Abstract
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) initiates the infection process by binding to the viral cellular receptor angiotensin-converting enzyme 2 through the receptor-binding domain (RBD) in the S1 subunit of the viral spike (S) protein. This event is followed by virus–cell membrane fusion mediated by the S2 subunit, which allows virus entry into the host cell. Therefore, the SARS-CoV-2 S protein is a key therapeutic target, and prevention and treatment of coronavirus disease 2019 (COVID-19) have focused on the development of neutralizing monoclonal antibodies (nAbs) that target this protein. In this review, we summarize the nAbs targeting SARS-CoV-2 proteins that have been developed to date, with a focus on the N-terminal domain and RBD of the S protein. We also describe the roles that binding affinity, neutralizing activity, and protection provided by these nAbs play in the prevention and treatment of COVID-19 and discuss the potential to improve nAb efficiency against multiple SARS-CoV-2 variants. This review provides important information for the development of effective nAbs with broad-spectrum activity against current and future SARS-CoV-2 strains.
Similar content being viewed by others
Introduction
Coronavirus disease 2019 (COVID-19) is a newly emerged infectious disease first identified in December 2019. The major clinical features of COVID-19 include fever, shortness of breath, cough, headache, and fatigue, which can lead to severe pneumonia, lung injury, acute respiratory/multiorgan failure, and death [1,2,3,4]. A variety of risk factors, including older age, hypertension, obesity, diabetes mellitus, and cardiovascular comorbidities, are associated with COVID-19 [5,6,7,8]. As of July 16, 2021, the WHO had reported over 188.6 million confirmed COVID-19 cases and more than 4 million deaths worldwide, resulting in devastating damage. Although several vaccines, including two mRNA vaccines (BNT162b2 and mRNA-1273) and one adenovirus-based vaccine (Ad.26.COV2.S), have been authorized by the U.S. FDA for immunizing persons 16 years and older (BNT162b2) or 18 years (mRNA-1273) and older to prevent COVID-19 [9,10,11], effective countermeasures are still needed to control the global COVID-19 pandemic.
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is the causative agent of COVID-19. It belongs to the Betacoronavirus genus of the Coronaviridae family in the order Nidovirales. SARS-CoV-1 and Middle East respiratory syndrome coronavirus (MERS-CoV), two other highly pathogenic coronaviruses first reported in 2002 and 2012, respectively [12, 13], are also betacoronaviruses. Both SARS-CoV-1 and MERS-CoV have limited transmission among humans, leading to case fatality rates of approximately 10% (774/8098) for SARS-CoV-1 [14] and approximately 34.4% (886/2574) for MERS-CoV. Compared to SARS-CoV-1 and MERS-CoV, SARS-CoV-2 exhibits superior human-to-human transmissibility [15,16,17], potentially contributing to its widespread infection and the global COVID-19 pandemic.
Similar to other coronaviruses, SARS-CoV-2 is an enveloped, single-stranded, and positive-sense RNA virus [18]. The viral genome encodes four major structural proteins (spike (S), membrane (M), envelope (E), and nucleocapsid (N)) (Fig. 1a), several nonstructural proteins (NSP1-16), and a series of accessory proteins, including 3a, 6, 7a, 7b, 8, and 10 [18, 19]. NSP proteins form replication-transcription complexes and mainly participate in biological processes, such as viral replication, protein processing, transcription, and proteolysis. Some function as RNA-dependent RNA polymerases, in addition to binding ATP and zinc ions [19, 20]. The E and M proteins may be involved in RNA packaging and virus assembly and release; the E protein has evolved to include a robust membrane topology, and it forms either a cation channel regulated by pH or an ion channel potentially inhibited by gliclazide and memantine [19, 21,22,23]. The N protein, which is located inside the virion, is responsible for RNA packaging and virus replication. It contains phosphorylation and protein kinase sites and might be important in modulating antiviral immunity and inhibiting interferon production by targeting the retinoic acid inducible gene-I-like receptor pathway [24,25,26].
Structural overview of the SARS-CoV-2 spike (S) protein. a Schematic diagram of the SARS-CoV-2 virion and its S protein. E envelope, M membrane, N nucleocapsid, ACE2 angiotensin-converting enzyme 2. b Cryo-EM structure of the SARS-CoV-2 S protein trimer (PDB 6VXX). The three subunits are colored orange, green, and blue. c Close-up views of the SARS-CoV-2 S receptor-binding domain (RBD) and N-terminal domain (NTD) in the S1 subunit. d Crystal structure of the SARS-CoV-2 RBD in complex with human ACE2 (PDB 6M0J). Human ACE2 is colored in light blue
SARS-CoV-2 S protein receptor binding and membrane fusion
Similar to the S proteins of other pathogenic coronaviruses, the S protein of SARS-CoV-2 plays the most important role in virus infection and pathogenesis and is therefore an important target for designing and developing effective COVID-19 vaccines and therapeutic antibodies. The S protein contains the S1 and S2 subdomains (Fig. 1a, b), which are responsible for host cell receptor binding and membrane fusion, respectively, mediated through the receptor-binding domain (RBD) in the S1 region (Fig. 1c) and heptad repeat region 1 (HR1) and HR2 in the S2 region [27, 28]. The N-terminal domain (NTD) is located on the S1 subunit (Fig. 1c) and has the potential to bind sialic acids or coreceptors [29, 30], but its specific function is still not fully understood. The RBD is composed of a core and a receptor-binding motif (RBM), and the latter is responsible for binding to the SARS-CoV-2 receptor [28, 31]. Similar to SARS-CoV-1 and some SARS-like bat coronaviruses, such as W1V1-CoV, SHC014-CoV, and RaTG13, SARS-CoV-2 utilizes angiotensin-converting enzyme 2 (ACE2) as its functional cellular receptor to enter target cells to initiate the virus infection process [28, 32]. In addition to ACE2, SARS-CoV-2 also uses the serine protease TMPRSS2 for S protein priming [33]. Moreover, neuropilin-1, a cofactor for binding furin-cleaved substrates, facilitates SARS-CoV-2 cell entry and infectivity [34].
Cryo-EM and/or crystal structures of the SARS-CoV-2 S-trimer/ACE2 and RBD/ACE2 complexes have been resolved (Fig. 1d). The prefusion conformation of the SARS-CoV-2 S protein in cryo-EM analysis presents as a trimeric structure consisting of three RBDs, with one RBD in the “up” conformation, allowing access to the ACE2 receptor [35, 36]. ACE2 recognizes the RBD through polar residues in its extracellular peptidase domain [37]. Analysis of the crystal structure of the RBD/ACE2 complex has identified critical residues in the RBD that are essential for binding to the ACE2 receptor, eight of which are identical in the SARS-CoV-2 and SARS-CoV-1 RBDs; the RBM contains most of the amino acid residues necessary for binding to ACE2 [31]. Relative to the SARS-CoV-1 S RBD, the SARS-CoV-2 S RBD has a higher binding affinity for ACE2 [28, 35, 38], partially explaining the more efficient human-to-human transmission of SARS-CoV-2.
During SARS-CoV-2 infection, the RBD in the S1 subunit of the S protein first binds the cellular receptor ACE2 to form an RBD/ACE2 complex (Figs 1 and 2). This binding event leads to conformational changes in the S protein and subsequent dissociation of the S1 and S2 subunits. Mediated by the HR1/HR2 domains in the S2 subunit, the viral and host cell membrane fusion process occurs, allowing the virus to enter the host cell and release viral RNA [39]. Subsequently, newly synthesized viral RNA–N complexes and S, E, and M proteins are further assembled in the endoplasmic reticulum–Golgi intermediate compartment to form mature virions, which are released from host cells [40]. Relative to SARS-CoV-1, SARS-CoV-2 demonstrates better membrane fusion ability, and changes in several amino acid residues in the HR1 domain potentially contribute to the increased protein–protein interactions with the HR2 domain [27]. A better understanding of the viral life cycle and associated infection process will be helpful for designing effective vaccines and therapeutic agents to control the COVID-19 pandemic.
Generation of SARS-CoV-2-targeting human neutralizing monoclonal antibodies (nAbs) and their mechanisms of action. nAbs specific to SARS-CoV-2 can be generated by single B cell cloning (a) or phage display library screening (b). c nAbs targeting the receptor-binding domain (RBD) of the spike (S) protein bind to the RBD in the S1 subunit and block its binding with the angiotensin-converting enzyme 2 (ACE2) receptor, thus preventing virus entry into host cells through endocytosis or cell surface fusion processes. d nAbs targeting the N-terminal domain (NTD) in the S1 subunit or S2 bind to the NTD or S2 subunit, thus inhibiting the conformational change of the S protein or NTD or the formation of a 6-helix bundle (6-HB) structure mediated by heptad repeat region 1 (HR1) and HR2 in the S2 subunit, further blocking membrane fusion and virus entry into the host cell
SARS-CoV-2-specific neutralizing monoclonal antibodies and their mechanisms of action
Neutralizing monoclonal antibodies (nAbs) serve as key therapeutic agents for the rapid prevention and treatment of SARS-CoV-2 infection. Anti–SARS-CoV-2 human nAbs can be isolated from antigen-specific B cells from the peripheral blood mononuclear cells of individuals who have been infected with SARS-CoV-2 (Fig. 2a). They can also be generated by screening naive or synthetic phage-displayed human antibody libraries (Fig. 2b) [41,42,43,44,45,46,47,48,49,50,51]. Most of these antibodies target the SARS-CoV-2 S protein, including its subdomains, and they present different mechanisms of action in the inhibition of SARS-CoV-2 infection.
