Abstract
Virus-like particles (VLPs) have become key tools in biology, medicine and even engineering. After their initial use to resolve viral structures at the atomic level, VLPs were rapidly harnessed to develop antiviral vaccines followed by their use as display platforms to generate any kind of vaccine. Most recently, VLPs have been employed as nanomachines to deliver pharmaceutically active products to specific sites and into specific cells in the body. Here, we focus on the use of VLPs for the development of vaccines with broad fields of indications ranging from classical vaccines against viruses to therapeutic vaccines against chronic inflammation, pain, allergy and cancer. In this review, we take a walk through time, starting with the latest developments in experimental preclinical VLP-based vaccines and ending with marketed vaccines, which earn billions of dollars every year, paving the way for the next wave of prophylactic and therapeutic vaccines already visible on the horizon.
Similar content being viewed by others
Introduction: the concept of virus-like particles (VLPs)
The term virus-like particles (VLPs) refers to particles that self-assemble as a result of the expression of proteins encoding capsids, cores or envelops of viruses or even preparations of monolayered particles derived from a multilayered virus [1]. Symmetrical particles formed from nonviral or artificial proteins can also be considered VLPs [2, 3]; in this case, symmetry refers to the way the capsomere units are geometrically organized. However, this category of VLPs is not discussed in this review. Additionally, VLPs self-assemble into particles that resemble or mimic the structure, size, and symmetry of original viruses, however VLPs cannot replicate as they lack a genome and replicases [1]. Detailed descriptions of VLP structure, immunogenicity and expression as they relate to vaccination are reviewed elsewhere [4, 5].
The structural proteins of hepatitis B virus, mainly the core (HBc) and the surface antigen (HBsAg), were among the first VLPs to be expressed in heterologous expression systems. These efforts resulted in the first recombinant human vaccine in 1986 against HBV that utilizes surface antigens [6]. Next, a vaccine against human papillomavirus (HPV), which causes cervical cancer, that uses the L1 structural protein was introduced. HPV vaccines entered the market in 2006 and 2007 [7, 8]. Following this success, a vaccine against hepatitis E virus (HEV) was approved in 2011 in China [9]. Overall, the use of VLPs as a conventional vaccine platform possesses several advantages, as listed in Table 1.
The current review aims to discuss some recent updates in VLP-based vaccine development by first describing some examples of VLP-based vaccines in preclinical development and clinical trials and finally describing the VLP-based vaccines currently on the market.
Preclinical development
VLPs have been extensively used as prophylactic or therapeutic vaccine platforms for a wide range of diseases. In this section, we aim to discuss some examples of the preclinical application of VLPs as a vaccine platform for successful induction of antibodies and/or T-cell responses in both prophylactic and therapeutic regimens in the following diseases: infectious diseases (examples discussed: Middle East respiratory syndrome, coronavirus disease 2019 (COVID-19), influenza, malaria and acquired immunodeficiency disease (AIDS), inflammation, allergy, pain, neurodegenerative diseases (Alzheimer’s and Parkinson’s disease) and cancer).
VLP-based vaccines for the induction of antibodies in prophylactic or therapeutic regimens
B cells can be optimally stimulated by particles with repetitive surfaces. It was found in the 1970s that an optimal B-cell response can be induced by at least 12–16 epitopes spaced 5–10 nm apart (e.g., haptens and artificial polymers), referred to as immunons [10, 11]. It was subsequently realized that typical RNA viruses exhibit the characteristics of multiple immunons, as they often have a diameter of 30 nm and 180 copies of a coat protein spaced ~5 nm apart, as illustrated in Fig. 1 [10]. The advantages of using such particles include the efficient cross-linking of B-cell receptors [10] and successful recognition by natural IgM antibodies that results in the activation of the classical complement pathway, causing the deposition of antigens on follicular dendritic cells (FDCs) [5, 12], enhancing germinal center (GC) formation [13], and inducing durable and long-lived plasma cells [14]. Accordingly, VLPs decorated with full-length antigens and loaded with RNA are considered an important tool for activation of B-cells and induction of long-lived B-cell responses [11]. In addition, for some VLPs, in particular RNA virus-derived VLPs, loading the particle with RNA or CpGs (DNA oligonucleotides rich in nonmethylated CG motifs) is possible, which results in TLR7/8 or TLR9 activation, respectively, in B cells. This augments IgG2a and IgA antibody responses and enhances plasma cell formation [15] and affinity maturation [16]. The type of RNA packaged in VLPs also matters, with bacterial RNA being the most efficient B-cell enhancer [17]. Below, we discuss some examples of prophylactic and therapeutic VLP-based vaccines that elicit efficient antibody responses, thus mediating protection against disease. In cases where the targeted antigen does not induce good TH cell responses (e.g., self-molecules or carbohydrates), TH against the VLPs is essential, as it will mediate intermolecular interactions, driving antibody responses against the targeted antigen as well.
Antigen processing is facilitated when antigens are particulate with repetitive surface epitopes spaced every 5–10 nm. Such repetitive structures are recognized by the immune system as Pathogen associated structural paterns (PASPs), which facilitate the cross-linking of B cells, natural IgM binding, complement activation, high-affinity long-lived antibody induction and GC formation. Created with BioRender.com
Infectious disease
Coronavirus disease
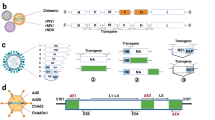
Coronaviruses are a family of viruses that have recently attracted much attention, mainly due to the ongoing pandemic caused by SARS-CoV-2. Coronaviruses are RNA viruses characterized by a single-stranded positive RNA molecule of approximately 30 kB, encoding, among others, four structural proteins, as illustrated in Fig. 2A: spike (S), membrane (M), nucleocapsid (N) and envelope (E). Extensive efforts have been made to develop vaccines against this family of viruses using conventional VLP platforms. However, the majority of these efforts are still in the preclinical stage, with the exception of some candidates against SARS-CoV-2 that are currently in phases I, II and III, as summarized in Table 2.
A A typical coronavirus based on four structural proteins: spike (S), envelope (E), membrane (M) and nucleocapsid (N). B Strategy of developing a mosaic VLP-based vaccine by genetically fusing the RBD of MERS-CoV into the optimized CuMVTT-VLPs, which incorporate a universal TT epitope and TLR7/8 ligand. Created with BioRender.com
Middle East Respiratory Syndrome (MERS): The Middle East respiratory syndrome coronavirus (MERS-CoV) causes severe respiratory disease in humans and continues to be a threat in more than 27 countries worldwide following the first registered pandemic in Saudi Arabia ten years ago. The mortality rate of MERS is very high and in the range of 40% [18]. MERS-CoV contains several immunogenic proteins, including the spike (S) protein, which mediates virus entry into host cells via its receptor dipeptidyl peptidase 4 (Dpp4). Accordingly, the receptor is used as the principle targeting agent against the virus in vaccine development. Preclinical studies were performed to develop vaccines against MERS-CoV using different platforms, including VLPs [19, 20].
We have recently optimized a plant-derived VLP called cucumber-mosaic virus-like particles (CuMVTT) as an improved vaccine platform incorporating a tetanus toxin (TT) epitope by genetic fusion. Incorporation of the TT epitope is thought to enhance the interaction between B cells and TH cells, especially in elderly people, due to the presence of preexisting TT-specific memory TH cells. Furthermore, CuMVTT-VLPs incorporate RNA from E. coli, which serves as a TLR7/8 agonist, during the expression process. This platform has proven to be highly immunogenic in mice, cats, dogs, rabbits and horses [21,22,23,24].
Using this new platform (CuMVTT), we have developed a scalable vaccine based on genetic fusion of the receptor-binding motif (RBM) of the MERS-CoV spike protein into CuMVTT-VLPs (Fig. 2B). The resultant mosaic VLP-based vaccine (mCuMVTT-MERS) was expressed in E. coli, allowing spontaneous packaging of the TLR7/8 ligand. mCuMVTT-MERS induced high levels of specific antibodies capable of neutralizing MERS-CoV/EMC/2012 isolate, demonstrating promising potential for translation into dromedaries and humans [25]. Wang et al. constructed a MERS-CoV-VLP vaccine using a baculovirus expression system that was tested in rhesus macaques. The developed vaccine could induce specific anti-RBD titers (ELISA) of 1:1280 in addition to T-cell-mediated immunity. The study demonstrated that MERS-CoV-VLPs have excellent immunogenicity and are promising vaccine candidates [26]. The same group also developed a chimeric VLP-based vaccine expressing RBD of MERS-CoV utilizing VP2 structural protein of canine parvovirus (CPV). Immunization with the chimeric vaccine also induced RBD-specific humoral and cellular immunity in murine models [27]. Unfortunately, no VLP-based vaccine has progressed to clinical trials thus far.
Severe Acute Respiratory Syndrome (COVID-19): The ongoing COVID-19 pandemic has overwhelmed the world’s health systems. Approximately thirty different vaccines have been approved for human use in different countries, and approximately eighty candidates are in clinical development, of which 19 are in phase III trials [28]. Nevertheless, with the continuous emergence of new variants of concern (VOCs), there is still a need for the development of effective, scalable, affordable and stable classical vaccines.
As an example, we have developed a COVID-19 vaccine by chemically coupling the receptor-binding domain (RBD) of SARS-CoV-2 to our immunologically optimized CuMVTT VLPs. We have utilized the most popular target for modification on the VLP surface, which contains accessible lysine residues (Fig. 3A). The displayed RBD recognized the viral receptor angiotensin-converting enzyme 2 (ACE2), confirming the correct conformational structure and resulting in a highly specific antibody response that neutralized SARS-CoV-2 [29] and VOCs [30] in vitro. A similar platform, CuMVTT-RBD, was tested for intranasal administration in a murine model. The vaccine candidate elicited a strong specific systemic and mucosal IgG and IgA response that efficiently neutralized different VOCs [31].
Schematic presentation of the following techniques. A Chemical techniques to conjugate an epitope to VLPs are mainly based on modifying a chemical group on the surface of the VLP. Lysine (Lys) residues on the surface of a VLP (shown as red dots) are the most commonly used target for modification. Epitopes should be modified to contain a free Cys for the chosen chemistry conjugation method. B A genetic fusion technique allowing the coexpression of both the VLP capsid proteins and the targeted epitope. Genetic fusion techniques may be considered a better option for GMP translational purposes and scaling up [37]. Created with BioRender.com
For translational purposes, we next designed a novel vaccine candidate by genetically grafting RBM of SARS-CoV-2 into CuMVTT-VLPs, called mosaic CuMVTT-RBM (Fig. 3B). The developed vaccine could be produced on a large scale (>2.5 million doses per 1000-liter fermenter run), showed high stability at 4 °C for 14 months and may be used for multiple booster doses. CuMVTT-RBM also induced high specific anti-RBD and anti-spike antibody titers in murine and rabbit models that were cross-reactive to mutant RBDs and VoCs [32]. Due to the high production rate and ease of storage at 4 °C, we expect that production and shipment costs will be orders of magnitude lower than for current RNA-based vaccines. Similar results were obtained with RBM fused to VLP AP205 [33]. In addition, we generated a double mosaic particle, which induced neutralizing antibodies against both RBM in S1 as well as the cleavage site in S2, a second, “minor” neutralizing epitope [34].
Using different techniques, other groups have also successfully developed vaccines against COVID-19 utilizing VLPs. For instance, Tan et al. designed and tested RBD-SpyAP205 vaccine in a prime/boost regimen in murine models. They used SpyTag/SpyCatcher technology to display RBD on the AP205-VLP platform. Their results showed that the vaccine candidate can elicit a neutralizing antibody response [35].
