Abstract
Importance
Macular telangiectasia type 2 (MacTel) is bilateral disease with characteristic alterations of the macular capillary network along with decreased macular pigment in the parafoveal area. The purpose of this study was to highlight that some eyes show microvascular changes which precede any visible neuronal changes on spectral-domain optical coherence tomography (SD-OCT).
Methods
This observational study was conducted at a tertiary eye institute. From a registry of 630 patients with MacTel, we identified 4 patients with typical MacTel characteristics in only one eye with no visible changes on colour photographs or SD-OCT in the fellow eye. These 4 patients had findings of MacTel documented by colour fundus photograph, multicolour imaging (MCI), fundus autofluorescence (FAF), confocal blue reflectance (CBR), SD-OCT, and OCT-Angiography (OCT-A) in one eye. OCT-A was performed in MacTel patients using the High-resolution Spectralis (Heidelberg Engineering) module with a full-spectrum probabilistic approach and we employed a 30° x 15° (~8.8 mm × 4.4 mm) scan pattern covering the macula. MCI was done at the end so as to avoid fading the confocal blue reflectance hyperreflectivity seen in MacTel.
Results
On OCT-A, all 4 fellow eyes showed telangiectasia and foveal avascular zone changes in the superficial and deep capillary plexuses with no changes on SD-OCT. None of the eyes showed typically increased reflectance on CBR around the foveal area.
Conclusion
These findings show that microvascular changes on OCT-A may occur before visible neurodegenerative changes on OCT, providing new insights into the course of the disease.
Similar content being viewed by others
Introduction
Macular telangiectasia type 2 (MacTel or perifoveal telangiectasia) is an idiopathic condition typically characterized by a visible dilated capillary network temporal to the fovea [1, 2]. The disease usually presents in the fifth and sixth decades and affects men and women equally [3]. Patients often report a progressive but slow bilateral visual loss. Clinically, it is initially characterized by loss of foveal transparency and macular pigment loss. As the disease progresses, typical lesions become more visible such as intraretinal crystalline deposition, hyperplastic retinal pigment epithelium migration, and telangiectasia of juxtafoveolar retinal vessels and right-angle vessels. Complications include subretinal or intraretinal neovascularization and retinal atrophy, and vascular invasion of the foveal avascular zone.
On spectral-domain optical coherence tomography (SD-OCT), optically empty spaces known as cavitation appear in the inner or outer neuronal layers of the retina leading to a collapse of the inner retina. A typical OCT finding is the internal limiting membrane drape. Gass and Blodi suggested that loss of retinal transparency is considered an early sign in MacTel, being the first detectable manifestation of the disease. Reflective imaging using short-wavelength confocal blue light reflectance imaging (BLR) provides a superior tool for detecting and recording this phenomenon [4, 5].
The exact pathophysiology of MacTel is unclear and concepts on its pathogenesis have evolved over the last decade. Gass et al. first assumed the vascular basis for MacTel and he described it as a disease being caused by vascular dysfunction and subsequent hypoperfusion [2]. Powner et al. studied histopathological changes in a post-mortem sample of a donor eye with MacTel and noticed abnormal dilation in the deep retinal vascular plexus and that these altered areas suffered from Müller cell depletion, having observed markedly reduced expression of Müller cell-specific markers in the central macula [6, 7]. Subsequent studies using multimodal imaging modalities suggested that MacTel is a neurodegenerative disorder [8, 9]. Although the pathophysiology of MacTel is not clearly understood, there is a consensus that the primary pathology is dysfunction or degeneration of Müller cells resulting in photoreceptor cell loss and decrease in macular pigment with secondary vascular changes [3].
Methods
This observational study was conducted in a tertiary care referral eye institute in South India. From a registry of 630 patients with MacTel, we included 4 patients in our study, which met inclusion and exclusion criteria. Eyes of participants with a confirmed diagnosis of MacTel in one eye were included in the study and we evaluated the clinically normal fellow eyes for early features of MacTel by multimodal imaging. We excluded the eyes with pre-existing vascular co-morbidities and in whom adequate multimodal imaging was not performed. The imaging modalities included SD-OCT, Confocal blue-light reflectance (CBR), Fundus autofluorescence (FAF), and OCT-A.
