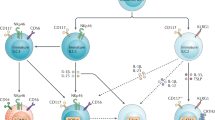
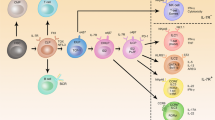
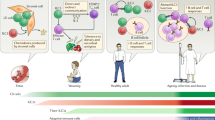
Abstract
Maintenance of homeostasis and immune protection rely on the coordinated action of different physiological systems. Bidirectional communication between the immune system and physiological systems is required to sense and restore any disruption of equilibrium. Recent transcriptomic analyses of innate lymphoid cells (ILCs) from different tissues have revealed that ILCs express a large array of receptors involved in the recognition of neuropeptides, hormones and metabolic signals. ILCs rapidly secrete effector cytokines that are central in the development and activation of early immune responses, but they also constitutively secrete mediators that are important for tissue homeostasis. To achieve these functions effectively, ILCs integrate intrinsic and extrinsic signals that modulate their constitutive and induced activity. Disruption of the regulation of ILCs by physiological regulators leads to altered immune responses with harmful consequences for the organism. An understanding of these complex interactions between the immune system and physiological mediators is crucial to decipher the events leading to the protective versus pathological effects of these cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Chrousos, G. P. & Gold, P. W. The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. JAMA 267, 1244–1252 (1992).
Medzhitov, R. Inflammation 2010: new adventures of an old flame. Cell 140, 771–776 (2010).
Vivier, E. et al. Innate lymphoid cells: 10 years on. Cell 174, 1054–1066 (2018).
Ishizuka, I. E. et al. Single-cell analysis defines the divergence between the innate lymphoid cell lineage and lymphoid tissue-inducer cell lineage. Nat. Immunol. 17, 269–276 (2016).
Constantinides, M. G., McDonald, B. D., Verhoef, P. A. & Bendelac, A. A committed precursor to innate lymphoid cells. Nature 508, 397–401 (2014).
Klose, C. S. N. et al. Differentiation of type 1 ILCs from a common progenitor to all helper-like innate lymphoid cell lineages. Cell 157, 340–356 (2014).
Seillet, C. et al. Differential requirement for Nfil3 during NK cell development. J. Immunol. 192, 2667–2676 (2014).
Yu, Y. et al. Single-cell RNA-seq identifies a PD-1(hi) ILC progenitor and defines its development pathway. Nature 539, 102–106 (2016).
Seillet, C. et al. Deciphering the innate lymphoid cell transcriptional program. Cell Rep. 17, 436–447 (2016).
Bando, J. K., Liang, H.-E. & Locksley, R. M. Identification and distribution of developing innate lymphoid cells in the fetal mouse intestine. Nat. Immunol. 16, 153–160 (2015).
Gasteiger, G., Fan, X., Dikiy, S., Lee, S. Y. & Rudensky, A. Y. Tissue residency of innate lymphoid cells in lymphoid and nonlymphoid organs. Science 350, 981–985 (2015).
Huang, Y. et al. IL-25-responsive, lineage-negative KLRG1(hi) cells are multipotential ‘inflammatory’ type 2 innate lymphoid cells. Nat. Immunol. 16, 161–169 (2015).
Stier, M. T. et al. IL-33 promotes the egress of group 2 innate lymphoid cells from the bone marrow. J. Exp. Med. 215, 263–281 (2018).
Huang, Y. et al. S1P-dependent interorgan trafficking of group 2 innate lymphoid cells supports host defense. Science 359, 114–119 (2018).
Ricardo-Gonzalez, R. R. et al. Tissue signals imprint ILC2 identity with anticipatory function. Nat. Immunol. 10, 1093–1099 (2018).
Gury-BenAri, M. et al. The spectrum and regulatory landscape of intestinal innate lymphoid cells are shaped by the microbiome. Cell 166, 1231–1246.e13 (2016).
Monticelli, L. A. et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat. Immunol. 12, 1045–1054 (2011).