Generally, nAbs targeting the S protein block receptor binding or membrane fusion, thereby preventing viral entry into host cells. Specifically, RBD-targeting nAbs bind to the RBD in the RBM region and inhibit RBD binding to the ACE2 receptor, thus blocking the subsequent viral entry process (Fig. 2c). Some non-ACE2 mimics target the RBD without inhibiting RBD-ACE2 binding and may instead inhibit SARS-CoV-2 infection by blocking S protein conformational changes. NTD-targeting nAbs bind to the NTD of the S protein, and the recognition sites may contain N-glycosylation sites [52, 53]. They neutralize SARS-CoV-2 infection by potentially preventing the interaction of the NTD with C-type lectin receptors, such as L-SIGN, inhibiting S protein or NTD conformational changes, or interrupting virus postattachment steps (cell–cell fusion) (Fig. 2d) [41, 52, 54,55,56]. However, these nAbs do not generally block the receptor binding and viral attachment steps. In contrast to NTD-specific nAbs that neutralize SARS-CoV-2 infection, some NTD-targeting non-nAbs can induce the open conformation of the RBD, resulting in enhanced binding of the RBD to the ACE2 receptor and thus increasing SARS-CoV-2 infectivity [57]. Both NTD- and RBD-targeting nAbs may activate Fc-mediated effector functions, antibody-dependent cellular phagocytosis, or antibody-dependent cellular cytotoxicity, which are potentially required for optimal protection against SARS-CoV-2 challenge [41, 54, 58, 59]. S2-targeting nAbs may inhibit HR1 or HR2 from forming a six-helix bundle structure, thereby blocking subsequent membrane fusion and the viral entry process (Fig. 2d).
Antigenic view of the NTD and RBD of SARS-CoV-2 S protein and nAb binding sites
Antigenic view of SARS-CoV-2 NTD and nAb binding sites
The NTD of the SARS-CoV-2 S protein is an immunodominant target to induce the production of neutralizing antibodies, although the neutralizing activity of nAbs targeting the NTD is generally much lower than that of the nAbs targeting the RBD. Antigenic and/or epitope mapping of the NTD has identified several supersites of SARS-CoV-2 recognized by NTD-specific nAbs [41, 52]. NTD-specific nAbs can be categorized into two distinct groups: a group with high potency against SARS-CoV-2 infection and a group with less potency but displaying glycan-dependent neutralizing activity [53].
Antigenic view of SARS-CoV-2 RBD and nAb binding sites
The RBD of the SARS-CoV-2 S protein is an immunodominant target for inducing the production of highly potent and specific neutralizing antibodies. Analysis of the binding between the RBD and RBD-specific nAbs, as well as the inhibition of RBD-ACE2 binding by nAbs, reveals different antigenic regions on the RBD. These regions can be clustered based on the site for antibody attachment that overlaps the ACE2 binding site, as well as the antibody binding site that does not compete with ACE2 binding [60]. Nevertheless, the N and C termini of the RBD generally have less ability to bind nAbs [60]. Accordingly, RBD-targeting nAbs are classified into different groups based on the recognized epitopes on the RBD. These groups include nAbs binding to the RBD site distal to the ACE2 receptor-binding site (normally having low neutralizing activity) and nAbs directly competing with the ACE2 binding site at the RBM region (generally presenting the most potent neutralizing activity); other RBD-specific nAbs recognize epitopes partially overlapping with or further distal from the above sites [53].
Following the COVID-19 pandemic, an increasing number of human nAbs targeting SARS-CoV-2 proteins were identified and developed as therapeutics. The majority of these nAbs target different regions of the S protein, including the NTD and RBD, whereas a few antibodies target other proteins. In the rest of this review, we will summarize the SARS-CoV-2-specific nAbs currently developed based on the domains they target in the viral proteins.
Neutralizing antibodies targeting the NTD of SARS-CoV-2 S protein
As part of the S1 domain of the S protein, the NTD is a key target for the development of COVID-19 nAbs, and several of the currently developed human nAbs bind to this region. These NTD-targeting nAbs may neutralize SARS-CoV-2 infection in vitro or protect animals from viral challenge (Table 1) [41, 44, 54, 55, 61]. However, it appears that none of these NTD-targeting nAbs have been tested in humans.
Binding ability of SARS-CoV-2 NTD-targeting nAbs
NTD-targeting nAbs bind to the SARS-CoV-2 NTD and S1 fragments of the S protein and/or S protein trimer. Cryo-EM and crystal structure analyses have identified critical neutralizing epitopes for several SARS-CoV-2 NTD-targeting nAbs (Fig. 3a–f) [41, 44, 55, 56]. For example, cryo-EM and crystal structures of the S2L28/S, S2M28/S, S2X28/S, S2X333/S, and S2M28/NTD complexes reveal that these nAbs recognize overlapping epitopes within a structurally identified antigenic supersite in the NTD of the S protein [41]. In addition, cryo-EM and/or crystal structures of NTD-specific nAbs, such as 5-24, 1-87, and 2-51, in complex with S or the NTD reveal a single supersite surrounded by glycans at N17, N74, N122, and N149, which is located at the periphery of S facing away from the viral membrane [56]. Thus, structural analysis of nAb/S and nAb/NTD complexes is helpful for determining which residues are important for nAb binding to the NTD of S and identifying neutralizing epitopes recognized by these nAbs.
Structures of SARS-CoV-2 spike (S) in complex with N-terminal domain (NTD)-targeting neutralizing monoclonal antibodies (nAbs). a–e Cryo-EM structures of the SARS-CoV-2 S trimer bound to NTD-targeting nAbs a S2L28 (PDB 7LXZ), b S2M28 (PDB 7LY2), c S2X333 (PDB 7LXY), d 4-8 (PDB 7LQV), and e 4A8 (PDB 7C2L). f Crystal structure of the SARS-CoV-2 NTD in complex with the nAb S2M28 (PDB 7LY3)
Neutralizing activity of SARS-CoV-2 NTD-targeting nAbs
SARS-CoV-2 NTD-targeting nAbs can neutralize wild-type pseudotyped and/or authentic SARS-CoV-2 infection in vitro. Pseudotyped and authentic SARS-CoV-2 have been found to be neutralized by nAbs 4-8 and 5-24 with a half-maximal inhibitory concentration (IC50) of 8–9 ng/ml [44]. nAb 4A8 also has been shown to neutralize both pseudovirus and live SARS-CoV-2 infection with a half-maximal effective concentration (EC50) of 0.61 and 49 μg/ml, respectively [55]. 89C8 has been found to neutralize infection of pseudotyped SARS-CoV-2, with an IC50 of 4.5 nM [61], whereas nAbs BLN1, BLN12, and P008_056 have been shown to neutralize authentic SARS-CoV-2 infection with IC50 values of 8, 8, and 14 ng/ml, respectively [52, 53]. Moreover, S2L28, S2M28, S2X28, and S2X333 have been found to potently neutralize infection of both pseudotyped and live SARS-CoV-2 with an IC50 value as low as 2 ng/ml [41]. The differences in potency of these nAbs against SARS-CoV-2 infection might result in part from differences in the neutralization assays and virus strains used for detecting neutralizing activity.
Protective efficacy of SARS-CoV-2 NTD-targeting nAbs
Several NTD-targeting nAbs have demonstrated prophylactic and therapeutic protective efficacy against SARS-CoV-2 infection in animal models, such as hACE2-transgenic (hACE2-Tg) mice and hamsters [41, 54]. For example, COV2-2676 and COV2-2489, which bind to a common antigenic site on the NTD and inhibit pseudotyped and authentic SARS-CoV-2 infection in vitro, have been found to prophylactically and therapeutically protect hACE2-Tg mice from SARS-CoV-2 infection. These animals exhibited reduced weight loss, decreased viral titers in the upper and lower respiratory tracts and heart, or reduced cytokine and chemokine levels in the lung [54]. NTD-targeting nAbs 159, BLN12, and BLN14 have been shown to therapeutically protect against SARS-CoV-2 infection in antibody-treated hACE2-Tg mice, preventing weight loss, with reduced viral titers and/or RNA copies in the nasal washes, lung, and other tissues [52, 60]. Moreover, S2X333 has been found to prophylactically protect hamsters from SARS-CoV-2 infection; these animals had reduced viral RNA copies and/or viral titers in the lung [41].
Cross-reactivity and cross-neutralizing activity of SARS-CoV-2 NTD-targeting nAbs
SARS-CoV-2 NTD-targeting nAbs may cross-react with other coronaviruses and/or cross-neutralize their infection to some extent. This phenomenon is observed for S2L28, S2M28, S2X28, and S2X333, which strongly bind to the S protein of RaTG13 (a bat coronavirus most closely related to SARS-CoV-2) and neutralize infection by pseudotyped RaTG13 expressing the viral S protein. However, their ability to bind to other coronavirus S proteins, such as those from Pangolin Guangxi 2017 and Pangolin Guangdong 2019, is reduced, and none of them neutralize infection by human SARS-CoV-1, Pangolin Guangxi 2017, Pangolin Guangdong 2019, bat W1V1, or bat W1V16 pseudoviruses [41]. These differences in binding affinity and subsequent viral neutralization may result from sequence variation within the S protein NTD regions for each coronavirus.
Neutralizing antibodies targeting the RBD of SARS-CoV-2 S protein
The SARS-CoV-2 S protein RBD is a critical target for the development of effective COVID-19 antibodies. SARS-CoV-2 RBD-targeting nAbs have been extensively studied, and most of the currently developed human nAbs are specific to this region. These nAbs bind to the RBD of SARS-CoV-2 S, neutralize SARS-CoV-2 infection in vitro, and/or protect animals from SARS-CoV-2 challenge (Table 1). However, most of these nAbs are in preclinical development, and only a few have progressed into human clinical trials [62].
