Abstract
Current treatment strategies for cancer, especially advanced cancer, are limited and unsatisfactory. One of the most substantial advances in cancer therapy, in the last decades, was the discovery of a new layer of immunotherapy approach, immune checkpoint inhibitors (ICIs), which can specifically activate immune cells by targeting immune checkpoints. Immune checkpoints are a type of immunosuppressive molecules expressed on immune cells, which can regulate the degree of immune activation and avoid autoimmune responses. ICIs, such as anti-PD-1/PD-L1 drugs, has shown inspiring efficacy and broad applicability across various cancers. Unfortunately, not all cancer patients benefit remarkably from ICIs, and the overall response rates to ICIs remain relatively low for most cancer types. Moreover, the primary and acquired resistance to ICIs pose serious challenges to the clinical application of cancer immunotherapy. Thus, a deeper understanding of the molecular biological properties and regulatory mechanisms of immune checkpoints is urgently needed to improve clinical options for current therapies. Recently, circular RNAs (circRNAs) have attracted increasing attention, not only due to their involvement in various aspects of cancer hallmarks, but also for their impact on immune checkpoints in shaping the tumor immune microenvironment. In this review, we systematically summarize the current status of immune checkpoints in cancer and the existing regulatory roles of circRNAs on immune checkpoints. Meanwhile, we also aim to settle the issue in an evidence-oriented manner that circRNAs involved in cancer hallmarks regulate the effects and resistance of ICIs by targeting immune checkpoints.
Similar content being viewed by others
Facts
-
ICIs has achieved prominent efficacy in many types of cancer and revolutionized the status of cancer immunotherapy.
-
Primary and acquired resistance limit the clinical application of ICIs in cancer immunotherapy.
-
CircRNAs as emerging modulators of various cancer hallmarks are associated with the regulation of ICIs by targeting immune checkpoints.
Questions
-
Can circRNAs directly serve as immune checkpoints to regulate immune responses against cancer cells?
-
Can circRNAs involved in cancer hallmarks such as proliferation and cell death mediate ICIs resistance?
-
Is resistance to ICIs mediated by circRNAs in tumor cells or immune cells, or a combination of both?
-
Can synthetic circRNAs regulate the cascades involved in immune checkpoint pathways and serve as new potential cancer immunotherapy regimens?
Introduction
Cancer immunotherapy with ICIs by inhibition of negative immune regulation to enhance immune activity against cancer cells, which won the 2018 Nobel Prize in Physiology or Medicine, has revolutionized the treatment dilemma of various human malignancies [1,2,3,4]. The first immune checkpoint inhibitor approved by FDA named lpilimumab that targets cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) to potentiate the T cell-mediated antitumor immune response, have remarkably improved the overall survival of metastatic melanoma patients [5,6,7,8]. Subsequently, the FDA approved programmed cell death protein 1 (PD-1) inhibitor nivolumab for the treatment of advanced melanoma due to its favorable overall survival, durable tumor remission after drug discontinuation, and acceptable long-term safety [9, 10]. Since then, the gate of immunotherapy has been thoroughly opened, and more monoclonal antibody drugs blocking PD-1 or its ligand PD-L1 have been approved in succession by the FDA alone or in combination with other drugs to treat numerous malignancies, such as melanoma, lung cancer, lymphoma, esophageal carcinoma, gastric cancer, and hepatocellular carcinoma [11,12,13]. Despite their prominent clinical efficacy, currently approved ICIs only benefit a subset of patients, even with response biomarkers like PD-L1 expression [14, 15]. The application of single-agent ICIs often leads to primary resistance, and some responsive patients develop acquired resistance to ICIs [16]. Additionally, immune-related adverse events (irAEs) owing to ICIs can induce excessive immune system activation or multiple organ disorders, and hyperprogression caused by ICIs pose the serious clinical challenges to immunotherapy [17,18,19,20,21]. Thus, identifying the molecular biological properties and regulatory mechanisms of immune checkpoints is crucial for optimizing therapeutic options and controlling adverse effects.
Immune checkpoints, a series of molecules that are expressed on immune cells and can tune up the degree of immune activation, act as gatekeepers during the body’s immune response to prevent overactivation of the immune system [22,23,24]. Over the past few decades, numerous immune checkpoint molecules have been identified, including but not limited to PD-1/PD-L1, CTLA-4, lymphocyte-activation gene 3 (LAG-3), T-cell immunoglobulin and mucin-domain containing-3 (TIM-3) [25]. Although the number of newly discovered immune checkpoints is considerable, the ICIs approved by FDA remain limited due to the complexity of their mechanisms. Recently, an increasing number of studies in molecular oncology have revealed the complicated regulatory mechanisms of immune checkpoint expression. Non-coding RNAs (ncRNAs), an abundant component of the human transcriptome involved in all cancer hallmarks, are highly correlated with the expression regulation of immune checkpoints [26,27,28]. One of the most unexpected discoveries in ncRNA field, with the advent of high-throughput sequencing techniques in recent years, was the identification of a new class of stable loop-structured molecules with both coding and non-coding properties, circRNAs [29]. They represent a novel category of transcripts that exhibit tissue- and spatio-temporal specific expression patterns, widely involved in multiple physiological and pathological processes [30]. Growing evidence has confirmed that circRNAs can serve as star molecules and play vital roles in cell signaling pathways regulation in human cancers [31,32,33,34]. During cancer progression, the body’s systemic immune landscape suffers from alterations in many aspects [35]. Meanwhile, the biological functions in anti-tumor immunity and potential applications in cancer immunotherapy have been gradually discovered [36,37,38]. However, circRNAs-mediated regulation of immune checkpoint molecules has not been well evaluated. Accordingly, this review will focus on the current immune checkpoints commonly applied in cancer and the molecular mechanisms by which circRNAs interfere with ICIs therapeutic strategies by regulating immune checkpoint pathways.
Status quo of frequent immune checkpoints and ICIs in cancer
PD-1/PD-L1
PD-1/PD-L1 is the most well-characterized immune checkpoint pathway that has been widely employed in clinical cancer immunotherapy. PD-1 (also known as CD279), a member of CD28 superfamily, belongs to type I transmembrane glycoprotein with a single extracellular immunoglobulin variable domain, transmembrane region, and cytoplasmic domain containing immunoreceptor tyrosine-based inhibitory motif (ITIM) and immunoreceptor tyrosine-based switch motif (ITSM) [39]. It has been reported that PD-1 is a coinhibitory receptor, mainly expressed on activated T cells, but also on B cells, natural killer (NK) cells, myeloid cells, monocytes, neutrophils, and dendritic cells (DCs) [25, 40, 41]. PD-L1 (also known as CD274 or B7-H1), the dominant ligand of PD-1, belongs to the B7 family and can be expressed not only by immune cells, such as T cells, B cells, macrophages, DCs, and mast cells, but also by normal tissue cells and aberrant tumor cells [42, 43]. When PD-1 binds to PD-L1 on the surface of immune effector cells such as T cells, its cytoplasmic ITIM and ITSM can be phosphorylated, and then recruit Src homology 2 domain-containing tyrosine phosphatase 2 (SHP2) to further suppress T cell receptor (TCR) signaling transduction [44]. PD-1/PD-L1 pathway contributes to tumor immune escape, which has been identified as a main mechanism of adaptive immune resistance (AIR) to enable tumors resistant to immune response [45,46,47].
Monoclonal antibodies (mAbs) blocking the PD-1/PD-L1 pathway have been widely applied for clinical immunotherapy to fight against a fraction of advanced cancer (Fig. 1). The approved or ongoing clinical trials mAbs that regulate PD-1/PD-L1 pathway were listed in Table 1. Nivolumab was the first mAb against PD-1 approved by FDA for the treatment of unresectable or metastatic melanoma based on its remarkable survival benefits in clinical trials [48]. Subsequently, the second PD-1 antibody, pembrolizumab, was designated for the treatment of advanced melanoma due to its manageable irAEs and significant improvements in prognosis and fewer treatment-related adverse events than chemotherapy [49]. This therapeutic effect was also found in patients with non-small-cell lung cancer (NSCLC), locally advanced or metastatic esophageal cancer and in patients with advanced or metastatic gastric or gastroesophageal junction cancer with microsatellite instability-high (MSI-H) status [50,51,52]. In addition, PD-1 antibodies include Cemiplimab, Dostarlimab and Toripalimab, whose therapeutic effects have been confirmed in various solid tumors [53,54,55]. Except for the encouraging performance of PD-1 antibodies, the PD-L1 antibodies have also shown obvious clinical benefits for multilple malignancies. For instance, avelumab, atezolizumab, and durvalumab have been proven beneficial activity and safety for treatment of locally advanced or metastatic urothelial cancer [56,57,58]. In the context of NSCLC, envafolimab, durvalumab, and sugemalimab were confirmed to achieve obvious improvements in prognosis with limited drug toxicity [59,60,61]. Nevertheless, it has also been demonstrated that not all patients with solid malignancies can benefit from PD-1/PD-L1 antibodies, and some patients experience hyperprogression, which is a remaining challenge in immunotherapy [62,63,64]. Therefore, the current research direction of PD-1/PD-L1 is to select individualized treatment plans, find potential benefit groups, combine multiple targeted ICIs, and timely adjust appropriate immunotherapy after immune resistance.
When PD-1 binds to PD-L1 on the surface of immune effector cells such as T cells, T cell receptor (TCR) signaling transduction was suppressed. Monoclonal antibodies (mAbs) blocking the PD-1/PD-L1 pathway have been widely applied for clinical immunotherapy to fight against a fraction of advanced cancer.
CTLA-4
CTLA-4 (also known as CD152) is another transmembrane receptor that shares the B7 ligand with co-stimulatory receptor CD28. CTLA-4 is mainly expressed on the surface of immunosuppressive T regulatory cells (Tregs) and only on the activated conventional T lymphocytes [65]. In view of the homology of CTLA-4 and CD28, they both bind to the same B7 ligands CD80 (B7-1) and CD86 (B7-2), but CTLA-4 has a higher affinity than CD28 [66]. CTLA-4 mainly plays a role in the inhibitory modulation of T cell activation and induction of T cell immune tolerance. It has been verified that CTLA-4 can impede T cell proliferation and differentiation, induce cell cycle arrest, and reduce IL-2 production to exert the inhibitory function in T cell modulation [67]. CTLA-4 has been reported to mediate negative regulation of T cell responses through several ways, such as attenuating the T cell receptor (TCR) and CD28 signaling by competively binding to B7 with CD28 or recruiting phosphatases to the intracellular domain of CTLA-4, and another mechanism is the induction of Treg development and function. Additionally, CTLA-4 was found to decrease CD80 and CD86 expression on antigen presenting cells (APCs) or remove them from APCs by transendocytosis to hinder the participation of CD28 in T cell activation process [68, 69]. Therefore, the application of monoclonal antibody to block CTLA-4 can relieve its inhibitory effects on T cells, reactivate T cell proliferation and differentiation into cytotoxic T lymphocytes (CTLs), thereby exerting anti-tumor immune effects (Fig. 2).
Currently, the mainstream monoclonal antibodies targeting CTLA-4 are ipilimumab and tremelmumab (Table 1). Ipilimumab is a humanized IgG1 monoclonal antibody that binds to CTLA-4 in the B7 interaction domain, mediating steric hindration to block the binding of CTLA-4 to B7, and is the first CTLA-4 antibody drug used in advanced melanoma. A number of clinical studies have shown that Ipilimumab has a high rate of disease control and benifits for improving prognosis [70,71,72]. Moreover, most existing studies have focused on the combination therapy of ICIs. In NSCLC patients, compared with neoadjuvant nivolumab, the combination therapy of nivolumab and ipilimumab can significantly improve the pathological complete response rates, tumor immune infiltrates and immune memory effects [73]. Another multicenter, randomized Phase III clinical trial (CheckMate 9LA) also confirmed the feasibility of this treatment [74, 75]. Tremelimumab is another humanized IgG2 monoclonal antibody against CTLA-4, which binds to CTLA-4 and blocks its interaction with ligands CD80 and CD86, thereby enhancing T cell response to tumor cells [76]. In October 2022, the FDA approved tremelimumab in combination with durvalumab for the treatment of adult patients with unresectable hepatocellular carcinoma (uHCC) based on data from HIMALAYA’s Phase III clinical trial [77,78,79]. In November 2022, based on the Phase III POSEIDON clinical trial, tremelimumab, durvalumab, and platinum-containing chemotherapy combinations are also approved by the FDA for the treatment of metastatic NSCLC without EGFR-sensitive mutations and ALK rearrangements [77, 80]. However, the overall rate of adverse events in patients receiving ICIs combination therapy is higher than in the monotherapy group, and not every specific cancer patient will benefit from it [81]. Therefore, future studies may be urgently needed to improve the clinical outcomes of such combination therapies and reduce the incidence of adverse events.
LAG-3
LAG-3 (also known as CD223), the third immune checkpoint applied in clinical practice after CTLA-4 and PD-1/PD-L1, is an inhibitory transmembrane receptor found on the surface of effector T cells, regulatory T cells, natural killer cells, activated B cells, and plasmacytoid dendritic cells [82]. The LAG-3 gene includes eight exons and its encoded protein can be divided into extracellular, transmembrane, and intracellular domains, which is closely related to CD4 [83]. The typical ligand of LAG-3 is MHC Class II (MHC-II), which interacts stably with LAG-3 through the D1 domain. It was initially thought that high-affinity LAG-3/MHC-II binding competes with CD4/MHC-II interactions to inhibit the proliferation, activation, cytotoxicity and cytokine production of CD4+ and CD8 + T lymphocytes [84, 85]. However, whether the LAG-3/MHC-II interaction alone is responsible for LAG-3 immunosuppressive signaling remains controversial, given the recent discovery of other ligands, including galactin-3 (Gal-3), fibrinin-as-protein 1 (FGL1), and LSECtin. Gal-3 is expressed in a variety of cancer cells and activated T cells, and it can interact with LAG-3 to inhibit cytotoxicity of CD8 + T lymphocytes [84,85,86]. Another newly discovered LAG-3 functional ligand, FGL1, is a member of the liver-secreted fibrinogen family of proteins and is highly expressed in a variety of tumor cells, such as melanoma, lung cancer, and colorectal cancer cells [87]. The binding of FGL1 and LAG-3 inhibits antigen-specific T cell immune response, and blocking the interaction between FGL1 and LAG-3 can reactivate the anti-tumor immune ability of T cells [87]. Interestingly, upregulation of FGL1 expression may also be associated with poor prognosis and anti-PD-1 resistance [87]. Thus, blocking LAG-3 with monoclonal antibodies mitigated the T-cell suppressive immune response triggered by the interaction between LAG-3 and its ligand (Fig. 3).