The images were acquired by trained retina specialists. Colour fundus photos were taken using the Carl Zeiss (FF 450 plus IR) fundus photo machine. Spectral-domain OCT volume scans of the retina were acquired using the Heidelberg Spectralis OCT machine (Heidelberg Engineering HRA, Heidelberg, Germany). Raster volume scans (in detail scan mode) covered a retinal area of at least 15 × 10 degrees in size, centred on the fovea, with a maximum B-scan interval of 30 microns. Multicolour imaging (MCI) with its component infrared (820 nm), green (518 nm), and blue (488 nm) light reflectance images were done at the end so as to avoid fading the confocal blue reflectance hyperreflectivity seen in MacTel. Confocal blue light reflectance images were acquired separately using a Heidelberg Spectralis system (Heidelberg Engineering, Heidelberg, Germany) employing the ‘red free’ setting (488 nm blue laser). En-face OCT-A imaging was done using the high-resolution Heidelberg Spectralis OCT module (Heidelberg Engineering, Heidelberg, Germany) with a full-spectrum probabilistic approach. Images were recorded with a 30 × 15° angle using the high-resolution mode resulting in a retinal section of 8.8 × 4.4 mm. We analysed changes in the superficial vascular plexus (SVP), deep capillary plexus (DCP), and avascular complex as well as the foveal avascular zones (FAZs) in SVP and DCP.
Results
The study included 8 eyes of 4 patients. The mean age of the 3 females (75%) and 1 male (25%) were 52.75 ± 8.25. The involved eye was the right eye (100%) in all our cases. Visual acuity ranged between 20/20 to 20/40 in eyes with established MacTel, while it was 20/20 in the fellow eyes. The 4 eyes with established MacTel had a nonproliferative disease.
In all our cases, we analysed fellow eyes of patients with asymmetric MacTel and found out that early changes of MacTel start in the microvascular layers of the retina in the absence of any neuronal changes visible on OCT. We present 4 cases where microvascular changes are seen on OCT-A in the absence of any clinically detectable neurodegenerative changes in patients with asymmetric MacTel.
The multimodal imaging features along with the changes in OCT-A of each of the 4 cases is described in detail. Table 1 gives a summary of the SD-OCT and OCT-A features of the 4 cases.
Case 1
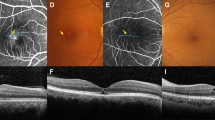
A 59-year-old woman presented with a blurring of vision in the right eye for 5 months. She had a history of ischemic heart disease on treatment. There was no history of ocular surgery or trauma. Her family history was also unremarkable. Best-corrected visual acuity was 20/30 in the right eye and 20/20 in the left eye. Colour fundus photography of the right eye showed a depigmented lesion at the fovea [Fig. 1-A (a)], while the left eye [Fig. 1-B (a)] was unremarkable. Confocal blue reflectance (CBR) showed a small area of increased reflectance within the foveal area in the left eye [Fig. 1-B (b)]. SD-OCT of the right eye showed a small inner retinal cavitation with ellipsoid zone (EZ) disruption at fovea [Fig. 1-A (c)], while OCT of the left eye was normal [Fig. 1-B (c)]. OCT-A of the right eye demonstrated telangiectatic vessels in the deep capillary plexus predominantly temporal to fovea [Fig. 1-A (e)], with the superficial vascular plexus [(Fig. 1-A (d)] being normal. OCT-A of the left eye showed few telangiectatic vessels in the deep capillary plexus [(Fig. 1-B (e)], while the superficial vascular plexus [(Fig. 1-B (d)] showed rarefaction of vessels temporally.