Binding and cross-reactivity of SARS-CoV-2 RBD-targeting nAbs
RBD-targeting SARS-CoV-2 nAbs generally have high binding affinities to the RBD of SARS-CoV-2 S. Most of the RBD-targeting nAbs, including P2C-1F11, P2B-2F6, P2C-IA3, P5A-1B8, P5A-2G7, MD62, MD65, COVA1-18, COVA2-15, IgG1 ab1, CV07-209, MV05, CA1, CB6, CT-P59, Ab6, ab8, VH-Fc ab8, m397, and 1212C2, bind to the ACE2-binding region of the RBD, directly blocking the binding interaction between the RBD and the cellular receptor ACE2 [43, 47,48,49, 59, 63,64,65,66,67,68]. Other nAbs, including TAU-1109, MD29, and MD47, strongly bind to the RBD but at regions distal to the ACE2 binding site; hence, they do not compete with ACE2 for RBD binding [48]. Only a few SARS-CoV-2 RBD-specific nAbs, such as CV38-142, COVA1-07, COVA1-16, COVA2-02, COVA2-44, and CoV2-12, cross-react against SARS-CoV-1 S and/or RBD proteins, potentially because they recognize conserved epitopes within the SARS-CoV-2 and SARS-CoV-1 RBDs [43, 51, 64].
Cryo-EM and crystal structures are available for several SARS-CoV-2 RBD-targeting nAbs in complex with the SARS-CoV-2 S protein RBD (Fig. 4) [44, 47, 49, 64, 65, 69]. Cryo-EM structures of the nAb Fab or IgG and SARS-CoV-2 S trimer complexes reveal that the binding of nAbs with the three RBDs can result in multiple conformations: three “down” RBDs, one “up” and two “down” RBDs, one or two “up” RBDs, two “up” RBDs, or two or three “up” RBDs (Fig. 4a–e) [44, 63]. Relative to the Fab nAb, the IgG nAb exhibits more potency in neutralizing SARS-CoV-2 infection [63]. Crystal structures of CT-P59, CV07-250, or CV07-270 (Fab/IgG) and RBD complexes show that CT-P59 and CV07-250 directly interact with or obscure the ACE2 binding sites, while CV07-270 only partially overlaps with these binding sites (Fig. 4f–g) [47, 64]. Crystal analysis of the P2B-2F6 Fab/RBD complex reveals that the epitopes recognized by the nAb are in the RBM region (Fig. 4h), demonstrating that the blockage of viral entry by this nAb is achieved through inhibiting ACE2 binding [49]. The crystal structure of the CV30 Fab/RBD complex also indicates that CV30 binds to epitopes necessary for ACE2 receptor binding (Fig. 4i) [69]. A crystal structure of the CB6 Fab/RBD complex reveals an epitope on the SARS-CoV-2 S protein RBD that overlaps with the ACE2 binding sites (Fig. 4j) [65]. Accordingly, structural analyses of the binding interactions between human nAbs and the SARS-CoV-2 S protein and/or RBD can map the common neutralizing epitopes recognized by these nAbs within the RBD, reveal the similarities between nAb/RBD binding and ACE2/RBD binding, and explain the differences, if any, between these antibodies in competing with the ACE2 receptor [64, 70,71,72]. These structures also help to elucidate the mechanisms by which these nAbs neutralize SARS-CoV-2 infection.
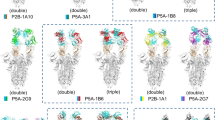
Structures of SARS-CoV-2 spike (S) in complex with receptor-binding domain (RBD)-targeting monoclonal antibodies (mAbs) with neutralizing activity (nAbs). a–e Cryo-EM structures of the SARS-CoV-2 S trimer bound to nAbs. a 2-4 with all three RBDs in the down conformation (PDB 6XEY), b P5A-1B9 with one RBD in the up conformation (PDB 7CZX), c P5A-3C12 with one or two RBDs in the up conformation, respectively (PDBs 7D0B and 7D0D), d P5A-2G7 with two RBDs in the up conformation (PDB 7CZW), and e P5A-1B8 with two or three RBDs in the up conformation, respectively (PDBs 7CZR and 7CZS). f–j Crystal structures of the SARS-CoV-2 RBD in complex with the nAbs f CT-P59 (PDB 7CM4), g CV07-270 (PDB 6XKP), h P2B-2F6 (PDB 7BWJ), i CV30 (PDB 6XE1), and j CB6 (PDB 7C01)
Neutralizing and cross-neutralizing activity of SARS-CoV-2 RBD-targeting nAbs
Most, if not all, nAbs specific to the SARS-CoV-2 S protein RBD potently neutralize wild-type SARS-CoV-2 pseudovirus and/or live virus infection in vitro, and they present more potent neutralizing activity than nAbs targeting non-RBD epitopes. These nAbs, including COVA1-18, COVA2-15, CV07-209, CC6.29, CC6.30, CC12.1, P008_108, 1212C2, and VH-Fc ab8, have been found to neutralize pseudovirus and/or live SARS-CoV-2 infection with high potency (IC50 ≤ 40 ng/ml) [43, 53, 64, 67, 68, 73]. The TAU-1145, TAU-2189, TAU-2230, and TAU-2303 nAbs, which recognize a key residue (G466) in the RBD, have been shown to neutralize both pseudotyped and live SARS-CoV-2 infection, inhibiting cell death or syncytium formation [45]. It has been noted that some RBD-targeting nAbs also neutralize infection by mutant variants of SARS-CoV-2 but at different potencies. CT-59 has been found to neutralize wild-type SARS-CoV-2 and the D614G mutant with IC50 values of 8.4 and 5.7 ng/ml, respectively [47]. Other nAbs, such as J08, I14, F05, G12, C14, and B07, have been shown to neutralize D614G and B.1.1.7 variants to different extents, and J08 has been found to also neutralize the E484K variant, albeit with relatively lower neutralizing activity [62]. Among the identified SARS-CoV-2 RBD-targeting nAbs, CC6.33, COVA2-02, and COVA1-16 also neutralize SARS-CoV-1 but at relatively high IC50 values of 162, 610, and 2,500 ng/ml, respectively [43, 73]. Most other SARS-CoV-2 RBD-targeting nAbs appear to have low or no cross-neutralizing activity against infection with SARS-CoV-1, MERS-CoV, or other coronaviruses [49].
Protective efficacy of SARS-CoV-2 RBD-targeting nAbs
A number of SARS-CoV-2 RBD-specific nAbs have demonstrated protective efficacy in different animal models, including mice (wild-type mice, adenovirus-hACE2 (AdV-hACE2)-transduced mice, and hACE2-Tg mice), hamsters, ferrets, and nonhuman primates (NHPs) [44, 47, 59, 64, 67, 68, 74,75,76]. RBD-targeting nAbs without Fc engineering protect against SARS-CoV-2 infection in vivo. For instance, IgG ab1 and VH-Fc ab8 have been found to prophylactically protect wild-type mice from infection with mouse-adapted SARS-CoV-2 and/or hACE2-Tg mice from infection with authentic SARS-CoV-2 [51, 59, 67]. IgG ab1, VH-Fc ab8, 1212C2, CV07-209, and CC12.1 have been shown to prophylactically and/or therapeutically protect hamsters from wild-type SARS-CoV-2 infection [59, 64, 67, 68, 73]. CT-59 has been found to exhibit therapeutic efficacy in preventing authentic SARS-CoV-2 infection in ferret, hamster, and NHP models, whereas 2-15, COV2-2196, and COV2-2381 have been shown to prophylactically protect hamsters or rhesus macaques from SARS-CoV-2 infection by inhibiting viral replication [44, 47, 75]. It has been reported that Fc-engineered SARS-CoV-2 RBD-targeting nAbs also provide protection against SARS-CoV-2 infection in vivo. For example, J08-MUT, which contains an Fc-engineered fragment of J08, has been found to prophylactically and therapeutically protect hamsters from wild-type SARS-CoV-2 infection [62]. In addition, the Fc-engineered MV05 and CB6 nAbs, with LALA mutations in the Fc domain, namely, MV05-LALA and CB6(LALA), respectively, have been shown to prophylactically and therapeutically protect NHPs from SARS-CoV-2 viral challenge [65, 74]. In addition to using the intraperitoneal (I.P.) route and parenteral routes, including intramuscular, subcutaneous, and intravenous injections, SARS-CoV-2 RBD-targeting nAbs can be administered via inhalation without reducing protective efficacy [47, 59, 62, 68, 73, 74]. For example, inhalation administration of Fc-engineered 1212C2 has been shown to eliminate viral loads in the lungs of SARS-CoV-2-challenged hamsters, resulting in better efficiency than injection via the I.P. route [68]. It is worth noting that the effective concentrations of nAbs required for the protection of animals against SARS-CoV-2 infection vary depending on the differences in nAb potency, viral titers, and strains and the animal models used for the challenge. It has been reported that serum concentrations of ~22 and 12 μg/ml (pseudovirus neutralization IC50×1160 and 630, respectively) of CC12.1 nAb protect hamsters challenged with SARS-CoV-2 (USA-WA1/2020 strain, 1 × 106 plaque-forming units) from weight loss (100% or 50%, respectively) [73]. It appears that the neutralization potency of nAbs in vitro does not always correlate well with their protective efficacy in vivo [77]. This discrepancy might be because the Fc effector function of nAbs affects their ability to neutralize SARS-CoV-2 in vivo; thus, identification of optimal nAb concentrations in monotherapy or combinatorial therapy is needed to maximize nAb protection against SARS-CoV-2 infection [77].