As discussed above, SARS-CoV-2 contains four essential structural proteins that can assemble into VLPs in mammalian cells without the viral genome. Yilmaz et al. described a VLP-based vaccine expressing hexaproline prefusion-stabilized spike (S-6p) as well as N, M and E structural proteins of SARS-CoV-2. The vaccine was absorbed to alum and formulated with K-type CpG ODN [36]. HEK293 cells were used to express the vaccine candidate, and vesicular VLPs that expressed the four structural proteins were generated. The results demonstrated effective production of anti-S, anti-RBD and anti-N IgG and prevention of lung pathology in vaccinated animals. This vaccine candidate is currently in a phase II clinical trial; please refer to Table 2.
In general, recombinant protein-based vaccines can be produced using different host expression systems, such as insect cells, yeast, mammalian cells and plants. Recently, plant-based vaccine development has become more accepted in the field following the COVID-19 pandemic. A recently published study described the successful construction of a SARS-CoV-2 vaccine based on VLPs in plants. The authors cloned the M, N and E genes of SARS-CoV-2 into a geminiviral vector and transiently expressed them in N. benthamiana plants. The process resulted in plant-derived VLPs that exhibited a similar shape and size as the native SARS-CoV-2 virion, without the spike protein. Figure 4 illustrates a plant-expression system process using the agroinfilitration method in tobacco plants. The authors indicated that such VLPs could serve in the future as a platform to carry S antigens as well [37].
The plant-derived VLP-production method has also been utilized by Medicago Inc. for the development of their SARS-CoV-2 vaccine (CoVLPs vaccine; Covifenz). CoVLPs were produced from the expression of a modified full-length S protein, which, upon expression in plant cells, trimerizes and moves to lipid rafts in the plasma membrane to spontaneously assemble into VLPs that “bud” off the surface of the plant cell. In macaques, these VLPs induced strong and lasting IgG responses, particularly in the presence of AS03 adjuvants, and boosted T-cell responses, resulting in protection of primates against viral infection [38]. CoVLPs are currently in late-stage clinical trials and have been approved in Canada for use in humans (Table 2).
Influenza
Influenza virus is an enveloped RNA virus belonging to the family Orthomyxoviridae. The virus surface glycoproteins hemagglutinin (HA) and neuraminidase (NA) are the major targets for vaccine development. HA consists of a globular head and a stem. The current marketed seasonal vaccines against influenza target the globular head of HA for neutralization; however, rapid antigenic variation in the globular head abrogates the affinity of the induced antibodies, making yearly or biyearly development of novel vaccines necessary [39]. Conversely, the stem of HA is more conserved; however, due to the immunodominance of the globular head and the steric hinderance of the stem, it may be challenging but not impossible to mount an efficient immune response against the stem [40]. Targeting NA, the second major protein in influenza virus, can prevent virus entry into the host cell, decrease disease severity, and inhibit virus replication [39]. Similar to HA and NA, the M2 protein is an integral surface protein of influenza virus, and the extracellular domain of M2 is highly conserved in influenza A [41]. Matrix protein (M) and nucleoprotein (NP) are the major internal proteins of the virus. Fig. 5 illustrates the structure and major antigens of influenza virus.
VLPs constitute an attractive, alternative vaccine platform to traditional influenza vaccine formulations, as they mimic the native virus but lack any genetic materials. Buffin et al. showed that the use of mammalian cells can offer many advantages for vaccine production, including the maintenance of glycosylation patterns. They developed a VLP vaccine containing HA, matrix (M1) and NA proteins. The expressed VLPs emulate the exterior surface of the authentic virus and showed high immunogenicity [42].
Using the bacteriophage Qß, we designed a VLP vaccine against various forms of the globular domain of HA. The globular domains were chemically coupled to the surface of the VLP [43]. Essentially, all vaccine candidates induced protective antibodies in mice that also cross-reacted with drifted strains. Interestingly, the HA globular domain was produced in bacteria, indicating that glycosylation was not essential for the induction of protective antibodies, as levels of hemagglutination inhibiting antibodies in mice were similar to those obtained with a classical vaccine [44]. In a next step, this vaccine was tested in a phase I study, revealing that this fully bacterial-produced vaccine was at least as immunogenic in humans as a classical, virus-based vaccine for both antibodies [45] and T cells [46]. Another interesting approach is based on HA displayed on ferritin nanoparticles, which induce high levels of broadly neutralizing antibodies against H1N1 influenza virus strains [47].
As HA proteins are very variable and poorly conserved, a vaccine based on conserved structures such as the extracellular domain of M2 (eM2) or the HA stem may be an interesting alternative to HA- and NA-based vaccines. Indeed, early experiments displaying eM2 on HBcAg by genetic fusion [41, 48, 49] or Qβ by chemical conjugation [50] induced antibodies that could protect against multiple clades of influenza A viruses. The Qβ-based M2e vaccine was also tested for its ability to induce protective antibodies upon intranasal administration. Vaccination using this route induced high levels of protective IgG and IgA antibodies in the serum and lungs of mice [51].
Similar to Qβ, AP205-VLPs derived from the bacteriophage AP205 have also shown high induction of specific antibodies against M2 protein displayed at the N-terminus by genetic fusion [52]. In the context of eM2-based vaccines against influenza virus, it is interesting to note that we have provided evidence in a murine influenza model that VLPs carrying prokaryotic RNA, a TLR7/8 ligand, and displaying eM2 induced better protective IgG subclasses than VLPs carrying eukaryotic RNA [17], highlighting the importance of the type of packaged RNA and confirming that the subclass of anti-M2 IgG antibodies is essential for protection [53].
The stem region of HA, which is more conserved than the globular domain of HA, may be an additional attractive target. Indeed, it is possible to express the stem region as a trimer suitable for chemical conjugation to VLPs [54], and when displayed on HBcAg, it can induce strong IgG responses in mice [55]. In addition, HA stem displayed on ferritin nanoparticles induced broadly neutralizing and protective antibodies in mice [56]. Thus, VLP-based vaccine candidates targeting conserved structures of influenza virus, HA or M2 proteins, may be a suitable alternative to the currently used influenza vaccines for battling seasonal and future influenza pandemics [57].
As an alternative method to induce broader immune coverage against influenza subtypes, a mosaic, multisubtype VLP vaccine has been developed containing 3 or 4 full-length HA proteins from H5N1, H7N2 and H9N2. The multiclade VLP vaccines were tested intranasally in ferrets and showed efficacy and immunogenicity [58]. Several VLP-based vaccines against H9N2 have also been described [59].
Malaria
The WHO estimated that there were 241 million malaria cases in 2020 in approximately 85 malaria-endemic countries in comparison to 227 million cases in 2019 [60]. Mortality cases increased in Sudan by 49% between 2016 and 2020, and 80% of reported cases were due to Plasmodium falciparum (P. falciparum) [60]. Vaccination against malaria can be categorized into three approaches: preerythrocytic vaccines, blood-stage vaccines, and transmission-blocking vaccines (TBV) [61]. The most advanced preerythrocytic vaccine is RTS,S (MosquirixTM), a VLP-based vaccine that was recommended by the WHO in October 2021 for use in high-risk areas [60]. RTS,S is discussed below in Section “Marketed VLP-based vaccines”.
Chan et al. designed and developed a transmission-blocking vaccine by utilizing duck HBV VLPs that incorporate large Pfs230 and Pfs25 proteins. The vaccine was expressed in the Hansenulla cell line compatible with cGMP. Induced antibodies could recognize the native protein on the gametocyte surface and reduce the transmission activity in a standard membrane feeding assay [62]. The same group recently used the small surface antigen (dS) of duck HBV to display circumsporozoite protein (CSP) in a larger portion on the surface of the scaffold. With the goal of overcoming RTS,S drawbacks, CSP is present on only a small portion of the vaccine surface [63]. The Barillas Mury group has also focused on developing a TBV by conjugating Pfs47 (a protein expressed on the surface of gametocytes) to an Acinetobacter phage (AP205-VLPs) using the Spycatcher-SpyTag technique. The candidate vaccine induced a high antibody titer with high affinity for Pfs47, confirming its potential [64].
Inflammation
VLPs have been used extensively for the development of therapeutic vaccines against different chronic inflammatory diseases. In this respect, proofs of concept for different preclinical vaccines in different animal species have been generated.
In insect-bite hypersensitivity (IBH), horses suffer from chronic allergic dermatitis caused by type-I/type-IV allergic reactions mediated by eosinophils and caused by midge (Culicoides) bites. Interleukin-5 (IL-5) is considered a key stimulator of eosinophils [65, 66] (Fig. 6).
IL-31 and IL-5 are central mediators of allergic itching. Typically, allergens penetrate the skin triggering mast cells and T-helper cells type 2 (TH2). Secretion of IL-31 stimulates peripheral sensory neurons which results in itching. IL-5 is the master cytokine required for both generation and activation of eosinophils. Created with BioRender.com
Monoclonal antibodies (mAbs) against IL-5 are successfully used to treat human diseases with an eosinophilic component, particularly eosinophilic asthma [67]. It may therefore be expected that lowering levels of IL-5 by vaccination reduces eosinophilic diseases in general, including eosinophilic infiltration of the horse skin in IBH [68], extending earlier observations in mice [69, 70]. To this end, we covalently linked equine IL-5 to our CuMVTT platform and tested the vaccine in a placebo-controlled study of thirty-four Iceland horses. The vaccine was formulated without adjuvants and did not show any safety concerns but induced anti-IL-5 autoantibodies in 89% of the vaccinated horses, which translated into clinical improvement [71]. Efficacy rates could be increased to 100% and maintained over several seasons by yearly booster injections, essentially eliminating symptoms almost completely [68]. This approach may be considered a breakthrough therapy targeting a chronic inflammatory disease in horses and has the potential for future translation to humans.
In addition to IL-5, IL-31 likewise plays a major role in IBH as well as in allergic pruritis in humans, dogs, monkeys and mice (Fig. 6). In contrast to IL-5, IL-31 mainly promotes itching; this itching, however, this results in chronic scratching causing local inflammation, typically exacerbated by infection [72]. Based on CuMVTT platform, an IL-31 equine vaccine was generated and tested in horses. The results demonstrated the safety of the vaccine and revealed the efficacy of the approach, resulting in strongly reduced clinical scores in the treatment group compared with the placebo group [24]. Atopic dermatitis is the most common allergic disease in dogs, causing extensive scratching and loss of fur as well as secondary infections. Canine IL-31 displayed on CuMVTT was tested in house dust mite-sensitized dogs who developed typical symptoms of atopic dermatitis upon topical challenge with allergen. Immunized dogs showed strongly reduced itching following immunization, which correlated with the induced specific antibody titers. However, a less pronounced response was noticed upon the decline in antibody titers, indicating that maintenance of IgG titers is an area for future improvement [23]. The performed study lays the foundation for a therapeutic modality for the effective treatment of atopic dermatitis in dogs. Further studies to increase and prolong the efficacy have yet to be performed, and the results with monoclonal anti-IL-31 antibodies will direct the path of the development of a vaccine targeting IL-31 in humans.
In addition to IL-5, VLPs displaying IL-13 may be an additional interesting modality to be broadly used to treat atopic dermatitis and are an avenue that should be explored in more detail together with VLPs targeting IL-4 [73].
