Abstract
Objectives
This study evaluated the efficacy of intravitreal bevacizumab (IVB) monotherapy for aggressive posterior retinopathy of prematurity (APROP) and Type 1 retinopathy of prematurity (ROP), along with recurrence rates and treatment outcomes for recurrences.
Methods
This retrospective cohort study reviewed the records of infants with ROP (Type 1 and APROP), who received IVB treatment between March 2013 and February 2018.
Results
A total of 257 eyes from 130 cases (unilateral eyes in three cases) were included. Cases were followed for 121.7 ± 45.7 weeks (range: 70–260 weeks). Recurrence requiring treatment was determined in 14.8% of all eyes at a mean of 9.6 ± 2.7 weeks (range: 6–15 weeks) after initial treatment and a mean of 42.3 ± 2.2 weeks (range: 38–48 weeks) postmenstrual age. Recurrence requiring treatment was observed in 20.8% of APROP and 5.8% of Type 1 ROP eyes at a statistically significant difference (p = 0.001). Persistent avascular areas were found in 54 eyes (25.8%) at the corrected age of 1 year, and prophylactic laser treatment was applied. This was statistically significantly higher in APROP (38.6%) than in Type 1 ROP (10.5%) (p < 0.001). An unfavourable structural outcome (progression to retinal detachment) occurred in one eye (0.4%), which developed insufficient regression and progression.
Conclusions
IVB monotherapy is effective for APROP and Type 1 ROP with Zone 1 and posterior Zone 2 localisation. However, because of recurrences requiring treatment and persistent peripheral avascular areas, severe, late complications must be considered, and follow-up examinations must be made. Prophylactic laser treatment for persistent avascular areas seems effective for minimising long-term complications.
Similar content being viewed by others
Introduction
Retinopathy of prematurity (ROP) is a vasoproliferative disease characterised by abnormal development of the immature retina [1, 2]. Despite ongoing developments in neonatal and ophthalmic care, ROP remains the leading cause of blindness in childhood [3].
In the 1990s, laser photocoagulation (LP) became the primary ROP treatment method, and it has been reported to be effective in more than 90% of cases [4]. However, in aggressive posterior retinopathy of prematurity (APROP) and ROP cases with Zone 1 localisation, high rates of insufficient regression, negative structural outcomes, recurrences requiring treatment and refractive defects have been reported after LP treatment [5,6,7,8].
Vascular endothelial growth factor (VEGF) is a strong pro-angiogenic factor in the pathological angiogenesis of ROP [9]. Therefore, it has increasingly been used in ROP treatment. Anti-VEGF injections have several advantages. They produce rapid clinical responses, are less troublesome than laser treatment, eliminate anaesthesia risks in infants and maintain the vascular development of the immature retina [10, 11]. Bevacizumab was the first anti-VEGF drug used for this purpose. The 2011 Bevacizumab Eliminates the Angiogenic Threat of ROP (BEATROP) study—which was the first prospective, randomised, controlled comparison of intravitreal bevacizumab (IVB) with conventional laser treatment—reports that IVB is more effective than laser treatment, especially in cases with Zone 1 localisation [7].
However, unexpected and delayed recurrences, observed in the follow-ups of infants treated with IVB monotherapy, have recently become a serious problem [12]. Due to the possibility of long-term sequelae following IVB treatment for ROP, early diagnosis and treatment of recurrence or treatment failure are of utmost importance during follow-up. Therefore, this study evaluated the efficacy of IVB treatment for ROP, as well as the rate and outcome of recurrences.
Materials and methods
This two-centre, non-randomised, retrospective study was conducted in the Ophthalmology Department of the School of Medicine at Kahramanmaras Sutcu Imam University and the Ophthalmology Department at Gaziantep Children’s Hospital, which are reference centres for ROP screening and treatment. The study was approved by the Local Ethics Committee of Kahramanmaras Sutcu Imam University, and all procedures complied with the Declaration of Helsinki. The parents of all studied infants were informed about the effects of the IVB injections and the possible ocular and systemic side effects, and their written informed consent was obtained before each treatment was performed.