Sawa, S. et al. RORgammat+ innate lymphoid cells regulate intestinal homeostasis by integrating negative signals from the symbiotic microbiota. Nat. Immunol. 12, 320–326 (2011).
Weizman, O. E. et al. ILC1 confer early host protection at initial sites of viral infection. Cell 171, 795–808 e712 (2017).
Abt, M. C. et al. Innate immune defenses mediated by two ILC subsets are critical for protection against acute Clostridium difficile infection. Cell Host Microbe 18, 27–37 (2015).
Jiao, Y., Huntington, N. D., Belz, G. T. & Seillet, C. Type 1 innate lymphoid cell biology: lessons learnt from natural killer cells. Front Immunol. 7, 426 (2016).
Boulenouar, S. et al. Adipose type one innate lymphoid cells regulate macrophage homeostasis through targeted cytotoxicity. Immunity 46, 273–286 (2017).
Fallon, P. G. et al. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J. Exp. Med. 203, 1105–1116 (2006).
Seillet, C., Belz, G. T. & Mielke, L. A. Complexity of cytokine network regulation of innate lymphoid cells in protective immunity. Cytokine 70, 1–10 (2014).
Chang, Y.-J. et al. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nat. Immunol. 12, 631–638 (2011).
Molofsky, A. B. et al. Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J. Exp. Med. 210, 535–549 (2013).
Behnsen, J. et al. The cytokine IL-22 promotes pathogen colonization by suppressing related commensal bacteria. Immunity 40, 262–273 (2014).
Pickard, J. M. et al. Rapid fucosylation of intestinal epithelium sustains host-commensal symbiosis in sickness. Nature 514, 638–641 (2014).
Satoh-Takayama, N. et al. The chemokine receptor CXCR6 controls the functional topography of interleukin-22 producing intestinal innate lymphoid cells. Immunity 41, 776–788 (2014).
Rankin, L. C. et al. The transcription factor T-bet is essential for the development of NKp46(+) innate lymphocytes via the Notch pathway. Nat. Immunol. 14, 389–395 (2013).
Sawada, M., Itoh, Y., Suzumura, A. & Marunouchi, T. Expression of cytokine receptors in cultured neuronal and glial cells. Neurosci. Lett. 160, 131–134 (1993).
Neumann, H. et al. Tumor necrosis factor inhibits neurite outgrowth and branching of hippocampal neurons by a rho-dependent mechanism. J. Neurosci. 22, 854–862 (2002).
Gougeon, P. Y. et al. The pro-inflammatory cytokines IL-1beta and TNFalpha are neurotrophic for enteric neurons. J. Neurosci. 33, 3339–3351 (2013).
Klose, C. S. N. et al. The neuropeptide neuromedin U stimulates innate lymphoid cells and type 2 inflammation. Nature 549, 282–286 (2017).
Howard, A. D. et al. Identification of receptors for neuromedin U and its role in feeding. Nature 406, 70–74 (2000).
Cardoso, V. et al. Neuronal regulation of type 2 innate lymphoid cells via neuromedin U. Nature 549, 277–281 (2017).
Wallrapp, A. et al. The neuropeptide NMU amplifies ILC2-driven allergic lung inflammation. Nature 549, 351–356 (2017).
Moriyama, M. et al. The neuropeptide neuromedin U promotes inflammation by direct activation of mast cells. J. Exp. Med. 202, 217–224 (2005).
Mashaghi, A. et al. Neuropeptide substance P and the immune response. Cell Mol. Life Sci. 73, 4249–4264 (2016).
Feistritzer, C. et al. Natural killer cell functions mediated by the neuropeptide substance P. Regul. Pept. 116, 119–126 (2003).
Lang, K., Drell, T. L., Niggemann, B., Zanker, K. S. & Entschladen, F. Neurotransmitters regulate the migration and cytotoxicity in natural killer cells. Immunol. Lett. 90, 165–172 (2003).