Figure 1-A Multimodal imaging features are described in the right eye of patient 1. Colour fundus photography: yellowish-white lesion at the fovea (a). Confocal blue reflectance: hypo reflective lesion at fovea surrounded by hyper-reflective halo temporal to it (b). Spectral-domain Optical Coherence Tomography (SD-OCT): inner retinal cavitation with ellipsoid zone (EZ) disruption at the fovea (c). Optical Coherence Tomography Angiography (OCT-A) superficial vascular plexus: normal (d); OCT-A deep capillary plexus: telangiectatic vessels, predominantly temporal to fovea [red square (e)] with rarefaction of vessels around the foveal avascular zone. Figure 1-B Multimodal imaging findings are described in the left eye of patient 1. Colour fundus photography: normal (a). Confocal blue reflectance image: unremarkable (b). Spectral-domain Optical Coherence Tomography (SD-OCT) sections taken through the macula: normal (c). Optical Coherence Tomography Angiography (OCT-A) superficial vascular plexus: rarefaction of vessels temporally [yellow arrows (d)]; OCT-A deep capillary plexus: few telangiectatic vessels [red squares (e)] with rarefaction of vessels temporal edge of the foveal avascular zone.
Case 2
A 40-year-old woman presented with a blurring of vision in the right eye for 1 year. Her medical history was significant for dyslipidaemia and spondyloarthritis on treatment. There was no history of ocular surgery or trauma. Her family history was unremarkable. The best-corrected visual acuity was 20/20 in both eyes. Multicolour photo of the right eye showed right angle dipping of vessel [Fig. 2-A (a)], while the left eye was unremarkable [Fig. 2-B (a)]. An increased circular area of parafoveal reflectance on CBR was seen in the right eye [more prominent temporally, Fig. 2-A (b)] compared to the left eye which showed small hyper-reflectance at the foveal centre [Fig. 2-B (b)]. SD-OCT of the right eye showed inner retinal cavitations and ellipsoid zone (EZ) disruption at the fovea with the collapse of outer retinal layers [Fig. 2-A (c)], while OCT of the left eye was normal [Fig. 2-B (c)]. OCT-A of the right eye showed telangiectatic vessels in both deep and superficial capillary plexus [Fig. 2-A (d)]. OCT-A of the left eye showed telangiectatic vessels (arranged linearly) temporal to the fovea in deep capillary plexus [Fig. 2-B (e)], while the superficial vascular plexus demonstrated rarefaction of vessels temporally [Fig. 2-B (d)].
Figure 2-A Multimodal imaging findings are described in the right eye of patient 2. Multicolour fundus photograph: crystalline and greyish deposits at the fovea with right angle dipping of a vessel [black arrow (a)] with a perifoveal white halo. Confocal blue reflectance: increased parafoveal reflectance, predominantly seen temporally (b). Spectral-domain Optical Coherence Tomography (SD-OCT): inner retinal cavitations with Ellipsoid Zone (EZ) disruption at the fovea with the collapse of outer retinal layers (c). Optical Coherence Tomography Angiography (OCT-A): telangiectatic vessels in superficial vascular [encircled yellow, (d)] and deep capillary plexuses [red squares, (e)]. Figure 2-B Multimodal imaging findings are described in the left eye of patient 2. Multicolour fundus photograph: unremarkable (a). Confocal blue reflectance: normal (b). Spectral-domain Optical Coherence Tomography (SD-OCT) sections through the macula: normal (c). Optical Coherence Tomography Angiography (OCT-A) superficial vascular plexus: rarefaction of vessels temporally [yellow arrows (d)]; OCT-A deep capillary plexus: telangiectatic vessels (arranged linearly) temporal to fovea [red arrows (e)]. Figure 2-C Multimodal imaging findings of the left eye at 3 years of follow-up. Multicolour fundus photograph: greyish discoloration parafoveally (a). Confocal blue reflectance: increased parafoveal reflectance temporally (b). Spectral-domain Optical Coherence Tomography (SD-OCT) sections through the macula: small inner retinal cavitation situated immediately above the outer retinal cavitation at the fovea with ellipsoid zone loss (c). Optical Coherence Tomography Angiography (OCT-A) superficial vascular plexus: increase in the rarefaction of vessels temporally [yellow arrows (d)]; OCT-A deep capillary plexus: telangiectatic vessels (arranged linearly) temporal to fovea [red arrows (e)] with an increase in the rarefaction of vessels temporally.