It has been shown that mAbs generated against the proinflammatory cytokine IL-17A are highly effective against psoriasis, which affects between 1 and 4% of global populations [74]. The high cost of IL-17 biologics restricts their access by patients. Targeting IL-17A in mice using vaccines based on IL-17 displayed on Qß or CMVTT has proven efficacious in preclinical models of myocarditis, [75], rheumatoid arthritis and multiple sclerosis [76] as well as psoriasis [77].
mAbs targeting TNF-α have demonstrated efficacy across a wide range of inflammatory diseases, such as psoriasis, Crohn’s disease, rheumatoid arthritis and ulcerative colitis [78, 79]. Unfortunately, patients treated with anti-TNF-α may develop resistance in the long term in addition to the large cost associated with the therapy. Hence, it may be attractive to generate a vaccine targeting TNF [80]. Two VLP-based vaccines have been described: one vaccine was based on HPV and displayed a TNF-derived peptide using biotin/streptavidin [80], and the other was based on full-length TNF or a TNF-derived peptide displayed on Qβ using SMPH chemistry and a free Cys introduced into TNF or the peptide [81]. Both strategies induced therapeutic antibodies capable of neutralizing TNF and abolishing disease in collagen-induced arthritis. Interestingly, the vaccine based on entire TNF linked to Qβ-VLPs induced antibodies that neutralized both transmembrane and soluble TNF. In contrast, the vaccine candidate based on 20 amino acids (a.a.) derived from the N-terminus of TNF (Qβ-C-TNF(4-23)) neutralized soluble TNF but not the membrane form. Nevertheless, both vaccines yielded specific antibodies that were capable of protecting mice from rheumatoid arthritis. In contrast, only mice immunized against full-length TNF (Qβ-C-TNF(1-156)) showed increased susceptibility to infection with Listeria monocytogenes as well as enhanced reactivation of latent Mycobacterium tuberculosis, while mice that received the peptide-based vaccine were not immunocompromised. Therefore, targeting soluble TNF-α alone can be an effective strategy for clinical translation that might overcome the risk of opportunistic infections [81]. The vaccine based on HPV and a TNF-derived peptide increased levels of anti-TNF antibodies by 1000-fold in comparison to the fusion peptide alone. As discussed above, this vaccine candidate also inhibited the development of collagen-induced arthritis in a mouse model [82]. Taking these findings into account, a clinical study was performed based on (Qβ-C-TNF(3-24)). The study showed disappointing results; however, the vaccine based on a murine TNF-derived peptide failed to induce TNF-neutralizing antibodies in humans (unpublished).
Allergy
Allergen-specific desensitization is currently the only available treatment for allergies. Such a strategy requires long-term application of allergens and can result in life-threatening anaphylactic shock [83]. Therapeutic nonreactogenic vaccines able to alleviate allergic symptoms after a few injections are therefore considered an attractive strategy. For the treatment of cat allergies, we have previously described a therapeutic vaccine that consists of the major cat allergen (Fel d 1) coupled to Qβ-VLPs. A single injection was capable of inducing protection against the type-1 allergic reaction. Additionally, allergen-induced systemic basophil degranulation was inhibited by allergen-specific IgG antibodies, a property that was enhanced by FcγRIIb [84]. Conjugating the allergen to VLPs not only increased allergen-specific IgG responses but also strongly reduced the ability of the allergens to cause allergic reactions, an important safety feature of this new specific immunotherapy approach [85]. Similar findings were made with allergens displayed on HBcAg [86].
To allow for rapid translation, we have developed an alternative strategy to treat Fel d1 allergy in humans by vaccinating cats against their own allergen. Using a conjugate vaccine based on CuMVTT, our results indicated tolerability and no overt toxicity in cats, and the vaccine also generated a strong and sustained specific IgG response with high affinity and neutralizing capacity. Humans and cats are expected to benefit from this treatment, which will reduce the risk of asthma in humans and will facilitate the interaction of owners with their cats [87].
Hirschberg et al. investigated the immune response induced following sensitization with the house dust mite Dermatophagoides pteronyssinus (Der p1) and vaccination with hybrid Ty-VLPs derived from the p1 protein of yeast retrotransposon carrying the immunodominant epitope Der p1. Their results demonstrate that the hybrid vaccine abrogated allergen-specific IL-5 production, and the effect was mediated by CD4+ T cells [88]. In an older study, it was shown that a Der p 1-derived peptide induced strong IgG responses in humans when it was chemically conjugated to Qβ-VLPs [89]. Recent evidence suggests that VLP-based vaccination against allergens may also be attractive for prophylactic intervention [90].
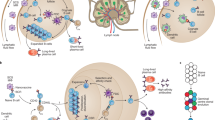
Peanut allergy is an increasingly frequent disease with a high burden, particularly in the US, and no treatment is currently available. CuMVTT-VLPs were used to display extracts of roasted peanut (Ara R) or purified single allergens Ara h1 or Ara h2 by chemical coupling and tested in a murine model of peanut allergy. One or two doses of all the generated vaccines could protect peanut-sensitized mice against anaphylactic reactions after challenge with the whole peanut extract. Surprisingly, all three vaccine candidates, also those based on single allergens, were able to confer protection against systemic as well as local challenge with the whole peanut extract, and all vaccines protected against eosinophil and mast cell infiltration in the gastrointestinal tract following an oral challenge with the complex extract [91]. These results demonstrate that vaccination against a single allergen can confer protection against challenge with a complex allergen mixture. Antibodies were established as the mode of action, as passive transfer of purified IgG or even a single monoclonal anti-Ara h 2 antibody conferred protection against allergic reactions in mice [91, 92]. The role of the FcγRIIb receptor on mast cells and basophils was also studied, and the obtained results demonstrate the critical role of this inhibitory receptor in blocking allergic reactions against the complex extract (Fig. 7). Briefly, the induction of high levels of specific IgG antibodies against a single allergen will result in the formation of immune complexes that will bind FcγRIIb, causing inhibition of IgE-mediated signals triggered also by other allergens [91, 93]. GMP material of the Ara h 2-based vaccine has been produced, and an IND in the US has been granted (Table 2) [94].
A cartoon illustrating the mechanism of inhibiting FcγRIIb on mast cells in a Peanut Allergy model. A Binding of peanut allergens to IgE bound to high-affinity FcƐ RI result in mast cells degranulation and allergic reactions. B Immunization with a VLP-based vaccine (CuMVTT-Arah2) induces specific-IgG antibodies for the single allergen Ara h2 forming an immune-complex that binds to FcγRIIb and inhibiting IgE-mediated signals. Created with BioRender.com
Pain
Nerve growth factor (NGF) is essential for early development of the nervous system, but at later stages, it becomes an essential mediator of pain. The mAb tanezumab has been shown to be efficacious in several human studies and is currently under registration [95], and a number of other mAbs are under development. Accordingly, developing a therapeutic vaccine targeting NGF may be of major interest. Indeed, we have shown the potent efficacy of a VLP-based vaccine against NGF to control pain in both rheumatoid arthritis and osteoarthritis in a preclinical study [96]. The developed vaccine was based on presenting murine NGF protein on CuMVTT VLPs. However, further studies are essential to establish the safety of a vaccine targeting NGF.
Neurodegenerative diseases
Alzheimer’s disease
Alzheimer’s disease (AD) is a major contributor to dementia, which is a disease that increases rapidly in frequency with aging. According to the Alzheimer Association, AD affects approximately 6.2 million people over the age of 65 in America, and that number is expected to increase to 13.8 million by 2060 [97]. Additionally, the current cost of therapy, particularly nursing aid, reached > $250 billion in 2020 in the USA. Active immunization against amyloid plaques is a promising therapeutic strategy [98]. However, there are currently no approved prophylactic or therapeutic vaccines against AD.
After the original breakthrough studies that put a potential vaccine against AD in the spotlight [99], a first VLP vaccine candidate was developed based on Qβ and the N-terminal end of Aβ (CAD106) [100]. Only the first 6 a.a. were chosen since clinical studies with the original vaccine candidate Aβ1-42 formulated in adjuvants induced self-specific T cells that induced meningo-encephalitis in up to 10% of the patients [101]. Preclinical evaluation demonstrated that immunization with CAD106 in the absence of adjuvants caused efficient removal of plaques in amyloid precursor protein (APP) transgenic mice, reproducing the original findings obtained with Aβ1-42 formulated in adjuvants. In addition, no signs of Aβ-specific T cells were observed. Subsequent clinical studies in AD patients demonstrated removal of plaques without signs of meningoencephalitis or microhemorrhages [102, 103]. However, there is currently no definitive evidence for improved performance of CAD106-immunized AD patients.
Using our immunologically optimized VLPs (CuMVTT-VLPs), as well as the Aβ1–6 peptide, we established a vaccine against AD. As discussed earlier, CuMVTT incorporates a TT epitope, which is proposed to enhance the response in aged individuals due to preexisting memory TH cells against tetanus. The developed vaccine may be used in both prophylactic and therapeutic settings. CuMVTT-Aβ1–6 showed high immunogenicity in young and old mice, and prepriming against tetanus enhanced the response against Aβ1-42. Indeed, displaying Aβ1–6 on the surface of CuMVTT-induced antibodies exhibiting the right specificity as sera from immunized mice could recognize AD plaques on postmortem brain sections as efficiently as a monoclonal antibody raised against Aβ peptide 1–17 [77]. Recent efforts have utilized HBc to produce a personalized AD vaccine tailored to a specific patient. Researchers have constructed a platform by inserting SpyCatcher into the major immunodominant region (MIR) of truncated HBc which could assemble into uniform VLPs readily binding to different SpyTag epitopes such as Aβ(1-6), Aβ(1-15), cAβ(1-7) cEP1, and cEP2 from β-amyloid monomer or oligomers and T294, pTau396-404, and pTau422 from tau proteins. The results of one study indicated that HBc-S-pTau422 alleviated cognitive deficits as well as neuropathy progression in transgenic mice [104]. Strong immunogenicity and a good safety profile were also observed for alternative Qß- [105], HPV- [106] and retroviral particle- [107] based vaccine candidates.
Aβ may not be the only target for treating AD. Indeed, Maphis et al. reported the development of a therapeutic vaccine against tauopathies, including AD. Tau peptide, phosphorylated at threonine 181, was chemically linked to Qβ-VLPs (pT181- Qβ). The vaccine induced a robust and long-lived specific antibody response recognizing postmortem human brain sections and was capable of reducing soluble and insoluble pTau in both the cortex and hippocampus of transgenic mice [108]. Hence, vaccines against Aβ or Tau or perhaps a combination of both may be promising ways forward for the development of AD therapies or perhaps even prophylaxis.
Parkinson’s disease
Parkinson’s disease is considered the second most frequent neurodegenerative disease and is associated with movement disorders, cognitive impairment and progressive disability. The disease affects 1% of people older than 60 years [109]. Neutralization of toxic alpha-synuclein (a-syn) oligomers is proposed to play a role in stopping the spread of oligomers and aggregates in the brain as well as cellular toxicity. Doucet et al. proposed an active vaccination strategy based on conjugating short peptides targeting the C-terminal region of α-syn to the Qβ platform. The obtained results demonstrated the ability of the vaccine to induce high antibody titers in wild-type mice and a-syn transgenic mice and recognize Lewy bodies. Despite the ability of the induced antibodies to specifically recognize oligomeric α-syn in solution, the vaccine failed to confer significant changes in a transgenic mouse model, showing no effect on the behavioral phenotype compared to the control condition [110]. Whether the absence of preclinical efficacy was due to the particular mouse model used remains unclear, and such results indicate the need for optimized preclinical models, which may be a challenging task.
Cancer
Antibodies have been studied and used in several tumors and have been demonstrated to be essential players in antitumour immunity. The effector function of antitumour antibodies includes induction of apoptosis, interference with tumor signaling pathways, antibody-dependent cellular cytotoxicity (ADCC) or activation of complement. Even though most antitumour antibodies are passively administered in the form of mAbs, vaccination may be a promising alternative for the active induction of antitumour antibodies. Nevertheless, most vaccines against cancer aim to induce T cells rather than antibodies, and the vaccines described below could be more accurately described as “niche candidates”. Prominent examples of antibody-inducing cancer vaccines are based on AP205, which has been used preclinically as a platform for a therapeutic vaccine for the induction of antibodies for cancer therapy. Using the SpyTag/SpyCatcher technique, the authors decorated VLPs with human epidermal growth factor receptor-2 (HER-2), a classical target for mAb therapy. Using this strategy, they could overcome B-cell tolerance, and potent anti-HER2 IgG antibodies were induced, hindering the progression of breast carcinoma tumors expressing HER-2 in mice [111]. Furthermore, overexpression of the xCT protein in triple-negative breast cancer tumors has been used as a target epitope and inserted into MS2-VLPs. The vaccine elicited a potent specific anti-IgG2a response, hampered tumor growth and prevented metastasis in an established 4T1 mouse model [91].