Study population and dataset
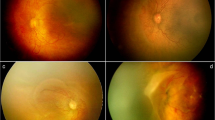
The study included preterm infants (≤34 weeks), screened between March 2013 and February 2018. Infants identified as pre-threshold (Type 1) ROP according to the Early Treatment ROP (ETROP) study [5], with Zone 1 and posterior Zone 2 localisation, or as APROP according to the International Classification of ROP (ICROP) criteria [13], and initially treated with IVB, were assessed. APROP was defined as dilatation and tortuosity of the vascular structures in all quadrants of the posterior pole, which is not proportional to peripheral retinopathy, with a flat network of shunts in Zone 1 or posterior Zone 2 within the retina or on the border of the vascular–avascular retina [13] (Fig. 1a–c). Type 1 ROP was defined, according to the current guidelines, as: Zone 1, any stage ROP with plus disease; Zone 1, stage 3 ROP with or without plus disease; Zone 2, stage 2 or 3 ROP with plus disease [5] (Fig. 1d–f).
Patients were excluded if they initially received laser treatment, received treatment at another centre or had been diagnosed with grade 4A, 4B or 5 ROP at the first screening. All data were obtained retrospectively from the infants’ medical records. The data included demographic characteristics, ROP type (APROP or Type 1 ROP), additional treatment type (IVB or LP), outcomes and local and systemic complications. Postmenstrual age (PMA) at the time of treatment, vascular maturation of the peripheral retina, duration of vascularisation, follow-up, recurrence and the interval between initial treatment and recurrence were also recorded.
Outcome measures
Primary outcome measures were rates of regression, recurrence and vascular maturation of the peripheral retina. Secondary outcome measures involved evaluating the results of laser treatment for recurrence and persistent avascular retina and their relation to unfavourable, abnormal structural outcomes. The effect of ROP type on outcomes was also analysed. Unfavourable structural outcomes were defined as retinal fold or retinal detachment including the macula, as stated in previous studies [14].
Ophthalmic examinations and treatment modalities
The preterm infants underwent ocular examinations 4–6 weeks postnatally, according to gestational age (GA). These examinations were conducted by a single, experienced ophthalmologist (AÇ) using an indirect ophthalmoscope and scleral depressor. Photographic documentation was collected via a Heine Video Omega® 2C indirect ophthalmoscope (Heine Optotechnik, Herrsching, Germany). ROP grading, localisation zones and plus disease findings were classified according to the revised ICROP criteria [13].
IVB was the primary treatment for Type 1 pre-threshold ROP (Zone 1 and posterior Zone 2 localisation) and APROP eyes, based on the BEATROP study [7]. The posterior Zone 2 was defined as the area surrounding the Zone 1 region, and the radius was threefold the distance between the centre of the optic disc and the centre of the macula [7].
IVB was administered in the operating room, accompanied by an experienced nurse. After treatment with topical anaesthesia (0.5% proparacaine hydrochloride), eyes and the periocular region were disinfected separately with 5% povidone iodine. Bevacizumab (0.625 mg/0.025 ml) was injected intravitreally, from the inferotemporal or superotemporal region, at 1 mm from the limbus with a 30-gauge needle. Vital signs were carefully monitored and recorded throughout the procedure. Following IVB injection, moxifloxacin drops were used every 4 h for 1 week as postoperative infection prophylaxis.
Follow-up examinations were performed 1 day, 3 days and 1 week after IVB application. Then, follow-up intervals were weekly for the first month post injection, every 2 weeks for the second and every 3–4 weeks thereafter, until ROP had regressed and vascularisation of the peripheral retina had reached Zone 3. Each examination evaluated disease regression (via indirect fundoscopic analysis), recurrence, the presence of tractional elements and peripheral vascularisation. Successful treatment was defined as remission of plus disease, good pupil dilation and reduced disease grade. Outcomes were further classified as insufficient regression (persistence of plus disease and neovascularisation at 3–5 days post injection), progression (post-injection intravitreal haemorrhage, increased neovascularisation and formation of tractional components) and recurrence requiring treatment (recurrence of plus disease and extraretinal fibrovascular proliferation, despite initial regression post injection) (Fig. 2). Patients with insufficient regression, progression and recurrence requiring treatment were treated with LP.
