Abstract
Background
Telomeres are repeats of DNA that contain the sequence TTAGGG at the ends of each chromosome, and their function is to protect DNA from damage. Little evidence exists regarding the relationship between dietary patterns and telomere length, especially derived applying longitudinal design. The aim was to study if overall dietary pattern is associated with leukocyte telomere length (LTL) or faster telomere attrition or both.
Methods
The setting was longitudinal and observational. Participants were 456 men and 590 women whose birth settled in between 1934 and 1944 and who participated in the Helsinki Birth Cohort Study. Baltic sea diet score (BSDS), modified Mediterranean diet score (mMED), and dietary inflammatory index (DII®) were calculated based on a 128-item food frequency questionnaire (FFQ) collected in 2001–2004. LTL was measured twice, in 2001–2004 and in 2011–2013 by quantitative real-time polymerase chain reaction. Association between the dietary patterns and LTL were analysed by general linear models with appropriate contrasts.
Results
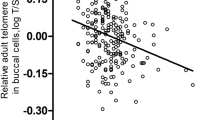
BSDS, mMED, and DII did not associate with LTL in the cross-sectional analysis in men or women. Higher mMED at baseline (2001–2004) was associated with slightly faster LTL shortening during the follow-up (standardized ß −0.08, 95% CI −0.15, −0.01). No association between mMED and LTL change was found in men. Adherence to BSDS and DII did not associate with LTL change in men or women.
Conclusion
Baltic sea diet, Mediterranean diet, and diet’s inflammatory potential seem to have only little impact on telomere length and telomere attrition in elderly Finnish men and women.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
von Zglinicki T, Martin-Ruiz CM. Telomeres as biomarkers for ageing and age-related diseases. Curr Mol Med. 2005;5:197–203.
Fitzpatrick AL, Kronmal RA, Kimura M, Gardner JP, Psaty BM, Jenny NS, et al. Leukocyte telomere length and mortality in the Cardiovascular Health Study. J Gerontol A Biol Sci Med Sci. 2011;66:421–9.
Hunt SC, Chen W, Gardner JP, Kimura M, Srinivasan SR, Eckfeldt JH, et al. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell. 2008;7:451–8.
Astuti Y, Wardhana A, Watkins J, Wulaningsih W, PILAR Research Network. Cigarette smoking and telomere length: a systematic review of 84 studies and meta-analysis. Environ Res. 2017;158:480–9.
Denham J, O’Brien BJ, Charchar FJ. Telomere length maintenance and cardio-metabolic disease prevention through exercise training. Sports Med. 2016;46:1213–37.
Furugori E, Hirayama R, Nakamura KI, Kammori M, Esaki Y, Takubo K. Telomere shortening in gastric carcinoma with aging despite telomerase activation. J Cancer Res Clin Oncol. 2000;126:481–5.
Fitzpatrick AL, Kronmal RA, Gardner JP, Psaty BM, Jenny NS, Tracy RP, et al. Leukocyte telomere length and cardiovascular disease in the cardiovascular health study. Am J Epidemiol. 2007;165:14–21.
Mundstock E, Sarria EE, Zatti H, Mattos Louzada F, Kich Grun L, Herbert Jones M, et al. Effect of obesity on telomere length: systematic review and meta-analysis. Obesity. 2015;23:2165–74.
Mathur MB, Epel E, Kind S, Desai M, Parks CG, Sandler DP, et al. Perceived stress and telomere length: a systematic review, meta-analysis, and methodologic considerations for advancing the field. Brain Behav Immun. 2016;54:158–69.
Marin C, Delgado-Lista J, Ramirez R, Carracedo J, Caballero J, Perez-Martinez P, et al. Mediterranean diet reduces senescence-associated stress in endothelial cells. Age. 2012;34:1309–16.