Human papillomavirus-related cancers account for ~4.5% of all types of malignancies and have been shown to affect more than half a million people every year [112]. Cancers associated with HPV include cervical carcinoma (99.7%) and squamous cell carcinoma of the vagina, anus, penis, vulva and oropharynx [113, 114]. The capsid L1 protein of HPV is a highly immunogenic epitope targeted in licensed prophylactic HPV vaccines (discussed later in 4). The structural L1 protein can self-assemble into VLPs that are morphologically similar to the parental native virus (Fig. 8). The minor capsid protein L2 is a highly conserved protein among HPV types and accordingly can be considered an appropriate target for the development of a next-generation Pan-HPV vaccine. However, L2-based vaccines have been shown to induce lower levels of neutralizing antibodies than L1-based vaccines [115]. Schellenbacher et al. have constructed a chimeric HPV vaccine expressing both L1 and L2 proteins (HPV16 L1-HPV16 L2 (chimera 17–36)) and adjuvanted VLPs induced broad-spectrum neutralizing antibodies directed against HPV types that are divergent from HPV16. These findings suggest the possibility for such a chimeric vaccine to protect against mucosal infection caused by high-risk, low-risk and beta HPV-associated diseases [116]. Clinical studies on RG1-VLP as another attempt to use L2 as a vaccine candidate will soon begin. RG1-VLPs contain the conserved amino acids 17–36 of HPV16L2, which are fused closely in the immunodominant surface loop of HPV16L1-VLP. Immunization experiments in small animal models have confirmed cross-protection against medically relevant high- and low-risk HPV types [117]. AAVLP-HPV is another chimeric vaccine that was constructed to display the L2 epitope [10,11,12,13, 15,16,17,18,19,20,21,22,23,24,25, 32, 118,119,120,121] from HPV16 and 31 onadeno-associated virus-derived VLPs. Immunization of mice and rabbits with these VLPs formulated in Montanide adjuvant induced specific antibody titers that were able to neutralize infection with several HPV types in a pseudovirion infection assay[122]. This chimeric vaccine successfully completed a phase I clinical trial (Table 2).
L1 and L2-capdid proteins of HPV A L1-capsid protein of HPV is a highly immunogenic epitope that can assemble into VLPs, example (licensed prophylactic HPV-based vaccines: Cervarix®, Gardasil® and Gardasil9®). B A chimeric HPV vaccine expressing both capsid proteins L1 and L2. Created with BioRender.Com
VLP-based therapeutic vaccines for the induction of protective T cells in a therapeutic regimen
VLPs decorated with T-cell epitopes are efficient tools for eliciting TH1 as well as cytotoxic T-cell lymphocytes (CTLs), even though VLPs do not incorporate genetic material [123]. Exogenous antigens preferably enter the major histocompatibility class-II (MHC-II) pathway to prime CD4+ T cells. However, previous studies have shown that particulate VLPs can be successfully taken up by APCs and cross-presented to enter the MHC-I pathway as well [124, 125]. Loading MHC-I can be achieved via a transporter associated with the antigen processing (TAP)-independent endosomal pathway or TAP-dependent endosome-to-cytosol pathway. Combining VLPs with stimuli for APCs results in CTL and TH1 responses [126, 127]. From all TLR ligands tested in mice, stimulation of TLR3, TLR7/8 and in particular TLR9 was most effective, while other TLRs, such as TLR2 and TLR4, had little ability to stimulate the CTL response [128]. Furthermore, Qß-VLPs loaded with TLR ligands, such as RNA or CpGs, were efficient at inducing both CTL and TH1 responses but failed to do so when loaded with polyglutamate or delivered empty [129]. Unexpectedly, we have shown that VLPs and TLR ligands can be delivered separately without a need for physical linkage for the generation of CTL responses in vivo as long as TLR ligands are formulated as particles [130, 131]. Depot-forming adjuvants may provide further opportunities to enhance the induced T-cell response [132].
Cancer
Cancer caused by human papilloma virus (HPV)
Prophylactic HPV vaccines aim to generate a humoral response against the late proteins L1 and L2. Accordingly, such vaccines would not show efficacy in a therapeutic setting, as virion capsid proteins are not detected in virus-infected proliferating cells. The main aim of an HPV therapeutic vaccine is to eliminate precancerous lesions and persistent HPV infection [133]. The early E-proteins (E6 and E7) are transcription factors that are responsible for driving the proliferation of infected cells [134]. Both proteins are consequently expressed at high levels in tumor cells, which makes them ideal targets for therapeutic vaccines. An increased CD4+:CD8+ ratio in the stroma, CD4+ T-cell response to E2 protein and E6- and E7-specific CD8+ T-cell infiltration have been detected in spontaneous regression of cervical interepithelial neoplasia (CIN) [133]. Several different preclinical approaches have been followed to develop a therapeutic HPV vaccine, including the use of VLPs. Some studies designed a T-cell-based vaccine by targeting the oncogenic proteins E6 and E7, and others developed a chimeric vaccine incorporating both capsid and oncogenic proteins. For example, Greenstone et al. demonstrated the ability of HPV-VLPs to induce cell-mediated immunity by generating chimeric VLPs consisting of the major capsid protein L1 plus the entire nonstructural E7 or E2 fused to minor capsid protein L2 [135]. HPV16 L1/L2-HPV16 E7 chimeric VLPs protected wild-type and MHC-II-deficient mice (lacking Th cells) from TC-1 tumor challenge [135]. Another study utilized a modified rabbit hemorrhagic disease virus VLP (RHDV-VLP) as a vaccine platform decorated with the E748–57 peptide. The developed vaccine was tested by using the TC-1-cell line expressing both E6 and E7 antigens in a murine model. The authors combined the vaccine with an anti-CTLA-4 checkpoint inhibitor or with anti-CD25 for Treg depletion. The results indicated a 50% reduction in tumor burden and a significantly enhanced survival rate [136]. We have shown that Qß-VLPs displaying or mixed with E7 protein could induce protection against tumor growth and enhance survival for more than 80% of vaccinated mice [131]. Another study generated a VLP-E7 vaccine incorporating a long E7 protein fragment into bursal disease virus VLPs. The VLP-E7 vaccine was tested in humanized transgenic mice expressing human HLA-A2 inoculated with the TC1/A2 cancer cell line in a therapeutic setting. The results showed complete eradication of established tumors as well as long-lasting immune responses [137].
Melanoma
A number of preclinical experiments have explored the efficacy of using VLPs as a therapeutic vaccine against melanoma.
Qβ-VLPs loaded with type-A CpGs (Qβ(G10), also called CMP-001) do not contain any tumor antigen and have been tested for anti-melanoma response upon intratumour injection. Sabree et al. demonstrated that CMP-001 induces the formation of anti-Qβ antibodies that opsonize Qβ VLPs, which are subsequently taken up by plasmacytoid dendritic cells (pDCs), leading to cytokine secretion and an antitumour T-cell response [138]. CMP-001 has been used in clinical trials, as shown in Table 2. In a recently published paper, Melhim et al. showed that CMP-001 alone or in combination with pembrolizumab (PD-1 inhibitor) in patients with advanced melanoma is tolerable and capable of reversing anti-PD1 resistance therapy with durable and strong clinical responses [139].
The Qβ(G10)MelanA vaccine was developed using a chemical coupling method, and potential efficacy was demonstrated in a phase I/II study in stage II-IV melanoma patients. More than 60% of the treated patients generated specific effector and memory T-cell responses as well as high IFN-γ, TNF-α and IL-2 cytokine production [140]. Recently, we have shifted our efforts toward developing personalized VLP-based vaccines targeting the patient’s tumor-specific epitopes for effective T-cell responses. Given that targeting a single antigen allows tumor cells to relapse by downregulating this single antigen, we designed a multitarget vaccine in a challenging transplanted murine melanoma model. Our results demonstrate that targeting both germline and mutated epitopes enhances the induced antitumour response [141]. We also tested the depot effect of microcrystalline (MCT) adjuvant in combination with CuMVTT-p33 vaccine. The results showed that MCT polarizes the response toward TH1 and enhances the induced antitumour response [132].
Mammary carcinoma
Since we have shown enhanced efficacy using the multitarget vaccine strategy in melanoma, we have expanded these findings to an aggressive metastatic breast carcinoma murine model. To further improve the active immunotherapy, we designed a personalized multitarget vaccine displaying elongated neoantigens using Qβ-VLPs packaging a TLR9 ligand by integrating mass spectrometry-based immunopeptidomics and whole-exome sequencing. Vaccination with long neoantigens was more effective than vaccination with short neoantigens, and the antitumor effect could significantly repolarize the tumor microenvironment, reduce lung metastasis and enhance survival [142].
Acquired Immunodeficiency Syndrome (AIDS)
Since its discovery as the etiological agent underlying acquired immunodeficiency syndrome (AIDS) in the 1980s, HIV has become a major global public health concern. Despite the ability of antiviral agents to keep the virus at bay, there is an urgent need for a therapeutic vaccine against HIV to eliminate long-term persistant HIV in patients on antiretroviral therapy. Insights into the pathogenesis of HIV-1 suggest that T-cell immunity plays a crucial role in controlling the acute phase of HIV infection [143], and long-term studies indicated that CD8+ CTL responses are associated with the control of HIV replication and infection clearance [144].
In 1996, Wolf et al. constructed and expressed a chimeric HIV-1 VLP vaccine. The 3rd variable region (V3) or CD4-binding domain of gp120 was inserted into Pr55gag-VLPs; this insertion did not interfere with the assembly ability of the VLPs. Mice were immunized using different routes and different chimeric Pr55gag/V3 VLPs without any adjuvants. The results showed a strong MHC-I (Dd) CTL response against a known epitope within the V3 region [145]. Furthermore, it has been shown that Gag-VLP vaccines were capable of inducing interferon alpha (IFN-α) in treated monocyte-derived dendritic cells (DCs) with increased expression of the mRNA encoding the proteins APOBEC3G and APOBEC3F, known inhibitors of HIV-1 [146]. In addition, yeast-derived VLPs containing the HIV V3 loop have been shown to be processed by murine dendritic cells for presentation in association with MHC class I [147].
Clinical development
Several prophylactic and therapeutic VLP-based vaccines are currently in the clinical development stage. We list the clinical trials related to the above discussed diseases in Table 2.
Marketed VLP-based vaccines
Human papilloma virus vaccine
Gardasil® was the 1st vaccine to be approved as a prophylactic vaccine against human-papilloma virus (HPV). Gardasil9®, covering 9 rather than 4 serotypes, has replaced Gardasil® in the US. The Gardasil®, Gardasil9® and Cervarix® vaccines are recombinantly manufactured VLPs of the L1-HPV epitope (Table 3). However, Gardasil9® contains a larger total antigenic load than Gardasil® to compensate for the larger number of different co-formulated VLPs [148]. Gardasil protects against 4 types of HPV (6, 11, 16 and 18), and Gardasil9® protects against 5 additional types (31, 33, 45, 52 and 58) [149]. The vaccines have been designed to elicit virus-neutralizing antibodies with the goal of preventing the initial HPV infection. These marketed vaccines have been shown to provide 100% protection against cervical precancerous as well as genital wart development with a protective effect expected to last a minimum of 4.5 years [150]. HPV vaccines are safe, and local adverse reactions such as swelling, pain and redness are mostly mild and reversible in the short term. Systemic reactions are rare and may involve nausea, fever, headache and fatigue [151]. The current cost of licensed HPV vaccines in yeasts or insect expression systems may be considered a barrier to their substantial implementation worldwide. Recent efforts to express low-cost HPV vaccines in E. coli have been successful. Cecolin® is a bivalent L1-HPV vaccine against HPV16 and HPV18 that was successfully expressed in E. coli and has shown high immunogenicity in mice, goats and rabbits [152]. On December 30, 2019, Cecolin® was officially approved by The National Medical Products Administration for use in China and was launched there in May 2020. In October 2021, the WHO granted prequalification for Cecolin®, making it the 1st China-manufactured HPV vaccine that received WHO prequalification [153].