Cases with no recurrence or recurrence not requiring treatment (grade 1 or grade 2 eyes, with Zone 2 or Zone 3 localisation, not accompanied by plus disease) were closely monitored until peripheral retinal vascularisation was completed. These cases were evaluated under sedation with detailed indirect fundoscopic examination at the corrected age of 1 year and prophylactic LP treatment was applied to the persistent avascular areas (peripheral avascular retina at the corrected age of 1 year was described as persistent avascular areas) to reduce the risk of late ROP reactivation [15,16,17,18].
Transpupillary diode LP was performed under operating theatre conditions with sedation or general anaesthesia. LP was performed in all quadrants from the vascularised retina region to the ora serrata, in close confluent form, using a 810-nm head-mounted diode laser (Iridex; Oculight SL, Mountainview, CA, US). Following LP, antibiotic and steroid drops were prescribed every 4 h for 1 week. Follow-up examinations were made weekly for the first month after LP and at 3–4-week intervals thereafter until the ROP findings receded. Follow-up examinations were continued at 3–4-month intervals after the patients turned 1 year old.
Cases with a second recurrence despite IVB and LP (i.e., plus disease with the presence of a ridge or fibrovascular proliferation over the laser scars) (Fig. 3) were treated with IVB again. Follow-up examinations were made weekly for the first month and at 3–4-week intervals thereafter until all recurrent ROP findings receded. Cases with progression or no regression despite all interventions were referred for vitreoretinal surgery (VRS).
Statistical analyses
Data were analysed via the Statistical Package for the Social Sciences 16.0 for Windows. When analysing numerical data conforming to the normal distribution and whose variances were homogeneous, Student’s t-test was performed for independent variables. The Mann–Whitney U test analysed numerical data that did not meet the parametric test assumptions. The χ2 test examined categorical data. Continuous data were stated as mean ± standard deviation (range), and categorical values were stated as number (n) and percentage (%). P < 0.05 was considered statistically significant.
Results
Records of 5200 preterm infants (≤34 weeks), screened between March 2013 and February 2018, were reviewed. Final analysis was performed on 257 eyes from 130 cases (unilateral in three cases) treated with IVB for Type 1 ROP or APROP. The mean GA was 28.2 ± 2.7 weeks (range: 22–34 weeks), and the mean birth weight (BW) was 1133.7 ± 356 g (range: 480–2300 g). APROP was diagnosed in 154 (59.9%) eyes and Type 1 ROP in 103 (40.1%) eyes. Zone 1 localisation was found in 112 (43.6%) eyes and posterior Zone 2 localisation in 145 (56.4%) eyes. No significant difference was determined between the groups in terms of GA, BW, gender or time of initial treatment (p = 0.530, p = 0.155, p = 0.126, p = 0.324, respectively). Table 1 shows the cases’ demographic characteristics.
All findings for ROP and plus disease had regressed in 247 (96.1%) eyes within 48–72 h. Ten eyes (3.9%) had insufficient regression and progression after initial injection (two eyes, Type 1 ROP; eight eyes, APROP). These eyes received LP 1 week after IVB. Except for one eye with APROP, in which progression was observed despite laser treatment, ROP findings regressed in all other eyes, and no recurrence was observed. In the eye with progression despite laser treatment, grade 4A ROP developed, and this case was referred for VRS. Table 2 shows the results after initial IVB treatment and recurrence rates.
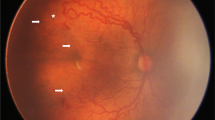
Figure 4 demonstrated fundus photographs from an infant who underwent LP treatment due to insufficient regression and subhyaloid haemorrhage after IVB.
No recurrences were found in 141 eyes (54.9%) in consecutive, regular follow-up examinations. A total of 78 (30.4%) eyes exhibited recurrence not requiring treatment, and they recovered spontaneously (Table 2). No recurrence was observed in 67% of Type 1 ROP eyes and in 46.8% of APROP eyes, and the difference was statistically significant (p = 0.001) (Table 2). Vascular maturation completed at PMA 57.7 ± 3.42 weeks (range: 52–66 weeks) in eyes with no recurrence and not requiring additional treatment.
