Abstract
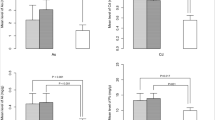
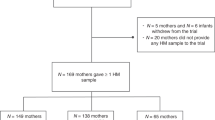
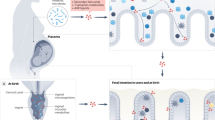
Tobacco smoking is still a widespread habit in pregnant and breastfeeding women. While the role of these risk factors on neonatal outcomes has been deeply studied, their effect on human milk composition is still not completely clear. This study aimed to report the most up to date evidence about the alteration of breast milk composition of smoking breastfeeding mothers compared to non-smoking ones. We performed a systematic review by searching PubMed, Embase, and Cochrane Library databases. Evaluated data were extracted and critically analyzed by two independent authors. PRISMA guidelines were applied, and the risk of bias was assessed (ROBINS), as was the methodological quality of the included studies (GRADE). After applying the inclusion criteria, we included 20 studies assessed as medium or high quality. In all the studies, we analyzed data regarding 1769 mothers (398 smokers and 971 nonsmokers). Smoking was associated with a lower content of lipids, calories, and proteins. Moreover, it was characterized by decreased antioxidant properties and an altered immune status. Smoking during pregnancy and breastfeeding is significantly associated with an alteration of milk metabolic properties. Further studies are needed to investigate how these changes can alter newborns’ development and outcomes and which molecular patterns are involved.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Slater L. Substance use in pregnancy. Pract Midwife. 2015;18:10–13.
EURO-PERISTAT. European Perinatal Health Report. 2015. http://www.europeristat.com.
Ashford KB, Hahn E, Hall L, Rayens MK, Noland M. Postpartum smoking relapse and secondhand smoke. Public Health Rep. 2009;124:515–26.
Scott-Goodwin AC, Puerto M, Moreno I. Toxic effects of prenatal exposure to alcohol, tobacco and other drugs. Reprod Toxicol. 2016;61:120–30.
Caputo C, Wood E, Jabbour L. Impact of fetal alcohol exposure on body systems: a systematic review. Birth Defects Res Part C—Embryo Today Rev. 2016;108:174–80.
Abraham M, Alramadhan S, Iniguez C, Duijts L, Jaddoe VWV, Den Dekker HT, et al. A systematic review of maternal smoking during pregnancy and fetal measurements with meta-analysis. PLoS ONE. 2017;12:e0170946.
WHO. Exclusive breastfeeding for 6 months best for babies everywhere. 2011.
Andreas NJ, Kampmann B, Mehring Le-Doare K. Human breast milk: a review on its composition and bioactivity. Early Hum Dev. 2015;91:629–35.
Zimmermann P, Curtis N. Breast milk microbiota: a complex microbiome with multiple impacts and conditioning factors. J Infect. 2020;81:17–47.
Napierala M, Mazela J, Merritt TA, Florek E. Tobacco smoking and breastfeeding: effect on the lactation process, breast milk composition and infant development. a critical review. Environ Res. 2016;151:321–38.
Napierala M, Merritt TA, Miechowicz I, Mielnik K, Mazela J, Florek E. The effect of maternal tobacco smoking and second-hand tobacco smoke exposure on human milk oxidant-antioxidant status. Environ Res. 2019;170:110–21.
Liberati A, Altman Douglas G, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700.
Richardson WS, Wilson MC, Nishikawa J, Hayward RS. The well-built clinical question: a key to evidence-based decisions. ACP J Club. 1995;123:A12–3.
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919.
Schünemann H, Brożek J, Guyatt G, Oxman A. Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. BMJ. 2013;332:1089–92.
Hascoët JM, Chauvin M, Pierret C, Skweres S, Van Egroo LD, Rougé C, et al. Impact of maternal nutrition and perinatal factors on breast milk composition after premature delivery. Nutrients. 2019;11:7–14.
Mello-Neto J, Rondó PHC, Morgano MA, Oshiiwa M, Santos ML, Oliveira JM. Iron concentrations in breast milk and selected maternal factors of human milk bank donors. J Hum Lact. 2010;26:175–9.
Agostoni C, Marangoni F, Grandi F, Lammardo AM, Giovannini M, Riva E, et al. Earlier smoking habits are associated with higher serum lipids and lower milk fat and polyunsaturated fatty acid content in the first 6 months of lactation. Eur J Clin Nutr. 2003;57:1466–72.
Ermis B, Yildirim A, Örs R, Tastekin A, Ozkan B, Akcay F. Influence of smoking on serum and milk malondialdehyde, superoxide dismutase, glutathione peroxidase, and antioxidant potential levels in mothers at the postpartum seventh day. Biol Trace Elem Res. 2005;105:27–36.