Monaco-Shawver, L. et al. Substance P inhibits natural killer cell cytotoxicity through the neurokinin-1 receptor. J. Leukoc. Biol. 89, 113–125 (2011).
Lighvani, S., Huang, X., Trivedi, P. P., Swanborg, R. H. & Hazlett, L. D. Substance P regulates natural killer cell interferon-gamma production and resistance to Pseudomonas aeruginosa infection. Eur. J. Immunol. 35, 1567–1575 (2005).
Sears, M. R. et al. Regular inhaled beta-agonist treatment in bronchial asthma. Lancet 336, 1391–1396 (1990).
Milano, C. A. et al. Enhanced myocardial function in transgenic mice overexpressing the beta 2-adrenergic receptor. Science 264, 582–586 (1994).
Sun, Z. et al. Norepinephrine inhibits the cytotoxicity of NK92MI cells via the beta2adrenoceptor/cAMP/PKA/pCREB signaling pathway. Mol. Med. Rep. 17, 8530–8535 (2018).
De Lorenzo, B. H., de Oliveira Marchioro, L., Greco, C. R. & Suchecki, D. Sleep-deprivation reduces NK cell number and function mediated by beta-adrenergic signalling. Psychoneuroendocrinology 57, 134–143 (2015).
Logan, R. W., Arjona, A. & Sarkar, D. K. Role of sympathetic nervous system in the entrainment of circadian natural-killer cell function. Brain Behav. Immun. 25, 101–109 (2011).
Moriyama, S. et al. beta2-adrenergic receptor-mediated negative regulation of group 2 innate lymphoid cell responses. Science 359, 1056–1061 (2018).
Wang, H. et al. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature 421, 384–388 (2003).
Borovikova, L. V. et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405, 458–462 (2000).
Dalli, J., Colas, R. A., Arnardottir, H. & Serhan, C. N. Vagal regulation of group 3 innate lymphoid cells and the immunoresolvent PCTR1 controls infection resolution. Immunity 46, 92–105 (2017).
Ramon, S. et al. The protectin PCTR1 is produced by human M2 macrophages and enhances resolution of infectious inflammation. Am. J. Pathol. 186, 962–973 (2016).
Said, S. I. & Rosenberg, R. N. Vasoactive intestinal polypeptide: abundant immunoreactivity in neural cell lines and normal nervous tissue. Science 192, 907–908 (1976).
Aton, S. J., Colwell, C. S., Harmar, A. J., Waschek, J. & Herzog, E. D. Vasoactive intestinal polypeptide mediates circadian rhythmicity and synchrony in mammalian clock neurons. Nat. Neurosci. 8, 476–483 (2005).
Ibrahim, H., Barrow, P. & Foster, N. Transcriptional modulation by VIP: a rational target against inflammatory disease. Clin. Epigenetics 2, 213–222 (2011).
Delgado, M. & Ganea, D. Vasoactive intestinal peptide: a neuropeptide with pleiotropic immune functions. Amino Acids 45, 25–39 (2013).
Nussbaum, J. C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature 502, 245–248 (2013).
Talbot, S. et al. Silencing nociceptor neurons reduces allergic airway inflammation. Neuron 87, 341–354 (2015).
Hoegger, M. J. et al. Impaired mucus detachment disrupts mucociliary transport in a piglet model of cystic fibrosis. Science 345, 818–822 (2014).
Sui, P. et al. Pulmonary neuroendocrine cells amplify allergic asthma responses. Science 360, eaan8546 (2018).
Xiang, Y. Y. et al. A GABAergic system in airway epithelium is essential for mucus overproduction in asthma. Nat. Med. 13, 862–867 (2007).
Ibiza, S. et al. Glial-cell-derived neuroregulators control type 3 innate lymphoid cells and gut defence. Nature 535, 440–443 (2016).
Ruzek, M. C., Pearce, B. D., Miller, A. H. & Biron, C. A. Endogenous glucocorticoids protect against cytokine-mediated lethality during viral infection. J. Immunol. 162, 3527–3533 (1999).