Combination treatments with SARS-CoV-2 RBD-targeting nAbs
Combination treatments with SARS-CoV-2 RBD-targeting nAbs have exhibited synergistic effects in preventing infection by escape mutants or improving their neutralization in vitro and/or protecting against SARS-CoV-2 infection in animal models. In vitro, a cocktail of CoV2-06 and CoV2-14 has been found to effectively prevent SARS-CoV-2 infection by escape mutants, but neither nAb had this effect individually [51]. The combination of TAU-2212 (which recognizes an unknown conformational epitope on the S protein) with an ACE2-competing, RBD-binding nAb (such as TAU-2230, TAU-2189, TAU-1145, or TAU-2303), and a non-ACE2-competing/RBD-targeting nAb (TAU-1099) has been shown to improve its efficiency in inhibiting SARS-CoV-2 infection [45]. Furthermore, the RBD-targeting nAbs COV2-2196 and COV2-2130, which block RBD-ACE2 binding but recognize the nonoverlapping sites on the RBD of SARS-CoV-2, have been shown to synergistically neutralize SARS-CoV-2 infection with improved neutralizing activity compared with each of the two nAbs alone [75]. In vivo, treatment with COV2-2196 and COV2-2130, individually or in combination, has been found to prophylactically protect AdV-hACE2–transduced mice from infection with wild-type SARS-CoV-2; these mice exhibited no weight loss and had reduced viral titers and inflammation in the lung. Prophylactic and therapeutic combined treatment with COV2-2196 and COV2-2130 has been shown to improve protection against authentic SARS-CoV-2 infection in AdV-hACE2-transduced mice and protect wild-type mice from infection by a mouse-adapted SARS-CoV-2 variant [75]. Hamsters and NHPs treated prophylactically or therapeutically with REGN-COV2, a combination of REGN10987 and REGN10933, exhibit a reduction in viral load and pathology [78]. Despite these promising results, the combinatorial concentrations of nAbs required for in vivo protection against SARS-CoV-2 infection still need to be optimized for individual nAbs.
Neutralizing antibodies targeting other regions of the SARS-CoV-2 S protein
Although a large number of nAbs identified to date target the SARS-CoV-2 RBD and the NTD, several nAbs target other regions (Table 1) [44, 55]. SARS-CoV-2 S2-targeting antibodies have been identified; however, the majority of them have no neutralizing activity. For example, among the 0304-3H3, 9A1, L19, P008_004, P008_005, P008_006, P008_016, P008_023, and P008_032 antibodies that bind to the SARS-CoV-2 S2 fragment, only L19 neutralizes infection by SARS-CoV-2, including variant strains (D614G and B.1.1.7) and the wild-type strain [53, 55, 62]. Several of these SARS-CoV-2 S2-targeting antibodies also cross-react with SARS-CoV-1 S protein, but none of them show neutralizing activity against SARS-CoV-1 infection [53]. In addition, nAbs binding to the S-trimer and other regions have been identified. For instance, the nAbs 2-43 and 2-51 bind to the SARS-CoV-2 S-trimer complex, recognizing non-NTD and non-RBD epitope(s) [44]. Other nAbs (such as I21, J13, D14, I15, F10, F20, and P008_060) bind to the S-trimer complex and/or non-RBD S1 proteins and are capable of neutralizing wild-type and/or the aforementioned variants in vitro [53, 62]. In general, the binding affinity and neutralizing activity of non-RBD and non-NTD epitope-specific nAbs against SARS-CoV-2 infection are lower than those of nAbs targeting the S protein NTD or RBD of SARS-CoV-2. It appears that no nAbs targeting non-NTD or non-RBD regions have yet been tested for protection against SARS-CoV-2 infection in vivo. It is worth noting that the structures of a few nAbs targeting the non-NTD and non-RBD regions of the SARS-CoV-2 S protein have been solved [44]. Identification of neutralizing epitopes outside the S protein NTD and RBD regions of SARS-CoV-2 is expected to lead to the design of novel and effective COVID-19 therapeutics and vaccines based on these epitopes.
Neutralizing antibodies targeting other SARS-CoV-2 proteins
Only a few monoclonal antibodies developed to date target other proteins of SARS-CoV-2, and most do not have neutralizing activity against SARS-CoV-2 infection. However, they can be used for other applications, such as diagnosis and epidemiology. For example, monoclonal antibodies specifically targeting the SARS-CoV-2 N protein increase the detection sensitivity of ELISAs, providing a tool for the early and accurate diagnosis based on clinical samples and for epidemiological studies of SARS-CoV-2 [79, 80]. In addition, the NP-monoclonal antibody system and related test strips can be utilized for large-scale screening of COVID-19 samples [81].
Multimeric neutralizing antibodies targeting multiple regions of SARS-CoV-2 proteins
SARS-CoV-2 nAbs can also be constructed by combining two or more neutralizing antibodies specific to SARS-CoV-2 proteins and/or antibody fusion with the receptor ACE2. The resultant multimeric (dimeric, trimeric, or tetravalent) nAbs may bind to their respective regions with neutralizing activity. A dimeric nAb, 89C8-ACE2, was designed by combining the SARS-CoV-2 NTD-targeting mAb 89C8 and the ACE2 ectodomain [61]. This nAb binds to the SARS-CoV-2 S protein RBD, inhibiting the S1–ACE2 interaction; it also neutralizes pseudotyped and authentic SARS-CoV-2 infection in vitro.
Neutralizing antibodies to prevent and treat COVID-19 in humans
A variety of non–SARS-CoV-2-specific nAbs, such as anti-CD6 (itolizumab), anti-IL-6 receptor (tocilizumab), anti-IL17A (ixekizumab), and anti-C5 complement antibodies, have been used in patients with COVID-19, with the purpose of reducing or eliminating SARS-CoV-2–induced cytokine storms and inflammatory responses or treating SARS-CoV-2–related pneumonia [54, 82,83,84,85,86,87,88,89,90,91,92,93]. By comparison, only a few SARS-CoV-2-specific human nAbs, including REGN-COV2, LY-CoV555, LY-CoV016, AZD7442, AGD20, 47D11, 2B04, and CT-P59, have been tested in clinical trials and/or approved for emergency use to prevent and/or treat COVID-19 disease in humans [94,95,96,97,98]. Almost all of the nAbs under clinical trials target the RBD of SARS-CoV-2.
REGN-COV2, a cocktail of two SARS-CoV-2 RBD-targeting nAbs, REGN10933 (casirivimab) and REGN10987 (imdevimab), has been studied in a phase 1–3 clinical trial for outpatients with COVID-19, and the treatment resulted in reduced viral load without notable safety problems [94]. A phase 3 trial with subcutaneous administration of this nAb combination showed prevention of COVID-19 progression from asymptomatic to symptomatic disease in early SARS-CoV-2 infection [99]. In addition, patients with mild-to-moderate COVID-19 exhibited reduced hospital utilization after receiving REGN-COV2 treatment within a few days of symptom onset, and a low-dose REGN-COV2 infusion has been shown to improve COVID-19 symptoms [100, 101]. In addition to treatment, REGN-COV2 has been tested for the possibility of preventing SARS-CoV-2 infection. Subcutaneously injected REGN-COV2 prevented SARS-CoV-2 infection and the presence of COVID-19 symptoms in high-risk individuals who had close contact with SARS-CoV-2-infected persons [102].
LY-CoV555, an RBD-specific nAb also known as LY3819253 or bamlanivimab, has been evaluated in treating outpatients with COVID-19, and a phase 2 trial of bamlanivimab concluded that it decreased the viral load in these patients [95, 103]. In a phase 2/3 clinical trial, combinatorial treatment with bamlanivimab and another RBD-specific nAb, LY-CoV016 (also known as LY3832479, etesevimab, or CB6), reduced the viral load in nonhospitalized patients with mild-to-moderate COVID-19 illness, whereas there was no significant reduction in the viral load with monotherapy bamlanivimab treatment [97]. However, when coinjected with remdesivir, bamlanivimab does not exhibit therapeutic efficiency in patients hospitalized with COVID-19 [104]. As prophylactic prevention, bamlanivimab has been shown to reduce the incidence of COVID-19 among residents and staff of skilled nursing and assisted living facilities in a randomized phase 3 trial [105].
Clinical trials based on other SARS-CoV-2 RBD-targeting nAbs are ongoing. A phase 3 trial (NCT04723394) was conducted for coadministration with AZD7442 (a nAb combination of AZD8895 + AZD1061) to assess its safety and efficacy for the treatment of outpatient adults with COVID-19 and prevention of severe COVID-19 or death. Two additional phase 3 trials (NCT04625725 and NCT04625972) are planned to evaluate the safety and efficacy of preexposure and postexposure prophylaxis of this mAb combination in adults with COVID-19. In addition, a phase 2/3 trial (NCT04859517) has recruited to determine the efficacy and safety of the nAb AGD20 in the prevention (preexposure and postexposure prophylaxis) of COVID-19, and a phase 2/3 trial (NCT04805671) is planned to evaluate the efficacy and safety of this nAb in treating ambulatory participants with mild or moderate COVID-19. 47D11 and 2B04 nAbs have been used as monotherapy or combinatorial therapy in a phase 1 trial (NCT04644120) to evaluate their safety, pharmacokinetics, and pharmacodynamics in adults with COVID-19, whereas the nAb CT-59 has been applied in a phase 2/3 trial (NCT04602000) to evaluate its therapeutic efficacy and safety in outpatients with mild-to-moderate symptoms of SARS-CoV-2 infection.