Recurrence requiring treatment was apparent in 38 eyes (14.8%) at 9.6 ± 2.7 weeks (range: 6–15 weeks) post IVB (Table 2). No statistically significant difference was observed between the groups in terms of PMA of recurrence development or time from first treatment to recurrence (p = 0.122, p = 0.892, respectively) (Table 2). Recurrence requiring treatment was observed in 32 (20.8%) of 154 eyes with APROP and in 6 (5.8%) of 103 eyes with Type 1 ROP, and the difference was statistically significant (p = 0.001).
LP was applied to all eyes with recurrence requiring treatment. After LP, ROP and plus disease findings regressed in all eyes. A second recurrence was determined in six eyes (2.3%) following laser treatment, and IVB injection was repeated, after which ROP findings regressed in all eyes. Table 3 shows the demographic characteristics and treatment results for cases with second recurrence despite IVB + LP treatment.
Persistent avascular areas were found in 54 (25.8%) out of 209 eyes, excluding those who underwent LP due to insufficient regression, progression and recurrence requiring treatment at the corrected age of 1 year and confluent prophylactic LP treatment was applied to all visible avascular areas. Persistent avascular areas were determined in 38.6% of APROP eyes and 10.5% of Type 1 ROP eyes, and the difference was statistically significant (p < 001) (Table 2). The mean follow-up period for patients who underwent prophylactic laser treatment for persistent avascular areas was 115.1 ± 54.3 weeks (range: 70–260 weeks), and none of these patients experienced unfavourable structural outcomes or late reactivation (tractional retinal detachment [TRD], recurrence of plus disease or fibrovascular proliferations).
Recurrence requiring treatment was determined in 16.7% of Type 1 ROP eyes and in 31.8% of APROP eyes with zone 1 localisation. Although the rate was higher in APROP eyes, the difference was not statistically significant (p = 0.203) (Table 2).
No severe ocular complications, such as retinal detachment, cataract or endophthalmitis, were observed in any eye after IVB. In three eyes, subhyaloid haemorrhage and intravitreal haemorrhage were observed on the first postoperative day, but these spontaneously recovered. All cases were followed up for a mean of 121.7 ± 45.7 weeks (range: 70–260 weeks), and, in the long term after follow-up, an unfavourable structural outcome was found in one eye (0.4%), which developed insufficient regression and progression.
Discussion
This research has demonstrated that IVB is successful in Type 1 ROP (Zone 1 and posterior Zone 2 localisation) and APROP, with similar regression rates. The recurrence rate was significantly higher in APROP eyes compared to Type 1 ROP. APROP is a severe form of ROP, which can rapidly progress to retinal detachment when early intervention is not made [13], and post-treatment success rates differ in combinations of anti-VEGF monotherapy with laser treatment or cryotherapy [5, 7, 14]. In previous studies, favourable structural outcomes (ranging from 71 to 84%) have been obtained in cases of developing APROP despite early LP [19, 20]. In an extensive case series, Yetik et al. [21] evaluate the results of applying 0.625 mg IVB to 122 pre-threshold, threshold and APROP cases. A 92% success rate was achieved in 31 APROP cases following 0.625 mg IVB monotherapy, and all ROP findings receded. However, repeated treatments were needed more often in APROP cases than in pre-threshold and threshold ROP cases. Garcia Gonzales et al. [22] have observed recurrence requiring treatment in 8 (44.4%) of 18 eyes with APROP and in 2 (4.2%) of 48 eyes with classic ROP, reporting that initial ROP type is a strong risk factor in ROP recurrence. Therefore, initial IVB treatment is effective in APROP eyes, but high recurrence rates must be considered.
In the current study, late recurrence rates and onset of late recurrence coincided with previous research. Martinez-Castellanos et al. [23] evaluate 672 ROP cases to which IVB was applied between 2005 and 2017; they have observed recurrence requiring treatment in 6.8% of cases at a mean of 12 weeks (range: 4–16 weeks) after initial treatment. In the BEATROP study [7], recurrences are observed at a mean of 16 ± 4.4 weeks (mean PMA: 51.2 ± 4.6 weeks) after the first injection. A shorter interval has, thus, been determined in the current study than in the BEATROP study.