Bereshchenko, O., Bruscoli, S. & Riccardi, C. Glucocorticoids, sex hormones, and immunity. Front Immunol. 9, 1332 (2018).
Quatrini, L. et al. Endogenous glucocorticoids control host resistance to viral infection through the tissue-specific regulation of PD-1 expression on NK cells. Nat. Immunol. 19, 954–962 (2018).
Quatrini, L. et al. Host resistance to endotoxic shock requires the neuroendocrine regulation of group 1 innate lymphoid cells. J. Exp. Med. 214, 3531–3541 (2017).
Kovats, S. Estrogen receptors regulate innate immune cells and signaling pathways. Cell Immunol. 294, 63–69 (2015).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Sorachi, K., Kumagai, S., Sugita, M., Yodoi, J. & Imura, H. Enhancing effect of 17 beta-estradiol on human NK cell activity. Immunol. Lett. 36, 31–35 (1993).
Souza, S. S. et al. Influence of menstrual cycle on NK activity. J. Reprod. Immunol. 50, 151–159 (2001).
Hao, S., Li, P., Zhao, J., Hu, Y. & Hou, Y. 17beta-estradiol suppresses cytotoxicity and proliferative capacity of murine splenic NK1.1+ cells. Cell. Mol. Immunol. 5, 357–364 (2008).
Bartemes, K., Chen, C. C., Iijima, K., Drake, L. & Kita, H. IL-33-responsive group 2 innate lymphoid cells are regulated by female sex hormones in the uterus. J. Immunol. 200, 229–236 (2018).
Laffont, S. et al. Androgen signaling negatively controls group 2 innate lymphoid cells. J. Exp. Med. 214, 1581–1592 (2017).
Townsend, E. A., Miller, V. M. & Prakash, Y. S. Sex differences and sex steroids in lung health and disease. Endocr. Rev. 33, 1–47 (2012).
Cephus, J. Y. et al. Testosterone attenuates group 2 innate lymphoid cell-mediated airway inflammation. Cell Rep. 21, 2487–2499 (2017).
Czarnewski, P., Das, S., Parigi, S. M. & Villablanca, E. J. Retinoic acid and its role in modulating intestinal innate immunity. Nutrients 9, 68 (2017).
Mora, J. R., Iwata, M. & von Andrian, U. H. Vitamin effects on the immune system: vitamins A and D take centre stage. Nat. Rev. Immunol. 8, 685–698 (2008).
Dimitrov, V. & White, J. H. Vitamin D signaling in intestinal innate immunity and homeostasis. Mol. Cell Endocrinol. 453, 68–78 (2017).
van de Pavert, S. A. et al. Maternal retinoids control type 3 innate lymphoid cells and set the offspring immunity. Nature 508, 123–127 (2014).
Mielke, L. A. et al. Retinoic acid expression associates with enhanced IL-22 production by gammadelta T cells and innate lymphoid cells and attenuation of intestinal inflammation. J. Exp. Med. 210, 1117–1124 (2013).
Spencer, S. P. et al. Adaptation of innate lymphoid cells to a micronutrient deficiency promotes type 2 barrier immunity. Science 343, 432–437 (2014).
Kim, M. H., Taparowsky, E. J. & Kim, C. H. Retinoic acid differentially regulates the migration of innate lymphoid cell subsets to the gut. Immunity 43, 107–119 (2015).
Bernink, J. H. et al. Interleukin-12 and -23 control plasticity of CD127(+) group 1 and group 3 innate lymphoid cells in the intestinal lamina propria. Immunity 43, 146–160 (2015).
Chen, J., Waddell, A., Lin, Y. D. & Cantorna, M. T. Dysbiosis caused by vitamin D receptor deficiency confers colonization resistance to Citrobacter rodentium through modulation of innate lymphoid cells. Mucosal Immunol. 8, 618–626 (2015).