Bachour P, Yafawi R, Jaber F, Choueiri E, Abdel-Razzak Z. Effects of smoking, mother’s age, body mass index, and parity number on lipid, protein, and secretory immunoglobulin a concentrations of human milk. Breastfeed Med. 2012;7:179–88.
Ozkan B, Ermis B, Tastekin A, Doneray H, Yildirim A, Ors R. Effect of smoking on neonatal and maternal serum and breast milk leptin levels. Endocr Res. 2005;31:177–83.
Karatas F. An investigation of orotic acid levels in the breastmilk of smoking and non-smoking mothers. Eur J Clin Nutr. 2002;56:958–60.
Szlagatys-Sidorkiewicz A, Zagierski M, Łuczak G, MacUr K, Ba̧czek T, Kamińska B. Maternal smoking does not influence vitamin A and e concentrations in mature breastmilk. Breastfeed Med. 2012;7:285–9.
Orhon FS, Ulukol B, Kahya D, Cengiz B, Başkan S, Tezcan S. The influence of maternal smoking on maternal and newborn oxidant and antioxidant status. Eur J Pediatr. 2009;168:975–81.
Pişkin IE, Karavar HN, Arasli M, Ermiş B. Effect of maternal smoking on colostrum and breast milk cytokines. Eur Cytokine Netw. 2012;23:187–90.
Szlagatys-Sidorkiewicz A, Martysiak-Zurowska D, Krzykowski G, Zagierski M, Kamińska B. Maternal smoking modulates fatty acid profile of breast milk. Acta Paediatr Int J Paediatr. 2013;102:353–9.
Zanardo V, Nicolussi S, Cavallin S, Trevisanuto D, Barbato A, Faggian D, et al. Effect of maternal smoking on breast milk interleukin-1α, β-endorphin, and leptin concentrations. Environ Health Perspect. 2005;113:1410–3.
Burch J, Karmaus W, Gangur V, Soto-Ramírez N, Yousefi M, Goetzl LM. Pre- and perinatal characteristics and breast milk immune markers. Pediatr Res. 2013;74:615–21.
Burianova I, Bronsky J, Pavlikova M, Janota J, Maly J. Maternal body mass index, parity and smoking are associated with human milk macronutrient content after preterm delivery. Early Hum Dev. 2019;137:104832.
Groer M, Davis M, Steele K. Associations between human milk SIgA and maternal immune, infectious, endocrine, and stress variables. J Hum Lact. 2004;20:153–8.
Kondo N, Suda Y, Nakao A, Oh-Oka K, Suzuki K, Ishimaru K, et al. Maternal psychosocial factors determining the concentrations of transforming growth factor-beta in breast milk. Pediatr Allergy Immunol. 2011;22:853–61.
Milnerowicz H, Chmarek M. Influence of smoking on metallothionein level and other proteins binding essential metals in human milk. Acta Paediatr Int J Paediatr. 2005;94:402–6.
Ermis B, Yildirim A, Tastekin A, Ors R. Influence of smoking on human milk tumor necrosis factor-α, interleukin-1β, and soluble vascular cell adhesion molecule-1 levels at postpartum seventh day. Pediatr Int. 2009;51:821–4.
Szlagatys-Sidorkiewicz A, Woś E, Aleksandrowicz E, Łuczak G, Zagierski M, Martysiak-Żurowska D, et al. Cytokine profile of mature milk from smoking and nonsmoking mothers. J Pediatr Gastroenterol Nutr. 2013;56:382–4.
Zagierski M, Szlagatys-Sidorkiewicz A, Jankowska A, Krzykowski G, Korzon M, Kaminska B. Maternal smoking decreases antioxidative status of human breast milk. J Perinatol. 2012;32:593–7.
Marangoni F, Colombo C, De Angelis L, Gambaro V, Agostoni C, Giovannini M, et al. Cigarette smoke negatively and dose-dependently affects the biosynthetic pathway of the n-3 polyunsaturated fatty acid series in human mammary epithelial cells. Lipids. 2004;39:633–7.
Martinez M. Tissue levels of polyunsaturated fatty acids during early human development. J Pediatr. 1992;120:PART 2.
Samper MP, Jiménez-Muro A, Nerín I, Marqueta A, Ventura P, Rodríguez G. Maternal active smoking and newborn body composition. Early Hum Dev. 2012;88:141–5.
Sowan NA, Stember ML. Effect of maternal prenatal smoking on infant growth and development of obesity. J Perinat Educ. 2005;9:22–29.
Campbell C. Long-chain polyunsaturated fatty acids, infant formula, and breastfeeding (multiple letters). Lancet. 1998;352:1703–4.
Agostoni C, Riva E, Giovannini M, Pinto F, Colombo C, Risé P, et al. Maternal smoking habits are associated with differences in infants’ long-chain polyunsaturated fatty acids in whole blood: a case-control study. Arch Dis Child. 2008;93:414–8.
