course of the disease showing phase shift was not described. And it is difficult to exclude the possibility that different entities were included in the assessed lesions since the report was published before the era of OCT. Feng Y, Conrady CD and Demirci H. [7] recently showed that subretinal fluid of an “active” FSN resolved over 6 weeks without intervention, suggesting an inflammatory granulomatous reaction associated with FSN. But other recent studies did not find FSN as an inflammatory lesion. Fung AT, Waldstein SM, Gal-Or O, Pellegrini M, Preziosa C, Shields JA, et al. [4] did not report any active lesions and considered subretinal fluid to be a sign of degeneration rather than a sign of disease activity. Duignan E, O’Day R, Moloney T, Rahman W and Damato B. [8] suggested the name “idiopathic scleroma” claiming that the lesions are intrascleral without any component of inflammation. In the present study, subretinal fluid was present in four (30.8%) lesions, of which three (75%) showed an absence of the choroidal layer at the site of lesion on OCT, suggesting that subretinal fluid is associated with the attenuation of choroidal function above the lesion. OCT angiography demonstrated decreased choroidal circulation, but preservation of retinal circulation in all of five (100%) lesions was examined, further supporting the hypothesis that subretinal fluid accumulation at the FSN lesion results from weakened function of the choroidal layer above the lesions. In addition, OCT angiography revealed that FSN lesions are avascular inside. We speculate that disrupted choroidal flow results in decreased function of the RPE layer, leading to accumulation of fluid as in other entities such as dome-shaped macula [9], whose subretinal fluid can spontaneously develop or resolve without signs of inflammation.
Patients with FSN lesions were followed for a minimum of 6 months and a maximum of 139 months. Overall, only three (20.0%) lesions showed minimal growth over time, and most of the lesions stayed stable over time, with none showing regression. None of the patients complained of visual symptoms possibly due to an intact macula and no lesion evolved into the point that it became symptomatic. The stable nature and predilection for location near optic disc may suggest that these lesions are congenital. The youngest patient with FSN was 3 years old in Shields JA, Shields JA, Shields CL, Demirci H and Hanovar S.’s report [2] and our study also includes a 3-year-old patient. Fung AT, Waldstein SM, Gal-Or O, Pellegrini M, Preziosa C, Shields JA, et al. [4] reported a 12-year-old patient with FSN. Because an FSN lesion at birth or a de novo FSN lesion has yet to be reported, the question remains unclear whether FSN is congenital, acquired, or both. Since the possibility of congenitality of FSN was suggested based on the fact that FSN “typically appears very uniform in size, shape, colour, and posterior location around the optic nerve,” [4] presence of the new bifocal or bilobed lesion seems to add plausibility to the possible acquired nature of the entity. However, clinical presentations other than bifocality were in clear accordance with previously presented features of FSN, and therefore the aetiology of the entity requires further cases to be clearly deciphered.
Limitations of this study included the retrospective design, the small number of cases, and the lack of complete multimodal imaging. Although most of the patients underwent fundus imaging and OCT scans, OCT angiography was not available for all patients.
In conclusion, FSN is an entity yet to be fully understood, but our study opens up a new spectrum of understanding FSN with the first evidence of its bifocality. Further assessment by multimodal imaging of FSN lesions and studies on the natural course of the disease should help avoid misdiagnosing or overtreating patients with such lesions.
Summary
What was known before:
-
Focal scleral nodule has been commonly misdiagnosed and overtreated before its clinical description.
What this study adds:
-
Our study demonstrates that bifocal lesions of FSN in the ipsilateral eye are possible and reaffirms the relative stability of this entity.
Change history
24 August 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41433-022-02203-2
References
Hong PH, Jampol LM, Dodwell DG, Hrisomalos NF, Lyon AT. Unifocal helioid choroiditis. Arch Ophthalmol. 1997;115:1007–13.
Shields JA, Shields CL, Demirci H, Hanovar S. Solitary idiopathic choroiditis: the Richard B. Weaver lecture. Arch Ophthalmol. 2002;120:311–9.
Fung AT, Kaliki S, Shields CL, Mashayekhi A, Shields JA. Solitary idiopathic choroiditis: findings on enhanced depth imaging optical coherence tomography in 10 cases. Ophthalmology 2013;120:852–8.
Fung AT, Waldstein SM, Gal-Or O, Pellegrini M, Preziosa C, Shields JA, et al. Focal scleral nodule: a new name for solitary idiopathic choroiditis and unifocal helioid choroiditis. Ophthalmology 2020;127:1567–77.
Goyal S, Ware GT, Petrovic V. Coxsackie virus a possible missing link to unifocal helioid choroiditis? Clin Exp Ophthalmol. 2015;43:377–9.
Kumar V, Khoo CT, Shields CL. SOLITARY IDIOPATHIC CHOROIDITIS IN THE SETTING OF EXTENSIVE ANIMAL EXPOSURE. Retin Cases Brief Rep. 2016;10:386–8.
Feng Y, Conrady CD, Demirci H. The evolution of an active solitary idiopathic choroiditis (focal scleral nodule): a case report of the natural course and a review of the literature. BMC Ophthalmol. 2021;21:130.
Duignan E, O’Day R, Moloney T, Rahman W, Damato B. A case series of “solitary idiopathic choroiditis” and proposal of a nomenclature change to “idiopathic scleroma”. Ocul Oncol Pathol. 2021;7:48–53.
Errera MH, Michaelides M, Keane PA, Restori M, Paques M, Moore AT, et al. The extended clinical phenotype of dome-shaped macula. Graefes Arch Clin Exp Ophthalmol. 2014;252:499–508.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea under 2019R1A2C2002393 (CSL). The funding organization had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Contributions
Conceptualization, C.S.L; methodology, H.S.P. and C.S.L.; validation, C.S.L.; investigation, H.S.P.; data curation, H.S.P. and C.S.L.; writing—original draft preparation, H.S.P.; writing—review and editing, C.S.L.; supervision, E.Y.C., Y.J.K., S.C.L., S.H.B., S.S.K. and C.S.L.; project administration, C.S.L. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The authors have found out that the “Neurosensory layer status, no. (%)” section of Table 2 had unnecessary typos which should have been removed (Normal = 1 Absent = 0 Thinning = 2 Thickening = 3 of the previous table), and that the some of the numbers of this section did not add up to 13. The table was revised, and the result section of the manuscript describing numbers of Table 2 was changed accordingly. Two additional sentences that were found to be including faulty information of the Table 2 were revised based on the information of Table 2. Furthermore, the authors would like the figures of the manuscript to be printed in color rather than black-and-white as it is now since the figure depicts lesions of which color, shape and size can be important factors. The authors are truly sorry for the fact that we were unable to detect the errors during the publication process and the inconvenience this might cause. We thank the editors for the time to be spent on this correction process.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Park, H.S., Kim, Y.J., Choi, E.Y. et al. Expanded spectrum of focal scleral nodule: focal scleral nodules can be bifocal. Eye 37, 773–778 (2023). https://doi.org/10.1038/s41433-022-02029-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-02029-y