Currently, bamlanivimab, etesevimab, and REGN-COV2 have been approved via Emergency Use Authorization (EUA) by the U.S. FDA for the therapeutic treatment of mild-to-moderate COVID-19 in adults and pediatric patients (≥12 years of age) from high-risk populations [106,107,108,109]. Moreover, the EUA also limits the use of these nAbs to treat only individuals who are hospitalized due to COVID-19 or other health concerns.
Potential challenges and improvement of SARS-CoV-2-specific nAbs
Similar to other coronaviruses, including SARS-CoV-1 and MERS-CoV, SARS-CoV-2 has continually developed mutations during the COVID-19 pandemic, and different mutants have been found in all four structural proteins and other viral proteins [110]. Multiple strains that are variants of concern (VOCs) (B.1.1.7, B.1.351, P.1, and B.1.617.2) have been identified that carry single or combined amino acid mutations or deletions, including K417N/T, E484K, N501Y, L452R, and T478K in the RBD; L18F, T20N, P26S, 69-70del, D138Y, 144del, 156-157del, R158G, R190S, and 241-243del in the NTD; and D614G in S1 of the S protein [111, 112]. In addition, variant of interest (VOI) strains (B.1.427, B.1.429, B.1.525, B.1.526, B.1.617.1, B.1.617.3, and P.2) have also been defined, which carry mutations, such as L452R, S477N, E484K, and E484Q in the RBD; 69-70del, T95I, G142D, 144del, W152C, E154K, F157S, and D253G in the NTD; and D614G in S1 of the S protein [111, 112]. Among these variants, P.1 and P.2 belong to the B.1.1.28 lineage. These VOC and VOI strains may exhibit enhanced viral replication and/or transmission. Furthermore, other mutations that could cause escape from antibody recognition have been found in the S protein RBD or NTD regions through mapping, recombinant chimeric VSV/SARS-CoV-2 reporter viruses, polyclonal human plasmas antibodies, and other approaches [113,114,115]. Mutations in the SARS-CoV-2 RBD or NTD may lead to escape from antibody recognition and decreased neutralizing activity of nAbs against virus infection [113].
Many of the currently available nAbs specific to the prototypic SARS-CoV-2, including those targeting the NTD or RBD of the S protein, have reduced neutralizing activity against newly emerging SARS-CoV-2 variants with substitutions in the NTD or RBD, particularly when they are used alone [41, 116,117,118,119,120]. For example, the RBD-targeting nAbs 2-15 and C121 exhibit a complete loss of neutralizing activity against the P.1 variant [121]. Neutralization of the B.1.1.7 variant by the RBD-targeting nAbs COVA2-15, COVA1-18, and S309 has also been found to be reduced, which might be potentially due to the N501Y mutation [119]; nevertheless, this mutation has minimal effect on the neutralizing activity of the nAbs IgG ab1 and VH-Fc ab8 [122]. In contrast to RBD-specific nAb ab6, which can neutralize a pseudotyped SARS-CoV-2 variant containing a triple mutation (K417N, E484K, N501Y), the nAbs COV2-2196, COV2-3025, COV2-2381, and S2E12 exhibit reduced neutralizing ability against the B.1.351 variant containing the E484K and N501Y mutations [117, 123]. Notably, the RBD-targeting nAbs COV2-2050, 1B07, COVOX-384, and S2H58 exhibit loss of neutralizing activity against the Wash SA-B.1.351 variant containing the full S sequence of the South African strain [117]. In addition, the NTD-specific nAbs COV2-2676 and COVD-2489 also exhibit loss of neutralizing activity against the Wash SA-B.1.351 variant [117]. Other NTD-specific nAbs, such as 5-24 and 4-8, show completely abolished neutralizing activity against the B.1.351 variant, whereas the 159 nAb exhibits reduced neutralizing ability against the P.1 variant, potentially due to the mutations in the residues recognized by this nAb (L18F, T20N, P26S, D138Y, and R190S) [121, 124].
Notably, clinically approved RBD-targeting nAbs, including LY-CoV555, LY-CoV016, REGN10933, and AZD8895, exhibit abolished neutralizing activity against the SARS-CoV-2 P.1, B.1.351, or B.1.1.7 VOCs in vitro [121, 124]. This phenomenon could be explained by the fact that these nAbs are sensitive to mutations at residues 417, 484, and/or 501 of the SARS-CoV-2 RBD [124, 125]. In addition, LY-CoV555 and its combination with LY-CoV016 have been shown to exhibit reduced or loss of protective activity against VOCs, including B.1.351 and/or B.1.128, in hACE2-Tg mouse models [125]. A combination of clinically trialed nAbs, 2B04/47D11, have been shown to have decreased protective efficacy against the B.1.1.7, B.1.351, and B.1.128 variant strains in hACE2-Tg mice, and the protection was dose-dependent [125]. Apparently, the loss of neutralizing activity or protection by clinically approved nAbs against SARS-CoV-2 VOC strains will significantly affect the clinical use of these nAbs to treat COVID-19 patients, particularly when persons are infected with variants carrying related mutations. It is plausible to identify the neutralizing activity of nAbs approved via EUA against all potential VOC and VOI variants, test their combinatorial effects with other appropriate nAbs, or optimize injection dosages before use for clinical treatment of COVID-19 patients.
nAbs are engineered or fused to improve neutralizing activity to prevent and treat infection caused by SARS-CoV-2 mutant strains. Remarkably, combinatorial nAbs can be constructed by fusing the heavy chain (HC) of one nAb with the engineered light chain (LC) of another nAb targeting conserved epitopes on the NTD, RBD, or S2 regions of the S protein of multiple SARS-CoV-2 strains. It has been shown that the RBD-targeting nAb 222 with somatic mutations at the LC (222LC) maintains neutralizing activity against P.1, B.1.1.7, and B.1.351 variants and that a chimeric nAb (150HC/222LC) fused to the LC of the nAb 222 with the HC of another RBD-targeting nAb (150) restores or improves neutralization potency of the naive nAb against these SARS-CoV-2 VOC strains [124]. In addition, cocktail treatments combining nAbs recognizing epitopes on the NTD, RBD, or S2 or different epitopes on the same fragments are expected to exhibit synergistic effects and reduce the prevalence of antibody escape mutants, potentially improving nAb efficacy in preventing COVID-19. Indeed, NTD-targeting nAbs may neutralize mutant viruses that exhibit RBD-targeting antibody escape, and vice versa [54]. A combination of RBD- and NTD-targeting nAbs limits the development of escape mutants [126]. In addition, combinations of two RBD-targeting nAbs (such as REGN10933 and REGN10987), particularly three noncompeting RBD-specific nAbs (such as REGN10933, REGN10987, and REGN10985), improve the efficiency of individual nAbs in neutralizing SARS-CoV-2 VOC and VOI strains, preventing the development of escape mutants [127]. Moreover, treatment of hACE2-Tg mice, 129S2 immunocompetent mice, and hamsters with combinations of RBD-targeting nAbs, such as S309/S2E12, COV2-2130/COV2-2196, and/or REGN10933/REGN10987, prevents infection with multiple SARS-CoV-2 variants, including B.1.1.7, B.1.351, B.1.128, and SARS-CoV-2 containing N501Y/D614G mutations [125]. Crystal and cryo-EM structural analyses of the binding between nAbs and the SARS-CoV-2 S protein, its functional fragments, or other proteins provide an effective tool to rapidly identify novel and conserved neutralizing epitopes across SARS-CoV-2 variants, which will allow for the design of innovative countermeasures.
Conclusions and prospects
The global COVID-19 pandemic has promoted rapid development and human testing of nAbs to prevent and treat SARS-CoV-2 infection. Among all of the SARS-CoV-2 proteins, S, particularly its RBD fragment, is the major target for the development of potent COVID-19 nAbs, and nAbs targeting this region are generally more potent than those targeting other regions, including the NTD. Most of these nAbs have been evaluated preclinically and exhibit neutralizing activity against SARS-CoV-2 in vitro and/or prophylactic or therapeutic protection of animals from SARS-CoV-2 challenge. Some of these nAbs also prevent infection or can be used to treat SARS-CoV-2-infected individuals. Nevertheless, the emergence of mutations in the antibody target regions, particularly in the S protein RBD, combined with the presence of antibody escape variants, makes it essential to design monovalent and multivalent nAbs or nAb cocktails with improved neutralizing activity and protective efficacy. Moreover, it would be advantageous to generate large amounts of monoclonal antibodies with relatively low cost for clinical use. Hopefully, new nAbs with broad-spectrum neutralizing activity against multiple SARS-CoV-2 variants and escape mutant strains will be developed, and more cost-effective manufacturing practices will be established. Ideally, these nAbs will provide safe and effective prophylactic and therapeutic agents that can be used to prevent and treat infection caused by current SARS-CoV-2 strains and future variants that may develop.
References
Elezkurtaj S, Greuel S, Ihlow J, Michaelis EG, Bischoff P, Kunze CA, et al. Causes of death and comorbidities in hospitalized patients with COVID-19. Sci Rep. 2021;11:4263.
Papoutsi E, Giannakoulis VG, Xourgia E, Routsi C, Kotanidou A, Siempos II. Effect of timing of intubation on clinical outcomes of critically ill patients with COVID-19: a systematic review and meta-analysis of non-randomized cohort studies. Crit Care. 2021;25:121.
Synowiec A, Szczepański A, Barreto-Duran E, Lie LK, Pyrc K. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): a systemic infection. Clin Microbiol Rev. 2021;34:e00133–20.
Piwowarczyk P, Szczukocka M, Kutnik P, Borys M, Mikłaszewska A, Kiciak S, et al. Risk factors and outcomes for acute respiratory failure in coronavirus disease 2019: an observational cohort study. Adv Clin Exp Med. 2021;30:165–71.
