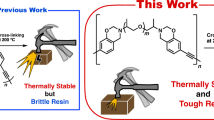
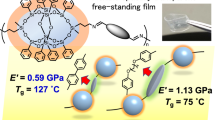
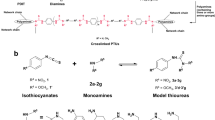
Abstract
Thermal curing of maleimidobenzoxazines (MB-Rs) with n-butyl, 2-ethylhexyl, allyl, and phenyl substituents was carried out in the presence of tetrafunctional thiols with pentaerythritol and glycolurea cores (PEMP and TSG, respectively). The thermosets were prepared by the following curing processes: mixtures of MB-R and thiol compounds were directly cured at 140–160 °C and a building block, which was synthesized by a ring-opening reaction involving a benzoxazine moiety of MB-R and thiol compounds at room temperature, was postcured. To synthesize the building blocks, furan was used to protect the maleimide moiety of the MB-Rs to selectively promote the reaction between benzoxazine and mercapto groups. The effects of the network polymer structures on the thermal stability and mechanical properties of the produced thermosets were investigated by thermogravimetric analysis, dynamic mechanical analysis, and a mechanical tensile test.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout














Similar content being viewed by others
References
Sun C, Liu J, Gong Y, Wilkinson DP, Zhang J. Recent advances in all-solid-state rechargeable lithium batteries. Nano Ener. 2017;33:363–86.
Simon P, Gogotsi Y. Perspectives for electrochemical capacitors and related devices. Nat Mater. 2020;19:1151–63.
Xing W, Xu Y, Song C, Deng T. Recent advances in thermal interface materials for thermal management of high-power electronics. Nanomater. 2022;12:3365.
Wang X, Guo W, Song L, Hu Y. Intrinsically flame retardant bio-based epoxy thermosets: A review. Comp B Eng. 2019;179:107487.
Quirino RL, Monroe K, Fleischer CH III, Biswas E, Kessler MR. Review on thermosetting polymers from renewable sources. Polym Int. 2021;70:167–80.
Luo J, Demchuk Z, Zhao X, Saito T, Tian M, Sokolov AP, et al. Elastic vitrimers: Beyond thermoplastic and thermoset elastomers. Matter. 2022;5:1391–422.
Nair CPR. Advances in addition-cure phenolic resins. Prog Polym Sci. 2004;29:401–98.
Asim M, Saba N, Jawaid M, Nasir M, Pervaiz M, Alothman OY. A review on phenolic resin and its composites. Curr Anal Chem. 2018;14:185–97.
Xu Y, Guo L, Zhang H, Zhai H, Ren H. Research status, industrial application demand and prospects of phenolic resin. RSC Adv. 2019;9:28924–35.
Ishida H, Agag T. (Editors) Handbook of benzoxazine resins, Elsevier: Amsterdam, 2011.
Takeichi T, Agag T. High performance polybenzoxazines as novel thermosets. High Perform Polym. 2006;18:777–97.
Ghosh NN, Kiskan B, Yagci Y. Polybenzoxazines. New high performance thermosetting resins: Synthesis and properties. Prog Polym Sci. 2007;32:1344–91.
Takeichi T, Kawauchi T, Agag T. High performance polybenzoxazines as a novel type of phenolic resin. Polym J. 2008;40:1121–31.
Yagci Y, Kiskan B, Ghosh NN. Recent advancement on polybenzoxazine-A newly developed high performance thermoset. J Polym Sci Part A Polym Chem. 2009;47:5565–76.
Lyu Y, Ishida H. Natural-sourced benzoxazine resins, homopolymers, blends and composites: A review of their synthesis, manufacturing and applications. Prog Polym Sci. 2019;99:101168.
Machado I, Shaer C, Hurdle K, Calado V, Ishida H. Towards the development of green flame retardancy by polybenzoxazines. Prog Polym Sci. 2021;121:101435.