The high-affinity LAG-3Ig/MHC-II binding competes with CD4/MHC-II interaction to mediate the suppressed T cell proliferation and inhibitory immune response. The second ligand of LAG-3 is Gal-3, which has been found to be expressed in multiple cancer cells and activated T cells. Gal-3 can interact with LAG-3 and this binding is essential for the inhibitory cytotoxicity of CD8 + T lymphocytes. Other newly identified LAG-3 functional ligands include FGL1 and LSECtin. The blockade of LAG-3 with mAbs can relieve the T cell inhibitory immune response triggered by the interaction between LAG-3 and its ligands.
The efficacy of LAG-3 mAbs is currently being studied in a variety of tumors. Relatlimab is a first-of-its-kind human IgG4 anti-LAG-3 antibody that binds to LAG-3 and restores T cell function [88]. Meanwhile, studies have found that LAG-3 and PD-1 were widely co-expressed on CD4+ and CD8 + T cells, especially on tumor-infiltrating T cells [89, 90]. LAG-3 in combination with the typical inhibitory immune checkpoint PD-1/PD-L1 can co-mediate immune homeostasis, eliminate autoimmune diseases, and enhance tumor-induced tolerance [90, 91]. The FDA has approved Relatlimab in combination with PD-1 mAb Nivolumab for the treatment of unresectable or metastatic melanoma [92, 93] (Table 1). In addition, LAG-3 mAb and PD-1 mAb have also been shown to have significant synergistic effects in colorectal cancer [94, 95]. In spite of the potential application value of LAG-3 in cancer immunotherapy, its functional mechanisms remains obscure. It is important to have a deep understanding of the action mechanism of LAG-3 for identifying its unknown effects and developing better therapeutic methods to block or replicate LAG-3 function, as well as searching for suitable biomarkers and selecting suitable patients to maximize the effect of LAG-3 mAb.
TIM-3
TIM-3, also known as hepatitis A virus cell receptor 2 (HAVCR2) or CD366, is another promising inhibitory receptor among numerous emerging immune checkpoints and has achieved initial anti-tumor effects in clinical trials of its monotherapy or combination with anti PD-1/PD-L1 drugs [96]. TIM-3 as an important member of the TIM family protein, it contains a C-terminal cytoplasmic tail, a single transmembrane domain signal peptides, a mucinlike domain and an extracellular N-terminal variable immunoglobulin (IgV) domain [97]. TIM-3 has been reported to have multiple ligands, such as galectin-9 (Gal-9), carcinoembryonic antigen related cell adhesion molecule 1 (CEACAM1), phosphatidylserine (PtdSer) and high mobility group box 1 (HMGB1), and each of them binds to different regions of the extracellular IgV domain [98]. The ligand Gal-9 binding to TIM-3 was found to induce apoptosis of Th1 effector cells and reduce IFN-γ production [99]. The ligand CEACAM1 binding to TIM-3 plays a crucial role in regulating cell-mediated autoimmunity and anti-tumor immunity [100]. The ligand PtdSer binding to TIM-3 promotes clearance of apoptotic vesicles to maintain immune tolerance [101]. TIM-3 is preferentially expressed on CD4 + T cell subsets (Th1/Th17), CD8 + T cell subsets (Tc1) and regulatory T cells (Tregs) to generate inhibitory signals, resulting in inhibition of T cell activation and proliferation, reducing the production of cytokines that positively regulate immune responses, and playing a negative regulatory role in the process of anti-tumor immunity [102]. Additionally, it is demonstrated that TIM-3 is also expressed on the surface of DCs, which can mediate the phagocytosis of apoptotic cells and the cross-presentation of antigens [103]. Researchers found that TIM-3 on DCs but not on CD4+ or CD8 + T cells has singular function to restrain anti-tumor immunity through impeding the maintenance of effector T cells and stem-like CD8 + T cells, resulting in inhibition of protective immune effects and reduction of NLRP3 inflammasome activation [104]. Except for DCs, TIM-3 was also detected on the surface of other innate immune cells, such as NK cells, monocytes and macrophages [105]. When TIM-3 is not bound to the ligand, it interacts with HLA-B associated transcription factor 3 (BAT3) and maintains T cell activation through Tyrosine kinase LCK recruitment. Once TIM-3 binds to the ligand, BAT3 is released from TIM-3 and Fyn binds to it, allowing TIM-3 to exert its inhibitory function [106, 107]. Most antibodies targeting TIM-3 hinder the binding of ligands to TIM-3, thereby maintaining the binding state between TIM-3 and BAT3 (Fig. 4). However, the affinity and function of TIM-3 binding to its ligand have not yet been fully elucidated.
When TIM-3 is not bound to the ligand, it interacts with BAT3 and maintains T cell activation through Tyrosine kinase LCK recruitment. Once TIM-3 binds to the ligand, BAT3 is released from TIM-3 and Fyn binds to it, allowing TIM-3 to exert its inhibitory function. Most mAbs targeting TIM-3 hinder the binding of ligands to TIM-3, thereby maintaining the binding state between TIM-3 and BAT3.
TIM-3 can serve as a biomarker for T cell exhaustion, and targeting TIM-3 with monoclonal antibodies can restore T cell vitality, allowing it to fight against tumor cells [108] (Table 1). Sabatolimab (MBG453) is a humanized representative anti-TIM-3 IgG4 antibody developed by Novartis that directly targets the TIM-3 receptor with immuno-myeloid activity to fight against myeloid cell neoplasms [109]. As a single agent, TIM-3 antibody has no substantial clinical benefits, but when combined with other checkpoint inhibitors, such as PD-1 antibody, it can elicit a stronger immune response. A prospective clinical study involving 219 cancer patients showed that MBG453 in combination with PD-1 mAb spartalizumab presented beneficial clinical activity and was also well tolerated with no dose-limiting toxicities observed [110]. Researchers also found that T cells co-expressing TIM-3/PD-1 showed more severe exhausted phenotype, deprived of proliferative viability and unable to produce IL-2, TNF, and IFN-γ. The study further confirmed that combined targeting TIM-3 and PD-1 showed stronger effects to restore anti-tumor immunne response and restrict tumor growth than blocking a single target alone [111]. Cobolimab (TSR-022) is another selective TIM-3 targeting monoclonal antibody developed by Tesaro that can activate immune cell function. When used in combination with anti PD-1 drugs such as dostarlimab, it can elicit strong anti-tumor activity without severe adverse events [96]. Currently, a phase 2 clinical trial (NCT03680508) is addressing the safety and efficacy of cobolimab in combination with dostarlimab in the context of liver cancer [96]. Another phase 2/3 clinical trial (NCT04655976) is evaluating its combination application with docetaxel and dostarlimab in NSCLC patients with prior progressive disease to anti-PD-1/PD-L1 drugs [96]. Although TIM-3 is currently one of the most studied immune checkpoints, there are no commercially available drugs nationally and internationally. This may be due to the complexity of the mechanism and the unclear beneficiaries. Therefore, the study of the combined application of anti TIM-3 and anti PD-1/PD-L1 drugs may ameliorate the resistance of PD-1/PD-L1, save T cell failure, and improve the prognosis of patients.
Other immune checkpoints
Except for the above major well-known immune checkpoints, there are several other emerging molecules on the investigation for negative immune regulation with immunotherapy targeting potential. For example, T cell immunoglobulin and ITIM domain (TIGIT), also known as WUCAM, Vstm3, VSIG9, is an inhibitory immune checkpoint molecule belonging to the poliovirus receptor (PVR)/Nectin family and is expressed on CD4 + T cells, CD8 + T cells, Treg cells, and NK cells [112, 113]. TIGIT can bind to its ligands CD155 and CD112 to inhibit innate and adaptive immune responses via different mechanisms [114]. There are currently various mAbs targeting TIGIT on the investigation either as a monotherapy or combined therapy with PD-1/PD-L1 antibodies. Combination therapy of TIGIT mAb tiragolumab and PD-L1 mAb atezolizumab has been reported to enhance overall response rate in the context of metastatic NSCLC with PD-L1 positive expression [115].
V domain Ig inhibitory factor activated by T cells (VISTA, also known as VSIR, Gi24, Dies-1, PD-1H, B7-H5, SISP1, or DD1α), is another inhibitory immune checkpoint that is predominantly expressed in myeloid cells (monocytes, macrophages, neutrophils and DCs), but also found in T lymphocytes and NK cells [116, 117]. VSIG-3 (also known as BTIgSF, IgSF11 or Igsf13) is a VISTA ligand and this interaction inhibits T cell responses [117]. The blockade of VISTA enhances T cell activation and the upregulates the production of many inflammatory cytokines, such as IFN-γ, IL-2, IL-17, CCL-3, CCL-5, and CXCL-11 [118]. Furthermore, VISTA has the potential to reinforce the function of myeloid-derived immune suppressor cells, maintaining the survival of Tregs, restraining the antigen presentation ability of APCs [119]. However, the exact regulatory functions and mechanisms of VISTA in tumor immunity remain to be fully elucidated. Other suppressive immune checkpoints that are being explored as promising immunotherapeutic targets include signal regulatory protein α (SIRPα), B and T lymphocyte attenuator (BTLA), leukocyte immunoglobulin-like receptor subfamily B member 4 (LILRB4), and sialic acid-binding immunoglobulin-like lectin 7 (Siglec-7).
Apart from these inhibitory immune checkpoints, some stimulant checkpoint molecules with the potential of positive immunoregulation have also been considered for anti-tumor immunotherapy. CD28 and immune co-stimulator (ICOS) belonging to B7-CD28 superfamily members are critical activating receptors expressed on T cells and participate in the survival, expansion, and function of effector T cells and Tregs [120,121,122]. The ICOS agonist mAb vopratelimab has been reported to present a beneficial safety profile alone and in combination with nivolumab, but the clinical activity was limited and only effective in a portion of patients with specific pharmacodynamic biomarkers of vopratelimab [123]. Additionally, checkpoint molecules belonging to the tumor necrosis factor receptor superfamily (TNFR-SF), such as glucocorticoid-induced TNFR-related gene (GITR/TNFRSF18/CD357), TNFRSF4 (OX40/CD134), TNFRSF5 (CD40), TNFRSF7 (CD27), and TNFRSF9 (CD137/4-1BB), have also been defined as stimulatory immune checkpoints and under evaluation as potential targets for intervention in the field of cancer immunotherapy [124]. We have summarized the common stimulatory and inhibitory immune checkpoint receptors with positive and negative regulatory effects on anti-tumor immunity in Fig. 5. These newly discovered immune checkpoints provide ideas to address primary and acquired resistance to PD-1/PD-L1 therapy, and may even replace existing PD-1/PD-L1 therapy in the future. It is also possible to develop new ICIs drugs by focusing on the immune checkpoints of the remaining cells in the immune microenvironment, including NK cells, DCs and etc, thereby extending the survival time of patients and improving their prognosis.
CircRNAs are involved in the hallmarks of cancer
For a long time, circRNAs have been mistakenly thought to be by-products of mRNA splicing, with no specific function. Until 2012, this dark matter was unexpectedly found to be ubiquitously expressed in eukaryotic genomes, which revealed fundamental issues respecting the promising biological function of circRNAs [125]. A deep sequencing study of 2000+ clinical tissue samples and cell lines from more than 10 cancer types revealed that circRNAs with stable structures may serve as the cancer biomarkers in blood or urine [126]. As research on circRNAs and cancer hallmarks in tumors has grown significantly over the past decade, it is impossible for this review to cover every reported study. For each hallmark, we will present the most representative examples. We have summarized the targets and mechanisms by which selected circRNAs are involved in the hallmarks of cancer in Table 2.
CircRNAs involvement in proliferation, migration, invasion and metastasis
Cancer cells have been found to continuously expand and grow by releasing a large number of growth-related signaling factors or expressing excessive growth signaling receptors [127]. A large number of studies have shown that circRNAs can affect the function of tumor proliferation, migration, invasion and metastasis. CircTP63 competitively bind to miR-873-3p to upregulate the expression of FOXM1, which further increase the level of downstream molecules CENPA and CENPB, thereby facilitating cell proliferation in lung squamous cell carcinoma [128]. Similarly, circPVT1 was found to promote cell proliferation in gastric cancer [129]. In addition, there are also various circRNAs that display an inhibitory role in tumor proliferation. CircVAMP3 inhibited the proliferation and migration of hepatocellular carcinoma by promoting the phase separation of CAPRIN1 protein to form stress granules in cells to prevent the translation of c-Myc mRNA [130]. CircFAM120A [131], circPCNX [132], circPINTexon2 [133], BCRC-3 [134] and circHIPK3 [135] have been shown to inhibit tumor proliferation in hepatocellular carcinoma, cervical carcinoma, glioblastoma, bladder cancer and esophageal squamous cell carcinoma, respectively.
Migration, invasion and metastasis are considered to be a complex cascade of biological events in cancer, involved in the spread and dissemination of cancer cells from the primary site to local site and foreign tissues, and tumors tend to exhibit the same trends in all three functions. In recent years, accumulated circRNAs have been reported to play important roles in cancer migration, invasion and metastasis through diverse mechanisms [34]. For instance, circPTCH1 (hsa_circ_0139402) was reported to facilitate migration, invasion and metastasis in renal cell carcinoma (RCC) through modulating miR-485-5p/MMP14 signaling pathway [136]. CircNHSL1 (hsa_circ_0006835) can act as a competitive endogenous RNA (ceRNA) to sponge miR-1306-3p to regulate SIX1/vimentin pathway, leading to enhanced migration, invasion and metastasis ability of gastric cancer cells in vitro and in vivo [137]. CircHIPK3 (hsa_circ_0000284) [138,139,140], hsa_circ_0001361 (circ0001361) [141], circLONP2 (hsa_circ_0008558) [142] and circANKS1B (hsa_circ_0007294) [143] can promote tumor migration, invasion and metastasis through the sponge mechanism. However, there are also circRNAs can inhibit tumor migration, invasion and metastasis. For example, circPTEN1 (hsa_circ_0002232) could attenuate invasion and metastasis of colorectal cancer through binding the MH2 domain of Smad4 to block its interaction with p-Smad2/3, further disrupting TGF-β/Smad signaling pathway [144].