Similar to the present research and in contrast with the BEATROP study, Hwang et al. [24] determined reactivation to occur at a mean of 9.0 ± 5.7 weeks after the first injection, and Ling et al. [25] reported this interval to be a mean of 8.8 ± 3.9 weeks (mean PMA: 43.4 ± 3.5 weeks). These differences could be due to ethnic composition and demographic variations. In the BEATROP study [7], the patients’ mean GA and BW are 24.7 ± 1.7 weeks and 714 ± 200 g, respectively, while in the current study, the patients were older and had higher BWs: 28.2 ± 2.7 weeks and 1133.7 ± 356 g, respectively. This, as well as the divergent patient management practices in neonatal intensive care units, could have caused the difference in the ROP reactivation interval. Inter-specialist differences may also have been seen in the clinical diagnosis of ROP recurrence; for example, various thresholds may be adopted to define pre-plus and plus disease, which could cause variations in treatment criteria [25].
Previous studies have shown that anti-VEGF treatments cause persistent avascular retina, and delayed recurrences and TRD may appear even 2.5 years later [17, 18, 26]. However, no consensus has been reached concerning how these avascular areas can be followed up or treated. In the current study, persistent avascular areas were determined in 25.8% of eyes without peripheral laser treatment at the corrected age of 1 year, and this rate was significantly higher in APROP eyes (38.6%) than in Type 1 ROP eyes (10.5%). In a previous study using fluorescein angiography (FA) to examine recurrence rates and peripheral avascular areas following IVB in APROP and classic ROP eyes, permanent non-perfusion areas and more extensive leakage were observed in APROP eyes, and all of those eyes underwent prophylactic laser treatment at a PMA of 60–70 weeks. In the long-term follow-up (mean: 125 weeks PMA) no abnormal structural outcomes were observed [22]. In a study in Turkey [27], retinal vascular structures were evaluated with FA at a mean of 95 postmenstrual weeks in 58 APROP patients. Peripheral avascular areas and late leakages were found in 10.3% of the cases, and confluent LP treatment was applied. Non-perfusion areas and leakage in cases initially treated with Bevacizumab could cause late reactivation and poor structural outcomes. Therefore, to minimise risks of future complication, and because of previously reported late complications [15, 18], prophylactic LP treatment was applied to such eyes in the current study, and no unfavourable structural outcomes were observed in the long-term follow-ups.
This study found an unfavourable structural outcome in one eye (0.4%), which progressed to retinal detachment, despite laser treatment, because of insufficient regression following IVB. In the ETROP study [5], unfavourable structural outcomes are observed in 4.9% of 325 cases in which LP treatment was applied. Ling et al. [25] also determined unfavourable structural outcomes in 3 of 61 eyes (4.9%) to which laser treatment was applied, and this rate was found to be lower (0.9%) when IVB was used. The risk of progression to retinal detachment is lower following IVB. However, continued VEGF expression from peripheral avascular areas may increase risks of long-term vitreoretinal complications.
Contrary to the desired outcomes of anti-VEGF applications in APROP eyes, adverse treatment effects, such as incomplete retinal vascularisation, very late reactivation and TRD, have been reported recently [8, 28,29,30,31]. In the current study, a second recurrence was observed, despite intense confluent laser treatment, in three APROP cases to which laser treatment was applied in response to late reactivation post IVB injection. These cases also received a second round of IVB injections. Tanaka et al. [32] treated a case series of extremely preterm APROP infants with combined IVB and laser treatments, and extensive disease activity with fibrovascular proliferation was seen.
In extremely preterm infants with immature vascular structures, anti-VEGF treatment could cause not only widespread loss in the capillary beds resulting in vascularised posterior retinas with wide non-perfusion areas, but also loss in the original retinal capillary beds and new vascular abnormalities—including aneurysm-like capillary formations and looped capillary vessels. This could cause persistent VEGF expression and ROP reactivation [32]. Similarly, a neonatal animal study has shown that impaired VEGF gradients lead to incorrect retinal vessel development, resulting in aneurysm-like formations [33]. Therefore, in APROP cases treated with anti-VEGF agents, because of late reactivation and persistent avascular areas, even if laser treatment is applied, the possibility of severe, repeated, late recurrence must be taken seriously, and regular, close follow-up examinations must be made in these cases.