Olivares F, Muñoz D, Fica A, Delama I, Alvarez I, Navarrete M, et al. Clinical features of 47 patients infected with COVID-19 admitted to a regional reference center. Rev Med Chil. 2020;148:1577–88.
Salter A, Fox RJ, Newsome SD, Halper J, Li D, Kanellis P, et al. Outcomes and risk factors associated with SARS-CoV-2 infection in a north American registry of patients with multiple sclerosis. JAMA Neurol. 2021;78:699–708.
Zhang T, et al. Erratum to risk factors and predictors associated with the severity of COVID-19 in China: a systematic review, meta-analysis, and meta-regression. J Thorac Dis. 2021;13:503–4.
Kwok S, Adam S, Ho JH, Iqbal Z, Turkington P, Razvi S, et al. Obesity: a critical risk factor in the COVID-19 pandemic. Clin Obes. 2020;10:e12403.
Oliver SE, Gargano JW, Marin M, Wallace M, Curran KG, Chamberland M, et al. The Advisory Committee on immunization practices’ interim recommendation for use of Moderna COVID-19 vaccine – United States, December 2020. MMWR Morb Mortal Wkly Rep. 2021;69:1653–6.
Oliver SE, Gargano JW, Marin M, Wallace M, Curran KG, Chamberland M, et al. The Advisory Committee on immunization practices’ interim recommendation for use of Pfizer-BioNTech COVID-19 vaccine – United States, December 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1922–4.
Oliver SE, Gargano JW, Scobie H, Wallace M, Hadler SC, Leung J, et al. The Advisory Committee on immunization practices’ interim recommendation for use of Janssen COVID-19 vaccine – United States, February 2021. MMWR Morb Mortal Wkly Rep. 2021;70:329–32.
Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–20.
Zhong NS, Zheng BJ, Li YM, Poon, Xie ZH, Chan KH, et al. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People’s Republic of China, in February, 2003. Lancet. 2003;362:1353–8.
Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The spike protein of SARS-CoV-a target for vaccine and therapeutic development. Nat Rev Microbiol. 2009;7:226–36.
Lee EC, Wada NI, Grabowski MK, Gurley ES, Lessler J. The engines of SARS-CoV-2 spread. Science. 2020;370:406–7.
Johansson MA, Quandelacy TM, Kada S, Prasad PV, Steele M, Brooks JT, et al. SARS-CoV-2 transmission from people without COVID-19 symptoms. JAMA Netw Open. 2021;4:e2035057.
Metlay JP, Haas JS, Soltoff AE, Armstrong KA. Household transmission of SARS-CoV-2. JAMA Netw Open. 2021;4:e210304.
Wang N, Shang J, Jiang S, Du L. Subunit vaccines against emerging pathogenic human coronaviruses. Front Microbiol. 2020;11:298.
Naqvi AAT, et al. Insights into SARS-CoV-2 genome, structure, evolution, pathogenesis and therapies: structural genomics approach. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165878.
Khan MT, Irfan M, Ahsan H, Ahmed A, Kaushik AC, Khan AS, et al. Structures of SARS-CoV-2 RNA-binding proteins and therapeutic targets. Intervirology. 2021;64:55–68.
Duart G, García-Murria MJ, Mingarro I. The SARS-CoV-2 envelope (E) protein has evolved towards membrane topology robustness. Biochim Biophys Acta Biomembr. 2021;1863:183608.
Cabrera-Garcia D, Bekdash R, Abbott GW, Yazawa M, Harrison NL. The envelope protein of SARS-CoV-2 increases intra-Golgi pH and forms a cation channel that is regulated by pH. J Physiol. 2021;599:2851–68.
Singh Tomar PP, Arkin IT. SARS-CoV-2 E protein is a potential ion channel that can be inhibited by gliclazide and memantine. Biochem Biophys Res Commun. 2020;530:10–14.
Gao T, Gao Y, Liu X, Nie Z, Sun H, Lin K, et al. Identification and functional analysis of the SARS-COV-2 nucleocapsid protein. BMC Microbiol. 2021;21:58.
Oh SJ, Shin OS. SARS-CoV-2 nucleocapsid protein targets RIG-I-Like receptor pathways to inhibit the induction of interferon response. Cells. 2021;10:530.
Chen K, Xiao F, Hu D, Ge W, Tian M, Wang W, et al. SARS-CoV-2 nucleocapsid protein interacts with RIG-I and represses RIG-mediated IFN-β production. Viruses. 2020;13:47.
Xia S, Liu M, Wang C, Xu W, Lan Q, Feng S, et al. Inhibition of SARS-CoV-2 (previously 2019-nCoV) infection by a highly potent pan-coronavirus fusion inhibitor targeting its spike protein that harbors a high capacity to mediate membrane fusion. Cell Res. 2020;30:343–55.
Shang J, Ye G, Shi K, Wan Y, Luo C, Aihara H, et al. Structural basis of receptor recognition by SARS-CoV-2. Nature. 2020;581:221–4.
Behloul N, Baha S, Shi R, Meng J. Role of the GTNGTKR motif in the N-terminal receptor-binding domain of the SARS-CoV-2 spike protein. Virus Res. 2020;286:198058.
Seyran M, Takayama K, Uversky VN, Lundstrom K, Palù G, Sherchan SP, et al. The structural basis of accelerated host cell entry by SARS-CoV-2. FEBS J. 2020. https://doi.org/10.1111/febs.15651.
Lan J, Ge J, Yu J, Shan S, Zhou H, Fan S, et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature. 2020;581:215–20.
Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–3.
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–80. e278
Cantuti-Castelvetri L, Ojha R, Pedro LD, Djannatian M, Franz J, Kuivanen S, et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science. 2020;370:856–60.
Wrapp D, Wang N, Corbett KS, Goldsmith JA, Hsieh CL, Abiona O, et al. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–3.
Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:281–92. e286
Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science. 2020;367:1444–8.
Tai W, He L, Zhang X, Pu J, Voronin D, Jiang S, et al. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol Immunol. 2020;17:613–20.
Jiang S, Zhang X, Du L. Therapeutic antibodies and fusion inhibitors targeting the spike protein of SARS-CoV-2. Expert Opin Ther Targets. 2021;25:415–21.
Jiang S, Hillyer C, Du L. Neutralizing antibodies against SARS-CoV-2 and other human coronaviruses. Trends Immunol. 2020;41:355–9.
McCallum M, De Marco A, Lempp FA, Tortorici MA, Pinto D, Walls AC, et al. N-terminal domain antigenic mapping reveals a site of vulnerability for SARS-CoV-2. Cell. 2021;184:2332–47.
Zost SJ, Gilchuk P, Chen RE, Case JB, Reidy JX, Trivette A, et al. Rapid isolation and profiling of a diverse panel of human monoclonal antibodies targeting the SARS-CoV-2 spike protein. Nat Med. 2020;26:1422–7.
Brouwer PJM, Caniels TG. Potent neutralizing antibodies from COVID-19 patients define multiple targets of vulnerability. Science. 2020;369:643–50.
Liu L, Wang P, Nair MS, Yu J, Rapp M, Wang Q, et al. Potent neutralizing antibodies against multiple epitopes on SARS-CoV-2 spike. Nature. 2020;84:450–6.
Mor M, Werbner M, Alter J, Safra M, Chomsky E, Lee JC, et al. Multi-clonal SARS-CoV-2 neutralization by antibodies isolated from severe COVID-19 convalescent donors. PLoS Pathog. 2021;17:e1009165.
Parray HA, Chiranjivi AK, Asthana S, Yadav N, Shrivastava T, Mani S, et al. Identification of an anti-SARS-CoV-2 receptor-binding domain-directed human monoclonal antibody from a naïve semisynthetic library. J Biol Chem. 2020;295:12814–21.
Kim C, Ryu DK, Lee J, Kim YI, Seo JM, Kim YG, et al. A therapeutic neutralizing antibody targeting receptor binding domain of SARS-CoV-2 spike protein. Nat Commun. 2021;12:288.
Noy-Porat T, Makdasi E, Alcalay R, Mechaly A, Levy Y, Bercovich-Kinori A, et al. A panel of human neutralizing mAbs targeting SARS-CoV-2 spike at multiple epitopes. Nat Commun. 2020;11:4303.
Ju B, Zhang Q, Ge J, Wang R, Sun J, Ge X, et al. Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature. 2020;584:115–9.
Kim YJ, Lee MH, Lee SR, Chung HY, Kim K, Lee TG, et al. Neutralizing human antibodies against severe acute respiratory syndrome coronavirus 2 isolated from a human synthetic Fab phage display library. Int J Mol Sci. 2021;22:1913.
Ku Z, Xie X. Molecular determinants and mechanism for antibody cocktail preventing SARS-CoV-2 escape. Nat Commun. 2021;12:469.
Noy-Porat T, Mechaly A, Levy Y, Makdasi E, Alcalay R, Gur D, et al. Therapeutic antibodies, targeting the SARS-CoV-2 spike N-terminal domain, protect lethally infected K18-hACE2 mice. iScience. 2021;24:102479.
Graham C, Seow J, Huettner I, Khan H, Kouphou N, Acors S, et al. Neutralization potency of monoclonal antibodies recognizing dominant and subdominant epitopes on SARS-CoV-2 Spike is impacted by the B.1.1.7 variant. Immunity. 2021;54:1276–89. e1276
Suryadevara N, Shrihari S, Gilchuk P, VanBlargan LA, Binshtein E, Zost SJ, et al. Neutralizing and protective human monoclonal antibodies recognizing the N-terminal domain of the SARS-CoV-2 spike protein. Cell. 2021;184:2316–31.