In terms of the above mentioned effects of circRNAs on biological functions in various tumors by different mechanisms, we systematically summarized the signaling pathways involved in circRNAs regulating tumor proliferation and metastasis as follows. The PI3K/AKT signaling pathway is an intracellular signaling pathway that regulates various cellular functions, such as metabolism, proliferation, cell survival, growth, and angiogenesis (Fig. 6). In esophageal cancer, circVRK1/miR-624-3p/PTEN, circLARP4/miR-1323/PTEN, and circ_0007624/miR-224-5p/CPEB3 axis were confirmed to regulate PI3K/AKT signaling pathway participating in cell proliferation [145,146,147,148]. In gastric cancer, ciRS-7/miR-7/PTEN, circPVT1/miR-152-3p/HDGF, and circPIP5K1A/miR-671-5p/KRT80 axis were reported to involve in cell proliferation [149,150,151]. In colorectal cancer, circIL4R, circ_0008285, and circ-0001313 have been verified to modulate proliferation phenotype [152,153,154]. CircCDYL, circCDK13 and circEPHB4 were verified to involve in PI3K/AKT signal regulation to alter hepatocellular carcinoma proliferation ability [155,156,157]. CircBFAR/miR-34b-5p/MET, circEIF6/miR-557/SLC7A11, and circNFIB1/miR-486-5p/PIK3R1/VEGF-C axis have been reported to be a pancreatic cancer proliferation hallmarker [158,159,160]. In glioma, circ0014359, circHIPK3, circPIP5K1A, and circ0000215 were confirmed to regulate PI3K/AKT axis involving in cell proliferation [161,162,163,164].
The circRNAs target PI3K/AKT axis to regulate cell proliferation and metastasis in the context of various cancers, such as circVRK1/miR-624-3p/PTEN, circLARP4/miR-1323/PTEN, and circ_0007624/miR-224-5p/CPEB3 pathway targeting PI3K/AKT axis in esophageal cancer; ciRS-7/miR-7/PTEN, circPVT1/miR-152-3p/HDGF, and circPIP5K1A/miR-671-5p/KRT80 pathway targeting PI3K/AKT axis in gastric cancer; circIL4R/miR-761/TRIM29/PHLPP1, circ_0008285/miR-382-5p/PTEN, and circ-0001313/miRNA-510-5p/AKT2 pathway targeting PI3K/AKT axis in colorectal cancer; circCDYL/miR-892a and miR-328-3p, circCDK13/JAK/STAT, and circEPHB4/HIF-1ɑ pathway targeting PI3K/AKT axis in liver cancer; circBFAR/miR-34b-5p/MET, circEIF6/miR-557/SLC7A11, and circNFIB1/miR-486-5p/PIK3R1/VEGF-C pathway targeting PI3K/AKT axis in pancreatic cancer; circ0014359/miR-153, circHIPK3/miR-524-5p/KIF2A, circPIP5K1A/miR-515-5p/TCF12, and circ0000215/miR-495-3p/CXCR2 pathway targeting PI3K/AKT axis in glioma.
Additionally, the nuclear factor-kappaB (NF-κB) signaling is also considered as a proliferation related pathway in cancer, and multiple circRNAs, such as circ_0001821, hsa_circ_0021727 (circ-CD44), ciRS-7, circCYP24A1, circZFR, circLIFR, circCORO1C, circADAMTS6, circ_0002019, circGLIS2, circINTS4, circARHGEF28, circ_0001461, have been reported to regulate cell proliferation through modulation of NF-κB signaling axis in different tumors [165] (Fig. 7). In the settings of esophageal squamous cell carcinoma (ESCC), circ_0001821 acts as a miR-423-5p sponge to upregulate BTRC expression, thus inducing IKBA ubiquitination to affect NF-κB signaling and cell proliferation [166]; hsa_circ_0021727 (circ-CD44) promotes proliferation by sponging miR-23b-5p to upregulate the TAB1/NF-κB pathway [167]; ciRS-7 promotes ESCC growth and metastasis through miR-7/HOXB13 induced NF-κB pathway [168]; circCYP24A1 facilitates ESCC progression by interacting with PKM2 to regulate NF-κB signaling [169]. Under the context of hepatocellular carcinoma, circZFR promotes hepatocellular carcinoma development by inhibiting the STAT3/NF-κB pathway [170]; circLIFR promotes cell proliferation by interacting with TANK binding kinase 1 (TBK1), which is a serine/threonine kinase that regulates NF-κB pathway [171]; circCORO1C promotes hepatocellular carcinoma proliferation and metastasis via upregulating NF-κB pathway induced PD-L1 expression [172]. During glioblastoma progression, circADAMTS6 facilitates cell proliferation and tumor growth through regulation of ANXA2/NF-κB pathway in a TDP43-dependent way [173]. In gastric cancer, circ_0002019 enhances cell proliferation, migration, and invasion through TNFAIP6/NF-κB signaling [174]. In colorectal cancer, circGLIS2 promotes cell proliferation by functioning as a miR-671 sponge to activate NF-κB signaling pathway [175]. In bladder cancer, circINTS4 facilitates proliferation and tumorigenesis by activating the NF-kB signaling via miR-146b/CARMA3 axis [176]. In prostate cancer, circARHGEF28 inhibits cell proliferation by regulating NF-κB axis through miR-671-5p/LGALS3BP pathway [177]. During oral squamous cell carcinoma progression, circ_0001461 promotes cell proliferation and tumor growth through sponging miR-145 to modulate TLR4/NF-κB signaling axis [178].
The circRNAs target NF-κB signaling to regulate cell proliferation in the context of various cancers, such as circ_0001821/miR-423-5p/BTRC/IKBA, circ-CD44/miR-23b-5p/TAB1, ciRS-7/miR-7/HOXB13, circCYP24A1/PKM2 axis to regulate NF-κB signal in ESCC; circZFR/STAT3/NF-κB, circLIFR/TBK1/NF-κB, circCORO1C/NF-κB/PD-L1 in hepatocellular carcinoma; circADAMTS6/ANXA2/NF-κB in glioblastoma; circ_0002019/TNFAIP6/NF-κB in gastric cancer; circGLIS2/miR-671/NF-κB in colorectal cancer; circINTS4/miR-146b/CARMA3/NF-κB in bladder cancer. circARHGEF28/miR-671-5p/LGALS3BP/NF-κB in prostate cancer; circ_0001461/miR-145/TLR4/NF-κB axis in oral squamous cell carcinoma.
Wnt/β-catenin pathway is another signaling pathway related to tumor progression, which can be regulated by circRNAs through multiple mechanisms under different settings (Fig. 8). For instance, in the context of lung cancer, circ-EIF3I facilitates cell proliferation by targeting miR-1253/NOVA2 to regulate Wnt/β-catenin pathway; circVAPA promotes cell proliferation by targeting miR-876-5p/WNT5A to activate Wnt/β-catenin signaling; circ-ZNF124 enhances cell proliferation via miR-498/YES1 axis to activate Wnt/β-catenin signaling pathway; cir-ITCH suppresses tumor proliferation by serving as the sponge of miR-7 and miR-214 to upregulate ITCH and then inactivate Wnt/β-catenin pathway [179,180,181,182]. In the context of hepatocellular carcinoma, circMTO1 suppresses tumor proliferation by regulating miR-541-5p/ZIC1 axis to inactivate Wnt/β-catenin signaling pathway; hsa_circRNA_104348 promotes cell proliferation through miR-187-3p/RTKN2 axis to activate Wnt/β-catenin pathway; circRNA-SORE functions as the sponge of miR-103a-2-5p and miR-660-3p, thus competitively activating Wnt/β-catenin signaling; circZFR enhances proliferation by regulating miR-3619-5p/CTNNB1 to activate Wnt/β-catenin signaling pathway [183,184,185,186]. In gastric cancer, circAXIN1 promotes cell proliferation by encoding a protein named AXIN1-295aa, which competitively interacts with APC and then activates the Wnt/β-catenin signaling pathway; circ_SMAD4 facilitates cell proliferation and gastric carcinogenesis by targeting miR-1276/TCF4/CTNNB1 axis to activate Wnt/β-catenin pathway; cir-ITCH inhibits cell proliferation by sponging miR-17 to increase linear ITCH expression and thus negatively regulate the Wnt/β-catenin pathway; circCNIH4 inhibits cell proliferation through enhancing DKK2 and FRZB expression and then suppressing Wnt/β-catenin signaling pathway [187,188,189,190]. Under the setting of colorectal cancer, circ-ACAP2 facilitates cell proliferation through modulating miR-143-3p/FZD4 axis to activate Wnt/β-catenin pathway; circAGFG1 accelerates cell proliferation and tumor growth by directly sponging miR-4262 and miR-185-5p to enhance YY1 or CTNNB1 expression and then activate Wnt/β-catenin pathway; hsa_circ_0005615 (circ5615) promotes cell proliferation and arrest cell cycle through functioning as a miR-149-5p sponge to release TNKS and activate Wnt/β-catenin pathway; hsa_circ_0009361 suppresses cell proliferation by acting as a miR-582 sponge to upregulate APC2, thus inhibiting the Wnt/β-catenin signaling [191,192,193,194].
In lung cancer, circ-EIF3I/miR-1253/NOVA2, circVAPA/miR-876-5p/WNT5A, circ-ZNF124/miR-498/YES1, and cir-ITCH/miR-7, miR-214/ ITCH pathway have been confirmed to affect the proliferation phenotype by targeting Wnt/β-catenin signaling. In hepatocellular carcinoma, circMTO1/miR-541-5p/ZIC1, hsa_circRNA_104348/miR-187-3p/RTKN2, circRNA-SORE/miR-103a-2-5p and miR-660-3p, circZFR/miR-3619-5p/CTNNB1 pathways have been reported to modulate cell proliferation by affecting Wnt/β-catenin signaling. In gastric cancer, circAXIN1-encoded protein AXIN1-295aa/APC, circ_SMAD4/miR-1276/TCF4/CTNNB1, cir-ITCH/miR-17/ITCH, circCNIH4/DKK2/FRZB axis have been confirmed to regulate proliferation by targeting Wnt/β-catenin signaling pathway. In colorectal cancer, circ-ACAP2/miR-143-3p/FZD4 axis, circAGFG1/miR-4262 and miR-185-5p/YY1 and CTNNB1, circ5615/miR-149-5p/TNKS, hsa_circ_0009361/miR-582/APC2 pathway were reported to influence proliferation via modulating Wnt/β-catenin signaling.
The mitogen-activated protein kinase (MAPK) signaling pathway is a series of highly conserved enzymatic reaction cascades involved in various physiological activities including cell proliferation, differentiation, and apoptosis. In glioma, circ-MAPK4 affects cell proliferation and apoptosis by functioning as a miR-125a-3p sponge to activate p38/MAPK signaling pathway [195]; circular RNA Pleiotrophin (circ_PTN) promotes proliferation and carcinogenesis through modulating miR-122/SOX6 axis to activate MAPK/ERK pathway [196]; circ-TLK1 facilitates cell proliferation and tumor growth by sponging miR-17-5p to target PANX1/MAPK/ERK axis [197]. In hepatocellular carcinoma, circDHPR facilitates tumor growth and metastasis through interacting with miR-3194-5p to target the RASGEF1B/RAS/MAPK signaling pathway [198]; circASAP1 promotes cell proliferation and invasion via acting as the sponge of miR-326 and miR-532-5p to regulate MAPK1 expression [199]; circSETD3 inhibits tumor growth through targeting miR-421/MAPK14 signaling pathway [200]. In breast cancer, circDNAJC11 promotes cell proliferation and tumor progression by interacting with TAF15 to upregulate MAPK6 expression and activate the MAPK signaling [201]; circBACH2 (hsa_circ_0001625) promotes cell proliferation via regulating hsa-miR-944/HNRNPC axis and stimulating MAPK signaling pathway [202]; hsa_circRNA_0006528 (circ_0006528) promotes proliferation and growth phenotypes by sponging miR-7-5p to enhance Raf1 expression and then activating the MAPK/ERK signaling pathway [203]. During NSCLC progression, circRNA C190 promotes cell proliferation via modulating the EGFR/MAPK/ERK signaling pathway [204]; circ-ZKSCAN1 accelerates proliferation and progression through sponging miR-330-5p to upregulate FAM83A expression and subsequent inactivation of MAPK signal transduction pathway [205]; circular RNA 0001313 (circ0001313) promotes NSCLC cell proliferation and invasion through serving as a microRNA-452 sponge to regulate HMGB3/ERK/MAPK axis [206]. In gastric cancer, circMAPK1 exerts suppressive function through encoding a novel protein MAPK1-109aa, which competitively binds to MEK1 and then decreases the phosphorylation of MAPK1, thus inactivating MAPK1 and the downstream molecules in MAPK signaling pathway [207]; circRNA hsa_circ_0044301 promotes cell proliferation by acting as hsa-miR-188-5p sponge to upregulate DAXX (ERK1/2) expression and MAPK signaling pathway [208]; hsa-circ-0052001 promotes cell proliferation and invasion via sponging hsa-miR-608 to regulate the MAPK pathway [209] (Fig. 9). Meanwhile, there were many circRNAs involved in the regulation of other pathways to affect tumor development. Therefore, it was an approach worthy of attention to influence circRNAs, then modulate cancer-related signaling pathways, and ultimately affect tumor progression.
The circRNAs involving in MAPK signaling pathway to regulate cell proliferation under the settings of various tumors, such as circ-MAPK4/miR-125a-3p/p38/MAPK, circ_PTN/miR-122/SOX6/MAPK/ERK, and circ-TLK1/miR-17-5p/PANX1/MAPK/ERK axis in glioma; circDHPR/miR-3194-5p/RASGEF1B/RAS/MAPK, circASAP1/miR-326 and miR-532-5p/MAPK1, circSETD3/miR-421/MAPK14 pathway in hepatocellular carcinoma; circDNAJC11/TAF15/MAPK6, circBACH2/hsa-miR-944/HNRNPC/MAPK, and circ_0006528/miR-7-5p/Raf1/MAPK/ERK pathway in breast cancer; circRNA C190/EGFR/MAPK/ERK, circ-ZKSCAN1/miR-330-5p/FAM83A/MAPK, and circ0001313/microRNA-452/HMGB3/ERK/MAPK axis in NSCLC; circMAPK1- encoded protein MAPK1-109aa/MEK1/MAPK1, hsa_circ_0044301/hsa-miR-188-5p/DAXX, and hsa-circ-0052001/hsa-miR-608/MAPK pathway in gastric cancer.
CircRNAs involvement in cell death
Cell death is divided into two categories based on how quickly it occurs and whether a drug or gene is likely to have an effect on it: accidental cell death (ACD) and regulated cell death (RCD). ACD is an uncontrolled cell death process that is triggered by unintended noxious stimuli that exceed the regulatable capacity of the cell, leading to the onset of cell death. RCD is mediated by signal transduction pathways and well-defined mechanisms of action, and plays a crucial role in homeostasis maintenance and disease development. According to different morphological, biochemical, immunological and genetic characteristics, RCD can be divided into two categories: apoptotic and non-apoptotic. Non-apoptotic RCD can be subdivided into autophagy, pyroptosis, ferroptosis, and necroptosis [210].
