Abstract
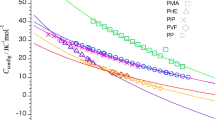
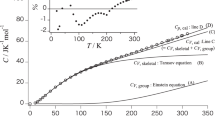
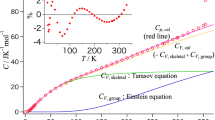
This study predicts the absolute values of heat capacities from the molecular formula per monomer for main-chain-type polymers below the glass transition temperature. The frequencies of the skeletal and group-vibration modes are calculated using the Tarasov and Einstein equations, respectively, and the heat-capacity differences at constant pressure and constant volume are used to correct the predicted heat capacity. The contributes of skeletal vibrations to the heat capacity can be expressed by one- and three-dimensional Tarasov equations, and the contribution of group vibrations can be determined by summing the group-vibration heat capacities for functional groups and atoms constituting the monomer as obtained from the Einstein equation. The absolute value of the heat capacity is predicted from this combination of equations. The heat capacities of poly(4-methyl-1-pentene) are predicted within an error range of 8.0% from 90 to 180 K and ±2.0% from 180 to 300 K. The heat capacities of poly(vinyl benzoate) are within ±2.0% agreement from 190 to 350 K, while for poly(1,4-butylene adipate), the agreement is within ±2.0% from 80 to 200 K.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Wagman DD, Evans WH, Parker VB, Schumm RH, Halow I, Bailey SM. The NBS tables of chemical thermodynamic properties: selected values for inorganic and C1 and C2 organic substances in SI units. J Phys Chem Ref Data. 1982;11:1–392.
Gopal ESR. Specific heats at low temperatures. London: Springer; 2012.
Wunderlich B. Thermal analysis of polymeric materials. Heidelberg: Springer; 2005.
Glassy, amorphous and nano-crystalline materials: thermal physics, analysis, structure and properties. In: Šesták J, Mareš JJ, Hubík P, editors. Hot topics in thermal analysis and calorimetry 8. New York: Springer Science & Business Media; 2010.
Gibson GE, Giauque WF. The third law of thermodynamics. Evidence from the specific heats of glycerol that the entropy of a glass exceeds that of a crystal at the absolute zero. J Am Chem Soc. 1923;45:93–104.
Haida O, Matsuo T, Suga H, Seki S. Calorimetric study of the glassy state X. Enthalpy relaxation at the glass-transition temperature of hexagonal ice. J Chem Thermodyn. 1974;6:815–25.
Tajima Y, Matsuo T, Suga H. Calorimetric study of phase transition in hexagonal ice doped with alkali hydroxides. J Phys Chem Solids. 1984;45:1135–44.
Kume Y, Muraoka H, Yamamuro O, Matsuo T. Deuteration-induced phase transition in ammonium hexachloroplumbate. J Chem Phys. 1988;108:4090–7.
Miyazaki Y, Wang Q, Sato A, Saito K, Yamamoto M, Kitagawa H, et al. Heat capacity of the halogen-bridged mixed-valence complex Pt2 (dta)4I (dta = CH3CS2−). J Phys Chem B. 2002;106:197–202.
Yamamura Y, Nakajima N, Tsuji T, Koyano M, Iwasa Y, Katayama S, et al. Low temperature heat capacities and Raman spectra of negative thermal expansion compounds ZrW2O8 and HfW2O8. Phys Rev B. 2002;66:014301.
Matsuo T, Maekawa T, Inaba A, Yamamuro O, Ohama M, Ichikawa M, et al. Isotope-dependent crystalline phases at ambient temperature: spectroscopic and calorimetric evidence for a deuteration-induced phase transition at 320 K in α-DCrO2. J Mol Struct. 2006;790:129–34.
Saito K, Sato A, Kikuchi K, Nishikawa H, Ikemoto I, Sorai M. Calorimetric study of metal-insulator transition in (DIMET) 2I 3. J Phys Soc Jpn. 2000;69:3602–6.
Yamamuro O, Tsukushi I, Lindqvist A, Takahara S, Ishikawa M, Matsuo T. Calorimetric study of glassy and liquid toluene and ethylbenzene: thermodynamic approach to spatial heterogeneity in glass-forming molecular liquids. J Phys Chem B. 1998;102:1605–9.
Pyda M, Bartkowiak M, Wunderlich B. Computation of heat capacities of solids using a general Tarasov equation. J Therm Anal. 1998;52:631–56.