Konya, V. et al. Vitamin D downregulates the IL-23 receptor pathway in human mucosal group 3 innate lymphoid cells. J. Allergy Clin. Immunol. 141, 279–292 (2018).
Ruiter, B., Patil, S. U. & Shreffler, W. G. Vitamins A and D have antagonistic effects on expression of effector cytokines and gut-homing integrin in human innate lymphoid cells. Clin. Exp. Allergy 45, 1214–1225 (2015).
Denison, M. S. & Nagy, S. R. Activation of the aryl hydrocarbon receptor by structurally diverse exogenous and endogenous chemicals. Annu. Rev. Pharmacol. Toxicol. 43, 309–334 (2003).
Stockinger, B., Di Meglio, P., Gialitakis, M. & Duarte, J. H. The aryl hydrocarbon receptor: multitasking in the immune system. Annu Rev. Immunol. 32, 403–432 (2014).
Fernandez-Salguero, P. et al. Immune system impairment and hepatic fibrosis in mice lacking the dioxin-binding Ah receptor. Science 268, 722–726 (1995).
Lee, J. S. et al. AHR drives the development of gut ILC22 cells and postnatal lymphoid tissues via pathways dependent on and independent of Notch. Nat. Immunol. 13, 144–151 (2011).
Kiss, E. A. et al. Natural aryl hydrocarbon receptor ligands control organogenesis of intestinal lymphoid follicles. Science 334, 1561–1565 (2011).
Qiu, J. et al. The aryl hydrocarbon receptor regulates gut immunity through modulation of innate lymphoid cells. Immunity 36, 92–104 (2012).
Schiering, C. et al. Feedback control of AHR signalling regulates intestinal immunity. Nature 542, 242–245 (2017).
Hughes, T. et al. The transcription factor AHR prevents the differentiation of a stage 3 innate lymphoid cell subset to natural killer cells. Cell Rep. 8, 150–162 (2014).
Conlon, M. A. & Bird, A. R. The impact of diet and lifestyle on gut microbiota and human health. Nutrients 7, 17–44 (2014).
Zelante, T. et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity 39, 372–385 (2013).
Sanos, S. L. et al. RORgammat and commensal microflora are required for the differentiation of mucosal interleukin 22-producing NKp46+ cells. Nat. Immunol. 10, 83–91 (2009).
Gomez de Aguero, M. et al. The maternal microbiota drives early postnatal innate immune development. Science 351, 1296–1302 (2016).
Shimizu, T. Lipid mediators in health and disease: enzymes and receptors as therapeutic targets for the regulation of immunity and inflammation. Annu Rev. Pharmacol. Toxicol. 49, 123–150 (2009).
Doherty, T. A. et al. Lung type 2 innate lymphoid cells express cysteinyl leukotriene receptor 1, which regulates TH2 cytokine production. J. Allergy Clin. Immunol. 132, 205–213 (2013).
von Moltke, J. et al. Leukotrienes provide an NFAT-dependent signal that synergizes with IL-33 to activate ILC2s. J. Exp. Med. 214, 27–37 (2017).
Lund, S. J. et al. Leukotriene C4 potentiates IL-33-induced group 2 innate lymphoid cell activation and lung inflammation. J. Immunol. 199, 1096–1104 (2017).
Salimi, M. et al. Cysteinyl leukotriene E4 activates human group 2 innate lymphoid cells and enhances the effect of prostaglandin D2 and epithelial cytokines. J. Allergy Clin. Immunol. 140, 1090–1100 e1011 (2017).
Yokomizo, T., Nakamura, M. & Shimizu, T. Leukotriene receptors as potential therapeutic targets. J. Clin. Invest. 128, 2691–2701 (2018).
Kim, A. S. & Doherty, T. A. New and emerging therapies for asthma. Ann. Allergy Asthma Immunol. 116, 14–17 (2016).
Zhou, Y. et al. Prostaglandin E2 inhibits group 2 innate lymphoid cell activation and allergic airway inflammation through E-prostanoid 4-cyclic adenosine monophosphate signaling. Front Immunol. 9, 501 (2018).