Apoptosis is the most classic form of cell death. A transcriptomic study using deep RNA sequencing identified differentially expressed circRNAs in HeLa cells under apoptotic conditions and manifested the potential regulatory role of circRNAs in apoptosis [211]. A well-studied circRNA derived from the tumor suppressor gene Foxo3, termed as circFoxo3 (hsa_circ_0006404), has been investigated to promote tumor cell apoptosis through various mechanisms in multiple types of human cancers [212]. In glioma, circ-MAPK4 (has_circ_0047688) was reported to inhibit cell apoptosis by functioning as miR-125a-3p sponge to regulate p38/MAPK signaling pathway [195]. It was also reported that circ-TTBK2 could act as miR-217 sponge to target HNF1β/Derlin-1 pathway and inhibit cell apoptosis in the glioma progression [213].
Autophagy is a self-digestive process that helps maintain the nutrient and energy needed by cells to limit damage in the short-term, but in the long-term it transforms into a self-destructive process that eventually leads to cell death [214]. Mounting evidence reveals that circRNAs can affect the biological behavior and treatment response of many tumors through the epigenetic regulation of autophagy [215]. Researchers found that an exosomal circRNA derived from ATG4B (circATG4B, hsa_circ_0007159) was increased in the exosomes secreted by drug-resistant colorectal cancer cells. Mechanistically, circATG4B encoded a novel protein (circATG4B-222aa), which could act as a decoy to competitively bind to TMED10 and prevent the interaction of TMED10 and ATG4B, contributing to promoted autophagy and inducing chemoresistance [216]. In pancreatic cancer, an autophagy-associated circRNA circ-autophagy related 7 (circATG7, has_circ_0064288) was reported to facilitate cell autophagy, proliferation and invasion through miR-766-5p/ATG7 and HUR/ATG7 axis, thereby promoting PC progression [217]. Furthermore, circCDYL (hsa_circ_0008285) was reported to augment autophagy of breast cancer cells via regulating the miR-1275-ATG7/ULK1 axis, which may be a potential biomarker for predicting the prognosis of breast cancer patients [218].
Pyroptosis is a newly discovered mode of programmed cell death that results in rapid cell lysis and the release of inflammatory substances, and is closely related to the progression of various cancers [219]. In lung adenocarcinoma (LUAD), circPIBF1 binds to nuclear factor erythroid 2-related factor 2 (Nrf2), which further promotes SOD2 expression and recruits EP300 to promote H3K27ac modification of SOD2, thus modulating the Nrf2/Keap-1 pathway and participating in pyroptosis-related carcinogenesis process [220]. Similarly, hsa_circ_0007312 (circ7312) was discovered to suppress lung adenocarcinoma cell pyroptosis and apoptosis via targeting the miR-764/MAPK1 axis [221]. Researchers discovered that circNEIL3 (hsa_circ_0001460) might mediate radiation-induced pyroptosis by sponging miR-1184 to upregulate PIF1 expression, providing a novel treatment option for lung adenocarcinoma therapy [222]. During the progression of ESCC, a mitochondria localized circular RNA derived from PUM1 (hsa_circ_0011240), termed as circPUM1, has been shown to play a scaffolding role in UQCRC1 and UQCRC2 interactions. Furthermore, knockdown of circPUM1 inhibited cellular oxidative phosphorylation levels, leading to pyroptosis of ESCC cell lines [223].
Ferroptosis is an iron-dependent programmed cell death manner and crucial tumor inhibition pathway, which is induced by the disturbance of intracellular lipid metabolism and accumulation of reactive oxygen species (ROS) on the membrane lipid [224, 225]. It has been demonstrated that a series of circRNAs can regulate ferroptosis via different mechanisms and then participate in cancer onset and development processes [226]. For instance, circSCN8A (hsa_circ_0026337) was found to be downregulated in NSCLC and induce cell ferroptosis through serving as a miR-1290 sponge to activate long-chain acyl-CoA synthetase-4 (ACSL4) expression [227]. In the context of bladder cancer, circST6GALNAC6 has been shown to bind to small heat shock protein 1 (HSPB1) and block erastin-induced phosphorylation site of HSPB1, protectively in response to ferroptosis stress (Ser-15 site), thus activating the P38 MAPK pathway and facilitating cell ferroptosis [228]. Additionally, researchers found that exosomal circRNA_101093 (cir93) could bind to fatty acid-binding protein 3 (FABP3) and increase its expression, desensitizing lung adenocarcinoma cells to ferroptosis [229].
CircRNAs involvement in inflammation
Inflammation is essentially a defensive response of the human body, which is a series of complex physiological reactions produced by the immune system for the protective purposes after the body is subjected to harmful stimuli. Inflammation can be divided into acute and chronic, among which acute inflammation is the initial response of the human body to stimuli, and its ideal outcome is timely regression until the body returns to homeostasis, which help to clear inflammatory substances and promote tissue repair. In this process, immune cells and the chemicals produced by them, such as antibodies and cytokines, are released into the blood or affected tissues to fight against foreign pathogens or inflammatory stimuli. The regression of inflammation is not a passive termination of the inflammatory response, but an active programmed process initiated in the first few hours after the onset of inflammation, involving multiple types of cells, anti-inflammatory factors and pro-regression mediators. When inflammatory factors persist, inflammation cannot subside in a timely manner, and it will transform into chronic inflammation. Chronic inflammation will produce a large number of inflammatory mediators, induce cell proliferation, DNA damage and repair disorders, ultimately leading to cancer. At present, research has determined that inflammation is closely related to the occurrence and development of cancer, and it can serve as a double-edged sword to promote or inhibit tumor progression. Moreover, the inflammation response triggered by anti-tumor therapy can in turn affect the clinical therapeutic efficacy of cancer to varying degrees [230, 231].
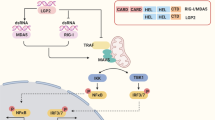
Growing evidence has demonstrated that circRNAs can modulate the interplay between inflammation and tumorigenesis through various regulatory mechanisms, such as sponging miRNAs, binding to RBPs, and encoding proteins to regulate inflammatory signaling pathways involves in cancer progression, including NF-κB, PI3K-AKT, MAPK, JAK-STAT, etc [232, 233]. For instance, circ-TPGS2 promotes tumor-associated inflammation, cell dissemination and metastasis of breast cancer through serving as a miR-7 sponge to increase TRAF6 expression, contributing to p65 phosphorylation and nuclear translocation, ultimately inducing the activation of NF-κB signaling pathway and dysregulation of tumor microenvironment [234]. During breast cancer progression, hsa_circRNA_002178 activates inflammation and accelerates tumor growth by sponging miR-328-3p to upregulate COL1A1 expression [235]. In the context of colitis-associated colorectal carcinoma (CAC), PRKAR2A-derived circRNA (mmu_circ_0001109) exacerbates the colitis progression through activating the JAK-STAT3 and NF-κB signaling pathways, and promotes the transition of inflammation to carcinoma via activating the Wnt/β-catenin signaling pathway [236]. In addition, circNEIL3 contributes to pyroptosis and activates AIM2 inflammasome by sponging miR-1184 to release the suppressed PIF1 in lung adenocarcinoma [222].
CircRNAs involvement in angiogenesis
One of the mechanisms closely related to the growth and metastasis of cancer is angiogenesis. Angiogenesis is necessary for tumors to obtain nutrients to grow and perform essential functions [237, 238]. It was reported that vascular endothelial growth factor (VEGF), angiopoietin, matrix metalloproteinases (MMPs), and fibroblast growth factor (FGF) could function as pro-angiogenic factors in cancer angiogenesis [239]. Numerous studies have confirmed that circRNAs mediated regulation of tumor angiogenesis [240]. For instance, circRNA-MYLK was found to function as miR-29a sponge to activate VEGFA/VEGFR2 and downstream Ras/ERK pathway to promote angiogenesis and metastasis of bladder cancer [241]. Circ3823 (hsa_circ_0001821) derived from PVT1 gene may promote colorectal cancer growth, metastasis and angiogenesis through targeting the miR-30c-5p/Tcf7 axis [242]. CircHIPK3 (hsa_circ_0000284) could function as the sponge of miR-558 to negatively regulate the expression of heparanase (HPSE), further inhibiting angiogenesis of bladder cancer cells [243]. Researchers also discovered a novel oncogenic circRNA circ-ASH2L that could act as a sponge for miR-34a to upregulate Notch 1 expression to promote tumor angiogenesis in pancreatic ductal adenocarcinoma [244].
Exosomal circRNAs also affect tumor angiogenesis by interacting with epigenetic regulators, acting as miRNA and RNA-binding protein (RBP) sponges. For example, exosomal cholangiocarcinoma (CCA)-associated circular RNA 1 (circ-CCAC1) derived from ERBB2 gene was discovered to sponge miR-514a-5p to upregulate Yin Yang 1 (YY1), inducing CCA angiogenesis, and regulate CCA tumorigenesis and metastasis [245]. Exosomal circSHKBP1 (hsa_circ_0000936) was upregulated in gastric cancer tissues and serum, but significantly decreased after gastrectomy. Functional experiments in vitro and in vivo showed that enforced expression of circSHKBP1 strengthened the proliferation, migration, invasion and angiogenesis capacities of gastric cancer. Mechanically, circSHKBP1 could sponge miR-582-3p to activate the HUR/VEGF signaling axis, and directly interact with HSP90 to disrupt the binding of HSP90 and STUB1, further restraining the ubiquitination and degradation of HSP90 [246].
CircRNAs involvement in tumor microenvironment
Tumor microenvironment (TME) is the internal environment in which tumor cells produce and survive, including tumor cells and their surrounding fibroblasts, immune and inflammatory cells, as well as the interstitial cells, microvessels, and biomolecules infiltrating in them [247]. Classical theory holds that cancer-causing mutations in malignant cells lead to the development of cancer [248]. Subsequently, surrounding untransformed cells are recruited and adapted, accompanied by the release of various intercellular communication factors, including cytokines, chemokines, and vesicles. Some studies have shown that chronic inflammation or wound healing processes activate cancer-causing signals in abnormal microenvironment and promote tumorigenesis [249, 250].
Increasing studies have shown that circRNAs are involved in various biological regulation of TME [251]. For example, overexpression of circSMARCC1 can promote CD163 expression in macrophages via the CCL20-CCR6 axis, inducing tumor-associated macrophages (TAMs) infiltration and M2 polarization, thus contributing to the progression of prostate cancer [252]. In addition, circNEIL3-overexpressed glioma cells drive macrophage infiltration into the TME. At the same time, circNEIL3 can be packaged into exosomes by binding to hnRNPA2B1 and delivered to infiltrating TAMs, allowing them to obtain immunosuppressive properties by stabilizing IGF2BP3, which in turn facilitates glioma progression [253].
CircRNAs involvement in other hallmarks of cancer
In addition to the major cancer features described above, circRNAs’ role in regulating other cancer hallmarks such as genome instability and metabolic reprogramming has also been discovered. The metabolic characteristics of tumor cells change during cancer progression to meet the needs of tumor growth and maintain TME homeostasis. The Warburg effect is a major metabolic characteristic regulated by many tumor cells and oncogenes. Tumor cells mainly use glycolysis pathway to provide energy under aerobic conditions, thereby improving the ability to escape apoptosis, invasion and resist chemotherapy [254, 255]. Many studies have shown that circRNAs can influence glucose metabolism in tumors through transcription factors or signaling pathways [256]. For example, CircECE1 can interact with c-Myc to prevent the speckle-type POZ protein from degrading c-Myc, thereby activating the Warburg effect of osteosarcoma via the c-Myc/TXNIP axis [257]. Highly expressed circNRIP1 in gastric cancer can act as the sponge of microRNA-149-5p to target AKT1/mTOR pathway and improve the glucose uptake capacity of gastric cancer cells [258]. In addition, circFOXP1 can interact with PTBP1 or sponge miR-370 to regulate PKLR, thus promoting the Warburg effect in gallbladder cancer progression [259].
Genome instability (GI) covers changes from a single nucleotide to the entire chromosome, contributing to the increase of genetic changes in cancer and is the driving force for cancer progression [260]. Petermann et al. showed that RNA-DNA hybridization products can be formed during transcription, DNA replication, and DNA repair, which can replace one of the DNA strands of double-stranded DNA to form an R-loop. R-loops pose a threat to genome stability and can cause DNA damage that promotes cancer progression [261]. A recent study has shown that circRNAs in the nucleus can directly regulate transcription and splicing by producing circRNAs-DNA hybrids. The team found that circRNAs enriched in mixed-lineage leukemia (MLL) recombinome and bound DNA to form a circR loop at its homologous site. These circR loops have been found to have the effect on promoting transcriptional suspension, inhibiting proteasome, chromatin translocations recombination, and DNA double-strand breaks (DSB). Together, these effects will lead to chromosomal translocations induced by endogenous RNA carcinogens in leukemia [262]. In addition, studies have shown that circRNAs, such as circBUB1B_544aa and circCHEK1_246aa, can activate the instability at the chromosomal level of multiple myeloma by phosphorylating CEP170, thus leading to the proliferation and drug resistance of tumor cells [263, 264].
CircRNAs regulate immune checkpoints and ICIs resistance
CircRNAs regulate PD-1/PD-L1 in cancer
Studies have shown that circRNAs, as competing endogenous RNAs (ceRNAs), can be involved in tumor immune escape process and immunotherapy resistance, thereby regulating PD-1/PD-L1 pathway [265]. Existing studies mainly reveal the mechanisms affecting the regulation of PD-L1 expression through two approaches. One approach is that circRNAs can directly regulate the expression of PD-L1 through its sponge effect on miRNA, the other approach is that circRNAs can affect the expression of downstream molecules through the sponge mechanism, thereby modulating the expression of PD-L1. Studies on the regulatory mechanism of PD-1 are mainly focused on anti-PD-1 immunotherapy resistance. All mechanisms were summarized in Table 3.
As a circRNA highly expressed in lung cancer, circCHST15 can directly inhibit the expression of PD-L1 by acting as miR-155-5p and miR-194-5p sponges to accelerate tumor growth and immune escape of lung cancer [266]. In addition, circCPA4 (hsa_circ_0082374) [267] and circ_0000284 [268] are highly expressed in lung cancer and could also upregulate PD-L1 expression. Both hsa_circ_0068252 (circ_0068252) [269] and circFOXK2 [270] can promote the expression of PD-L1 through sponge mechanism, activate PD-1/PD-L1 pathway-dependent immune escape, and play a pro-tumor biological role. Moreover, several circRNAs can affect the expression of PD-L1 through the sponge mechanism in various tumors, such as hepatocellular carcinoma (hsa_circ_0003288) [271], pancreatic cancer (hsa_circ_0046523) [272], colorectal cancer (CDR1-AS, hsa_circ_0136666, circEIF3K, has_circ_0020397) [273,274,275,276], head and neck squamous cell carcinoma (circ_0000052) [277], breast cancer (circ_0001598) [278], bladder cancer (circ_0001005) [279] and hematological system cancer (circPCBP2) [280]. Specifically, upregulated circVIM has been found to release its targeted inhibition of PD-L1 expression via sponging miR-124 to promote immune escape and various malignant biological behaviors of esophageal cancer [281]. Another circRNA, has_circ_0020397, was reported to be upregulated in colorectal cancer, promoting cell activity and invasion, but inhibiting apoptosis by acting as a miR-138 sponge to positively regulate its target genes telomerase reverse transcriptase (TERT) and PD-L1 [276]. In diffuse large B-cell lymphoma (DLBCL), circPCBP2 was found to be highly expressed in DLBCL tissues and cells. Functionally, circPCBP2 can inhibit its target gene PD-L1 by directly binding to miR-33a/b, thereby promoting DLBCL cell dryness and chemotherapy resistance to prednisone (CHOP) [280].