Einstein A. Die Plancksche Theorie der Strahlung und die Theorie der spezifischen Wärme. Ann Phys. 1907;327:180–90.
Debye P. Zur Theorie der spezifischen Wärmen. Ann Phys. 1912;344:789–839.
Nernst W, Lindemann FA. Spezifische Wärme und Quantentheorie. Z Elektrochem. 1911;17:817–27.
Tarasov VV. Heat capacity of anisotropic solids. Zh Fiz Khimii. 1950;24:111–28.
Tarasov VV, Yunitskii GA. Theory of heat capacity of layered-chain and structures. Russ J Phys Chem. 1965;39:1109–11.
Jianye W. Heat capacities of polymers in physical properties of polymers handbook. In: James E. Mark, editor. Chapter 9. New York: Springer; 2007. p. 145–54
Domalski ES, Hearing ED. Heat capacities and entropies of organic compounds in the condensed phase. Volume III. J Phys Chem Ref Data. 1996;25:1–525.
Wunderlich B. Motion in polyethylene. II. Vibrations in crystalline polyethylene. J Chem Phys. 1962;37:1207–16.
Pyda M, Nowak-Pyda E, Mays J, Wunderlich B. Heat capacity of poly (butylene terephthalate). J Polym Sci. 2004;42:4401–11.
Yoshida S, Suga H, Seki S. Thermodynamic studies of solid polyethers. II. Heat capacity of poly(oxacyclobutane), –[–(CH2)3O–]– n, between 1.4 and 330°K. Polym J. 1973;5:11–24.
Yoshida S, Suga H, Seki S. Thermodynamic studies of solid polyethers. III. Poly(tetrahydrofuran), –[–(CH2)4O–]–n. Polym J. 1973;5:25–32.
Yoshida S, Suga H, Seki S. Thermodynamic studies of solid polyethers. IV. Poly(octamethylene oxide), –[–(CH2)8O–]–n. Polym J. 1973;5:33–40.
Yokota M, Sugane K, Tsukushi I, Shibata M. Evaluation of the heat capacity of amorphous polymers composed of a carbon backbone below the glass-transition temperature, Polym J. 2020. https://doi.org/10.1038/s41428-020-0317-x.
Yokota M, Tsukushi I. Heat capacities of polymer solids composed of polyesters and poly(oxide)s, evaluated below the glass-transition temperature. Polym J. 2020. https://doi.org/10.1038/s41428-020-0364-3.
Karasz FE, Bair HE, O’Reilly JM. Thermodynamic properties of poly-4-methyl-pentene-1. Polymer. 1967;8:547–60.
Pasquini M, Melia TP, Marchetti A. Thermal properties of some substructured poly(vinyl benzoates). Therm Anal Proc Int Conf 4th. 1974;2:65.
Rabinovich IB, Nistratov VP, Babinkov AG, Shvetsova KG, Larina VN. Thermodynamic properties of polybutyleneglycol adipate. Polym Sci USSR 1984;26:826–31.
NIST Chemistry WebBook SRD69. 100 Bureau Drive Gaithersburg, MD 20899, 301-975-2000. 1901. https://webbook.nist.gov/cgi/cbook.cgi?ID=C106989&Units=SI&Mask=80#IR-Spec. Accessed 7 Nov 2019.
Kauzmann K. The nature of the glassy state and the behavior of liquids at low temperatures. Chem Rev. 1948;43:219–56.
Angell CA, Choi Y. Crystallization and vitrification in aqueous systems. J Microsc. 1986;141:251–6.
Abramowitz M, Stegun IA. Handbook of mathematical functions: with formulas, graphs, and mathematical tables. New York: Dover Publications; 1965. p. 796.
Acknowledgements
The authors would like to thank Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yokota, M., Tsukushi, I. Prediction of the heat capacity of main-chain-type polymers below the glass transition temperature. Polym J 52, 1113–1120 (2020). https://doi.org/10.1038/s41428-020-0365-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-020-0365-2
This article is cited by
-
Analysis of the configurational heat capacity of polystyrene and its monomer and oligomer above the glass transition temperature
Polymer Journal (2022)
-
Configurational heat capacity of various polymers above the glass transition temperature
Polymer Journal (2022)
-
Estimation of the configurational heat capacity of polyisobutylene, isobutane and 2,2,4-isomethylpentane above the glass transition temperature
Polymer Journal (2021)