A major limitation of this study is that it was retrospective, and detailed evaluation of peripheral persistent avascular areas, especially in eyes with recurrence, could not be made with FA. In addition, the mean follow-up period was 121.7 weeks and, to evaluate much longer-term results (particularly after prophylactic LP treatment), further studies are needed.
IVB is an effective treatment for APROP and Type 1 ROP with Zone 1 and posterior Zone 2 localisation. However, due to higher recurrence rates requiring treatment and persistent peripheral avascular areas in APROP eyes, the risk of severe, late complications must be considered, and intensive follow-up examinations must be made. Prophylactic laser treatment for persistent avascular areas seems an effective option for minimising the risk of long-term complications.
Summary
What was known before
-
Intravitreal bevacizumab (IVB) has been used extensively in the treatment of retinopathy of prematurity (ROP) in recent years, However, unexpected and delayed recurrences observed in the follow-up of infants treated with IVB monotherapy have recently become a serious problem.
What this study adds
-
IVB monotherapy is an effective treatment for both aggressive posterior retinopathy of prematurity (APROP) and Type 1 ROP with zone 1 and posterior zone 2.
-
Especially in patients with APROP, requiring treatment recurrences and persistent avascular areas after IVB monotherapy are more common than Type 1 ROP, and second recurrence can be seen despite additional laser treatment.
-
The application of prophylactic laser for persistent avascular areas after IVB seems to be an effective option to be able to reduce the risk of long-term complications to a minimum.
References
Sapieha P, Joyal J, Rivera JC, Kermorvant-Duchemin E, Sennlaub F, Hardy P, et al. Retinopathy of prematurity: understanding ischemic retinal vasculopathies at an extreme of life. J Clin Invest. 2010;120:3022–32. https://doi.org/10.1172/JCI42142
Cavallaro G, Filippi L, Bagnoli P, La Marca G, Cristofori G, Raffaeli G, et al. The pathophysiology of retinopathy of prematurity: an update of previous and recent knowledge. Acta Ophthalmol. 2014;92:2–20. https://doi.org/10.1111/aos.12049
Blencowe H, Lawn JE, Vazquez T, Fielder A, Gilbert C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013;1:35–49. https://doi.org/10.1038/pr.2013.205.4
Early Treatment For Retinopathy Of Prematurity Cooperative Group. Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol. 2003;121:1684–94.
Good WV, Early Treatment for Retinopathy of Prematurity Cooperative Group. Final results of the Early Treatment for Retinopathy of Prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc. 2004;102:233–48. discussion 248-50
Geloneck MM, Chuang AZ, Clark WL, Hunt MG, Norman AA, Packwood EA, et al. Refractive outcomes following bevacizumab monotherapy compared with conventional laser treatment: a randomized clinical trial. JAMA Ophthalmol. 2014;132:1327–33. https://doi.org/10.1001/jamaophthalmol.2014.2772. BEAT-ROP Cooperative Group
Mintz-Hittner HA, Kennedy KA, Chuang AZ, BEAT-ROP Cooperative Group. Efficacy of intravitreal bevacizumab for stage3+ retinopathy of prematurity. N Engl J Med. 2011;364:603–15. https://doi.org/10.1056/NEJMoa1007374
Vander Veen DK, Melia M, Yang MB, Hutchinson AK, Wilson LB, Lambert SR. Anti-vascular endothelial growth factor therapy for primary treatment of Type 1 retinopathy of prematurity: a report by the American Academy of Ophthalmology. Ophthalmology. 2017;124:619–33. https://doi.org/10.1016/j.ophtha.2016.12.025
Chen J, Smith LE. Retinopathy of prematurity. Angiogenesis. 2007;10:133–40. https://doi.org/10.1007/s10456-007-9066-0