Cetin I, Giovannini N, Alvino G, Agostoni C, Riva E, Giovannini M, et al. Intrauterine growth restriction is associated with changes in polyunsaturated fatty acid fetal-maternal relationships. Pediatr Res. 2002;52:750–5.
Agostoni C, Galli C, Riva E, Colombo C, Giovannini M, Marangoni F. Reduced docosahexaenoic acid synthesis may contribute to growth restriction in infants born to mothers who smoke. J Pediatr. 2005;147:854–6.
Chu DM, Antony KM, Ma J, Prince AL, Showalter L, Moller M, et al. The early infant gut microbiome varies in association with a maternal high-fat diet. Genome Med. 2016;8:77.
Ma J, Prince AL, Bader D, Hu M, Ganu R, Baquero K, et al. High-fat maternal diet during pregnancy persistently alters the offspring microbiome in a primate model. Nat Commun. 2014;5.
Oliveira E, Pinheiro CR, Santos-Silva AP, Trevenzoli IH, Abreu-Villça Y, Nogueira Neto JF, et al. Nicotine exposure affects mother’s and pup’s nutritional, biochemical, and hormonal profiles during lactation in rats. J Endocrinol. 2010;205:159–70.
Santos-Silva AP, Oliveira E, Pinheiro CR, Nunes-Freitas AL, Abreu-Villaça Y, Santana AC, et al. Effects of tobacco smoke exposure during lactation on nutritional and hormonal profiles in mothers and offspring. J Endocrinol. 2011;209:75–84.
Mcleod G, Sherriff J, Nathan E, Hartmann PE, Simmer K. Four-week nutritional audit of preterm infants born <33 weeks gestation. J Paediatr Child Health. 2013;49:E332–9.
Ebadi M, Leuschen MP, Refaey HEL, Hamada FM, Rojas P. The antioxidant properties of zinc and metallothionein. Neurochem Int. 1996;29:159–66.
Kunz C, Rodriguez-Palmero M, Koletzko B, Jensen R. Nutritional and biochemical properties of human milk, Part I: general aspects, proteins, and carbohydrates. Clin Perinatol. 1999;26:307–33.
Marangoni F, Agostoni C, Lammard AM, Giovannini M, Galli C, Riva E. Polyunsaturated fatty acid concentrations in human hindmilk are stable throughout 12-months of lactation and provide a sustained intake to the infant during exclusive breastfeeding: an Italian study. Br J Nutr. 2000;84:103–9.
Carnevale R, Cammisotto V, Pagano F, Nocella C. Effects of smoking on oxidative stress and vascular function. Smoking Prevention and Cessation, IntechOpen. 2018;2:25–47.
Groten JP, Sinkeldam EJ, Luten JB, van Bladeren PJ. Cadmium accumulation and metallothionein concentrations after 4-week dietary exposure to cadmium chloride or cadmium-metallothionein in rats. Toxicol Appl Pharm. 1991;111:504–13.
Amici A, Emanuelli M, Ferretti E, Raffaelli N, Ruggieri S, Magni G. Homogeneous pyrimidine nucleotidase from human erythrocytes: enzymic and molecular properties. Biochem J. 1994;304:987–92.
Bell SG, Vallee BL. The Metallothionein/thionein system: an oxidoreductive metabolic zinc link. ChemBioChem. 2009;10:55–62.
Ling XB, Wei HW, Wang J, Kong YQ, Wu YY, Guo JL, et al. Mammalian metallothionein-2A and oxidative stress. Int J Molecular Sci. 2016;17:1483.
Acknowledgements
We thank the Medical Student Research Academy for supporting this project with lessons about research methodology and manuscript preparation; without its network and support this project would not have been completed.
Author information
Authors and Affiliations
Contributions
MM organized the systematic process, wrote the discussion, and supervised the process. LB and SF reviewed the literature. LB, SF, and DA wrote the results. CA assisted in the review of literature and in writing of the review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Macchi, M., Bambini, L., Franceschini, S. et al. The effect of tobacco smoking during pregnancy and breastfeeding on human milk composition—a systematic review. Eur J Clin Nutr 75, 736–747 (2021). https://doi.org/10.1038/s41430-020-00784-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-020-00784-3
This article is cited by
-
Chemical Effects on Breast Development, Function, and Cancer Risk: Existing Knowledge and New Opportunities
Current Environmental Health Reports (2022)
-
Is maternal cigarette or water pipe use associated with stopping breastfeeding? Evidence from the Jordan population and family health surveys 2012 and 2017–18
International Breastfeeding Journal (2021)
-
Association of prenatal medical risk with breastfeeding outcomes up to 12 months in the All Our Families community-based birth cohort
International Breastfeeding Journal (2021)