We followed up with the patient after 3 years and performed a repeat multimodal imaging which showed progression of MacTel changes in the left eye, while the right eye remained similar as the last visit. Multicolour photo of the left eye showed greyish discoloration parafoveally [Fig. 2-C (a)]. CBR of the left eye showed increased parafoveal reflectance temporally [Fig. 2-C (b)] and extending superiorly along with increased reflectance immediately temporal to the foveal centre. SD-OCT of the left eye showed a small inner retinal cavitation and outer retinal cavitation at the fovea disrupting the ellipsoid zone but sparing the external limiting membrane (ELM) [Fig. 2-C (c)]. The OCT-A of the left eye demonstrated telangiectatic vessels in the deep capillary plexus with an increase in the rarefaction of vessels temporally in superficial and deep plexuses compared to the last visit [Fig. 2-C (d), (e)].
Case 3
A 61-year-old male complained of blurring of vision in the right eye for 1 year. His past medical history was significant for ischemic heart disease. There was no history of ocular surgery or trauma. His family history was also unremarkable. Best-corrected visual acuity was 20/40 in the right eye and 20/20 in the left eye. Colour fundus photography of the right eye showed loss of retinal transparency with early pigmentation temporal to fovea [Fig. 3-A (a)], while the left eye was unremarkable [Fig. 3-B (a)]. SD-OCT of the right eye showed inner retinal cavitations, EZ loss at the fovea, the collapse of outer retinal layers, and pigment clumping [Fig. 3-A (b)], while SD-OCT of the left eye was normal [Fig. 3-B (b)]. Confocal blue reflectance was not done for this patient. OCT-A of the right eye showed telangiectatic vessels in deep capillary plexus with an enlarged foveal avascular zone (horizontal diameter 1359 µ) and areas of vessel rarefaction perifoveally in superficial vascular and deep capillary plexuses [Fig. 3-A (c, d)]. OCT-A of the left eye demonstrated telangiectatic vessels in the deep capillary plexus [Fig. 3-B (d)] with telangiectasia and right angle dipping of vessels in the superficial vascular plexus [(Fig. 3-B (c)].
Figure 3-A Multimodal imaging characteristics are described in the right eye of patient 3. Colour fundus photography: dark greyish lesions at the fovea (a). Spectral-domain Optical Coherence Tomography (SD-OCT): inner retinal cavitations with Ellipsoid Zone (EZ) loss at the fovea, the collapse of outer retinal layers, and retinal pigment epithelial (RPE) clumping (b). Optical Coherence Tomography Angiography (OCT-A) superficial vascular plexus: rarefaction of vessels [yellow arrow (c)] perifoveally; OCT-A deep capillary plexus: telangiectatic vessels (red squares), rarefaction of vessels (red arrows), and an enlarged foveal avascular zone (d). Figure 3-B Multimodal imaging characteristics are described in the left eye of patient 3. Colour fundus photography: normal (a). Spectral-domain Optical Coherence Tomography (SD-OCT): normal (b). Optical Coherence Tomography Angiography (OCT-A) superficial vascular plexus: Telangiectatic vessel with right angle dipping of vein [encircled yellow, (c)]; OCT-A deep capillary plexus: telangiectatic vessels [red squares, (d)].
Case 4
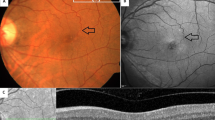
A 51-year-old woman presented with a blurring of vision in the right eye for 3 weeks. Her medical history was significant for hypothyroidism. There was no history of ocular surgery or trauma. Her family history was unremarkable. Best-corrected visual acuity was 20/30 in the right eye and 20/20 in the left eye. The fundus of the right eye showed broad foveal reflex while the left eye was normal. Fundus autofluorescence (FAF) of the right eye showed right angle dipping of a vessel with increased autofluorescence at fovea [Fig. 4-A (a)], while FAF of the left eye was normal [Fig. 4-B (a)]. SD-OCT of the right eye showed outer retinal cavitation at the fovea [Fig. 4-A (b)], while SD-OCT of the left eye was normal [Fig. 4-B (b)]. Confocal blue reflectance was not done for this patient. OCT-A of the right macula showed telangiectasia (predominantly temporally) in the deep capillary plexus [Fig. 4-A (d)], while the superficial vascular plexus showed right angle dipping of vessel [Fig. 4-A (c)]. OCT-A of the left eye showed few telangiectatic vessels temporal to the fovea in the deep capillary plexus [Fig. 4-B (d)], while the superficial vascular plexus showed right angle dipping of vessel [Fig. 4-B (c)].