CircRNAs involved in another regulatory mechanism of PD-L1 include circFGFR1 (hsa_circ_0084003) [282], circ_0014235 [283], and circIGF2BP3 (hsa_circ_0079587) [284], and all of them are highly expressed in lung cancer. CircFGFR1 activates its downstream gene C-X-C motif chemokine receptor 4 (CXCR4) through spongy adsorption of miR-381-3p, promoting tumor malignant progression and anti-PD-1 immunotherapy resistance [282]. Circ_0014235 alleviates the inhibition of YAP by directly sponging miR-146b-5p, thereby further upregating the expression of PD-L1, thus promoting gefitinib resistance and harmful biological behavior in NSCLC [283]. Mechanism experiments demonstrated that N6-methyladenosine-modified circIGF2BP3 by METTL3 could serve as miR-328-3p and miR-3173-5p sponge to competitively elevate the expression of plakophilin 3 (PKP3), which binds with FXR1 protein to further stabilize OTUB1 mRNA, and OTUB1 enhances PD-L1 abundance by promoting its deubiquitination [284].
CircRNA_001678 (circ_001678) [285] and hsa_circ_0003222 [286] are circRNAs that regulate the high expression of PD-1 in lung cancer. CircRNA_001678 could enhance ZEB1 expression through sponging miR-326 to activate PD-1/PD-L1 pathway-dependent immune escape, induce apoptosis of CD8 + T cells, and play a pro-tumor biological role [285]. Hsa_circ_0003222 has been studied to alleviate the inhibited expression of PHF21B and its downstream gene β-catenin by acting as a molecular sponge of miR-527, thereby promoting proliferation, migration, invasion, stemness and anti-PD-L1 immunotherapy resistance [286]. According to a recent study, circSOD2 could sponge miR-497-5p to upregulate the expression of ANXA11, which contributes to tumor malignant progression and anti-PD-1 immunotherapy resistance in hepatocellular carcinoma [287].
Except for serving as the aforementioned ceRNAs, circRNAs have also been reported to regulate the PD-1/PD-L1 pathway through interacting with proteins or other unclarified mechanisms. Ge et al. found that circBART2.2 encoded by Epstein-Barr virus (EBV) could bind to the helicase domain of RBP RIG-I in nasopharyngeal carcinoma (NPC) cells, and then activate the transcription factors IRF3 and NF-κB, resulting in enhanced expression of PD-L1 to induce tumor immune escape [288]. In addition, the circMGA/HNRNPL complex has also been found to significantly inhibit the growth of bladder cancer by stabilizing CCL5 and exerting synergistic effects with anti-PD-1 [289]. A recent publication demonstrated that heat shock protein 90 alpha (HSP90A) derived circHSP90A was significantly upregulated in NSCLC and could facilitate cell growth, stemness, and immune evasion by recruiting ubiquitin specific peptidase 30 (USP30) to further stabilize HSP90A protein, which then activates the STAT3 signaling pathway. Moreover, circ-HSP90A has also been shown to modulate the PD-1/PD-L1 checkpoint in this context by sponging miR-424-5p to target PD-L1 expression [290]. Hsa_circ_0000190 has also been reported to accelerate the tumor progression and immune evasion of NSCLC through increasing soluble PD-L1 expression, and its high expression could serve as an effective indicator for anti-PD-L1 immunotherapy resistance and poor prognosis of NSCLC patients [291]. CircCORO1C, which was highly expressed in hepatocellular carcinoma, could upregulate the expression of c-Myc and COX-2 by stimulating the NF-κB pathway and enhancing P65 phosphorylation to induce PD-L1 expression, thus promoting immune escape and tumor growth [172].
CircRNAs regulate CTLA-4 in cancer
Compared with PD-1/PD-L1, there is relatively little research on the regulation of CTLA-4 by circRNAs, which may be due to the fact that most CTLA-4 targeting ICIs are immature and have no effective approved clinical transformation results. A total of three circRNAs have been studied to participate in the regulation of CTLA-4 (Table 4). CircUBAP2 (hsa_circ_0007367) was shown to repress antigen presentation and induce immune escape in pancreatic adenocarcinoma (PAAD) through sponging miR-494 to upregulate CXCR4 and ZEB1 proteins, then leading to increased expression of CTLA-4 and PD-1 [292]. CircWDR25 (hsa_circ_004310) and circQSOX1 (hsa_circ_0015497) could also promote the increased expression of CTLA-4 and PD-L1 through the sponge mechanism in hepatocellular carcinoma and colorectal cancer, respectively, thus driving tumor immune escape and promoting cancer progression [293, 294]. In these studies, we found that circRNAs can affect the expression of CTLA-4 and PD-L1, and whether the regulation of specific circRNAs can achieve the same therapeutic effect as the combined application of anti-CTLA-4 and anti-PD-L1 ICIs in clinical practice is the direction of future research.
CircRNAs regulate other immune checkpoints in cancer
Analogous with CTLA-4, there have been few studies on the regulation by circRNAs of other newly discovered immune checkpoints in recent years, such as TIM-3, SIRPα and CD47, which may be due to the fact that the mechanism of these immune checkpoints is not yet fully elucidated, and the exploration of ICIs targeting them as cancer immunotherapy is still at an early stage. As for the regulation of TIM-3, Zhang et al. reported that under the setting of hepatocellular carcinoma, circUHRF1 upregulated the expression of TIM-3 in NK cells by sponging miR-449c-5p, and inhibited the secretion of IFN-γ and TNF-α by NK cells, thus promoting tumor immune escape [295]. Another recently studied immune checkpoint, CD47, is a transmembrane immunoglobulin that is expressed on the surface of traditional human cells as a ligand for SIRPα and is overexpressed on a variety of tumor cells. The formation of complexes with CD47 and SIRPα transmits an antiphagocytic signal to macrophages, and the CD47-SIRPα pathway serves as an innate immune checkpoint to function as a druggable target, which enables cancer cells to escape from phagocytosis mediated by macrophages [296]. The impact of circRNAs targeting CD47 on cancer has been reviewed in the reference [28], which mentions that circRERE (hsa_circ_0009581) promotes the resistance to chemotherapy drug bortezomib (BTZ) in multiple myeloma (MM) via sponging miR-152-3p to upregulate the expression of CD47 [297]. Besides that, hsa_circ_0000276 could also regulate the expression of CD47 in cervical cancer and participate in the regulation of immune infiltration and trigger carcinogenic function [298]. These studies suggest that since circRNAs can influence tumor resistance by modulating novel immune checkpoints, circRNAs may be useful as biomarkers to help predict the effectiveness of ICIs. At the same time, circRNA targeting therapy may also achieve simultaneous regulation of multiple immune checkpoints.
Conclusion and perspective
Over the past decade, ICIs have been developed and approved for clinical use in multiple cancer types at an unprecedented rate. Despite tremendous progress, ICIs have not completely solved the problem of cancer treatment. Besides discovering biomarkers to predict the efficacy and safety of ICIs, more immune checkpoint molecules may need to be identified in the next decade to provide more options for immunotherapy and bring treatment opportunities to more patients, which depends on our insights into the mechanisms of immune checkpoint regulation. This review summarizes the status quo of frequent immune checkpoints and ICIs commonly applied in cancer, such as anti-PD-1/PD-L1, anti-CTLA-4, anti-LAG-3, anti-TIM-3, and other immune checkpoints targeted mAbs. The regulatory mechanisms of immune checkpoints are complex and unclear, which stem from its complicated and diverse influencing factors, involving ncRNAs, proteins, and other elements. As a class of abundant component of the human transcriptome involved in cancer hallmarks, ncRNAs are closely associated with the expression regulation of immune checkpoints. In view of the structure’s stability, circRNAs present significantly greater advantages and potential as diagnostic and prognostic biomarkers for cancer management than other types of ncRNAs. Also, circRNAs have been confirmed to involve in various cancer hallmarks including proliferation, cell death, inflammation, angiogenesis, invasion and metastasis through functioning as miRNA sponge, interacting with proteins, encoding proteins, or other unclarified mechanisms.
In this present review, we emphasized on the molecular mechanisms by which circRNAs interfere with the therapeutic strategy of ICIs by regulating immune checkpoint pathways. PD-1/PD-L1, CTLA-4, TIM-3, CD47/SIRPɑ, and other immune checkpoints have been shown to be regulated by circRNAs, mostly through serving as ceRNAs, and sometimes through interacting with proteins or encoding proteins. Meanwhile, we found that circRNAs may regulate the expression of two or more immune checkpoints at the same time, and when the tumor developed resistance to one immune checkpoint inhibitor, circRNAs may resist the development of resistance by regulating the other immune checkpoint. Therefore, in future studies, the mechanisms by which circRNAs regulate the interaction of multiple immune checkpoints to affect the resistance of ICIs, and the question of other adverse reactions that circRNAs may cause as well as resistance to ICIs are the main directions of research. In addition, the exact contribution of circRNAs-mediated epigenetic regulation of immune checkpoints and resistance to cancer immunotherapy have not been clearly elucidated. Many critical questions about the precise molecular mechanisms of circRNAs in regulating immune checkpoints and mediating resistance to ICIs therapy remain to be resolved. For instance, considering the abundant expression of circRNAs in tumor cells and immune cells, can they directly serve as immune checkpoints to regulate tumor immunity? Is resistance to ICIs therapy mediated by circRNAs in tumor cells or immune cells, or a combination of both? Can we design synthetic RNAs specifically targeting circRNAs to regulate the cascade reactions involved in immune checkpoint pathway, thus achieving the development of new potential cancer immunotherapy regimens? Moreover, hyperprogression is a new challenge in the treatment of ICIs, and its mechanism is not yet clear. Current studies have shown that important causes of hyperprogression may include overactivation or mutation of cancer related genes or pathways in tumor cells themselves, such as the amplification of MDM2/MDM4 and EGFR mutations [299, 300], and changes in immune cells in the TME, such as increased number of regulatory T cells and M2-like differentiation of TAMs [301, 302]. However, the role of circRNAs in this process still needs to be further studied.
At present, various treatment regimens such as chemotherapy, radiotherapy, anti-angiogenic therapy targeting VEGF, interleukin therapy, epigenetic modulators, and protein kinase inhibitors have been proven to boost the body’s anti-tumor immunity. The reasonable combination application of these treatment approaches with ICIs will improve the current status of tumor immunotherapy. In addition, tumor vaccines have become the most promising anti-tumor immunotherapy method because of their specificity, safety and good tolerance. With the development of mRNA vaccines winning the Nobel Prize, circRNA vaccine research has also made great progress [303]. The circRNA vaccine, which contains internal ribosome entry site (IRES) and open reading frame (ORF), has better safety, stability, ease of production and scalability than the first-generation mRNA vaccine [304]. In recent studies, circRNA cancer vaccines delivered by lipid nanoparticle (LNP) system have shown robust immune responses and better anti-tumor effects in mouse models [38]. However, research on the use of circRNA vaccines for tumor immunotherapy is still limited. In the future, is there a possibility of using synthetic circRNAs or circRNA inhibitors as an alternative therapy for ICIs remedy or as a combination therapy with ICIs? Recent research advances in single-cell transcriptomics, spatial transcriptomics and epigenetics may provide important information for circRNAs’ function, phenotype in cells and location in TME, and discover additional circRNAs potentially related to immune checkpoints regulation, which takes a key step forward in the right direction for our understanding progress in tumor immunity and immunotherapy resistance.
References
Kraehenbuehl L, Weng CH, Eghbali S, Wolchok JD, Merghoub T. Enhancing immunotherapy in cancer by targeting emerging immunomodulatory pathways. Nat Rev Clin Oncol. 2022;19:37–50.
Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018;359:1350–5.
Sharma P, Allison JP. Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell. 2015;161:205–14.
Huang PW, Chang JW. Immune checkpoint inhibitors win the 2018 Nobel Prize. Biomed J. 2019;42:299–306.
Leach DR, Krummel MF, Allison JP. Enhancement of antitumor immunity by CTLA-4 blockade. Science. 1996;271:1734–6.
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711–23.
Ledford H. Melanoma drug wins US approval. Nature. 2011;471:561.
Korman AJ, Garrett-Thomson SC, Lonberg N. The foundations of immune checkpoint blockade and the ipilimumab approval decennial. Nat Rev Drug Discov. 2022;21:509–28.
Topalian SL, Sznol M, McDermott DF, Kluger HM, Carvajal RD, Sharfman WH, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2023;41:943–54.
O’Sullivan Coyne G, Madan RA, Gulley JL. Nivolumab: promising survival signal coupled with limited toxicity raises expectations. J Clin Oncol. 2014;32:986–8.
Pan C, Liu H, Robins E, Song W, Liu D, Li Z, et al. Next-generation immuno-oncology agents: current momentum shifts in cancer immunotherapy. J Hematol Oncol. 2020;13:29.
Sharma P, Siddiqui BA, Anandhan S, Yadav SS, Subudhi SK, Gao J, et al. The next decade of immune checkpoint therapy. Cancer Discov. 2021;11:838–57.
Kubli SP, Berger T, Araujo DV, Siu LL, Mak TW. Beyond immune checkpoint blockade: emerging immunological strategies. Nat Rev Drug Discov. 2021;20:899–919.
Doroshow DB, Bhalla S, Beasley MB, Sholl LM, Kerr KM, Gnjatic S, et al. PD-L1 as a biomarker of response to immune-checkpoint inhibitors. Nat Rev Clin Oncol. 2021;18:345–62.
Wang DR, Wu XL, Sun YL. Therapeutic targets and biomarkers of tumor immunotherapy: response versus non-response. Signal Transduct Target Ther. 2022;7:331.
Syn NL, Teng MWL, Mok TSK, Soo RA. De-novo and acquired resistance to immune checkpoint targeting. Lancet Oncol. 2017;18:e731–41.
Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Prim. 2020;6:38.
Dougan M, Luoma AM, Dougan SK, Wucherpfennig KW. Understanding and treating the inflammatory adverse events of cancer immunotherapy. Cell. 2021;184:1575–88.
Sullivan RJ, Weber JS. Immune-related toxicities of checkpoint inhibitors: mechanisms and mitigation strategies. Nat Rev Drug Discov. 2022;21:495–508.