Garcia-Calzon S, Zalba G, Ruiz-Canela M, Shivappa N, Hebert JR, Martinez JA, et al. Dietary inflammatory index and telomere length in subjects with a high cardiovascular disease risk from the PREDIMED-NAVARRA study: cross-sectional and longitudinal analyses over 5 years. Am J Clin Nutr. 2015;102:897–904.
Gu Y, Honig LS, Schupf N, Lee JH, Luchsinger JA, Stern Y, et al. Mediterranean diet and leukocyte telomere length in a multi-ethnic elderly population. Age. 2015;37:24-015–9758-0.
Perez LM, Amaral MA, Mundstock E, Barbe-Tuana FM, Guma FTCR, Jones MH, et al. Effects of diet on telomere length: systematic review and meta-analysis. Public Health Genom. 2017;20:286–92.
Tiainen AM, Mannisto S, Blomstedt PA, Moltchanova E, Perala MM, Kaartinen NE, et al. Leukocyte telomere length and its relation to food and nutrient intake in an elderly population. Eur J Clin Nutr. 2012;66:1290–4.
Nettleton JA, Diez-Roux A, Jenny NS, Fitzpatrick AL, Jacobs DR Jr. Dietary patterns, food groups, and telomere length in the Multi-Ethnic Study of Atherosclerosis (MESA). Am J Clin Nutr. 2008;88:1405–12.
Leung CW, Laraia BA, Needham BL, Rehkopf DH, Adler NE, Lin J, et al. Soda and cell aging: associations between sugar-sweetened beverage consumption and leukocyte telomere length in healthy adults from the National Health and Nutrition Examination Surveys. Am J Public Health. 2014;104:2425–31.
Xu Q, Parks CG, DeRoo LA, Cawthon RM, Sandler DP, Chen H. Multivitamin use and telomere length in women. Am J Clin Nutr. 2009;89:1857–63.
Cassidy A, De Vivo I, Liu Y, Han J, Prescott J, Hunter DJ, et al. Associations between diet, lifestyle factors, and telomere length in women. Am J Clin Nutr. 2010;91:1273–80.
Lee JY, Jun NR, Yoon D, Shin C, Baik I. Association between dietary patterns in the remote past and telomere length. Eur J Clin Nutr. 2015;69:1048–52.
Shivappa N, Wirth MD, Hurley TG, Hebert JR. Association between the dietary inflammatory index (DII) and telomere length and C-reactive protein from the National Health and Nutrition Examination Survey-1999–2002. Mol Nutr Food Res. 2017; https://doi.org/10.1002/mnfr.201600630.
Crous-Bou M, Fung TT, Prescott J, Julin B, Du M, Sun Q, et al. Mediterranean diet and telomere length in Nurses’ Health Study: population based cohort study. BMJ. 2014;349:g6674.
Garcia-Calzon S, Martinez-Gonzalez MA, Razquin C, Aros F, Lapetra J, Martinez JA, et al. Mediterranean diet and telomere length in high cardiovascular risk subjects from the PREDIMED-NAVARRA study. Clin Nutr. 2016;35:1399–405.
Garcia-Calzon S, Martinez-Gonzalez MA, Razquin C, Corella D, Salas-Salvado J, Martinez JA, et al. Pro12Ala polymorphism of the PPARgamma2 gene interacts with a mediterranean diet to prevent telomere shortening in the PREDIMED-NAVARRA randomized trial. Circ Cardiovasc Genet. 2015;8:91–99.
Kanerva N, Loo BM, Eriksson JG, Leiviska J, Kaartinen NE, Jula A, et al. Associations of the Baltic Sea diet with obesity-related markers of inflammation. Ann Med. 2014;46:90–96.
Yliharsila H, Kajantie E, Osmond C, Forsen T, Barker DJ, Eriksson JG. Body mass index during childhood and adult body composition in men and women aged 56–70 years. Am J Clin Nutr. 2008;87:1769–75.