Figure 4-A Multimodal imaging findings are described in the right eye of patient 4. Fundus autofluorescence (FAF): Increased autofluorescence at the fovea with right angle dipping of vessel [Yellow arrows (a)]. Spectral-domain Optical Coherence Tomography (SD-OCT): outer retinal cavitation at the fovea (b). OCT-A superficial vascular plexus: right angle dipping of vessel [encircled yellow, (c)]; OCT-A deep capillary plexus: few telangiectatic vessels temporally [Red squares (d)]. Figure 4-B Multimodal imaging findings are described in the left eye of patient 4. FAF: unremarkable (a). Spectral-domain Optical Coherence Tomography (SD-OCT) sections taken through macula: normal (b). OCT-A superficial vascular plexus right angle dipping of a vessel [encircled yellow, (c)]; OCT-A deep capillary plexus: few telangiectatic vessels temporal to fovea [Red squares, (d)].
Summary of the results
As described in the above 4 cases, we analysed patients with very asymmetric MacTel. The right eye was the eye with established MacTel while the left eye was the fellow eye in all 4 cases. The fellow eyes were those which had normal fundus and SD-OCT findings with no parafoveal CBR reflectance changes. It was on OCT-A that we were able to notice changes of MacTel in the fellow eyes. Each of the fellow eyes showed a characteristic pattern of microvascular changes on OCT-A with telangiectasias in the deep capillary plexus. We also noticed changes in the superficial vascular plexus of the fellow eye of patients 3 and 4 in the form of right angle dipping of a vessel which did not correspond to any changes on SD-OCT.
Discussion
OCT-A enables visualization of retinal and choroidal circulations and enables the study of both the superficial and deep retinal vascular plexus [10]. Thorell et al. were the first to employ the use of OCT-A to study microvascular changes in MacTel [11]. Spaide et al. suggested that more prominent alterations on OCT-A such as dilation and telangiectasias in early stages to retinal thinning and ectasia in the advanced stages of the disease occur in the deep retinal capillary plexus [12]. Zeimer et al. used OCT-A to image patients with MacTel and noted reduced vascular density in the superficial vascular plexus and dilatation and telangiectasias in the deep capillary plexus associated with vascular proliferation of the outer retina [13].
As per the Gass and Blodi staging which analysed morphological findings of MacTel based only on fundoscopy and FFA, the presence of right angle vessels (RAV’s) on fundoscopy was considered a sign of advanced disease (stage 3 out of 5) [2]. Tzaridis et al. employed OCT-A to show that RAV’s could be detected in early disease (stages 1 to 2) even before they became visible in fundoscopy. In their study, RAV’s in the SVC and DCP of OCT-A corresponded to intraretinal hyper-reflective lesions in the outer retina on SD-OCT even in eyes with well preserved retinal architecture [14]. In our study, we detected RAV’s in the SVC of OCT-A in the fellow eye of 2 patients (patients 3 and 4), but no correlation between RAV’s and SD-OCT changes in the fellow eyes were seen.
Confocal blue reflectance (CBR) imaging using the confocal scanning laser ophthalmoscope (cSLO; HRA2, Heidelberg Engineering, Heidelberg, Germany) has been a valuable tool for the diagnosis of MacTel. CBR was performed on two of our patients. Increased parafoveal reflectance on CBR was observed in the eyes with established MacTel. However, none of the fellow eyes showed similar parafoveal reflectance changes on CBR. Instead, a small area of foveal hyper-reflectance involving the temporal edge on CBR was observed in both cases and these did not correlate to the microvascular changes. This finding could not be attributed to abnormal metabolic handling of luteal pigment in the macula in early disease. Central blue light‐induced fundus autofluorescence (FAF) and confocal blue light fundus reflectance (CBLR) are thought to better delineate the distribution of macular pigments. In previous studies, Dual (488 + 512 nm) wavelength autofluorescence (DWAF) imaging have demonstrated a redistribution of macular pigment (lutein) in a specific pattern which starts with a wedge-shaped depletion of pigment just temporal to the centre of the fovea, which progresses centripetally to involve the foveal centre, finally encompassing an oval area centred on the fovea, with a band of increased pigment density along with its boundaries [15,16,17].