Johnson DB, Nebhan CA, Moslehi JJ, Balko JM. Immune-checkpoint inhibitors: long-term implications of toxicity. Nat Rev Clin Oncol. 2022;19:254–67.
de Miguel M, Calvo E. Clinical challenges of immune checkpoint inhibitors. Cancer Cell. 2020;38:326–33.
He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020;30:660–9.
Sharma P, Allison JP. Immune checkpoint therapy: forging ahead. Sci Transl Med. 2022;14:eadf2947.
Kalaora S, Nagler A, Wargo JA, Samuels Y. Mechanisms of immune activation and regulation: lessons from melanoma. Nat Rev Cancer. 2022;22:195–207.
Shibru B, Fey K, Fricke S, Blaudszun AR, Fürst F, Weise M, et al. Detection of immune checkpoint receptors - a current challenge in clinical flow cytometry. Front Immunol. 2021;12:694055.
Vishnubalaji R, Shaath H, Elango R, Alajez NM. Noncoding RNAs as potential mediators of resistance to cancer immunotherapy. Semin Cancer Biol. 2020;65:65–79.
Shek D, Read SA, Akhuba L, Qiao L, Gao B, Nagrial A, et al. Non-coding RNA and immune-checkpoint inhibitors: friends or foes? Immunotherapy. 2020;12:513–29.
Jiang Y, Zhao L, Wu Y, Deng S, Cao P, Lei X, et al. The Role of NcRNAs to regulate immune checkpoints in cancer. Front Immunol. 2022;13:853480.
Liu CX, Chen LL. Circular RNAs: characterization, cellular roles, and applications. Cell. 2022;185:2016–34.
Misir S, Wu N, Yang BB. Specific expression and functions of circular RNAs. Cell Death Differ. 2022;29:481–91.
Liu J, Li D, Luo H, Zhu X. Circular RNAs: the star molecules in cancer. Mol Asp Med. 2019;70:141–52.
Chen L, Shan G. CircRNA in cancer: fundamental mechanism and clinical potential. Cancer Lett. 2021;505:49–57.
Almouh M, Razmara E, Bitaraf A, Ghazimoradi MH, Hassan ZM, Babashah S. Circular RNAs play roles in regulatory networks of cell signaling pathways in human cancers. Life Sci. 2022;309:120975.
Kristensen LS, Jakobsen T, Hager H, Kjems J. The emerging roles of circRNAs in cancer and oncology. Nat Rev Clin Oncol. 2022;19:188–206.
Hiam-Galvez KJ, Allen BM, Spitzer MH. Systemic immunity in cancer. Nat Rev Cancer. 2021;21:345–59.
Xu Z, Li P, Fan L, Wu M. The potential role of circRNA in tumor immunity regulation and immunotherapy. Front Immunol. 2018;9:9.
Meng L, Ding P, Liu S, Li Z, Sang M, Shan B. The emerging prospects of circular RNA in tumor immunity. Ann Trans Med. 2020;8:1091.
Li H, Peng K, Yang K, Ma W, Qi S, Yu X, et al. Circular RNA cancer vaccines drive immunity in hard-to-treat malignancies. Theranostics. 2022;12:6422–36.
Ohaegbulam KC, Assal A, Lazar-Molnar E, Yao Y, Zang X. Human cancer immunotherapy with antibodies to the PD-1 and PD-L1 pathway. Trends Mol Med. 2015;21:24–33.
Postow MA, Callahan MK, Wolchok JD. Immune checkpoint blockade in cancer therapy. J Clin Oncol. 2015;33:1974–82.
Morad G, Helmink BA, Sharma P, Wargo JA. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell. 2021;184:5309–37.
Dong H, Zhu G, Tamada K, Chen L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med. 1999;5:1365–9.
Sun C, Mezzadra R, Schumacher TN. Regulation and Function of the PD-L1 Checkpoint. Immunity. 2018;48:434–52.
Yokosuka T, Takamatsu M, Kobayashi-Imanishi W, Hashimoto-Tane A, Azuma M, Saito T. Programmed cell death 1 forms negative costimulatory microclusters that directly inhibit T cell receptor signaling by recruiting phosphatase SHP2. J Exp Med. 2012;209:1201–17.
Kim TK, Vandsemb EN, Herbst RS, Chen L. Adaptive immune resistance at the tumour site: mechanisms and therapeutic opportunities. Nat Rev Drug Discov. 2022;21:529–40.
Patsoukis N, Wang Q, Strauss L, Boussiotis VA. Revisiting the PD-1 pathway. Sci Adv. 2020;6:eabd2712.
Yin S, Chen Z, Chen D, Yan D. Strategies targeting PD-L1 expression and associated opportunities for cancer combination therapy. Theranostics. 2023;13:1520–44.
Scott LJ. Nivolumab: a review in advanced melanoma. Drugs. 2015;75:1413–24.
Deeks ED. Pembrolizumab: a review in advanced melanoma. Drugs. 2016;76:375–86.
Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ, et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet. 2019;393:1819–30.
Kojima T, Shah MA, Muro K, Francois E, Adenis A, Hsu CH, et al. Randomized phase III KEYNOTE-181 study of pembrolizumab versus chemotherapy in advanced esophageal cancer. J Clin Oncol. 2020;38:4138–48.
Chao J, Fuchs CS, Shitara K, Tabernero J, Muro K, Van Cutsem E, et al. Assessment of pembrolizumab therapy for the treatment of microsatellite instability-high gastric or gastroesophageal junction cancer among patients in the KEYNOTE-059, KEYNOTE-061, and KEYNOTE-062 clinical trials. JAMA Oncol. 2021;7:895–902.
Lee A, Duggan S, Deeks ED. Cemiplimab: a review in advanced cutaneous squamous cell carcinoma. Drugs. 2020;80(Jun):813–9.
André T, Berton D, Curigliano G, Sabatier R, Tinker AV, Oaknin A, et al. Antitumor activity and safety of dostarlimab monotherapy in patients with mismatch repair deficient solid tumors: a nonrandomized controlled trial. JAMA Netw Open. 2023;6(Nov):e2341165.
Mai HQ, Chen QY, Chen D, Hu C, Yang K, Wen J, et al. Toripalimab plus chemotherapy for recurrent or metastatic nasopharyngeal carcinoma: the JUPITER-02 randomized clinical trial. JAMA. 2023;330:1961–70.
Iacovelli R, Ciccarese C, Brunelli M, Battelli N, Buttigliero C, Caserta C, et al. First-line avelumab for patients with PD-L1-positive metastatic or locally advanced urothelial cancer who are unfit for cisplatin. Ann Oncol. 2022;33:1179–85.
Powles T, Kockx M, Rodriguez-Vida A, Duran I, Crabb SJ, Van Der Heijden MS, et al. Clinical efficacy and biomarker analysis of neoadjuvant atezolizumab in operable urothelial carcinoma in the ABACUS trial. Nat Med. 2019;25:1706–14.
Powles T, van der Heijden MS, Castellano D, Galsky MD, Loriot Y, Petrylak DP, et al. Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2020;21:1574–88.
Markham A. Envafolimab: first approval. Drugs. 2022;82:235–40.
Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N. Engl J Med. 2018;379:2342–50.
Zhou C, Wang Z, Sun M, Cao L, Ma Z, Wu R, et al. Interim survival analysis of the randomized phase III GEMSTONE-302 trial: sugemalimab or placebo plus chemotherapy as first-line treatment for metastatic NSCLC. Nat Cancer. 2023;4:860–71.
Ferrara R, Mezquita L, Texier M, Lahmar J, Audigier-Valette C, Tessonnier L, et al. Hyperprogressive disease in patients with advanced non-small cell lung cancer treated with PD-1/PD-L1 inhibitors or with single-agent chemotherapy. JAMA Oncol. 2018;4:1543–52.
Kim CG, Kim KH, Pyo KH, Xin CF, Hong MH, Ahn BC, et al. Hyperprogressive disease during PD-1/PD-L1 blockade in patients with non-small-cell lung cancer. Ann Oncol. 2019;30:1104–13.
Hwang I, Park I, Yoon SK, Lee JL. Hyperprogressive disease in patients with urothelial carcinoma or renal cell carcinoma treated with PD-1/PD-L1 inhibitors. Clin Genitourin Cancer. 2020;18:e122–33.
Lingel H, Brunner-Weinzierl MC. CTLA-4 (CD152): a versatile receptor for immune-based therapy. Semin Immunol. 2019;42:101298.
Van Coillie S, Wiernicki B, Xu J. Molecular and cellular functions of CTLA-4. Adv Exp Med Biol. 2020;1248:7–32.
Buchbinder EI, Desai A. CTLA-4 and PD-1 pathways: similarities, differences, and implications of their inhibition. Am J Clin Oncol. 2016;39:98–106.
Zhang A, Ren Z, Tseng KF, Liu X, Li H, Lu C, et al. Dual targeting of CTLA-4 and CD47 on Treg cells promotes immunity against solid tumors. Sci Transl Med. 2021;13:eabg8693.
Hosseini A, Gharibi T, Marofi F, Babaloo Z, Baradaran B. CTLA-4: from mechanism to autoimmune therapy. Int Immunopharmacol. 2020;80:106221.
Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Rutkowski P, Lao CD, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2019;381:1535–46.
Robert C, Ribas A, Schachter J, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 2019;20:1239–51.
Tawbi HA, Forsyth PA, Hodi FS, Lao CD, Moschos SJ, Hamid O, et al. Safety and efficacy of the combination of nivolumab plus ipilimumab in patients with melanoma and asymptomatic or symptomatic brain metastases (CheckMate 204). Neuro Oncol. 2021;23:1961–73.
Cascone T, William WN Jr, Weissferdt A, Leung CH, Lin HY, Pataer A, et al. Neoadjuvant nivolumab or nivolumab plus ipilimumab in operable non-small cell lung cancer: the phase 2 randomized NEOSTAR trial. Nat Med. 2021;27:504–14.
Paz-Ares L, Ciuleanu TE, Cobo M, Schenker M, Zurawski B, Menezes J, et al. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): an international, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22:198–211.
Nikoo M, Rabiee F, Mohebbi H, Eghbalifard N, Rajabi H, Yazdani Y, et al. Nivolumab plus ipilimumab combination therapy in cancer: current evidence to date. Int Immunopharmacol. 2023;117:109881.
Tarhini AA, Kirkwood JM. Tremelimumab, a fully human monoclonal IgG2 antibody against CTLA4 for the potential treatment of cancer. Curr Opin Mol Ther. 2007;9:505–14.
Keam SJ. Tremelimumab: first approval. Drugs. 2023;83:93–102.
Akce M, El-Rayes BF, Bekaii-Saab TS. Frontline therapy for advanced hepatocellular carcinoma: an update. Ther Adv Gastroenterol. 2022;15:17562848221086126.
Bejjani AC, Finn RS. Hepatocellular carcinoma: pick the winner-tyrosine kinase inhibitor versus immuno-oncology agent-based combinations. J Clin Oncol. 2022;40:2763–73.
Johnson ML, Cho BC, Luft A, Alatorre-Alexander J, Geater SL, Laktionov K, et al. Durvalumab with or without tremelimumab in combination with chemotherapy as first-line therapy for metastatic non-small-cell lung cancer: the phase III POSEIDON study. J Clin Oncol. 2023;41:1213–27.
Rotte A. Combination of CTLA-4 and PD-1 blockers for treatment of cancer. J Exp Clin Cancer Res. 2019;38:255.
Maruhashi T, Sugiura D, Okazaki IM, Shimizu K, Maeda TK, Ikubo J, et al. Binding of LAG-3 to stable peptide-MHC class II limits T cell function and suppresses autoimmunity and anti-cancer immunity. Immunity. 2022;55:912–24.e8.
Triebel F, Jitsukawa S, Baixeras E, Roman-Roman S, Genevee C, Viegas-Pequignot E, et al. LAG-3, a novel lymphocyte activation gene closely related to CD4. J Exp Med. 1990;171:1393–405.
Chocarro L, Blanco E, Zuazo M, Arasanz H, Bocanegra A, Fernández-Rubio L, et al. Understanding LAG-3 signaling. Int J Mol Sci. 2021;22:5282.
Huard B, Prigent P, Pagès F, Bruniquel D, Triebel F. T cell major histocompatibility complex class II molecules down-regulate CD4+ T cell clone responses following LAG-3 binding. Eur J Immunol. 1996;26:1180–6.
Kouo T, Huang L, Pucsek AB, Cao M, Solt S, Armstrong T, et al. Galectin-3 shapes antitumor immune responses by suppressing CD8+ T cells via LAG-3 and inhibiting expansion of plasmacytoid dendritic cells. Cancer Immunol Res. 2015;3:412–23.
Wang J, Sanmamed MF, Datar I, Su TT, Ji L, Sun J, et al. Fibrinogen-like protein 1 is a major immune inhibitory ligand of LAG-3. Cell. 2019;176:334–47.e12.
Paik J. Nivolumab plus relatlimab: first approval. Drugs. 2022;82:925–31.
Tawbi HA, Schadendorf D, Lipson EJ, Ascierto PA, Matamala L, Castillo Gutiérrez E, et al. Relatlimab and nivolumab versus nivolumab in untreated advanced melanoma. N Engl J Med. 2022;386:24–34.
Okagawa T, Konnai S, Nishimori A, Maekawa N, Goto S, Ikebuchi R, et al. Cooperation of PD-1 and LAG-3 in the exhaustion of CD4+ and CD8+ T cells during bovine leukemia virus infection. Vet Res. 2018;49:50.
Woo SR, Turnis ME, Goldberg MV, Bankoti J, Selby M, Nirschl CJ, et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 2012;72:917–27.
LAG3-PD-1 combo impresses in melanoma. Cancer Discov. 2021;11:1605–6.
Seth R, Agarwala SS, Messersmith H, Alluri KC, Ascierto PA, Atkins MB, et al. Systemic therapy for melanoma: ASCO guideline update. J Clin Oncol. 2023;41:4794–820.
Makaremi S, Asadzadeh Z, Hemmat N, Baghbanzadeh A, Sgambato A, Ghorbaninezhad F, et al. Immune checkpoint inhibitors in colorectal cancer: challenges and future prospects. Biomedicines. 2021;9:1075.
Garralda E, Sukari A, Lakhani NJ, Patnaik A, Lou Y, Im SA, et al. A first-in-human study of the anti-LAG-3 antibody favezelimab plus pembrolizumab in previously treated, advanced microsatellite stable colorectal cancer. ESMO Open. 2022;7:100639.
Gomes de Morais AL, Cerdá S, de Miguel M. New checkpoint inhibitors on the road: targeting TIM-3 in solid tumors. Curr Oncol Rep. 2022;24:651–8.
Zeidan AM, Komrokji RS, Brunner AM. TIM-3 pathway dysregulation and targeting in cancer. Expert Rev Anticancer Ther. 2021;21:523–34.
