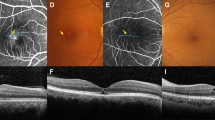
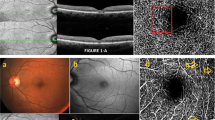
Abstract
Background
To study the relation between interdigitation zone (IDZ) and right-angle vessel (RAV) in Macular Telangiectasia Type-2 (MacTel).
Methods
A total of 43 eyes of 38 patients with presence of definite RAV on colour fundus photograph (Gass and Blodi-only stage-3) were confirmed on multimodal imaging. The relation of IDZ changes and associated ellipsoid zone (EZ) alterations on spectral-domain optical coherence tomography (OCT) with RAV were studied at baseline and these OCT changes were followed up in 15 eyes over a mean of 3.86 years.
Results
A total of 58 RAVs were found in the 43 eyes and 53/58 (91.3%) RAVs were associated with IDZ alterations in 39 eyes. On follow-up, IDZ attenuation progressed to IDZ loss and subsequent EZ attenuation and loss. A pre-existing IDZ loss was associated with subsequent EZ defect (P = 0.002). In 36 eyes that had OCT angiography, eyes with RAV showed deep capillary plexus telangiectasia in all 36 (100%) eyes and 32/36 (89%) eyes showed IDZ changes with or without EZ loss.
Conclusions
IDZ attenuation and/or loss are associated with RAV and may serve as predictor of EZ loss in MacTel.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this article (and its supplementary information files).
References
Gass JD, Blodi BA. Idiopathic juxtafoveolar retinal telangiectasis. Update classification follow- study Ophthalmol. 1993;100:1536–46.
Zandi R, Song J, Micevych PS, Fawzi AA. Topographic Relationship between Telangiectasia and Cone Mosaic Disruption in Macular Telangiectasia Type 2. J. Clin. Med. 2020;9:3149.
Chandran K, Giridhar A, Gopalakrishnan M, Sivaprasad S Microvascular changes precede visible neurodegeneration in fellow eyes of patients with asymmetric type 2 macular telangiectasia. Eye (Lond). 2021. https://doi.org/10.1038/s41433-021-01699-4.
Powner MB, Gillies MC, Tretiach M, Scott A, Guymer RH, Hageman GS, et al. Perifoveal Müller cell depletion in a case of macular telangiectasia type 2. Ophthalmology 2010;117:2407–16.
Powner MB, Gillies MC, Zhu M, Vevis K, Hunyor AP, Fruttiger M. Loss of Müller’s cells and photoreceptors in macular telangiectasia type 2. Ophthalmology 2013;120:2344–52.
Sallo FB, Peto T, Egan C, Wolf-Schnurrbusch UE, Clemons TE, Gillies MC, et al. “En face” OCT imaging of the IS/OS junction line in type 2 idiopathic macular telangiectasia. Investigative Ophthalmol Vis Sci. 2012;53:6145–52.
Pauleikhoff D, Gunnemann F, Book M, Rothaus K. Progression of vascular changes in macular telangiectasia type 2: Comparison between SD-OCT and OCT angiography. Graefes Arch Clin Exp Ophthalmol. 2019;257:1381–92.
Peto T, Heeren TFC, Clemons TE, Sallo FB, Leung I, Chew EY. et al.Correlation of clinical and structural progression with visual acuity loss in Macular Telangiectasia type 2: MacTel Project Report No. 6-The MacTel Research Group.Retina (Philadelphia. Pa). 2018;38:S8–S13.
Gaudric A, Krivosic V, Tadayoni R. Outer retina capillary invasion and ellipsoid zone loss in Macular Telangiectasia type 2 imaged by optical coherence tomography angiography. Retina 2015;35:2300–6.
Ong JX, Zandi R, Fawzi AA. Early-stage macular telangiectasia type 2 vascular abnormalities are associated with interdigitation zone disruption. PLoS One. 2021;16:e0259811.
Jonnal RS, Kocaoglu OP, Zawadzki RJ, Lee SH, Werner JS, Miller DT. The cellular origins of the outer retinal bands in optical coherence tomography images. Invest Ophthalmol Vis Sci. 2014;55:7904–18.
Spaide RF, Curcio CA. Anatomical correlates to the bands seen in the outer retina by optical coherence tomography: literature review and model. Retina 2011;31:1609–19.
Anderson DH, Stern WH, Fisher SK, Erickson PA, Borgula GA. Retinal detachment in the cat: the pigment epithelial-photoreceptor interface. Invest Ophthalmol Vis Sci. 1983;24:906–26.
Fisher SK, Lewis GP, Linberg KA, Barawid E, Verardo MR. Cellular Remodelling in Mammalian Retina Induced by Retinal Detachment. In: Kolb H, Fernandez E, Nelson R, editors. Webvision: The Organization of the Retina and Visual System. Salt Lake City (UT)1995.