Hepatitis B virus (HBV) vaccine
Several HBV vaccines against the potentially life-threatening disease hepatitis B are commercially available. HBV infection can cause a chronic infection and danger people with cirrhosis and liver cancer. The available vaccines show 98–100% protection against HBV.
The first-generation HBV vaccine was approved in 1981 based on the work of Bulmberg, who was awarded the Nobel Prize for the discovery of the Australian Antigen, now called hepatitis B surface antigen (HBsAg), in the serum of an infected patient [154]. The unique vaccine was based on obtaining HBsAg directly from human carriers. However, due to biosafety concerns, this blood-derived vaccine was replaced with an improved recombinant version of HBsAg in 1986 using the recombinant DNA method and production in yeast cells and currently also in mammalian cells. The HBV recombinant vaccine (second-generation) was initially produced using the yeast system. Available yeast-derived HBV vaccines are based on the self-assembly of HBsAg monomers into VLPs. Several studies have shown that purified yeast-derived HBsAg appears as ∼22 nm VLPs [155,156,157]. Sixty to seventy percent of the formed HBsAg VLPs consist of HBsAg monomer proteins, while the rest consist of lipids [158]. Overall, the formed VLPs are highly immunogenic and capable of eliciting potent neutralizing antibodies in addition to being a safe vaccine lacking any viral genome [159, 160]. Vaccination with a single antigen (small S antigen of HBsAg) has some limitations, including the prolonged time required to achieve seroprotection. Thirty to forty percent of adults reached seroprotection following 2 doses of the vaccine, but 10% may fail to achieve this even after administration of a 3rd dose [161]. The recently FDA-approved third-generation HBV vaccine PreHevBrio expresses three surface antigens of HBV, S, pre-S1 and pre-S2, and has been manufactured in mammalian cells. Phase III clinical trials confirmed high immunogenicity of the vaccine even in older people and those with chronic conditions [162]. The study concluded noninferiority of the vaccine in seroprotection rate when compared to a single antigen vaccine 4 weeks following the 3rd dose. The seroprotection rate was higher after the administration of 2 or 3 doses than after vaccination with the classical HBV vaccine (the control group). Rapid induction of a protective antibody profile was reported in more than 90% of participants after 2 doses; moreover, a good safety profile was observed. Table 4 summarizes the different HBV vaccines available, and Table 5 outlines the main differences between the second- and third-generation HBV vaccines.
Hepatitis E virus (HEV) vaccine
Hepatitis E virus is responsible for more than 50% of acute viral hepatitis in countries such as India China, Kenya, Sudan, Nepal and Bangladesh [163]. Studies have estimated that ∼35 million cases occur annually worldwide, with an average mortality rate of 0.2–4% and up to 25% in pregnant women [164]. HEV is a nonenveloped virus with a size of 27–35 nm and is divided into four different genotypes, I, II, III and IV [165]. Genotypes I and II are restricted to humans, while genotypes III and IV infect other mammals who can subsequently transmit the virus to humans. Despite this genetic diversity, all HEVs belong to the same serotype. The virus’s positive-stranded RNA genome contains three open reading frames, one of which encodes the viral capsid protein, the main target of neutralizing antibodies [166].
HEV239 (Hecolin®) was licensed as a vaccine against HEV in China in 2012. The vaccine is a recombinant VLP-based vaccine developed by Xiamen Innovax Biotech Co., Ltd. (China), and the encoding gene is from the ORF2 HEV genotype I strain. The produced recombinant protein comprising amino acids 368–606 (p239) 4-5 produced in an E. coli expression system, refolded and self-assembled into VLPs 20–30 nm in size [167]. The formed VLPs are highly immunogenic in both mice and rhesus macaques. Hecolin® has shown a good safety profile and 100% efficacy in humans. Studies are currently underway to assess vaccine safety and efficacy in high-risk groups for potential global use, as recommended by the WHO [168].
Malaria vaccine
RTS,S/AS01 (Mosquirix™) is considered the most advanced vaccine targeting the preerythrocytic malaria stage to date. The monovalent recombinant vaccine targets a fragment of circumsporozoite protein (CSP) protein, the major component of P. falciparum coat protein. The CSP protein fragment was fused to HBsAg as a carrier. In RTS,S, R: stands for the 19 central tandem repeats, T: stands for T epitope at the C-terminus, first S stands for modified surface antigen and the second S refers to the nonmodified surface antigen that allows for the presentation of CSP on the particle’s surface in mosaic HBsAg particles [169]. The construct assembles into VLPs and is formulated with AS01, a potent adjuvant. RTS,S/AS01 has been evaluated in a phase III clinical study; however, the results revealed only partial protection with some concerning safety issues, in particular a possible increase in mortality rates in young females [170]. RTS,S mosaic particles contain a 4-fold molar excess of nonmodified HBsAg subunits compared to CSP-HBsAg fusion protein. In contrast, the next-generation RTS,S/AS01 vaccine (R21) contains only CSP-HBsAg fusion protein and R21 therefore displays more CSP epitope on the surface and is capable of inducing sterile protection in mice [171]. Several late-stage clinical trials are ongoing to test the safety and efficacy of the next-generation R21 as well as to improve the efficacy of the first-generation RTS,S (Table 2). On October 6, 2021, the WHO recommended the widespread use of the RTS,S/AS01 vaccine among children in sub-Saharan Africa as well as in regions with moderate to high P. falciparum transmission [172].
Concluding remarks
VLPs exhibit a unique combination of high immunogenicity combined with excellent safety profiles, rendering them ideal platforms for vaccine design. Since the first description of VLP-based HBV vaccines, the field has made notable progress, and VLPs are now the basis of many marketed vaccines, such as those against HPV and HEV. Very exciting novel developments have been made regarding the use of VLPs as display platforms, not only for pathogen-derived antigens such as CSP displayed on HBsAg for immunization against malaria but also against self-molecules, such as Aβ for vaccines against Alzheimer’s disease or cytokines for the treatment of chronic diseases in companion animals and humans. We therefore expect several new products, both for humans and animals, to reach the market in the next few years.
References
Pumpens P, Pushko P. Virus-like particles, a comprehensive guide. Boca Raton, FL: CRC Press; 2022.
Nooraei S, Bahrulolum H, Hoseini ZS, Katalani C, Hajizade A, Easton AJ, et al. Virus-like particles: preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. J Nanobiotechnol. 2021;19:59.
Heddle JG, Chakraborti S, Iwasaki K. Natural and artificial protein cages: design, structure and therapeutic applications. Curr Opin Struct Biol. 2017;43:148–55.
Arevalo MT, Wong TM, Ross TM. Expression and purification of virus-like particles for vaccination. J Vis Exp. 2016;112:54041.
Mohsen MO, Gomes AC, Vogel M, Bachmann MF. Interaction of viral capsid-derived virus-like particles (VLPs) with the innate immune system. Vaccines. 2018;6:37.
Moradi Vahdat M, Hemmati F, Ghorbani A, Rutkowska D, Afsharifar A, Eskandari MH, et al. Hepatitis B core-based virus-like particles: a platform for vaccine development in plants. Biotechnol Rep. 2021;29:e00605.
Cheng L, Wang Y, Du J. Human papillomavirus vaccines: an updated review. Vaccines. 2020;8:391.
de Oliveira CM, Fregnani J, Villa LL. HPV vaccine: updates and highlights. Acta Cytol. 2019;63:159–68.
Cao Y, Bing Z, Guan S, Zhang Z, Wang X. Development of new hepatitis E vaccines. Hum Vaccin Immunother. 2018;14:2254–62.
Bachmann MF, Rohrer UH, Kundig TM, Burki K, Hengartner H, Zinkernagel RM. The influence of antigen organization on B cell responsiveness. Science. 1993;262:1448–51.
Dintzis HM, Dintzis RZ, Vogelstein B. Molecular determinants of immunogenicity: the immunon model of immune response. Proc Natl Acad Sci USA 1976;73:3671–5.
Link A, Zabel F, Schnetzler Y, Titz A, Brombacher F, Bachmann MF. Innate immunity mediates follicular transport of particulate but not soluble protein antigen. J Immunol. 2012;188:3724–33.
Zinkhan S, Ogrina A, Balke I, Resevica G, Zeltins A, de Brot S, et al. The impact of size on particle drainage dynamics and antibody response. J Control Rel. 2021;331:296–308.
Mohsen MO, Augusto G, Bachmann MF. The 3Ds in virus-like particle based-vaccines: “design, delivery and dynamics”. Immunol Rev. 2020;296:155–68.
Krueger CC, Thoms F, Keller E, Vogel M, Bachmann MF. Virus-specific secondary plasma cells produce elevated levels of high-avidity antibodies but are functionally short lived. Front Immunol. 2019;10:1831.
Chang X, Krenger P, Krueger CC, Zha L, Han J, Yermanos A, et al. TLR7 signaling shapes and maintains antibody diversity upon virus-like particle immunization. Front Immunol. 2021;12:827256.
Gomes AC, Roesti ES, El-Turabi A, Bachmann MF. Type of RNA packed in VLPs impacts IgG class switching-implications for an influenza vaccine design. Vaccines. 2019;7:47.
Ebrahim SH, Maher AD, Kanagasabai U, Alfaraj SH, Alzahrani NA, Alqahtani SA, et al. MERS-CoV Confirmation among 6,873 suspected persons and relevant Epidemiologic and Clinical Features, Saudi Arabia-2014 to 2019. Eclinicalmedicine. 2021;41:101191.
Rabaan AA, Bazzi AM, Al-Ahmed SH, Al-Tawfiq JA. Molecular aspects of MERS-CoV. Front Med. 2017;11:365–77.
Alharbi NK. Vaccines against Middle East respiratory syndrome coronavirus for humans and camels. Rev Med Virol. 2017;27:e1917.
Cabral-Miranda G, Lim SM, Mohsen MO, Pobelov IV, Roesti ES, Heath MD, et al. Zika virus-derived E-DIII protein displayed on immunologically optimized VLPs induces neutralizing antibodies without causing enhancement of dengue virus infection. Vaccines Basel. 2019;7:72. 2020;8.
Thoms F, Jennings GT, Maudrich M, Vogel M, Haas S, Zeltins A, et al. Immunization of cats to induce neutralizing antibodies against Fel d 1, the major feline allergen in human subjects. J Allergy Clin Immun. 2019;144:193–203.
Bachmann MF, Zeltins A, Kalnins G, Balke I, Fischer N, Rostaher A, et al. Vaccination against IL-31 for the treatment of atopic dermatitis in dogs. J Allergy Clin Immun. 2018;142:279–81.
Olomski F, Fettelschoss V, Jonsdottir S, Birkmann K, Thoms F, Marti E, et al. Interleukin 31 in insect bite hypersensitivity-Alleviating clinical symptoms by active vaccination against itch. Allergy. 2020;75:862–71.
Mohsen MO, Rothen D, Balke I, Martina B, Zeltina V, Inchakalody V, et al. Neutralization of MERS coronavirus through a scalable nanoparticle vaccine. NPJ Vaccines. 2021;6:107.
Wang C, Zheng X, Gai W, Zhao Y, Wang H, Wang H, et al. MERS-CoV virus-like particles produced in insect cells induce specific humoural and cellular imminity in rhesus macaques. Oncotarget. 2017;8:12686–94.