Kozłowski M, Borzyszkowska D, Cymbaluk-Płoska A. The role of TIM-3 and LAG-3 in the microenvironment and immunotherapy of ovarian cancer. Biomedicines. 2022;10:2826.
Zhu C, Anderson AC, Schubart A, Xiong H, Imitola J, Khoury SJ, et al. The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol. 2005;6:1245–52.
Huang YH, Zhu C, Kondo Y, Anderson AC, Gandhi A, Russell A, et al. CEACAM1 regulates TIM-3-mediated tolerance and exhaustion. Nature. 2015;517:386–90.
Das M, Zhu C, Kuchroo VK. Tim-3 and its role in regulating anti-tumor immunity. Immunol Rev. 2017;276:97–111.
Zhao L, Cheng S, Fan L, Zhang B, Xu S. TIM-3: an update on immunotherapy. Int Immunopharmacol. 2021;99:107933.
de Mingo Pulido Á, Hänggi K, Celias DP, Gardner A, Li J, Batista-Bittencourt B, et al. The inhibitory receptor TIM-3 limits activation of the cGAS-STING pathway in intra-tumoral dendritic cells by suppressing extracellular DNA uptake. Immunity. 2021;54:1154–67.e7.
Dixon KO, Tabaka M, Schramm MA, Xiao S, Tang R, Dionne D, et al. TIM-3 restrains anti-tumour immunity by regulating inflammasome activation. Nature. 2021;595:101–6.
Chen H, Zha J, Tang R, Chen G. T-cell immunoglobulin and mucin-domain containing-3 (TIM-3): Solving a key puzzle in autoimmune diseases. Int Immunopharmacol. 2023;121:110418.
Rangachari M, Zhu C, Sakuishi K, Xiao S, Karman J, Chen A, et al. Bat3 promotes T cell responses and autoimmunity by repressing Tim-3–mediated cell death and exhaustion. Nat Med. 2012;18:1394–400.
Wolf Y, Anderson AC, Kuchroo VK. TIM3 comes of age as an inhibitory receptor. Nat Rev Immunol. 2020;20:173–85.
Cheng L, Ruan Z. Tim-3 and Tim-4 as the potential targets for antitumor therapy. Hum Vaccin Immunother. 2015;11:2458–62.
Schwartz S, Patel N, Longmire T, Jayaraman P, Jiang X, Lu H, et al. Characterization of sabatolimab, a novel immunotherapy with immuno-myeloid activity directed against TIM-3 receptor. Immunother Adv. 2022;2:ltac019.
Curigliano G, Gelderblom H, Mach N, Doi T, Tai D, Forde PM, et al. Phase I/Ib clinical trial of sabatolimab, an anti-TIM-3 antibody, alone and in combination with spartalizumab, an anti-PD-1 antibody, in advanced solid tumors. Clin Cancer Res. 2021;27:3620–9.
Sakuishi K, Apetoh L, Sullivan JM, Blazar BR, Kuchroo VK, Anderson AC. Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J Exp Med. 2010;207:2187–94.
Stanietsky N, Simic H, Arapovic J, Toporik A, Levy O, Novik A, et al. The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc Natl Acad Sci USA. 2009;106:17858–63.
Lozano E, Dominguez-Villar M, Kuchroo V, Hafler DA. The TIGIT/CD226 axis regulates human T cell function. J Immunol. 2012;188:3869–75.
Harjunpää H, Guillerey C. TIGIT as an emerging immune checkpoint. Clin Exp Immunol. 2020;200:108–19.
Cho BC, Abreu DR, Hussein M, Cobo M, Patel AJ, Secen N, et al. Tiragolumab plus atezolizumab versus placebo plus atezolizumab as a first-line treatment for PD-L1-selected non-small-cell lung cancer (CITYSCAPE): primary and follow-up analyses of a randomised, double-blind, phase 2 study. Lancet Oncol. 2022;23:781–92.
Blando J, Sharma A, Higa MG, Zhao H, Vence L, Yadav SS, et al. Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc Natl Acad Sci USA. 2019;116:1692–7.
Wang J, Wu G, Manick B, Hernandez V, Renelt M, Erickson C, et al. VSIG-3 as a ligand of VISTA inhibits human T-cell function. Immunology. 2019;156:74–85.
Hosseinkhani N, Derakhshani A, Shadbad MA, Argentiero A, Racanelli V, Kazemi T, et al. The role of V-Domain Ig suppressor of T cell activation (VISTA) in cancer therapy: lessons learned and the road ahead. Front Immunol. 2021;12:676181.
Martin AS, Molloy M, Ugolkov A, von Roemeling RW, Noelle RJ, Lewis LD, et al. VISTA expression and patient selection for immune-based anticancer therapy. Front Immunol. 2023;14:1086102.
Fu T, He Q, Sharma P. The ICOS/ICOSL pathway is required for optimal antitumor responses mediated by anti-CTLA-4 therapy. Cancer Res. 2011;71:5445–54.
Liakou CI, Kamat A, Tang DN, Chen H, Sun J, Troncoso P, et al. CTLA-4 blockade increases IFNgamma-producing CD4+ICOShi cells to shift the ratio of effector to regulatory T cells in cancer patients. Proc Natl Acad Sci USA. 2008;105:14987–92.
Burmeister Y, Lischke T, Dahler AC, Mages HW, Lam KP, Coyle AJ, et al. ICOS controls the pool size of effector-memory and regulatory T cells. J Immunol. 2008;180:774–82.
Yap TA, Gainor JF, Callahan MK, Falchook GS, Pachynski RK, LoRusso P, et al. First-in-human phase I/II ICONIC trial of the ICOS agonist vopratelimab alone and with nivolumab: ICOS-high CD4 T-cell populations and predictors of response. Clin Cancer Res. 2022;28:3695–708.
Müller D. Targeting co-stimulatory receptors of the TNF superfamily for cancer immunotherapy. BioDrugs. 2023;37:21–33.
Salzman J. Circular RNA expression: its potential regulation and function. Trends Genet. 2016;32:309–16.
Vo JN, Cieslik M, Zhang Y, Shukla S, Xiao L, Zhang Y, et al. The landscape of circular RNA in cancer. Cell. 2019;176:869–81.e13.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74.
Cheng Z, Yu C, Cui S, Wang H, Jin H, Wang C, et al. circTP63 functions as a ceRNA to promote lung squamous cell carcinoma progression by upregulating FOXM1. Nat Commun. 2019;10:3200.
Chen J, Li Y, Zheng Q, Bao C, He J, Chen B, et al. Circular RNA profile identifies circPVT1 as a proliferative factor and prognostic marker in gastric cancer. Cancer Lett. 2017;388:208–19.
Chen S, Cao X, Zhang J, Wu W, Zhang B, Zhao F. circVAMP3 drives CAPRIN1 phase separation and inhibits hepatocellular carcinoma by suppressing c-Myc translation. Adv Sci (Weinh). 2022;9:e2103817.
Li S, Li X, Xue W, Zhang L, Yang LZ, Cao SM, et al. Screening for functional circular RNAs using the CRISPR-Cas13 system. Nat Methods. 2021;18:51–9.
Tsitsipatis D, Grammatikakis I, Driscoll RK, Yang X, Abdelmohsen K, Harris SC, et al. AUF1 ligand circPCNX reduces cell proliferation by competing with p21 mRNA to increase p21 production. Nucleic Acids Res. 2021;49:1631–46.
Zhang M, Zhao K, Xu X, Yang Y, Yan S, Wei P, et al. A peptide encoded by circular form of LINC-PINT suppresses oncogenic transcriptional elongation in glioblastoma. Nat Commun. 2018;9:4475.
Xie F, Li Y, Wang M, Huang C, Tao D, Zheng F, et al. Circular RNA BCRC-3 suppresses bladder cancer proliferation through miR-182-5p/p27 axis. Mol Cancer. 2018;17:144.
Zheng Q, Bao C, Guo W, Li S, Chen J, Chen B, et al. Circular RNA profiling reveals an abundant circHIPK3 that regulates cell growth by sponging multiple miRNAs. Nat Commun. 2016;7:11215.
Liu H, Hu G, Wang Z, Liu Q, Zhang J, Chen Y, et al. circPTCH1 promotes invasion and metastasis in renal cell carcinoma via regulating miR-485-5p/MMP14 axis. Theranostics. 2020;10:10791–807.
Zhu Z, Rong Z, Luo Z, Yu Z, Zhang J, Qiu Z, et al. Circular RNA circNHSL1 promotes gastric cancer progression through the miR-1306-3p/SIX1/vimentin axis. Mol Cancer. 2019;18:126.
Wen J, Liao J, Liang J, Chen XP, Zhang B, Chu L. Circular RNA HIPK3: a key circular RNA in a variety of human cancers. Front Oncol. 2020;10:773.
Zeng K, Chen X, Xu M, Liu X, Hu X, Xu T, et al. CircHIPK3 promotes colorectal cancer growth and metastasis by sponging miR-7. Cell Death Dis. 2018;9:417.
Jin P, Huang Y, Zhu P, Zou Y, Shao T, Wang O. CircRNA circHIPK3 serves as a prognostic marker to promote glioma progression by regulating miR-654/IGF2BP3 signaling. Biochem Biophys Res Commun. 2018;503:1570–4.
Liu F, Zhang H, Xie F, Tao D, Xiao X, Huang C, et al. Hsa_circ_0001361 promotes bladder cancer invasion and metastasis through miR-491-5p/MMP9 axis. Oncogene. 2020;39:1696–709.
Han K, Wang FW, Cao CH, Ling H, Chen JW, Chen RX, et al. CircLONP2 enhances colorectal carcinoma invasion and metastasis through modulating the maturation and exosomal dissemination of microRNA-17. Mol Cancer. 2020;19:60.
Zeng K, He B, Yang BB, Xu T, Chen X, Xu M, et al. The pro-metastasis effect of circANKS1B in breast cancer. Mol Cancer. 2018;17:160.
Zheng L, Liang H, Zhang Q, Shen Z, Sun Y, Zhao X, et al. circPTEN1, a circular RNA generated from PTEN, suppresses cancer progression through inhibition of TGF-β/Smad signaling. Mol Cancer. 2022;21:41.
Xue C, Li G, Lu J, Li L. Crosstalk between circRNAs and the PI3K/AKT signaling pathway in cancer progression. Signal Transduct Target Ther. 2021;6:400.
He Y, Mingyan E, Wang C, Liu G, Shi M, Liu S. CircVRK1 regulates tumor progression and radioresistance in esophageal squamous cell carcinoma by regulating miR-624-3p/PTEN/PI3K/AKT signaling pathway. Int J Biol Macromol. 2019;125:116–23.
Chen Z, Yao N, Gu H, Song Y, Ye Z, Li L, et al. Circular RNA_LARP4 sponges miR-1323 and hampers progression of esophageal squamous cell carcinoma through modulating PTEN/PI3K/AKT pathway. Dig Dis Sci. 2020;65:2272–83.
Cheng J, Ma H, Yan M, Zhang Z, Xing W. Circ_0007624 suppresses the development of esophageal squamous cell carcinoma via targeting miR-224-5p/CPEB3 to inactivate the EGFR/PI3K/AKT signaling. Cell Signal. 2022;99:110448.
Pan H, Li T, Jiang Y, Pan C, Ding Y, Huang Z, et al. Overexpression of circular RNA ciRS-7 abrogates the tumor suppressive effect of miR-7 on gastric cancer via PTEN/PI3K/AKT signaling pathway. J Cell Biochem. 2018;119:440–6.
Wang X, Zhang Y, Li W, Liu X. Knockdown of cir_RNA PVT1 elevates gastric cancer cisplatin sensitivity via sponging miR-152-3p. J Surg Res. 2021;261:185–95.
Song H, Xu Y, Xu T, Fan R, Jiang T, Cao M, et al. CircPIP5K1A activates KRT80 and PI3K/AKT pathway to promote gastric cancer development through sponging miR-671-5p. Biomed Pharmacother. 2020;126:109941.
Jiang T, Wang H, Liu L, Song H, Zhang Y, Wang J, et al. CircIL4R activates the PI3K/AKT signaling pathway via the miR-761/TRIM29/PHLPP1 axis and promotes proliferation and metastasis in colorectal cancer. Mol Cancer. 2021;20:167.
Wang J, Luo J, Liu G, Li X. Circular RNA hsa_circ_0008285 inhibits colorectal cancer cell proliferation and migration via the miR-382-5p/PTEN axis. Biochem Biophys Res Commun. 2020;527:503–10.
Tu FL, Guo XQ, Wu HX, He ZY, Wang F, Sun AJ, et al. Circ-0001313/miRNA-510-5p/AKT2 axis promotes the development and progression of colon cancer. Am J Transl Res. 2020;12:281–91.
Wei Y, Chen X, Liang C, Ling Y, Yang X, Ye X, et al. A noncoding regulatory RNAs network driven by Circ-CDYL acts specifically in the early stages hepatocellular carcinoma. Hepatology. 2020;71:130–47.
Lin Q, Ling YB, Chen JW, Zhou CR, Chen J, Li X, et al. Circular RNA circCDK13 suppresses cell proliferation, migration and invasion by modulating the JAK/STAT and PI3K/AKT pathways in liver cancer. Int J Oncol. 2018;53:246–56.
Tan Y, Du B, Zhan Y, Wang K, Wang X, Chen B, et al. Antitumor effects of circ-EPHB4 in hepatocellular carcinoma via inhibition of HIF-1α. Mol Carcinog. 2019;58:875–86.
Guo X, Zhou Q, Su D, Luo Y, Fu Z, Huang L, et al. Circular RNA circBFAR promotes the progression of pancreatic ductal adenocarcinoma via the miR-34b-5p/MET/Akt axis. Mol Cancer. 2020;19:83.
Zhang T, Li M, Lu H, Peng T. Up-regulation of circEIF6 contributes to pancreatic cancer development through targeting miR-557/SLC7A11/PI3K/AKT signaling. Cancer Manag Res. 2021;13:247–58.
Kong Y, Li Y, Luo Y, Zhu J, Zheng H, Gao B, et al. circNFIB1 inhibits lymphangiogenesis and lymphatic metastasis via the miR-486-5p/PIK3R1/VEGF-C axis in pancreatic cancer. Mol Cancer. 2020;19:82.
Shi F, Shi Z, Zhao Y, Tian J. CircRNA hsa-circ-0014359 promotes glioma progression by regulating miR-153/PI3K signaling. Biochem Biophys Res Commun. 2019;510:614–20.
Yin H, Cui X. Knockdown of circHIPK3 facilitates temozolomide sensitivity in glioma by regulating cellular behaviors through miR-524-5p/KIF2A-mediated PI3K/AKT pathway. Cancer Biother Radiopharm. 2021;36:556–67.