Perala MM, von Bonsdorff M, Mannisto S, Salonen MK, Simonen M, Kanerva N, et al. A healthy Nordic diet and physical performance in old age: findings from the longitudinal Helsinki Birth Cohort Study. Br J Nutr. 2016;115:878–86.
Fossati P, Prencipe L. Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin Chem. 1982;28:2077–80.
Lie RF, Schmitz JM, Pierre KJ, Gochman N. Cholesterol oxidase-based determination, by continuous-flow analysis, of total and free cholesterol in serum. Clin Chem. 1976;22:1627–30.
Lopes-Virella MF, Stone P, Ellis S, Colwell JA. Cholesterol determination in high-density lipoproteins separated by three different methods. Clin Chem. 1977;23:882–4.
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502.
Paalanen L, Mannisto S, Virtanen MJ, Knekt P, Rasanen L, Montonen J, et al. Validity of a food frequency questionnaire varied by age and body mass index. J Clin Epidemiol. 2006;59:994–1001.
Mannisto S, Virtanen M, Mikkonen T, Pietinen P. Reproducibility and validity of a food frequency questionnaire in a case-control study on breast cancer. J Clin Epidemiol. 1996;49:401–9.
Reinivuo H, Hirvonen T, Ovaskainen ML, Korhonen T, Valsta LM. Dietary survey methodology of FINDIET 2007 with a risk assessment perspective. Public Health Nutr. 2010;13:915–9.
Kanerva N, Kaartinen NE, Schwab U, Lahti-Koski M, Mannisto S. The Baltic Sea Diet Score: a tool for assessing healthy eating in Nordic countries. Public Health Nutr. 2014;17:1697–705.
Trichopoulou A, Orfanos P, Norat T, Bueno-de-Mesquita B, Ocke MC, Peeters PH, et al. Modified Mediterranean diet and survival: EPIC-elderly prospective cohort study. BMJ. 2005;330:991.
Shivappa N, Steck SE, Hurley TG, Hussey JR, Ma Y, Ockene IS, et al. A population-based dietary inflammatory index predicts levels of C-reactive protein in the Seasonal Variation of Blood Cholesterol Study (SEASONS). Public Health Nutr. 2014;17:1825–33.
Shivappa N, Steck SE, Hurley TG, Hussey JR, Hebert JR. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 2014;17:1689–96.
Guzzardi MA, Iozzo P, Salonen M, Kajantie E, Eriksson JG. Rate of telomere shortening and metabolic and cardiovascular risk factors: a longitudinal study in the 1934–1944 Helsinki Birth Cohort Study. Ann Med. 2015;47:499–505.
Cawthon RM. Telomere measurement by quantitative PCR. Nucleic Acids Res. 2002;30:e47.
Cawthon RM. Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucleic Acids Res. 2009;37:e21.
Willett W, Stampfer MJ. Total energy intake: implications for epidemiologic analyses. Am J Epidemiol. 1986;124:17–27.
Cohen J. Statistical power analysis for the behavioral sciences. 2nd edition. New York, NY: Lawrence Erlbaum Associates; 1988.
Boccardi V, Esposito A, Rizzo MR, Marfella R, Barbieri M, Paolisso G. Mediterranean diet, telomere maintenance and health status among elderly. PLoS One. 2013;8:e62781.
De Meyer T, Bekaert S, De Buyzere ML, De Bacquer DD, Langlois MR, Shivappa N, et al. Leukocyte telomere length and diet in the apparently healthy, middle-aged Asklepios population. Sci Rep. 2018;8:6540-018–24649-9.
Raulio S, Erlund I, Mannisto S, Sarlio-Lahteenkorva S, Sundvall J, Tapanainen H, et al. Successful nutrition policy: improvement of vitamin D intake and status in Finnish adults over the last decade. Eur J Public Health. 2017;27:268–73.