Zhou, W. et al. Prostaglandin I2 signaling and inhibition of group 2 innate lymphoid cell responses. Am. J. Respir. Crit. Care Med. 193, 31–42 (2016).
Wojno, E. D. et al. The prostaglandin D(2) receptor CRTH2 regulates accumulation of group 2 innate lymphoid cells in the inflamed lung. Mucosal Immunol. 8, 1313–1323 (2015).
Chang, J. E., Doherty, T. A., Baum, R. & Broide, D. Prostaglandin D2 regulates human type 2 innate lymphoid cell chemotaxis. J. Allergy Clin. Immunol. 133, 899–901 e893 (2014).
Xue, L. et al. Prostaglandin D2 activates group 2 innate lymphoid cells through chemoattractant receptor-homologous molecule expressed on TH2 cells. J. Allergy Clin. Immunol. 133, 1184–1194 (2014).
Nagata, K. et al. Selective expression of a novel surface molecule by human Th2 cells in vivo. J. Immunol. 162, 1278–1286 (1999).
Mjosberg, J. M. et al. Human IL-25- and IL-33-responsive type 2 innate lymphoid cells are defined by expression of CRTH2 and CD161. Nat. Immunol. 12, 1055–1062 (2011).
Boyce, J. A. Mast cells and eicosanoid mediators: a system of reciprocal paracrine and autocrine regulation. Immunol. Rev. 217, 168–185 (2007).
Fajt, M. L. et al. Prostaglandin D(2) pathway upregulation: relation to asthma severity, control, and TH2 inflammation. J. Allergy Clin. Immunol. 131, 1504–1512 (2013).
Barnig, C. et al. Lipoxin A4 regulates natural killer cell and type 2 innate lymphoid cell activation in asthma. Sci. Transl. Med. 5, 174ra126 (2013).
Maric, J. et al. Prostaglandin E2 suppresses human group 2 innate lymphoid cell function. J. Allergy Clin. Immunol. 141, 1761–1773 e1766 (2018).
Duffin, R. et al. Prostaglandin E(2) constrains systemic inflammation through an innate lymphoid cell-IL-22 axis. Science 351, 1333–1338 (2016).
Claar, D., Hartert, T. V. & Peebles, R. S. Jr. The role of prostaglandins in allergic lung inflammation and asthma. Expert Rev. Respir. Med. 9, 55–72 (2015).
Rumzhum, N. N. & Ammit, A. J. Cyclooxygenase 2: its regulation, role and impact in airway inflammation. Clin. Exp. Allergy 46, 397–410 (2016).
Hara, S. Prostaglandin terminal synthases as novel therapeutic targets. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 93, 703–723 (2017).
Acknowledgements
The authors apologize to all investigators whose works were not cited in this article due to space limitations. We are grateful to Kylie Luong for helpful comments and preparation of the manuscript. The figure was drawn using Servier Medical Art (https://smart.servier.com) and modified by the authors under the terms of the Creative Commons Attribution 3.0 Unported License. C.S. was supported by grants and fellowships from the National Health and Medical Research Council (NHMRC) of Australia (APP1165443) and the Australian Research Career Development Fellowship (APP1123000). N.J. was supported by Grant 1163990 awarded through the 2018 Priority-driven Collaborative Cancer Research Scheme and cofunded by Cancer Australia and Cure Cancer. This work was made possible through the Victorian State Government Operational Infrastructure Support and the Australian Government NHMRC IRIIS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Seillet, C., Jacquelot, N. Sensing of physiological regulators by innate lymphoid cells. Cell Mol Immunol 16, 442–451 (2019). https://doi.org/10.1038/s41423-019-0217-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-019-0217-1
Keywords
This article is cited by
-
AHR in the intestinal microenvironment: safeguarding barrier function
Nature Reviews Gastroenterology & Hepatology (2021)