Hagiwara A, Mitamura Y, Kumagai K, Baba T, Yamamoto S. Photoreceptor impairment on optical coherence tomographic images in patients with retinitis pigmentosa. Br J Ophthalmol. 2013;97:237–8.
Tsunoda K, Fujinami K, Miyake Y. Selective abnormality of cone outer segment tip line in acute zonal occult outer retinopathy as observed by spectral-domain optical coherence tomography. Arch Ophthalmol. 2011;129:1099–101.
Hirota K, Itoh Y, Rii T, Inoue M, Hirakata A. Correlation between foveal interdigitation zone band defect and visual acuity after surgery for macular pseudohole. Retina 2015;35:908–14.
Tzaridis S, Heeren T, Mai C, Thiele S, Holz FG, Charbel Issa P. Right-angled vessels in macular telangiectasia type 2. Br J Ophthalmol. 2021;105:1289–96.
Charbel Issa P, Gillies MC, Chew EY, Bird AC, Heeren TF, Peto T, et al. Macular telangiectasia type 2. Prog Retin Eye Res. 2013;34:49–77.
Sallo FB, Leung I, Zeimer M, Clemons TE, Dubis AM, Fruttiger M, et al. MacTel Study Group. Abnormal retinal reflectivity to short-wavelength light in type 2 idiopathic macular telangiectasia. Retina 2018;38:S79–S88.
Nalcı H, Şermet F, Demirel S, Özmert E. Optical coherence angiography findings in type 2 Macular Telangiectasia. Turkish J Ophthalmol. 2017;47:279–84.
Chidambara L, Gadde SG, Yadav NK, Jayadev C, Bhanushali D, Appaji AM, et al. Characteristics and quantification of vascular changes in macular telangiectasia type 2 on optical coherence tomography angiography. Br J Ophthalmol. 2016;100:1482–8.
Spaide RF, Klancnik JM, Cooney MJ. Retinal vascular layers in macular telangiectasia type 2 imaged by optical coherence tomographic angiography. JAMA Ophthalmol. 2015;133:66–73.
Hu W, Jiang A, Liang J, Meng H, Chang B, Gao H, et al. Expression of VLDLR in the retina and evolution of subretinal neovascularization in the knockout mouse model’s retinal angiomatous proliferation. Investigative Ophthalmol Vis Sci. 2008;49:407–15.
Hasegawa E, Sweigard H, Husain D, Olivares AM, Chang B, Smith KE, et al. Characterization of a spontaneous retinal neovascular mouse model. PLoS One. 2014;9:e106507.
Zhao M, Andrieu-Soler C, Kowalczuk L, Paz Cortes M, Berdugo M, Dernigoghossian M, et al. A new CRB1 rat mutation links Muller glial cells to retinal telangiectasia. J Neurosci: Off J Soc Neurosci. 2015;35:6093–106.
Luo L, Uehara H, Zhang X, Das SK, Olsen T, Holt D, et al. Photoreceptor avascular privilege is shielded by soluble VEGF receptor-1. eLife 2013;2:e00324.
Micevych PS, Lee HE, Fawzi AA. Overlap between telangiectasia and photoreceptor loss increases with progression of macular telangiectasia type 2. PLoS One. 2019;14:e0224393.
Jacob J, Paques M, Krivosic V, Dupas B, Couturier A, Kulcsar C, et al. Meaning of visualizing retinal cone mosaic on adaptive optics images. Am J Ophthalmol. 2015;159:118–123.e1.
Scoles D, Flatter JA, Cooper RF, Langlo CS, Robison S, Neitz M, et al. Assessing Photoreceptor Structure Associated with Ellipsoid Zone Disruptions Visualized with Optical Coherence Tomography. Retina 2016;36:91–103.
Litts KM, Okada M, Heeren TFC, Kalitzeos A, Rocco V, Mastey RR, et al. Longitudinal assessment of remnant foveal cone structure in a case series of early macular telangiectasia type 2. Transl Vis Sci Technol. 2020;9:27.
Funding
This work was funded by the Global Challenges Research Fund and UK Research and Innovation through the Medical Council Grant number MR/P027881/1.
Author information
Authors and Affiliations
Contributions
The four authors are justifiably credited with authorship, according to the current ICMJE criteria for authorship. KC, AG, MG and SS conceived the study. KC was involved in the multimodal imaging, design, analysis, interpretation of data and drafting of manuscript. AG and MG provided guidance and expert inputs during manuscript writing and also helped in critical revisions of the manuscript. SS co-ordinated and mentored the study and helped in critical manuscript revisions. All authors reviewed the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
SS reports consultancy and payments for lectures from Bayer, Boehringer Ingelheim, Novartis, Oxurion, Roche, Allergan, Apellis, outside the submitted work. The remaining authors have no conflicting interests to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chandran, K., Giridhar, A., Gopalakrishnan, M. et al. Relation of interdigitation zone changes and right-angle vessels in Macular Telangiectasia Type-2 (MacTel). Eye 37, 2977–2986 (2023). https://doi.org/10.1038/s41433-023-02453-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-023-02453-8