Farzaneh-Far R, Lin J, Epel E, Lapham K, Blackburn E, Whooley MA. Telomere length trajectory and its determinants in persons with coronary artery disease: longitudinal findings from the heart and soul study. PLoS One. 2010;5:e8612.
Ehrlenbach S, Willeit P, Kiechl S, Willeit J, Reindl M, Schanda K, et al. Influences on the reduction of relative telomere length over 10 years in the population-based Bruneck Study: introduction of a well-controlled high-throughput assay. Int J Epidemiol. 2009;38:1725–34.
Verhulst S, Aviv A, Benetos A, Berenson GS, Kark JD. Do leukocyte telomere length dynamics depend on baseline telomere length? An analysis that corrects for ‘regression to the mean’. Eur J Epidemiol. 2013;28:859–66.
Teixeira MT, Arneric M, Sperisen P, Lingner J. Telomere length homeostasis is achieved via a switch between telomerase- extendible and -nonextendible states. Cell. 2004;117:323–35.
Willett WC. Nutritional epidemiology. 3rd ed. Oxford: Oxford University Press; 2012.
Lin J, Smith DL, Esteves K, Drury S. Telomere length measurement by qPCR—summary of critical factors and recommendations for assay design. Psychoneuroendocrinology 2018;99:271–278.
Southern EM. A preparative gel electrophoresis apparatus for large scale separations. Anal Biochem. 1979;100:304–18.
Acknowledgements
This study was supported by grants from Finska Läkaresällskapet, the Finnish Special Governmental Subsidy for Health Sciences, Academy of Finland, Samfundet Folkhälsan, Liv och Hälsa, the Signe and Ane Gyllenberg Foundation, EU FP7 [Developmental Origins of Healthy Aging (DORIAN)] project number 278603, and EU H2020-PHC-2014-DynaHealth grant 633595 (all for the Helsinki Birth Cohort Study).
Funding
Supported by grants from Finska Läkaresällskapet, the Finnish Special Governmental Subsidy for Health Sciences, Academy of Finland, Samfundet Folkhälsan, Liv och Hälsa, the Signe and Ane Gyllenberg Foundation, EU FP7 [Developmental Origins of Healthy Aging (DORIAN)] project number 278603, and EU H2020-PHC-2014-DynaHealth grant 633595 (all for the Helsinki Birth Cohort Study).
Author contributions
JM, JGE, SM, MMP, and HK designed research, JGE, MMP, NK, SM, PI, MAG, NS, and JRH provided essential materials, JM and HK performed statistical analysis, JM wrote the paper, JM had primary responsibility of final content, JM, JGE, SM, MMP, NK, PI, MAG, HK, JRH, and NS provided inputs to the paper and approved the final version of the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. James R. Hébert owns controlling interest in Connecting Health Innovations LLC (CHI), a company planning to license the right to his invention of the Dietary Inflammatory Index (DII) from the University of South Carolina in order to develop computer and smart phone applications for patient counseling and dietary intervention in clinical settings. Dr. Nitin Shivappa is an employee of CHI. The remaining authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Meinilä, J., Perälä, MM., Kautiainen, H. et al. Healthy diets and telomere length and attrition during a 10-year follow-up. Eur J Clin Nutr 73, 1352–1360 (2019). https://doi.org/10.1038/s41430-018-0387-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-018-0387-4
This article is cited by
-
Aging Processes Are Affected by Energy Balance: Focused on the Effects of Nutrition and Physical Activity on Telomere Length
Current Nutrition Reports (2024)
-
Dietary inflammatory potential and biological aging among US adults: a population-based study
Aging Clinical and Experimental Research (2023)
-
Mediterranean diet and the hallmarks of ageing
European Journal of Clinical Nutrition (2021)
-
Telomere shortening associates with elevated insulin and nuchal fat accumulation
Scientific Reports (2020)
-
Dietary patterns and telomere length in community-dwelling Chinese older men and women: a cross-sectional analysis
European Journal of Nutrition (2020)