Wang C, Zheng X, Gai W, Wong G, Wang H, Jin H, et al. Novel chimeric virus-like particles vaccine displaying MERS-CoV receptor-binding domain induce specific humoral and cellular immune response in mice. Antivir Res. 2017;140:55–61.
J C. COVID-19 vaccine tracker. 2022. https://www.raps.org/news-and-articles/news-articles/2020/3/covid-19-vaccine-tracker.
Zha L, Chang X, Zhao H, Mohsen MO, Hong L, Zhou Y, et al. Development of a vaccine against SARS-CoV-2 based on the receptor-binding domain displayed on virus-like particles. Vaccines. 2021;9:395.
Chang X, Liu X, Mohsen MO, Zeltins A, Martina B, Vogel M, et al. Induction of broadly cross-reactive antibodies by displaying receptor binding domains of SARS-CoV-2 on virus-like particles. Vaccines. 2022;10:307.
Rothen DA, Krenger PS, Nonic A, Balke I, Vogt AS, Chang X, et al. Intranasal administration of a VLP-based vaccine induces neutralizing antibodies against SARS-CoV-2 and variants of concern. Allergy. 2022. https://doi.org/10.1111/all.15311.
Mohsen MO, Balke I, Zinkhan S, Zeltina V, Liu X, Chang X, et al. A scalable and highly immunogenic virus-like particle-based vaccine against SARS-CoV-2. Allergy. 2022;77:243–57.
Liu X, Chang X, Rothen D, Derveni M, Krenger P, Roongta S, et al. AP205 VLPs based on dimerized capsid proteins accommodate RBM domain of SARS-CoV-2 and serve as an attractive vaccine candidate. Vaccines (Basel). 2021;9:403.
Chang X, Zeltins A, Mohsen MO, Gharailoo Z, Zha L, Liu X, et al. A novel double mosaic virus-like particle-based vaccine against SARS-CoV-2 incorporates both receptor binding motif (RBM) and fusion domain. Vaccines (Basel). 2021;9:1287.
Tan TK, Rijal P, Rahikainen R, Keeble AH, Schimanski L, Hussain S, et al. A COVID-19 vaccine candidate using SpyCatcher multimerization of the SARS-CoV-2 spike protein receptor-binding domain induces potent neutralising antibody responses. Nat Commun. 2021;12:542.
Yilmaz IC, Ipekoglu EM, Bulbul A, Turay N, Yildirim M, Evcili I, et al. Development and preclinical evaluation of virus-like particle vaccine against COVID-19 infection. Allergy. 2022;77:258–70.
Moon KB, Jeon JH, Choi H, Park JS, Park SJ, Lee HJ, et al. Construction of SARS-CoV-2 virus-like particles in plant. Sci Rep. 2022;12:1005.
Pillet S, Arunachalam PS, Andreani G, Golden N, Fontenot J, Aye PP, et al. Safety, immunogenicity, and protection provided by unadjuvanted and adjuvanted formulations of a recombinant plant-derived virus-like particle vaccine candidate for COVID-19 in nonhuman primates. Cell Mol Immunol. 2022;19:222–33.
Jazayeri SD, Poh CL. Development of universal influenza vaccines targeting conserved viral proteins. Vaccines (Basel). 2019;7:169.
Krammer F, Garcia-Sastre A, Paleseu P. Is it possible to develop a “universal” influenza virus vaccine? Potential target antigens and critical aspects for a universal influenza vaccine. Cold Spring Harb Perspect Biol. 2018;10:a028845.
Neirynck S, Deroo T, Saelens X, Vanlandschoot P, Jou WM, Fiers W. A universal influenza A vaccine based on the extracellular domain of the M2 protein. Nat Med. 1999;5:1157–63.
Buffin S, Peubez I, Barriere F, Nicolai MC, Tapia T, Dhir V, et al. Influenza A and B virus-like particles produced in mammalian cells are highly immunogenic and induce functional antibodies. Vaccine. 2019;37:6857–67.
Jegerlehner A, Zabel F, Langer A, Dietmeier K, Jennings GT, Saudan P, et al. Bacterially produced recombinant influenza vaccines based on virus-like particles. PLoS One. 2013;8:e78947.
Skibinski DA, Hanson BJ, Lin Y, von Messling V, Jegerlehner A, Tee JB, et al. Enhanced neutralizing antibody titers and Th1 polarization from a novel Escherichia coli derived pandemic influenza vaccine. PLoS One. 2013;8:e76571.
Low JG, Lee LS, Ooi EE, Ethirajulu K, Yeo P, Matter A, et al. Safety and immunogenicity of a virus-like particle pandemic influenza A (H1N1) 2009 vaccine: results from a double-blinded, randomized Phase I clinical trial in healthy Asian volunteers. Vaccine. 2014;32:5041–8.
Skibinski DAG, Jones LA, Zhu YO, Xue LW, Au B, Lee B, et al. Induction of human T-cell and cytokine responses following vaccination with a novel influenza vaccine. Sci Rep. 2018;8:18007.
Kanekiyo M, Wei CJ, Yassine HM, McTamney PM, Boyington JC, Whittle JR, et al. Self-assembling influenza nanoparticle vaccines elicit broadly neutralizing H1N1 antibodies. Nature. 2013;499:102–6.
Tsybalova LM, Stepanova LA, Kuprianov VV, Blokhina EA, Potapchuk MV, Korotkov AV, et al. Development of a candidate influenza vaccine based on virus-like particles displaying influenza M2e peptide into the immunodominant region of hepatitis B core antigen: broad protective efficacy of particles carrying four copies of M2e. Vaccine. 2015;33:3398–406.
Fiers W, De Filette M, Birkett A, Neirynck S, Min Jou WA. “universal” human influenza A vaccine. Virus Res. 2004;103:173–6.
Jegerlehner A, Schmitz N, Storni T, Bachmann MF. Influenza A vaccine based on the extracellular domain of M2: weak protection mediated via antibody-dependent NK cell activity. J Immunol. 2004;172:5598–605.
Bessa J, Schmitz N, Hinton HJ, Schwarz K, Jegerlehner A, Bachmann MF. Efficient induction of mucosal and systemic immune responses by virus-like particles administered intranasally: implications for vaccine design. Eur J Immunol. 2008;38:114–26.
Tissot AC, Renhofa R, Schmitz N, Cielens I, Meijerink E, Ose V, et al. Versatile virus-like particle carrier for epitope based vaccines. PLoS One. 2010;5:e9809.
Schmitz N, Beerli RR, Bauer M, Jegerlehner A, Dietmeier K, Maudrich M, et al. Universal vaccine against influenza virus: linking TLR signaling to anti-viral protection. Eur J Immunol. 2012;42:863–9.
Lu IN, Kirsteina A, Farinelle S, Willieme S, Tars K, Muller CP, et al. Structure and applications of novel influenza HA tri-stalk protein for evaluation of HA stem-specific immunity. PLoS One. 2018;13:e0204776.
Kazaks A, Lu IN, Farinelle S, Ramirez A, Crescente V, Blaha B, et al. Production and purification of chimeric HBc virus-like particles carrying influenza virus LAH domain as vaccine candidates. BMC Biotechnol. 2017;17:79.
Yassine HM, Boyington JC, McTamney PM, Wei CJ, Kanekiyo M, Kong WP, et al. Hemagglutinin-stem nanoparticles generate heterosubtypic influenza protection. Nat Med. 2015;21:1065–70.
Lopez-Macias C. Virus-like particle (VLP)-based vaccines for pandemic influenza: performance of a VLP vaccine during the 2009 influenza pandemic. Hum Vaccin Immunother. 2012;8:411–4.
Tretyakova I, Pearce MB, Florese R, Tumpey TM, Pushko P. Intranasal vaccination with H5, H7 and H9 hemagglutinins co-localized in a virus-like particle protects ferrets from multiple avian influenza viruses. Virology. 2013;442:67–73.
Pushko P, Tretyakova I. Influenza virus like particles (VLPs): opportunities for H7N9 vaccine development. Viruses. 2020;12:518.
WHO. World malaria report 2021. 2021. https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2021.
Beeson JG, Kurtovic L, Dobano C, Opi DH, Chan JA, Feng G, et al. Challenges and strategies for developing efficacious and long-lasting malaria vaccines. Sci Transl Med. 2019;11:eaau1458.
Chan JA, Wetzel D, Reiling L, Miura K, Drew DR, Gilson PR, et al. Malaria vaccine candidates displayed on novel virus-like particles are immunogenic and induce transmission-blocking activity. PLoS One. 2019;14:e0221733.
Kurtovic L, Wetzel D, Reiling L, Drew DR, Palmer C, Kouskousis B, et al. Novel virus-like particle vaccine encoding the circumsporozoite protein of plasmodium falciparum is immunogenic and induces functional antibody responses in mice. Front Immunol. 2021;12:641421.
Yenkoidiok-Douti L, Williams AE, Canepa GE, Molina-Cruz A, Barillas-Mury C. Engineering a virus-like particle as an antigenic platform for a Pfs47-targeted malaria transmission-blocking vaccine. Sci Rep. 2019;9:16833.
Kurotaki T, Narayama K, Oyamada T, Yoshikawa H, Yoshikawa T. Immunopathological study on equine insect hypersensitivity (Kasen) in Japan. J Comp Pathol. 1994;110:145–52.
Kurotaki T, Narayama K, Arai Y, Arai S, Oyamada T, Yoshikawa H, et al. Langerhans cells within the follicular epithelium and the intradermal sweat duct in equine insect hypersensitivity “Kasen”. J Vet Med Sci. 2002;64:539–41.
Hammad H, Lambrecht BN. The basic immunology of asthma. Cell. 2021;184:2521–2.
Fettelschoss-Gabriel A, Fettelschoss V, Olomski F, Birkmann K, Thoms F, Buhler M, et al. Active vaccination against interleukin-5 as long-term treatment for insect-bite hypersensitivity in horses. Allergy. 2019;74:572–82.
Zou Y, Sonderegger I, Lipowsky G, Jennings GT, Schmitz N, Landi M, et al. Combined vaccination against IL-5 and eotaxin blocks eosinophilia in mice. Vaccine. 2010;28:3192–200.
Thrane S, Janitzek CM, Matondo S, Resende M, Gustavsson T, de Jongh WA, et al. Bacterial superglue enables easy development of efficient virus-like particle based vaccines. J Nanobiotechnology. 2016;14:30.
Fettelschoss-Gabriel A, Fettelschoss V, Thoms F, Giese C, Daniel M, Olomski F, et al. Treating insect-bite hypersensitivity in horses with active vaccination against IL-5. J Allergy Clin Immunol. 2018;142:1194–205. e3
Datsi A, Steinhoff M, Ahmad F, Alam M, Buddenkotte J. Interleukin-31: The “itchy” cytokine in inflammation and therapy. Allergy. 2021;76:2982–97.
Foerster J, Moleda A. Virus-like particle-mediated vaccination against interleukin-13 may harbour general anti-allergic potential beyond atopic dermatitis. Viruses. 2020;12:438.
Farahnik B, Beroukhim K, Nakamura M, Abrouk M, Zhu TH, Singh R, et al. Anti-IL-17 agents for psoriasis: a review of phase III data. J Drugs Dermatol. 2016;15:311–6.
Sonderegger I, Rohn TA, Kurrer MO, Iezzi G, Zou Y, Kastelein RA, et al. Neutralization of IL-17 by active vaccination inhibits IL-23-dependent autoimmune myocarditis. Eur J Immunol. 2006;36:2849–56.
Rohn TA, Jennings GT, Hernandez M, Grest P, Beck M, Zou Y, et al. Vaccination against IL-17 suppresses autoimmune arthritis and encephalomyelitis. Eur J Immunol. 2006;36:2857–67.
Zeltins A, West J, Zabel F, El Turabi A, Balke I, Haas S, et al. Incorporation of tetanus-epitope into virus-like particles achieves vaccine responses even in older recipients in models of psoriasis, Alzheimer’s and cat allergy. NPJ Vaccines. 2017;2:30.