Chi X, Yan R, Zhang J, Zhang G, Zhang Y, Hao M, et al. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science. 2020;369:650–5.
Cerutti G, Guo Y, Zhou T, Gorman J, Lee M, Rapp M, et al. Potent SARS-CoV-2 neutralizing antibodies directed against spike N-terminal domain target a single supersite. Cell Host Microbe. 2021;29:819–33. e817
Liu Y, Soh WT, Kishikawa JI, Hirose M, Nakayama EE, Li S, et al. An infectivity-enhancing site on the SARS-CoV-2 spike protein targeted by antibodies. Cell. 2021;184:3452–66. e3418
Winkler ES, Gilchuk P, Yu J, Bailey AL, Chen RE, Chong Z, et al. Human neutralizing antibodies against SARS-CoV-2 require intact Fc effector functions for optimal therapeutic protection. Cell. 2021;184:1804–20. e1816
Li W, Chen C, Drelich A, Martinez DR, Gralinski LE, Sun Z, et al. Rapid identification of a human antibody with high prophylactic and therapeutic efficacy in three animal models of SARS-CoV-2 infection. Proc Natl Acad Sci USA. 2020;117:29832–8.
Dejnirattisai W, Zhou D, Ginn HM, Duyvesteyn H, Supasa P, Case JB, et al. The antigenic anatomy of SARS-CoV-2 receptor binding domain. Cell. 2021;184:2183–2200. e2122
Miao X, Luo Y, Huang X, Lee S, Yuan Z, Tang Y, et al. A novel biparatopic hybrid antibody-ACE2 fusion that blocks SARS-CoV-2 infection: implications for therapy. MAbs. 2020;12:1804241.
Andreano E, Nicastri E, Paciello I, Pileri P, Manganaro N, Piccini G, et al. Extremely potent human monoclonal antibodies from COVID-19 convalescent patients. Cell. 2021;184:1821–35. e1816
Yan R, Wang R, Ju B, Yu J, Zhang Y, Liu N, et al. Structural basis for bivalent binding and inhibition of SARS-CoV-2 infection by human potent neutralizing antibodies. Cell Res. 2021;31:517–25.
Kreye J, Reincke SM, Kornau HC, Sánchez-Sendin E, Corman VM, Liu H, et al. A Therapeutic non-self-reactive SARS-CoV-2 antibody protects from lung pathology in a COVID-19 hamster model. Cell. 2020;183:1058–69. e1019
Shi R, Shan C, Duan X, Chen Z, Liu P, Song J, et al. A human neutralizing antibody targets the receptor-binding site of SARS-CoV-2. Nature. 2020;584:120–4.
Sun Z, Chen C, Li W, Martinez DR, Drelich A, Baek DS, et al. Potent neutralization of SARS-CoV-2 by human antibody heavy-chain variable domains isolated from a large library with a new stable scaffold. MAbs. 2020;12:1778435.
Li W, Schäfer A, Kulkarni SS, Liu X, Martinez DR, Chen C, et al. High potency of a bivalent human V(H) domain in SARS-CoV-2 animal models. Cell. 2020;183:429–41. e416
Piepenbrink MS, Park JG, Oladunni FS, Deshpande A, Basu M, Sarkar S, et al. Therapeutic activity of an inhaled potent SARS-CoV-2 neutralizing human monoclonal antibody in hamsters. Cell Rep Med. 2021;2:100218.
Hurlburt NK, Seydoux E, Wan YH, Edara VV, Stuart AB, Feng J, et al. Structural basis for potent neutralization of SARS-CoV-2 and role of antibody affinity maturation. Nat Commun. 2020;11:5413.
Barnes CO, West AP, Huey-Tubman KE, Hoffmann M, Sharaf NG, Hoffman PR, et al. Structures of human antibodies bound to SARS-CoV-2 spike reveal common epitopes and recurrent features of antibodies. Cell. 2020;182:828–42. e816
Piccoli L, Park YJ, Tortorici MA, Czudnochowski N, Walls AC, Beltramello M, et al. Mapping neutralizing and immunodominant sites on the SARS-CoV-2 spike receptor-binding domain by structure-guided high-resolution serology. Cell. 2020;183:1024–42. e1021
Ge J, Wang R, Ju B, Zhang Q, Sun J, Chen P, et al. Antibody neutralization of SARS-CoV-2 through ACE2 receptor mimicry. Nat Commun. 2021;12:250.
Rogers TF, Zhao F, Huang D, Beutler N, Burns A, He WT, et al. Isolation of potent SARS-CoV-2 neutralizing antibodies and protection from disease in a small animal model. Science. 2020;369:956–63.
Wang S, Peng Y, Wang R, Jiao S, Wang M, Huang W, et al. Characterization of neutralizing antibody with prophylactic and therapeutic efficacy against SARS-CoV-2 in rhesus monkeys. Nat Commun. 2020;11:5752.
Zost SJ, Gilchuk P, Case JB, Binshtein E, Chen RE, Nkolola JP, et al. Potently neutralizing and protective human antibodies against SARS-CoV-2. Nature. 2020;584:443–9.
Jones BE, Brown-Augsburger PL, Corbett KS, Westendorf K, Davies J, Cujec TP, et al. The neutralizing antibody, LY-CoV555, protects against SARS-CoV-2 infection in non-human primates. Sci Transl Med. 2021;13:eabf1906.
Schäfer A, Muecksch F, Lorenzi J, Leist SR, Cipolla M, Bournazos S, et al. Antibody potency, effector function, and combinations in protection and therapy for SARS-CoV-2 infection in vivo. J Exp Med. 2021;218:e20201993.
Baum A, Ajithdoss D, Copin R, Zhou A, Lanza K, Negron N, et al. REGN-COV2 antibodies prevent and treat SARS-CoV-2 infection in rhesus macaques and hamsters. Science. 2020;370:1110–5.
Hodge CD, Rosenberg DJ, Grob P, Wilamowski M, Joachimiak A, Hura GL, et al. Rigid monoclonal antibodies improve detection of SARS-CoV-2 nucleocapsid protein. MAbs. 2021;13:1905978.
Yamaoka Y, Miyakawa K, Jeremiah SS, Funabashi R, Okudela K, Kikuchi S, et al. Highly specific monoclonal antibodies and epitope identification against SARS-CoV-2 nucleocapsid protein for antigen detection tests. Cell Rep Med. 2021;2:100311.
Liu D, Wu F, Cen Y, Ye L, Shi X, Huang Y, et al. Comparative research on nucleocapsid and spike glycoprotein as the rapid immunodetection targets of COVID-19 and establishment of immunoassay strips. Mol Immunol. 2021;131:6–12.
Díaz Y, Ramos-Suzarte M, Martín Y, Calderón NA, Santiago W, Viñet O, et al. Use of a humanized anti-CD6 monoclonal antibody (itolizumab) in elderly patients with moderate COVID-19. Gerontology. 2020;66:553–61.
Perrone F, Piccirillo MC, Ascierto PA, Salvarani C, Parrella R, Marata AM, et al. Tocilizumab for patients with COVID-19 pneumonia. The single-arm TOCIVID-19 prospective trial. J Transl Med. 2020;18:405.
Stone JH, Frigault MJ, Serling-Boyd NJ, Fernandes AD, Harvey L, Foulkes AS, et al. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020;383:2333–44.
Hermine O, Mariette X, Tharaux PL, Resche-Rigon M, Porcher R, Ravaud P, et al. Effect of tocilizumab vs usual care in adults hospitalized with COVID-19 and moderate or severe pneumonia: a randomized clinical trial. JAMA Intern Med. 2021;181:32–40.
Salvarani C, Dolci G, Massari M, Merlo DF, Cavuto S, Savoldi L, et al. Effect of tocilizumab vs standard care on clinical worsening in patients hospitalized with COVID-19 pneumonia: a randomized clinical trial. JAMA Intern Med. 2021;181:24–31.
Gupta S, Wang W, Hayek SS, Chan L, Mathews KS, Melamed ML, et al. Association between early treatment with tocilizumab and mortality among critically ill patients with COVID-19. JAMA Intern Med. 2021;181:41–51.
Malekzadeh R, Abedini A, Mohsenpour B, Sharifipour E, Ghasemian R, Javad-Mousavi SA, et al. Subcutaneous tocilizumab in adults with severe and critical COVID-19: a prospective open-label uncontrolled multicenter trial. Int Immunopharmacol. 2020;89:107102.
Panoskaltsis N, McCarthy NE, Stagg AJ, Mummery CJ, Husni M, Arebi N, et al. Immune reconstitution and clinical recovery following anti-CD28 antibody (TGN1412)-induced cytokine storm. Cancer Immunol Immunother. 2021;70:1127–42.
Martínez-Sanz J, Muriel A, Ron R, Herrera S, Pérez-Molina JA, Moreno S, et al. Effects of tocilizumab on mortality in hospitalized patients with COVID-19: a multicentre cohort study. Clin Microbiol Infect. 2021;27:238–43.
Cotter A, Wallace D, McCarthy C, Feeney E, O'Neill L, Stack J, et al. The COVIRL002 trial-tocilizumab for management of severe, non-critical COVID-19 infection: a structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21:758.
Jordan SC, Zakowski P, Tran HP, Smith EA, Gaultier C, Marks G, et al. Compassionate use of tocilizumab for treatment of SARS-CoV-2 pneumonia. Clin Infect Dis. 2020;71:3168–73.