Ning X, Ishida H. Phenolic materials via ring-opening polymerization: synthesis and characterization of bisphenol-A based benzoxazines and their polymers. J Polym Sci Part A Polym Chem. 1994;32:1121–9.
Ishida H, Low HY. A study on the volumetric expansion of benzoxazine-based phenolic resin. Macromolecules. 1997;30:1099–106.
Low HY, Ishida H. Structural effects of phenols on the thermal and thermo-oxidative degradation of polybenzoxazines. Polymer. 1999;40:4365–76.
Kolanadiyil SN, Minami M, Endo T. Synthesis and thermal properties of difunctional benzoxazines with attached oxazine ring at the para-, meta-, and ortho-position. Macromolecules. 2017;50:3476–88.
Kolanadiyil SN, Minami M, Endo T. Implementation of meta-positioning in tetrafunctional benzoxazines: Synthesis, properties, and differences in the polymerized structure. Macromolecules. 2020;53:6866–86.
Murai Y, Uemura T, Chen Y, Kawauchi T, Takeichi T. Synthesis of high-molecular-weight benzoxazines from various combinations of bisphenols and diamines via Mannich condensation and properties of their thermosets. Polym J. 2021;53:439–47.
Shaer C, Oppenheimer L, Lin A, Ishida H. Advanced carbon materials derived from polybenzoxazines: A review. Polymers (Basel). 2021;13:3775.
Kumar KSS, Nair CPR, Sadhana R, Ninan KN. Benzoxazine–bismaleimide blends: Curing and thermal properties. Eur Polym J. 2007;43:5084–96.
Chou C-I, Liu Y-L. High performance thermosets from a curable Diels–Alder polymer possessing benzoxazine groups in the main chain. J Polym Sci Part A Polym Chem. 2008;46:6509–17.
Lochab B, Varma IK, Bijwe J. Blends of benzoxazine monomers: Effect of structure and composition on polymer properties. J Therm Anal Calorim. 2013;111:1357–64.
Wang Y, Kou K, Wu G, Zhuo L, Li J, Zhang Y. The curing reaction of benzoxazine with bismaleimide/cyanate ester resin and the properties of the terpolymer. Polymer. 2015;77:354–60.
Wang H, Dayo AD, Wang J, Wang J-Y, Liu W-B. Trifunctional quinoxaline-based maleimide and its polymer alloys with benzoxazine: Synthesis, characterization, and properties. J Appl Polym Sci. 2021;138:49694.
Lochab B, Monisha M, Amarnath N, Sharma P, Mukherjee S, Ishida H. Review on the accelerated and low-temperature polymerization of benzoxazine resins: addition polymerizable sustainable polymers. Polymers (Basel). 2021;13:1260.
Agag T, Takeichi T. Novel benzoxazine monomers containing p-phenyl propargyl ether: Polymerization of monomers and properties of polybenzoxazines. Macromolecules. 2001;34:7257–63.
Agag T, Takeichi T. Synthesis and characterization of novel benzoxazine monomers containing p-allyl groups and their high performance thermosets. Macromolecules. 2003;36:6010–17.
Takeichi T, Thongpradith S, Kawauchi T. Copolymers of vinyl-containing benzoxazine with vinyl monomers as precursors for high performance thermosets. Molecules. 2015;20:6488–503.
Kobayashi T, Muraoka M, Goto M, Minami M, Sogawa H, Sanda F. Main-chain type benzoxazine polymers consisting of polypropylene glycol and phenyleneethynylene units: spacer effect on curing behavior and thermomechanical properties. Polym J. 2022;54:133–41.
Muraoka M, Goto M, Minami M, Zhou D, Suzuki T, Yajima T, et al. Ethynylene-linked multifunctional benzoxazines: the effect of the ethynylene group and packing on thermal behavior. Polym Chem. 2022;13:5590–6.
Kobayashi T, Goto M, Minami M, Sanda F. Synthesis and crosslinking reaction of a novel polymer containing benzoxazine and phenyleneethynylene moieties in the main chain. J Polym Sci Part A Polym Chem. 2019;57:2581–9.