We did not have access to DWAF or devices to measure macular pigment loss or photoreceptor loss. Reports using adaptive optics scanning light ophthalmoscopy (AOSLO) have shown early cone photoreceptor loss. Ooto et al. [18] noted a decrease in parafoveal cone count while Song et al. [19] observed a foveal decrease in cones. All our patients with early microvascular changes in the apparently normal eyes also had perfect visual acuity. We can only hypothesise that there may be clinically invisible dysfunction of the Müller cells and/or photoreceptor cells in these eyes.
As deep vascular plexus contribute nourishment to the photoreceptors and the Müller cells, our findings suggest a possible primary pathology of the deep vascular plexus in MacTel. Similar changes in the deep vascular plexus has been observed in one of the cases reported by Hilal Nalcı et al. [20]. Charbel Issa et al. detected foveal pit asymmetry and temporal foveal thinning as being associated with very early disease [21]. These eyes did not have OCTA. In a study by Alex D et al. temporal retinal thinning and the presence of hyperreflective outer retinal dots in the fellow eye on SD-OCT were considered as being the earliest signs of MacTel [22]. In our study eyes, there was a widening of foveal contour in few apparently normal eyes but no obvious foveal pit asymmetry or other abnormalities on OCT, suggesting that changes in deep plexus precede thinning of temporal parafoveal area. Our observations suggest that vascular changes in the deep capillary plexus occur very early in MacTel.
Conclusion
Early microvascular changes in MacTel occur in deep retinal capillary plexus before any clinically visible OCT changes suggestive of neuronal or glial abnormalities. Further studies with larger samples in early MacTel utilising OCTA, AOSLO, and macular pigment measurements are required to decipher the relationship between microvascular and neuroglial changes in MacTel.
SUMMARY
What was known before
-
In the published literature, OCTA features in eyes with established MacTel have been studied but there is limited information on OCTA changes occurring in very early MacTel.
What this study adds
-
Since the incidence of unilateral MacTel is small, we were able to collect a limited number of cases from our large MacTel repository.
-
In that, we were able to find out that vascular changes in the deep capillary plexus occur very early in MacTel.
-
These vascular changes were seen before any visible neurodegenerative changes on SD-OCT.
References
Yannuzzi LA, Bardal AM, Freund KB, Chen KJ, Eandi CM, Blodi B. Idiopathic macular telangiectasia. Arch Ophthalmol. 2006;124:450–60.
Gass JD, Blodi BA. Idiopathic juxtafoveolar retinal telangiectasis. Update classification follow- study Ophthalmol. 1993;100:1536–46.
Wu L, Evans T, Arevalo JF. Idiopathic macular telangiectasia type 2 (idiopathic juxtafoveolar retinal telangiectasis type 2A, Mac Tel 2). Surv Ophthalmol. 2013;58:536–59.
Charbel Issa P, Finger RP, Helb HM, Holz FG, Scholl HP. A new diagnostic approach in patients with type 2 macular telangiectasia: Confocal reflectance imaging. Acta Ophthalmol. 2008;86:464–5.
Charbel Issa P, Berendschot TT, Staurenghi G, Holz FG, Scholl HP. Confocal blue reflectance imaging in type 2 idiopathic macular telangiectasia. Invest Ophthalmol Vis Sci. 2008;49:1172–7.
Powner MB, Gillies MC, Tretiach M, Scott A, Guymer RH, Hageman GS, et al. Perifoveal Müller cell depletion in a case of macular telangiectasia type 2. Ophthalmology 2010;117:2407–16.
Powner MB, Gillies MC, Zhu M, Vevis K, Hunyor AP, Fruttiger M. Loss of Müller’s cells and photoreceptors in macular telangiectasia type 2. Ophthalmology. 2013;120:2344–52.
Wu Lihteh. Multimodality imaging in macular telangiectasia 2. Indian J Ophthalmol. 2015;63:394–8.