Liu P, Huang Z, Yin M, Liu C, Chen X, Pan P, et al. Safety and efficacy of ixekizumab and antiviral treatment for patients with COVID-19: A structured summary of a study protocol for a pilot randomized controlled trial. Trials. 2020;21:999.
Weinreich DM, Sivapalasingam S, Norton T, Ali S, Gao H, Bhore R, et al. REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19. N Engl J Med. 2021;384:238–51.
Chen P, Nirula A, Heller B, Gottlieb RL, Boscia J, Morris J, et al. SARS-CoV-2 neutralizing antibody LY-CoV555 in outpatients with Covid-19. N Engl J Med. 2021;384:229–37.
Dhand A, Lobo SA, Wolfe K, Feola N, Nabors C. Bamlanivimab for treatment of COVID-19 in solid organ transplant recipients: early single-center experience. Clin Transpl. 2021;35:e14245.
Gottlieb RL, Nirula A, Chen P, Boscia J, Heller B, Morris J, et al. Effect of bamlanivimab as monotherapy or in combination with etesevimab on viral load in patients with mild to moderate COVID-19: a randomized clinical trial. JAMA. 2021;325:632–44.
Tuccori M, Ferraro S, Convertino I, Cappello E, Valdiserra G, Blandizzi C, et al. Anti-SARS-CoV-2 neutralizing monoclonal antibodies: clinical pipeline. MAbs. 2020;12:1854149.
O'brien MP, Forleo-Neto E, Sarkar N, Isa F, Hou P, Chan KC, et al. Subcutaneous REGEN-COV antibody combination in early SARS-CoV-2 infection. medRxiv [Preprint]. 2021. https://doi.org/10.1101/2021.06.14.21258569.
Verderese JP, Stepanova M, Lam B, Racila A, Kolacevski A, Allen D, et al. Neutralizing monoclonal antibody treatment reduces hospitalization for mild and moderate COVID-19: a real-world experience. Clin Infect Dis. 2021:ciab579. https://doi.org/10.1093/cid/ciab579.
Phan AT, Gukasyan J, Arabian S, Wang S, Neeki MM. Emergent inpatient administration of casirivimab and imdevimab antibody cocktail for the treatment of COVID-19 pneumonia. Cureus. 2021;13:e15280.
O’brien MP, Forleo-Neto E, Musser BJ, Isa F, Chan KC, Sarkar N, et al. Subcutaneous REGEN-COV antibody combination to prevent Covid-19. N Engl J Med. 2021. https://doi.org/10.1056/NEJMoa2109682.
Casadevall A, Joyner MJ, Pirofski LA. Neutralizing antibody LY-CoV555 for outpatient Covid-19. N Engl J Med. 2021;384:189.
ACTIV-/TICO LY- Study Group, Lundgren JD, Grund B, Barkauskas CE, Holland TL, Gottlieb RL, et al. A neutralizing monoclonal antibody for hospitalized patients with Covid-19. N Engl J Med. 2021;384:905–14.
Cohen MS, Nirula A, Mulligan MJ, Novak RM, Marovich M, Yen C, et al. Effect of bamlanivimab vs placebo on incidence of COVID-19 among residents and staff of skilled nursing and assisted living facilities: a randomized clinical trial. JAMA. 2021;326:46–55.
Wolf J, Abzug MJ, Wattier RL, Sue PK, Vora SB, Zachariah P, et al. Initial guidance on use of monoclonal antibody therapy for treatment of COVID-19 in children and adolescents. J Pediatr Infect Dis Soc. 2021;10:629–34.
Emergency Use Authorization for bamlanivimab in mild to moderate COVID-19: implications for CNS Practice. Clin Nurse Spec. 2021;35:E3.
O’Malley PA. Emergency Use Authorization for bamlanivimab in mild to moderate COVID-19: implications for clinical nurse specialist practice. Clin Nurse Spec. 2021;35:112–5.
An EUA for bamlanivimab-A monoclonal antibody for COVID-19. JAMA. 2021;325:880–1.
Troyano-Hernáez P, Reinosa R, Holguín Á. Evolution of SARS-CoV-2 envelope, membrane, nucleocapsid, and spike structural proteins from the beginning of the pandemic to September 2020: a global and regional approach by epidemiological week. Viruses. 2021;13:243.
Centers for Disease Control and Prevention. SARS-CoV2 variant classifications and definitions. Updated on Aug. 17, 2021. https://www.cdc.gov/coronavirus/2019-ncov/variants/variant-info.html.
World Health Organization. Tracking SARS-CoV-2 variants. Updated on Aug. 13, 2021. https://www.who.int/en/activities/tracking-SARS-CoV-2-variants/.
Greaney AJ, Starr TN, Gilchuk P, Zost SJ, Binshtein E, Loes AN, et al. Complete mapping of mutations to the SARS-CoV-2 spike receptor-binding domain that escape antibody recognition. Cell Host Microbe. 2021;29:44–57. e49
Weisblum Y, Schmidt F, Zhang F, DaSilva J, Poston D, Lorenzi JC, et al. Escape from neutralizing antibodies by SARS-CoV-2 spike protein variants. Elife. 2020;9:e61312.
Greaney AJ, Loes AN, Crawford K, Starr TN, Malone KD, Chu HY, et al. Comprehensive mapping of mutations in the SARS-CoV-2 receptor-binding domain that affect recognition by polyclonal human plasma antibodies. Cell Host Microbe. 2021;29:463–76. e466
Wang P, Nair MS, Liu L, Iketani S, Luo Y, Guo Y, et al. Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7. Nature. 2021;593:130–5.
Chen RE, Zhang X, Case JB, Winkler ES, Liu Y, VanBlargan LA, et al. Resistance of SARS-CoV-2 variants to neutralization by monoclonal and serum-derived polyclonal antibodies. Nat Med. 2021;27:717–26.
Diamond M, Chen R, Xie X, Case J, Zhang X, VanBlargan L, et al. SARS-CoV-2 variants show resistance to neutralization by many monoclonal and serum-derived polyclonal antibodies. Res Sq [Preprint]. 2021;rs.3.rs-228079.
Shen X, Tang H, McDanal C, Wagh K, Fischer W, Theiler J, et al. SARS-CoV-2 variant B.1.1.7 is susceptible to neutralizing antibodies elicited by ancestral spike vaccines. Cell Host Microbe. 2021;29:529–39. e523
Starr TN, Greaney AJ, Dingens AS, Bloom JD. Complete map of SARS-CoV-2 RBD mutations that escape the monoclonal antibody LY-CoV555 and its cocktail with LY-CoV016. Cell Rep Med. 2021;2:100255.
Wang P, Casner RG, Nair MS, Wang M, Yu J, Cerutti G, et al. Increased resistance of SARS-CoV-2 variant P.1 to antibody neutralization. Cell Host Microbe. 2021;29:747–51. e744
Zhu X, Mannar D, Srivastava SS, Berezuk AM, Demers JP, Saville JW, et al. Cryo-electron microscopy structures of the N501Y SARS-CoV-2 spike protein in complex with ACE2 and 2 potent neutralizing antibodies. PLoS Biol. 2021;19:e3001237.
Sun Z, Kim A, Sobolewski MD, Enick N, Chen C, Adams C, et al. Neutralization of European, South African, and United States SARS-CoV-2 mutants by a human antibody and antibody domains. bioRxiv [Preprint]. 2021. https://doi.org/10.1101/2021.03.22.436481.
Dejnirattisai W, Zhou D, Supasa P, Liu C, Mentzer AJ, Ginn HM, et al. Antibody evasion by the P.1 strain of SARS-CoV-2. Cell. 2021;184:2939–54. e2939
Starr TN, Greaney AJ, Addetia A, Hannon WW, Choudhary MC, Dingens AS, et al. Prospective mapping of viral mutations that escape antibodies used to treat COVID-19. Science. 2021;371:850–4.
Haslwanter D, Dieterle ME, Wec AZ, O’Brien CM, Sakharkar M, Florez C, et al. A combination of RBD and NTD neutralizing antibodies limits the generation of SARS-CoV-2 spike neutralization-escape mutants. bioRxiv [Preprint]. 2021. https://doi.org/10.1101/2021.06.10.447999.
Copin R, Baum A, Wloga E, Pascal KE, Giordano S, Fulton BO, et al. The monoclonal antibody combination REGEN-COV protects against SARS-CoV-2 mutational escape in preclinical and human studies. Cell. 2021;184:3949–61.
Acknowledgements
This study was supported by the NIH grants R01AI139092, R01AI137472, and R01AI157975.
Author information
Authors and Affiliations
Contributions
L.D. wrote and revised the manuscript. Y.Y. and X.Z. prepared the figures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Du, L., Yang, Y. & Zhang, X. Neutralizing antibodies for the prevention and treatment of COVID-19. Cell Mol Immunol 18, 2293–2306 (2021). https://doi.org/10.1038/s41423-021-00752-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-021-00752-2
Keywords
This article is cited by
-
Therapeutic proteins: developments, progress, challenges, and future perspectives
3 Biotech (2024)
-
A high-throughput screening system for SARS-CoV-2 entry inhibition, syncytia formation and cell toxicity
Biological Procedures Online (2023)
-
Safety, tolerability, pharmacokinetics, and immunogenicity of JMB2002–an antibody against COVID-19: a phase 1 clinical trial in healthy Chinese adults
BMC Infectious Diseases (2023)
-
Influence of variant-specific mutations, temperature and pH on conformations of a large set of SARS-CoV-2 spike trimer vaccine antigen candidates
Scientific Reports (2023)
-
Production of neutralizing antibody fragment variants in the cytoplasm of E. coli for rapid screening: SARS-CoV-2 a case study
Scientific Reports (2023)