Lu Y, Yu X, Evans CJ, Yang S, Zhang K. Elucidating the role of acetylene in ortho-phthalimide functional benzoxazines: Design, synthesis, and structure-property investigations. Polym Chem. 2021;12:5059–68.
Gorodisher I, DeVoe RJ, Webb RJ. Catalytic Opening of Lateral Benzoxazine Rings by Thiols. In ref. 10. Chapter 11, pp.211–234.
Kawaguchi AW, Sudo A, Endo T. Synthesis of highly polymerizable 1,3-benzoxazine assisted by phenyl thio ether and hydroxyl moieties. J Polym Sci Part A Polym Chem. 2012;50:1457–61.
Beyazkilic Z, Kahveci MU, Aydogan B, Kiskan B, Yagci Y. Synthesis of polybenzoxazine precursors using thiols: Simultaneous thiol-ene and ring-opening reactions. J Polym Sci Part A Polym Chem. 2012;50:4029–36.
Kawaguchi AW, Sudo A, Endo T. Polymerization-depolymerization system based on reversible addition-dissociation reaction of 1,3-benzoxazine with thiol. ACS Macro Lett. 2013;2:1–4.
Semerci E, Kiskan B, Yagci Y. Thiol reactive polybenzoxazine precursors: A novel route to functional polymers by thiol-oxazine chemistry. Eur Polym J. 2015;69:636–41.
Liu Y-L, Yu J-M, Chou C-I. Preparation and properties of novel benzoxazine and polybenzoxazine with maleimide groups. J Polym Sci Part A Polym Chem. 2004;42:5954–63.
Ishida H, Ohba S. Synthesis and characterization of maleimide and norbornene functionalized benzoxazines. Polymer. 2005;46:5588–95.
Chaisuwan T, Ishida T. High-performance maleimide and nitrile-functionalized benzoxazines with good processibility for advanced composites applications. J Appl Polym Sci. 2006;101:548–58.
Ishida H, Ohba S. Thermal analysis and mechanical characterization of maleimide-functionalized benzoxazine/epoxy copolymers. J Appl Polym Sci. 2006;101:1670–7.
Agag T, Takeichi T. Preparation, characterization, and polymerization of maleimidobenzoxazine monomers as a novel class of thermosetting resins. J Polym Sci Part A Polym Chem. 2006;44:1424–35.
Liu Y-L, Yu J-M. Cocuring behaviors of benzoxazine and maleimide derivatives and the thermal properties of the cured products. J Polym Sci Part A Polym Chem. 2006;44:1890–9.
Gacal B, Cianga L, Agag T, Takeichi T, Yagci Y. Synthesis and characterization of maleimide (co)polymers with pendant benzoxazine groups by photoinduced radical polymerization and their thermal curing. J Polym Sci Part A Polym Chem. 2007;45:2774–86.
Agag T, Arza CR, Maurer FHJ, Ishida H. Primary amine-functional benzoxazine monomers and their use for amide-containing monomeric benzoxazines. Macromolecules. 2010;43:2748–58.
Jin L, Agag T, Ishida H. Bis(benzoxazine-maleimide)s as a novel class of high performance resin: Synthesis and properties. Eur Polym J. 2010;46:354–63.
Chaisuwan T, Ishida T. Highly processible maleimide and nitrile functionalized benzoxazines for advanced composites applications. J Appl Polym Sci. 2010;117:2559–65.
Zhong H, Lu Y, Chen J, Xu W, Liu X. Preparation, characterization, and polymerization of novel maleimidobenzoxazine containing carboxylic moiety and its cocuring behaviors with epoxy resin. J Appl Polym Sci. 2010;118:705–10.
Ke L, Hu D, Lu Y, Feng S, Xie Y, Xu W. Copolymerization of maleimide-based benzoxazine with styrene and the curing kinetics of the resultant copolymer. Polym Degrad Stabil. 2012;97:132–8.