Spaide RF, Klancnik JM, Cooney MJ. Retinal vascular layers in macular telangiectasia type 2 imaged by optical coherence tomographic angiography. JAMA Ophthalmol. 2015;133:66–73.
Yali Jia, Ou Tan, Jason Tokayer, Benjamin Potsaid, Yimin Wang, Jonathan JLiu, et al. “Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt Express. 2012;20:4710–25.
Thorell MR, Zhang Q, Huang Y, An L, Durbin MK, Laron M, et al. Swept-source OCT angiography of macular telangiectasia type 2. Ophthalmic Surg Lasers Imaging. Retina. 2014;45:369–80.
Spaide RF, Klancnik JM, Cooney MJ. Retinal Vascular Layers imaged by fluorescein angiography and Optical Coherence Tomographic Angiography. JAMA Ophthalmol. 2015;133:45–50.
Zeimer M, Gutfleisch M, Heimes B, Spital G, Lommatzsch A, Pauleikhoff D. Association between changes in macular vasculature in optical coherence tomography and fluorescein-angiography and distribution of macular pigment in type-2 idiopathic macular telangiectasia. Retina. 2015;35:2307–16.
Tzaridis S, Heeren T, Mai C, Thiele S, Holz FG, Charbel Issa P, et al. Right-angled vessels in macular telangiectasia type 2. Br J Ophthalmol. 2019;0:1–8.
Helb HM, Charbel Issa P, Van der Veen RL, Berendschot TT, Scholl HP, Holz FG, et al. Abnormal macular pigment distribution in type 2 idiopathic macular telangiectasia. Retina. 2008;28:808–16.
Zeimer MB, Padge B, Heimes B, Pauleikhoff D. Idiopathic macular telangiectasia type 2: distribution of macular pigment and functional investigations. Retina. 2010;30:586–95.
Esposti SD, Egan C, Bunce C, Moreland JD, Bird AC, Robson AG, et al. Macular pigment parameters in patients with macular telangiectasia (MacTel) and normal subjects: implications of a novel analysis. Invest Ophthalmol Vis Sci. 2012;53:6568–75.
Ooto S, Hangai M, Takayama K, Arakawa N, Tsujikawa A, Koizumi H, et al. High-resolution photoreceptor imaging in idiopathic macular telangiectasia type 2 using adaptive optics scanning laser ophthalmoscopy. Invest Ophthalmol Vis Sci. 2011;52:5541–50.
Song H, Rossi EA, Williams DR. Reduced foveal cone density in early idiopathic macular telangiectasia. BMJ Open Ophthalmol. 2021;6:e000603.
Nalcı H, Şermet F, Demirel S, Özmert E. Optic coherence angiography findings in type-2 macular telangiectasia. Turk J Ophthalmol. 2017;47:279–84.
Charbel Issa P, Heeren TF, Kupitz EH, Holz FG, Berendschot TT. Very early disease manifestations of macular telangiectasia Type 2. Retina. 2016;36:524–34.
Alex D, Giridhar A, Gopalakrishnan M, Manayath G, Amar S, Raman R, et al. Early spectral-domain optical coherence tomography biomarkers to confirm fellow eye changes in asymmetric type-2 macular telangiectasia: a case-control study (India Macular Telangiectasia Report 1). Retina. 2021;41:471–9.
Acknowledgements
This work was funded by Global Challenges Research Fund and UK Research and Innovation through the Medical Research Council grant number MR/P027881/1.
Author information
Authors and Affiliations
Contributions
The four authors are justifiably credited with authorship, according to the current ICMJE criteria for authorship. KC, AG, MG and SS conceived the study. KC was involved in the multimodal imaging, design, analysis, interpretation of data and drafting of the manuscript; AG and MG provided guidance and expert inputs during manuscript writing and also helped in critical revisions of the manuscript; SS co-ordinated and mentored the study and helped in critical manuscript revisions. All authors reviewed the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chandran, K., Giridhar, A., Gopalakrishnan, M. et al. Microvascular changes precede visible neurodegeneration in fellow eyes of patients with asymmetric type 2 macular telangiectasia. Eye 36, 1623–1630 (2022). https://doi.org/10.1038/s41433-021-01699-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01699-4