Chames P, Van Regenmortel M, Weiss E, Baty D. Therapeutic antibodies: successes, limitations and hopes for the future. Br J Pharm. 2009;157:220–33.
Beck A, Wurch T, Bailly C, Corvaia N. Strategies and challenges for the next generation of therapeutic antibodies. Nat Rev Immunol. 2010;10:345–52.
Dalum I, Butler DM, Jensen MR, Hindersson P, Steinaa L, Waterston AM, et al. Therapeutic antibodies elicited by immunization against TNF-alpha. Nat Biotechnol. 1999;17:666–9.
Spohn G, Guler R, Johansen P, Keller I, Jacobs M, Beck M, et al. A virus-like particle-based vaccine selectively targeting soluble TNF-alpha protects from arthritis without inducing reactivation of latent tuberculosis. J Immunol. 2007;178:7450–7.
Chackerian B, Lowy DR, Schiller JT. Conjugation of a self-antigen to papillomavirus-like particles allows for efficient induction of protective autoantibodies. J Clin Invest. 2001;108:415–23.
Cappella A, Durham SR. Allergen immunotherapy for allergic respiratory diseases. Hum Vacc Immunother. 2012;8:1499–512.
Schmitz N, Dietmeier K, Bauer M, Maudrich M, Utzinger S, Muntwiler S, et al. Displaying Fel d1 on virus-like particles prevents reactogenicity despite greatly enhanced immunogenicity: a novel therapy for cat allergy. J Exp Med. 2009;206:1941–55.
Engeroff P, Caviezel F, Storni F, Thoms F, Vogel M, Bachmann MF. Allergens displayed on virus-like particles are highly immunogenic but fail to activate human mast cells. Allergy. 2018;73:341–9.
Sani MZ, Bargahi A, Momenzadeh N, Dehghani P, Moghadam MV, Maleki SJ, et al. Genetically engineered fusion of allergen and viral-like particle induces a more effective allergen-specific immune response than a combination of them. Appl Microbiol Biotechnol. 2021;105:77–91.
Thoms F, Jennings GT, Maudrich M, Vogel M, Haas S, Zeltins A, et al. Immunization of cats to induce neutralizing antibodies against Fel d 1, the major feline allergen in human subjects. J Allergy Clin Immunol. 2019;144:193–203.
Hirschberg S, Layton GT, Harris SJ, Savage N, Dallman MJ, Lamb JR. CD4(+) T cells induced by virus-like particles expressing a major T cell epitope down-regulate IL-5 production in an ongoing immune response to Der p 1 independently of IFN-gamma production. Int Immunol. 1999;11:1927–34.
Kundig TM, Senti G, Schnetzler G, Wolf C, Prinz Vavricka BM, Fulurija A, et al. Der p 1 peptide on virus-like particles is safe and highly immunogenic in healthy adults. J Allergy Clin Immunol. 2006;117:1470–6.
Soongrung T, Mongkorntanyatip K, Peepim T, Jitthamstaporn S, Pitakpolrat P, Kaewamatawong T, et al. Virus-like particles displaying major house dust mite allergen Der p 2 for prophylactic allergen immunotherapy. Allergy. 2020;75:1232–6.
Storni F, Zeltins A, Balke I, Heath MD, Kramer MF, Skinner MA, et al. Vaccine against peanut allergy based on engineered virus-like particles displaying single major peanut allergens. J Allergy Clin Immunol. 2020;145:1240–53. e3
Storni F, Cabral-Miranda G, Roesti E, Zha L, Engeroff P, Zeltins A, et al. A single monoclonal antibody against the peanut allergen Ara h 2 protects against systemic and local peanut allergy. Int Arch Allergy Immunol. 2020;181:334–41.
Bachmann MF, Mohsen MO, Kramer MF, Heath MD. Vaccination against allergy: a paradigm shift? Trends Mol Med. 2020;26:357–68.
Saiba. FDA approves IND application for peanut allergen vaccine. Phase I study in peanut allergy patients to begin May 2022. 2022. https://ir.q4europe.com/solutions/allergytherapeutics2018tf/3856/newsArticle.aspx?storyid=15315077.
Berenbaum F, Blanco FJ, Guermazi A, Miki K, Yamabe T, Viktrup L, et al. Subcutaneous tanezumab for osteoarthritis of the hip or knee: efficacy and safety results from a 24-week randomised phase III study with a 24-week follow-up period. Ann Rheum Dis. 2020;79:800–10.
von Loga IS, El-Turabi A, Jostins L, Miotla-Zarebska J, Mackay-Alderson J, Zeltins A, et al. Active immunisation targeting nerve growth factor attenuates chronic pain behaviour in murine osteoarthritis. Ann Rheum Dis. 2019;78:672–5.
2021 Alzheimer’s disease facts and figures. Alzheimers Dement. 2021;17:327–406. https://doi.org/10.1002/alz.12328.
Bertram L, Lill CM, Tanzi RE. The genetics of Alzheimer disease: back to the future. Neuron. 2010;68:270–81.
Schenk D, Barbour R, Dunn W, Gordon G, Grajeda H, Guido T, et al. Immunization with amyloid-beta attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature. 1999;400:173–7.
Wiessner C, Wiederhold KH, Tissot AC, Frey P, Danner S, Jacobson LH, et al. The second-generation active Abeta immunotherapy CAD106 reduces amyloid accumulation in APP transgenic mice while minimizing potential side effects. J Neurosci. 2011;31:9323–31.
Nicoll JA, Wilkinson D, Holmes C, Steart P, Markham H, Weller RO. Neuropathology of human Alzheimer disease after immunization with amyloid-beta peptide: a case report. Nat Med. 2003;9:448–52.
Wang CY, Wang PN, Chiu MJ, Finstad CL, Lin F, Lynn S, et al. UB-311, a novel UBITh((R)) amyloid beta peptide vaccine for mild Alzheimer’s disease. Alzheimers Dement. 2017;3:262–72.
Vandenberghe R, Riviere ME, Caputo A, Sovago J, Maguire RP, Farlow M, et al. Active Abeta immunotherapy CAD106 in Alzheimer’s disease: a phase 2b study. Alzheimers Dement. 2017;3:10–22.
Ji M, Xie XX, Liu DQ, Lu S, Zhang LX, Huang YR, et al. Engineered hepatitis B core virus-like particle carrier for precise and personalized Alzheimer’s disease vaccine preparation via fixed-point coupling. Appl Mater Today. 2020;19:100575.
Chackerian B, Rangel M, Hunter Z, Peabody DS. Virus and virus-like particle-based immunogens for Alzheimer’s disease induce antibody responses against amyloid-beta without concomitant T cell responses. Vaccine. 2006;24:6321–31.
Zamora E, Handisurya A, Shafti-Keramat S, Borchelt D, Rudow G, Conant K, et al. Papillomavirus-like particles are an effective platform for amyloid-beta immunization in rabbits and transgenic mice. J Immunol. 2006;177:2662–70.
Bach P, Tschape JA, Kopietz F, Braun G, Baade JK, Wiederhold KH, et al. Vaccination with Abeta-displaying virus-like particles reduces soluble and insoluble cerebral Abeta and lowers plaque burden in APP transgenic mice. J Immunol. 2009;182:7613–24.
Maphis NM, Peabody J, Crossey E, Jiang S, Jamaleddin Ahmad FA, Alvarez M, et al. Qss Virus-like particle-based vaccine induces robust immunity and protects against tauopathy. NPJ Vaccines. 2019;4:26.
Nussbaum RL, Ellis CE. Alzheimer’s disease and Parkinson’s disease. N Engl J Med. 2003;348:1356–64.
Doucet M, El-Turabi A, Zabel F, Hunn BHM, Bengoa-Vergniory N, Cioroch M, et al. Preclinical development of a vaccine against oligomeric alpha-synuclein based on virus-like particles. PLoS One. 2017;12:e0181844.
Palladini A, Thrane S, Janitzek CM, Pihl J, Clemmensen SB, de Jongh WA, et al. Virus-like particle display of HER2 induces potent anti-cancer responses. Oncoimmunology. 2018;7:e1408749.
de Martel C, Plummer M, Vignat J, Franceschi S. Worldwide burden of cancer attributable to HPV by site, country and HPV type. Int J Cancer. 2017;141:664–70.
Arbyn M, Weiderpass E, Bruni L, de Sanjose S, Saraiya M, Ferlay J, et al. Estimates of incidence and mortality of cervical cancer in 2018: a worldwide analysis. Lancet Glob Health. 2020;8:e191–e203.
Smalley Rumfield C, Roller N, Pellom ST, Schlom J, Jochems C. Therapeutic vaccines for HPV-associated malignancies. Immunotargets Ther. 2020;9:167–200.
Yousefi Z, Aria H, Ghaedrahmati F, Bakhtiari T, Azizi M, Bastan R, et al. An update on human papilloma virus vaccines: history, types, protection, and efficacy. Front Immunol. 2021;12:805695.
Schellenbacher C, Roden R, Kirnbauer R. Chimeric L1-L2 virus-like particles as potential broad-spectrum human papillomavirus vaccines. J Virol. 2009;83:10085–95.
Huber B, Wang JW, Roden RBS, Kirnbauer R. RG1-VLP and other L2-based, broad-spectrum HPV vaccine candidates. J Clin Med. 2021;10:1044.
Lu Y, Chan W, Ko BY, VanLang CC, Swartz JR. Assessing sequence plasticity of a virus-like nanoparticle by evolution toward a versatile scaffold for vaccines and drug delivery. Proc Natl Acad Sci USA 2015;112:12360–5.
Tumban E, Muttil P, Escobar CA, Peabody J, Wafula D, Peabody DS, et al. Preclinical refinements of a broadly protective VLP-based HPV vaccine targeting the minor capsid protein, L2. Vaccine. 2015;33:3346–53.
Lang R, Winter G, Vogt L, Zurcher A, Dorigo B, Schimmele B. Rational design of a stable, freeze-dried virus-like particle-based vaccine formulation. Drug Dev Ind Pharm. 2009;35:83–97.
Vicente T, Roldao A, Peixoto C, Carrondo MJ, Alves PM. Large-scale production and purification of VLP-based vaccines. J Invertebr Pathol. 2011;107:S42–8.
Nieto K, Weghofer M, Sehr P, Ritter M, Sedlmeier S, Karanam B, et al. Development of AAVLP(HPV16/31L2) particles as broadly protective HPV vaccine candidate. PLoS One. 2012;7:e39741.
Deml L, Schirmbeck R, Reimann J, Wolf H, Wagner R. Recombinant human immunodeficiency Pr55gag virus-like particles presenting chimeric envelope glycoproteins induce cytotoxic T-cells and neutralizing antibodies. Virology. 1997;235:26–39.
Moron VG, Rueda P, Sedlik C, Leclerc C. In vivo, dendritic cells can cross-present virus-like particles using an endosome-to-cytosol pathway. J Immunol. 2003;171:2242–50.
Joffre OP, Segura E, Savina A, Amigorena S. Cross-presentation by dendritic cells. Nat Rev Immunol. 2012;12:557–69.
Bachmann MF, Oxenius A, Pircher H, Hengartner H, Ashton-Richardt PA, Tonegawa S, et al. TAP1-independent loading of class I molecules by exogenous viral proteins. Eur J Immunol. 1995;25:1739–43.
Ruedl C, Schwarz K, Jegerlehner A, Storni T, Manolova V, Bachmann MF. Virus-like particles as carriers for T-cell epitopes: limited inhibition of T-cell priming by carrier-specific antibodies. J Virol. 2005;79:717–24.
Schwarz K, Storni T, Manolova V, Didierlaurent A, Sirard JC, Rothlisberger P, et al. Role of Toll-like receptors in costimulating cytotoxic T cell responses. Eur J Immunol. 2003;33:1465–70.