Sankar MJ, Sankar J, Mehta M, Bhat V, Srinivasan R. Anti-vascular endothelial growth factor (VEGF) drugs for treatment of retinopathy of prematurity. Cochrane Database Syst Rev. 2016;2:CD009734 https://doi.org/10.1002/14651858.CD009734.pub2
Pertl L, Steinwender G, Mayer C, Hausberger S, Pöschl EM, Wackernagel W, et al. A systematic review and meta-analysis on the safety of vascular endothelial growth factor (VEGF) inhibitors for the treatment of retinopathy of prematurity. PLoS One. 2015;10:e0129383 https://doi.org/10.1371/journal.pone.0129383
Mintz-Hittner HA. Retinopathy of prematurity: intravitreal injections of bevacizumab: timing, technique, and outcomes. J AAPOS. 2016;20:478–80. https://doi.org/10.1016/j.jaapos.2016.10.002
International Committee for the Classification of Retinopathy of Prematurity. The international Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol. 2005;123:991–9. https://doi.org/10.1001/archopht.123.7.991
Palmer EA. Results of U.S. randomized clinical trial of cryotherapy for ROP(CRYO-ROP). Doc Ophthalmol. 1990;74:245–51. https://doi.org/10.1007/bf02482615
Ittiara S, Blair MP, Shapiro MJ, Lichtenstein SJ. Exudative retinopathy and detachment: a late reactivation of retinopathy of prematurity after intravitreal bevacizumab. J AAPOS. 2013;17:323–5. https://doi.org/10.1016/j.jaapos.2013.01.004.
Patel RD, Blair MP, Shapiro MJ, Lichtenstein SJ. Significant treatment failure with intravitreous bevacizumab for retinopathy of prematurity. Arch Ophthalmol. 2012;130:801–2. https://doi.org/10.1001/archophthalmol.2011.1802
Hu J, Blair MP, Shapiro MJ, Lichtenstein SJ, Galasso JM, Kapur R. Reactivation of retinopathy of prematurity after bevacizumab injection. Arch Ophthalmol. 2012;130(8):1000–6. https://doi.org/10.1001/archophthalmol.2012.592. Erratum in: Arch Ophthalmol. 2013;131:212.
Snyder LL, Garcia-Gonzalez JM, Shapiro MJ, Blair MP. Very late reactivation of retinopathy of prematurity after monotherapy with intravitreal bevacizumab. Ophthalmic Surg Lasers Imaging Retin. 2016;47:280–3. https://doi.org/10.3928/23258160-20160229-12.
Sanghi G, Dogra MR, Das P, Vinekar A, Gupta A, Dutta S. Aggressive posterior retinopathy of prematurity in Asian Indian babies: spectrum of disease and outcome after laser treatment. Retina. 2009;29:1335–9. https://doi.org/10.1097/IAE.0b013e3181a68f3a
Drenser KA, Trese MT, Capone A Jr. Aggressive posterior retinopathy of prematurity. Retina. 2010;30:37–40. https://doi.org/10.1097/IAE.0b013e3181cb6151
Yetik H, Gunay M, Sirop S, Salihoglu Z. Intravitreal bevacizumab monotherapy for type-1 prethreshold, threshold, and aggressive posterior retinopathy of prematurity – 27 month follow-up results from Turkey. Graefes Arch Clin Exp Ophthalmol. 2015;253:1677–83. https://doi.org/10.1007/s00417-014-2867-0
Garcia Gonzalez JM, Snyder L, Blair M, Rohr A, Shapiro M, Greenwald M. Prophylactic peripheral laser and fluorescein angiography after bevacizumab for retinopathy of prematurity. Retina. 2018;38:764–72. https://doi.org/10.1097/IAE.0000000000001581
Martínez-Castellanos MA, González-H León A, Romo-Aguas JC, Gonzalez-Gonzalez LA. A proposal of an algorithm for the diagnosis and treatment of recurrence or treatment failure of retinopathy of prematurity after anti-VEGF therapy based on a large case series. Graefes Arch Clin Exp Ophthalmol. 2020;258:767–72. https://doi.org/10.1007/s00417-020-04605-y
Hwang CK, Hubbard GB, Hutchinson AK, Lambert SR. Outcomes after intravitreal bevacizumab versus laser photocoagulation for retinopathy of prematurity: a 5-year retrospective analysis. Ophthalmology. 2015;122:1008–15. https://doi.org/10.1016/j.ophtha.2014.12.017
Ling KP, Liao PJ, Wang NK, Chao AN, Chen KJ, Chen TL, et al. Rates and risk factors for recurrence of retinopathy of prematurity after laser or intravitreal anti-vascular endothelial growth factor monotherapy. Retina. 2020;40:1793–803. https://doi.org/10.1097/IAE.0000000000002663.