Gao Y, Huang F, Zhou Y, Du L. Synthesis and characterization of a novel acetylene- and maleimide-terminated benzoxazine and its high-performance thermosets. J Appl Polym Sci. 2013;128:340–6.
Cheng Y, Qi T, Jin Y, Deng D, Xiao F. Highly cross-linked thermosetting resin of maleimidobenzoxazine functionalized with benzocyclobutene. Polymer. 2013;54:143–7.
Lin S-C, Wu C-S, Yeh J-M, Liu Y-L. Reaction mechanism and synergistic anticorrosion property of reactive blends of maleimidecontaining benzoxazine and amine-capped aniline trimer. Polym Chem. 2014;5:4235–44.
Zhang K, Liu Y, Ishida H. Polymerization of an AB-type benzoxazine monomer toward different polybenzoxazine networks: When Diels−Alder reaction meets benzoxazine chemistry in a single-component resin. Macromolecules. 2019;52:7386–95.
Otsu T, Matsumoto A, Kubota T, Mori S. Reactivity in radical polymerization of N-substituted maleimides and thermal stability of the resulting polymers. Polym Bull. 1990;23:43–50.
Otsu T, Matsumoto A, Kubota T. Radical polymerization of N-(alkyl-substituted phenyl)maleimides: Synthesis of thermally stable polymers soluble in nonpolar solvents. Macromolecules. 1990;23:4508–13.
Omayu A, Matsumoto A. Synthesis and thermal properties of alternating copolymers of N-methylmaleimide with olefins including cyclic and polar groups. Macromol Chem Phys. 2008;209:2312–19.
Takeda K, Matsumoto A. Thermosetting maleimide/isobutene alternating copolymer as a new class of transparent materials. Macromol Chem Phys. 2010;211:782–90.
Hisano M, Takeda K, Takashima T, Jin Z, Shiibashi A, Matsumoto A. Sequence controlled radical copolymerization of N-substituted maleimides with olefins and polyisobutene macromonomers to fabricate thermally stable and transparent maleimide copolymers with tunable glass transition temperatures and viscoelastic properties. Macromolecules. 2013;46:7733–44.
Hamerton I. High-performance thermoset-thermoset polymer blends: a review of the chemistry of cyanate ester-bismaleimide blends. High Perform Polym. 1996;8:83–95.
Gouri C, Nair CPR, Ramaswamy R. Reactive Alder-ene blend of diallyl bisphenol A novolac and bisphenol A bismaleimide: synthesis, cure and adhesion studies. Polym Int. 2001;50:403–13.
Anagwu FI, Thakur VK, Skordos AA. High-performance vitrimeric benzoxazines for sustainable advanced materials: Design, synthesis, and applications. Macromol Mater Eng. 2023;308:2200534.
Kurasaki Y, Suzuki Y, Matsumoto A. Synthesis of heat-resistant and high-strength polymers by thiol-ene reaction of N-allylmaleimide copolymers using glycolurea cross-linkers with rigid molecular structures. J Polym Sci. 2020;58:923–31.
Holly FW, Cope AC. Condensation products of aldehydes and ketones with o-aminobenzyl alcohol and o-hydroxybezylamine. J Am Chem Soc. 1944;66:1875–9.
Takeichi T, Saito Y, Agag T, Muto H, Kawauchi T. High-performance polymer alloys of polybenzoxazine and bismaleimide. Polymer. 2008;49:1173–9.
Andreu R, Reina JA, Ronda JC. Studies on the thermal polymerization of substituted benzoxazine monomers: Electronic effects. J Polym Sci Part A Polym Chem. 2008;46:3353–66.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takasu, M., Sugiura, K., Sugimoto, S. et al. Synthesis of thermosets from maleimidobenzoxazines and tetrafunctional thiols and their thermal and mechanical properties. Polym J 56, 17–30 (2024). https://doi.org/10.1038/s41428-023-00841-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-023-00841-1