Keller SA, Bauer M, Manolova V, Muntwiler S, Saudan P, Bachmann MF. Cutting edge: limited specialization of dendritic cell subsets for MHC class II-associated presentation of viral particles. J Immunol. 2010;184:26–9.
Mohsen MO, Gomes AC, Cabral-Miranda G, Krueger CC, Leoratti FM, Stein JV, et al. Delivering adjuvants and antigens in separate nanoparticles eliminates the need of physical linkage for effective vaccination. J Control Rel. 2017;251:92–100.
Gomes AC, Flace A, Saudan P, Zabel F, Cabral-Miranda G, Turabi AE, et al. Adjusted particle size eliminates the need of linkage of antigen and adjuvants for appropriated T cell responses in virus-like particle-based vaccines. Front Immunol. 2017;8:226.
Mohsen MO, Heath MD, Cabral-Miranda G, Lipp C, Zeltins A, Sande M, et al. Correction to: vaccination with nanoparticles combined with micro-adjuvants protects against cancer. J Immunother Cancer. 2019;7:137.
Wang R, Pan W, Jin L, Huang W, Li Y, Wu D, et al. Human papillomavirus vaccine against cervical cancer: opportunity and challenge. Cancer Lett. 2020;471:88–102.
Longworth MS, Laimins LA. Pathogenesis of human papillomaviruses in differentiating epithelia. Microbiol Mol Biol Rev. 2004;68:362–72.
Greenstone HL, Nieland JD, de Visser KE, De Bruijn MLH, Kirnbauer R, Roden RBS, et al. Chimeric papillomavirus virus-like particles elicit antitumor immunity against the E7 oncoprotein in an HPV16 tumor model. Proc Natl Acad Sci USA 1998;95:1800–5.
Jemon K, Young V, Wilson M, McKee S, Ward V, Baird M, et al. An enhanced heterologous virus-like particle for human papillomavirus type 16 tumour immunotherapy. PLoS One. 2013;8:e66866.
Martin Caballero J, Garzon A, Gonzalez-Cintado L, Kowalczyk W, Jimenez Torres I, Calderita G, et al. Chimeric infectious bursal disease virus-like particles as potent vaccines for eradication of established HPV-16 E7-dependent tumors. PLoS One. 2012;7:e52976.
Sabree SA, Voigt AP, Blackwell SE, Vishwakarma A, Chimenti MS, Salem AK, et al. Direct and indirect immune effects of CMP-001, a virus-like particle containing a TLR9 agonist. J Immunother Cancer. 2021;9:e002484.
Milhem M, Zakharia Y, Davar D, Buchbinder E, Medina T, Daud A, et al. Durable responses in anti-Pd-1 refractory melanoma following intratumoral injection of a toll-like receptor 9 (Tlr9) agonist, Cmp-001, in combination with pembrolizumab. J Immunother Cancer. 2020;8:A2–A3.
Speiser DE, Schwarz K, Baumgaertner P, Manolova V, Devevre E, Sterry W, et al. Memory and effector CD8 T-cell responses after nanoparticle vaccination of melanoma patients. J Immunother. 2010;33:848–58.
Mohsen MO, Vogel M, Riether C, Muller J, Salatino S, Ternette N, et al. Targeting mutated plus germline epitopes confers pre-clinical efficacy of an instantly formulated cancer nano-vaccine. Front Immunol. 2019;10:1015.
Mohsen MO, Speiser DE, Michaux J, Pak H, Stevenson BJ, Vogel M, et al. Bedside formulation of a personalized multi-neoantigen vaccine against mammary carcinoma. J Immunother Cancer. 2022;10:e002927.
Koup RA, Safrit JT, Cao Y, Andrews CA, McLeod G, Borkowsky W, et al. Temporal association of cellular immune responses with the initial control of viremia in primary human immunodeficiency virus type 1 syndrome. J Virol. 1994;68:4650–5.
Paul WE. Can the immune-response control hiv-infection. Cell. 1995;82:177–82.
Wagner R, Deml L, Schirmbeck R, Niedrig M, Reimann J, Wolf H. Construction, expression, and immunogenicity of chimeric HIV-1 virus-like particles. Virology. 1996;220:128–40.
Chang MO, Suzuki T, Yamamoto N, Watanabe M, Takaku H. HIV-1 Gag-virus-like particles inhibit HIV-1 replication in dendritic cells and T cells through IFN-alpha-dependent upregulation of APOBEC3G and 3F. J Innate Immun. 2012;4:579–90.
Bachmann MF, Lutz MB, Layton GT, Harris SJ, Fehr T, Rescigno M, et al. Dendritic cells process exogenous viral proteins and virus-like particles for class I presentation to CD8+ cytotoxic T lymphocytes. Eur J Immunol. 1996;26:2595–600.
Harper DM, DeMars LR. HPV vaccines-a review of the first decade. Gynecol Oncol. 2017;146:196–204.
Gupta AK, MacLeod MA, Abramovits W. GARDASIL 9 (Human Papillomavirus 9-Valent Vaccine, Recombinant). Skinmed. 2016;14:33–7.
Schiller JT, Lowy DR. Understanding and learning from the success of prophylactic human papillomavirus vaccines. Nat Rev Microbiol. 2012;10:681–92.
Phillips A, Patel C, Pillsbury A, Brotherton J, Macartney K. Safety of human papillomavirus vaccines: an updated review. Drug Saf. 2018;41:329–46.
Hu YM, Huang SJ, Chu K, Wu T, Wang ZZ, Yang CL, et al. Safety of an Escherichia coli-expressed bivalent human papillomavirus (types 16 and 18) L1 virus-like particle vaccine An open-label phase I clinical trial. Hum Vacc Immunother. 2014;10:469–75.
Organization WH. Vaccines Cecolin. https://extranet.who.int/pqweb/content/cecolin®. 2021.
Roldao A, Mellado MC, Castilho LR, Carrondo MJ, Alves PM. Virus-like particles in vaccine development. Expert Rev Vaccines. 2010;9:1149–76.
Gurramkonda C, Adnan A, Gabel T, Lunsdorf H, Ross A, Nemani SK, et al. Simple high-cell density fed-batch technique for high-level recombinant protein production with Pichia pastoris: application to intracellular production of Hepatitis B surface antigen. Micro Cell Fact. 2009;8:13.
Valenzuela P, Medina A, Rutter WJ, Ammerer G, Hall BD. Synthesis and assembly of hepatitis B virus surface antigen particles in yeast. Nature. 1982;298:347–50.
McAleer WJ, Buynak EB, Maigetter RZ, Wampler DE, Miller WJ, Hilleman MR. Human hepatitis B vaccine from recombinant yeast. 1984. Biotechnology. 1992;24:500–2.
Gavilanes F, Gonzalez-Ros JM, Peterson DL. Structure of hepatitis B surface antigen. Characterization of the lipid components and their association with the viral proteins. J Biol Chem. 1982;257:7770–7.
Bachmann MF, Jennings GT. Vaccine delivery: a matter of size, geometry, kinetics and molecular patterns. Nat Rev Immunol. 2010;10:787–96.
Grgacic EV, Anderson DA. Virus-like particles: passport to immune recognition. Methods. 2006;40:60–5.
Peters MG, Weinbaum C, Tan LLJ, Baine WB, Dienstag JL & Liang TJ. Advisory Committee on Immunization Practices and Centers for Disease Control and Prevention. A comprehensive immunization strategy to eliminate transmission of hepatitis B infection in the United States. Pediatrics. 2006;118:404.
Vesikari T, Finn A, van Damme P, Leroux-Roels I, Leroux-Roels G, Segall N, et al. Immunogenicity and safety of a 3-antigen hepatitis B vaccine vs a single-antigen hepatitis B vaccine: a phase 3 randomized clinical trial. JAMA Netw Open. 2021;4:e2128652.
Lanini S, Garbuglia AR, Lapa D, Puro V, Navarra A, Pergola C, et al. Epidemiology of HEV in the Mediterranean basin: 10-year prevalence in Italy. BMJ Open. 2015;5:e007110.
Wu X, Chen P, Lin H, Hao X, Liang Z. Hepatitis E virus: current epidemiology and vaccine. Hum Vaccin Immunother. 2016;12:2603–10.
Holla RP, Ahmad I, Ahmad Z, Jameel S. Molecular virology of hepatitis E virus. Semin Liver Dis. 2013;33:3–14.
Bradley DW. Hepatitis E virus: a brief review of the biology, molecular virology, and immunology of a novel virus. J Hepatol. 1995;22:140–5.
Zhang X, Wei M, Pan H, Lin Z, Wang K, Weng Z, et al. Robust manufacturing and comprehensive characterization of recombinant hepatitis E virus-like particles in Hecolin((R)). Vaccine. 2014;32:4039–50.
Mazalovska M, Kouokam JC. Progress in the production of virus-like particles for vaccination against hepatitis E virus. Viruses. 2020;12:826.
Arora N, Anbalagan LC, Pannu AK. Towards eradication of malaria: is the WHO’s RTS,S/AS01 vaccination effective enough? Risk Manag Health P. 2021;14:1033–9.
Klein SL, Shann F, Moss WJ, Benn CS, Aaby P. RTS,S malaria vaccine and increased mortality in girls. mBio. 2016;7:e00514–16.
Collins KA, Snaith R, Cottingham MG, Gilbert SC, Hill AVS. Enhancing protective immunity to malaria with a highly immunogenic virus-like particle vaccine. Sci Rep. 2017;7:46621.
WHO. WHO recommends groundbreaking malaria vaccine for children at risk. 2021. https://www.who.int/news/item/06-10-2021-who-recommends-groundbreaking-malaria-vaccine-for-children-at-risk.
Khudyakov YP, Pumpens P. Viral nanotechnology. Boca Raton, FL: CRC Press; 2021.
Mohsen MO, Zha L, Cabral-Miranda G, Bachmann MF. Major findings and recent advances in virus-like particle (VLP)-based vaccines. Semin Immunol. 2017;34:123–32.
Frietze KM, Peabody DS, Chackerian B. Engineering virus-like particles as vaccine platforms. Curr Opin Virol. 2016;18:44–9.
Fiedler JD, Higginson C, Hovlid ML, Kislukhin AA, Castillejos A, Manzenrieder F, et al. Engineered mutations change the structure and stability of a virus-like particle. Biomacromolecules. 2012;13:2339–48.
Acknowledgements
We would like to thank Paul Pumpens, the father of VLPs, for his very important insights in the field and for never getting tired of answering our questions.
Funding
Open access funding provided by University of Bern.
Author information
Authors and Affiliations
Contributions
Writing, revision and editing of manuscript: MOM and MFB (equal contribution).
Corresponding author
Ethics declarations
Competing interests
MFB is a board member of Saiba AG and holds the patent of CuMVTT. MOM received payments from Saiba AG to work on the development of vaccines against Dengue and SARS-CoV-2. MFB and MOM are shareholders of Saiba AG and DeepVax GmbH.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohsen, M.O., Bachmann, M.F. Virus-like particle vaccinology, from bench to bedside. Cell Mol Immunol 19, 993–1011 (2022). https://doi.org/10.1038/s41423-022-00897-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-022-00897-8
Keywords
This article is cited by
-
The quest for nanoparticle-powered vaccines in cancer immunotherapy
Journal of Nanobiotechnology (2024)
-
Nanotechnology’s frontier in combatting infectious and inflammatory diseases: prevention and treatment
Signal Transduction and Targeted Therapy (2024)
-
Virusähnliche Partikel – Impfstoffe, die den Eindringling imitieren
BIOspektrum (2024)
-
Virus-like particle-mediated delivery of structure-selected neoantigens demonstrates immunogenicity and antitumoral activity in mice
Journal of Translational Medicine (2024)
-
A recombinant virus-like particle vaccine against adenovirus-7 induces a potent humoral response
npj Vaccines (2023)