Mintz-Hittner HA, Geloneck MM, Chuang AZ. Clinical management of recurrent retinopathy of prematurity after intravitreal bevacizumab monotherapy. Ophthalmology. 2016;123:1845–55. https://doi.org/10.1016/j.ophtha.2016.04.028
Perente I, Eris E, Seymen Z, Cevik SG, Bekmez S. Aggressive posterior retinopathy of prematurity treated with intravitreal bevacizumab: late period fluorescein angiographic findings. Graefes Arch Clin Exp Ophthalmol. 2019;257:1141–6. https://doi.org/10.1007/s00417-019-04292-4
Tahija SG, Hersetyati R, Lam GC, Kusaka S, McMenamin PG. Fluorescein angiographic observations of peripheral retinal vessel growth in infants after intravitreal injection of bevacizumab as sole therapy for zone I and posterior zone II retinopathy of prematurity. Br J Ophthalmol. 2014;98:507–12. https://doi.org/10.1136/bjophthalmol-2013-304109
Lorenz B, Stieger K, Jäger M, Mais C, Stieger S, Andrassi-Darida M. Retinal vascular development with 0.312 MG intravitrealbevacizumab to treat severe posterior retinopathy OF prematurity: a longitudinal fluorescein angiographic study. Retina. 2017;37:97–111. https://doi.org/10.1097/IAE.0000000000001126
Yonekawa Y, Wu WC, Nitulescu CE, Chan RVP, Thanos A, Thomas BJ, et al. Progressive retinal detachment in infants with retinopathy of prematurity treated with intravitreal bevacizumab or ranibizumab. Retina. 2018;38:1079–83. https://doi.org/10.1097/IAE.0000000000001685
Chen TA, Shields RA, Bodnar ZH, Callaway NF, Schachar IH, Moshfeghi DM. A spectrum of regression following intravitreal bevacizumab in retinopathy of prematurity. Am J Ophthalmol. 2019;198:63–69. https://doi.org/10.1016/j.ajo.2018.09.039.
Tanaka S, Yokoi T, Katagiri S, Yoshida T, Nishina S, Azuma N. Severe recurrent fibrovascular proliferation after combined intravitreal bevacizumab injection and laser photocoagulation for aggressive posterior retinopathy of prematurity. Retin Cases Brief Rep. 2019. https://doi.org/10.1097/ICB.0000000000000887. Epub ahead of print 17 July
Gerhardt H, Golding M, Fruttiger M, Ruhrberg C, Lundkvist A, Abramsson A, et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J Cell Biol. 2003;161:1163–77. https://doi.org/10.1083/jcb.200302047
Acknowledgements
We would like to thank Betül Kızıldağ, MD, Associate Professor, Department of Radiology, Faculty of Medicine, Sutcu Imam University, and Nurten Akkeçeci, Associate Professor, Department of Physiology, Faculty of Medicine, Sutcu Imam University, Kahramanmaras, Turkey for their assistance in revision and for their comments that greatly improved the manuscript.
Author information
Authors and Affiliations
Contributions
AÇ is the lead author, and was responsible for the conception of the research idea. AÇ, YK, MCÖ, PÇ and OS have contributed to the literature review, data collection, analysis of results and formulation of the article. All authors read and confirmed the final article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Çömez, A., Karaküçük, Y., Özmen, M.C. et al. The results of intravitreal bevacizumab monotherapy for treating aggressive posterior retinopathy of prematurity and Type 1 retinopathy of prematurity. Eye 35, 3302–3310 (2021). https://doi.org/10.1038/s41433-021-01413-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01413-4