Zheng K, Xie H, Wu W, Wen X, Zeng Z, Shi Y. CircRNA PIP5K1A promotes the progression of glioma through upregulation of the TCF12/PI3K/AKT pathway by sponging miR-515-5p. Cancer Cell Int. 2021;21:27.
Mutalifu N, Du P, Zhang J, Akbar H, Yan B, Alimu S, et al. Circ_0000215 increases the expression of CXCR2 and promoted the progression of glioma cells by sponging miR-495-3p. Technol Cancer Res Treat. 2020;19:1533033820957026.
Mirzaei S, Zarrabi A, Hashemi F, Zabolian A, Saleki H, Ranjbar A, et al. Regulation of nuclear factor-kappaB (NF-κB) signaling pathway by non-coding RNAs in cancer: Inhibiting or promoting carcinogenesis? Cancer Lett. 2021;509:63–80.
Lin C, Wei Y, Duan X, Liu C, Du Y, Wang X, et al. Circ_0001821 affects proliferation and the cell cycle in esophageal squamous cell carcinoma by elevating BTRC-mediated IKBA ubiquitination. Mol Cancer Res. 2022;20:1686–96.
Meng F, Zhang X, Wang Y, Lin J, Tang Y, Zhang G, et al. Hsa_circ_0021727 (circ-CD44) promotes ESCC progression by targeting miR-23b-5p to activate the TAB1/NFκB pathway. Cell Death Dis. 2023;14:9.
Li RC, Ke S, Meng FK, Lu J, Zou XJ, He ZG, et al. CiRS-7 promotes growth and metastasis of esophageal squamous cell carcinoma via regulation of miR-7/HOXB13. Cell Death Dis. 2018;9:838.
Gu L, Sang Y, Nan X, Zheng Y, Liu F, Meng L, et al. circCYP24A1 facilitates esophageal squamous cell carcinoma progression through binding PKM2 to regulate NF-κB-induced CCL5 secretion. Mol Cancer. 2022;21:217.
Zhou Y, Tang W, Zhuo H, Zhu D, Rong D, Sun J, et al. Cancer-associated fibroblast exosomes promote chemoresistance to cisplatin in hepatocellular carcinoma through circZFR targeting signal transducers and activators of transcription (STAT3)/ nuclear factor -kappa B (NF-κB) pathway. Bioengineered. 2022;13:4786–97.
Ji J, Tang J, Ren P, Cai W, Shen M, Wang Q, et al. Downregulation of circLIFR exerts cancer-promoting effects on hepatocellular carcinoma in vitro. Front Genet. 2022;13:986322.
Wu F, Sun G, Zheng W, Tang W, Cheng Y, Wu L, et al. circCORO1C promotes the proliferation and metastasis of hepatocellular carcinoma by enhancing the expression of PD-L1 through NF-κB pathway. J Clin Lab Anal. 2021;35:e24003.
Zhao S, Li B, Zhao R, Pan Z, Zhang S, Qiu W, et al. Hypoxia-induced circADAMTS6 in a TDP43-dependent manner accelerates glioblastoma progression via ANXA2/NF-κB pathway. Oncogene. 2023;42:138–53.
Chen D, Shi L, Zhong D, Nie Y, Yang Y, Liu D. Hsa_circ_0002019 promotes cell proliferation, migration, and invasion by regulating TNFAIP6/NF-κB signaling in gastric cancer. Genomics. 2023;115:110641.
Chen J, Yang X, Liu R, Wen C, Wang H, Huang L, et al. Circular RNA GLIS2 promotes colorectal cancer cell motility via activation of the NF-κB pathway. Cell Death Dis. 2020;11:788.
Zhang X, Liu X, Jing Z, Bi J, Li Z, Liu X, et al. The circINTS4/miR-146b/CARMA3 axis promotes tumorigenesis in bladder cancer. Cancer Gene Ther. 2020;27:189–202.
Guo K, Shi J, Tang Z, Lai C, Liu C, Li K, et al. Circular RNA circARHGEF28 inhibited the progression of prostate cancer via the miR-671-5p/LGALS3BP/NF-κB axis. Cancer Sci. 2023;114:2907–19.
Ai Y, Song J, Wei H, Tang Z, Li X, Lv X, et al. circ_0001461 promotes oral squamous cell carcinoma progression through miR-145/TLR4/NF-κB axis. Biochem Biophys Res Commun. 2021;566:108–14.
Chen T, Feng G, Xing Z, Gao X. Circ-EIF3I facilitates proliferation, migration, and invasion of lung cancer via regulating the activity of Wnt/β-catenin pathway through the miR-1253/NOVA2 axis. Thorac Cancer. 2022;13:3133–44.
Zhao Y, Dai Q, Fu X, Chen Q, Tang Y, Gao X, et al. CircVAPA exerts oncogenic property in non-small cell lung cancer by the miR-876-5p/WNT5A axis. J Gene Med. 2021;23:e3325.
Gao F, Jia L, Han J, Wang Y, Luo W, Zeng Y. Circ-ZNF124 downregulation inhibits non-small cell lung cancer progression partly by inactivating the Wnt/β-catenin signaling pathway via mediating the miR-498/YES1 axis. Anticancer Drugs. 2021;32:257–68.
Wan L, Zhang L, Fan K, Cheng ZX, Sun QC, Wang JJ. Circular RNA-ITCH suppresses lung cancer proliferation via inhibiting the Wnt/β-catenin pathway. Biomed Res Int. 2016;2016:1579490.
Li D, Zhang J, Yang J, Wang J, Zhang R, Li J, et al. CircMTO1 suppresses hepatocellular carcinoma progression via the miR-541-5p/ZIC1 axis by regulating Wnt/β-catenin signaling pathway and epithelial-to-mesenchymal transition. Cell Death Dis. 2021;13:12.
Huang G, Liang M, Liu H, Huang J, Li P, Wang C, et al. CircRNA hsa_circRNA_104348 promotes hepatocellular carcinoma progression through modulating miR-187-3p/RTKN2 axis and activating Wnt/β-catenin pathway. Cell Death Dis. 2020;11:1065.
Xu J, Wan Z, Tang M, Lin Z, Jiang S, Ji L, et al. N6-methyladenosine-modified CircRNA-SORE sustains sorafenib resistance in hepatocellular carcinoma by regulating β-catenin signaling. Mol Cancer. 2020;19:163.
Tan A, Li Q, Chen L. CircZFR promotes hepatocellular carcinoma progression through regulating miR-3619-5p/CTNNB1 axis and activating Wnt/β-catenin pathway. Arch Biochem Biophys. 2019;661:196–202.
Peng Y, Xu Y, Zhang X, Deng S, Yuan Y, Luo X, et al. A novel protein AXIN1-295aa encoded by circAXIN1 activates the Wnt/β-catenin signaling pathway to promote gastric cancer progression. Mol Cancer. 2021;20:158.
Wang L, Li B, Yi X, Xiao X, Zheng Q, Ma L. Circ_SMAD4 promotes gastric carcinogenesis by activating wnt/β-catenin pathway. Cell Prolif. 2021;54:e12981.
Peng Y, Wang HH. Cir-ITCH inhibits gastric cancer migration, invasion and proliferation by regulating the Wnt/β-catenin pathway. Sci Rep. 2020;10:17443.
Shi Q, Zhou C, Xie R, Li M, Shen P, Lu Y, et al. CircCNIH4 inhibits gastric cancer progression via regulating DKK2 and FRZB expression and Wnt/β-catenin pathway. J Biol Res (Thessalon). 2021;28:19.
Zhang G, Liu Z, Zhong J, Lin L. Circ-ACAP2 facilitates the progression of colorectal cancer through mediating miR-143-3p/FZD4 axis. Eur J Clin Invest. 2021;51:e13607.
Zhang L, Dong X, Yan B, Yu W, Shan L. CircAGFG1 drives metastasis and stemness in colorectal cancer by modulating YY1/CTNNB1. Cell Death Dis. 2020;11:542.
Ma Z, Han C, Xia W, Wang S, Li X, Fang P, et al. circ5615 functions as a ceRNA to promote colorectal cancer progression by upregulating TNKS. Cell Death Dis. 2020;11:356.
Geng Y, Zheng X, Hu W, Wang Q, Xu Y, He W, et al. Hsa_circ_0009361 acts as the sponge of miR-582 to suppress colorectal cancer progression by regulating APC2 expression. Clin Sci (Lond). 2019;133:1197–213.
He J, Huang Z, He M, Liao J, Zhang Q, Wang S, et al. Circular RNA MAPK4 (circ-MAPK4) inhibits cell apoptosis via MAPK signaling pathway by sponging miR-125a-3p in gliomas. Mol Cancer. 2020;19:17.
Chen C, Deng L, Nie DK, Jia F, Fu LS, Wan ZQ, et al. Circular RNA Pleiotrophin promotes carcinogenesis in glioma via regulation of microRNA-122/SRY-box transcription factor 6 axis. Eur J Cancer Prev. 2020;29:165–73.
Wang Z, Chen X, Liang Q, An Y, Wei M, Shi W. Inhibiting of circ-TLK1 inhibits the progression of glioma through down-regulating PANX1 via targeting miR-17-5p. J Mol Histol. 2021;52:1007–20.
Guo Z, Xie Q, Wu Y, Mo H, Zhang J, He G, et al. Aberrant expression of circular RNA DHPR facilitates tumor growth and metastasis by regulating the RASGEF1B/RAS/MAPK axis in hepatocellular carcinoma. Cell Oncol (Dordr). 2023. https://doi.org/10.1007/s13402-023-00814-9.
Hu ZQ, Zhou SL, Li J, Zhou ZJ, Wang PC, Xin HY, et al. Circular RNA sequencing identifies CircASAP1 as a key regulator in hepatocellular carcinoma metastasis. Hepatology. 2020;72:906–22.
Xu L, Feng X, Hao X, Wang P, Zhang Y, Zheng X, et al. CircSETD3 (Hsa_circ_0000567) acts as a sponge for microRNA-421 inhibiting hepatocellular carcinoma growth. J Exp Clin Cancer Res. 2019;38:98.
Wang B, Chen H, Deng Y, Chen H, Xing L, Guo Y, et al. CircDNAJC11 interacts with TAF15 to promote breast cancer progression via enhancing MAPK6 expression and activating the MAPK signaling pathway. J Transl Med. 2023;21:186.
Lv W, Tan Y, Xiong M, Zhao C, Wang Y, Wu M, et al. Analysis and validation of m6A regulatory network: a novel circBACH2/has-miR-944/HNRNPC axis in breast cancer progression. J Transl Med. 2021;19:527.
Gao D, Qi X, Zhang X, Fang K, Guo Z, Li L. hsa_circRNA_0006528 as a competing endogenous RNA promotes human breast cancer progression by sponging miR-7-5p and activating the MAPK/ERK signaling pathway. Mol Carcinog. 2019;58:554–64.
Ishola AA, Chien CS, Yang YP, Chien Y, Yarmishyn AA, Tsai PH, et al. Oncogenic circRNA C190 promotes non-small cell lung cancer via modulation of the EGFR/ERK pathway. Cancer Res. 2022;82:75–89.
Wang Y, Xu R, Zhang D, Lu T, Yu W, Wo Y, et al. Circ-ZKSCAN1 regulates FAM83A expression and inactivates MAPK signaling by targeting miR-330-5p to promote non-small cell lung cancer progression. Transl Lung Cancer Res. 2019;8:862–75.
Zhang S, Liu J, Yuan T, Liu H, Wan C, Le Y. Circular RNA 0001313 knockdown suppresses non-small cell lung cancer cell proliferation and invasion via the microRNA-452/HMGB3/ERK/MAPK axis. Int J Gen Med. 2020;13:1495–507.
Jiang T, Xia Y, Lv J, Li B, Li Y, Wang S, et al. A novel protein encoded by circMAPK1 inhibits progression of gastric cancer by suppressing activation of MAPK signaling. Mol Cancer. 2021;20:66.
Jiang F, Liu G, Chen X, Li Q, Fang F, Shen X. Hsa_circ_0044301 regulates gastric cancer cell’s proliferation, migration, and invasion by modulating the Hsa-miR-188-5p/DAXX Axis and MAPK pathway. Cancers (Basel). 2022;14:4183.
Xu Q, Yao Y, Ni H, Gu J, Wang X, Jiang L, et al. Hsa-circ-0052001 promotes gastric cancer cell proliferation and invasion via the MAPK pathway. Cancer Med. 2023;12:7246–57.
Gao W, Wang X, Zhou Y, Wang X, Yu Y. Autophagy, ferroptosis, pyroptosis, and necroptosis in tumor immunotherapy. Signal Transduct Target Ther. 2022;7:196.
Yaylak B, Erdogan I, Akgul B. Transcriptomics analysis of circular RNAs differentially expressed in apoptotic HeLa cells. Front Genet. 2019;10:176.
Yang T, Li Y, Zhao F, Zhou L, Jia R. Circular RNA Foxo3: a promising cancer-associated biomarker. Front Genet. 2021;12:652995.
Zheng J, Liu X, Xue Y, Gong W, Ma J, Xi Z, et al. TTBK2 circular RNA promotes glioma malignancy by regulating miR-217/HNF1β/Derlin-1 pathway. J Hematol Oncol. 2017;10:52.
Feng J, Zhou J, Wu Y, Shen HM, Peng T, Lu GD. Targeting mitophagy as a novel therapeutic approach in liver cancer. Autophagy. 2023;19:2164–5.
Zandieh MA, Farahani MH, Rajabi R, Avval ST, Karimi K, Rahmanian P, et al. Epigenetic regulation of autophagy by non-coding RNAs in gastrointestinal tumors: Biological functions and therapeutic perspectives. Pharm Res. 2023;187:106582.
Pan Z, Zheng J, Zhang J, Lin J, Lai J, Lyu Z, et al. A novel protein encoded by exosomal CircATG4B induces oxaliplatin resistance in colorectal cancer by promoting autophagy. Adv Sci (Weinh). 2022;9:e2204513.
He Z, Cai K, Zeng Z, Lei S, Cao W, Li X. Autophagy-associated circRNA circATG7 facilitates autophagy and promotes pancreatic cancer progression. Cell Death Dis. 2022;13:233.
Liang G, Ling Y, Mehrpour M, Saw PE, Liu Z, Tan W, et al. Autophagy-associated circRNA circCDYL augments autophagy and promotes breast cancer progression. Mol Cancer. 2020;19:65.
Tuncer M, Alcan S. Pyroptosis: a new therapeutic strategy in cancer. Mol Biol Rep. 2023;50:6191–6200.
Zhang Z, Chen Q, Huang C, Rao D, Sang C, Zhu S, et al. Transcription factor Nrf2 binds to circRNAPIBF1 to regulate SOD2 in lung adenocarcinoma progression. Mol Carcinog. 2022;61:1161–76.
Dai C, Ma Z, Si J, An G, Zhang W, Li S, et al. Hsa_circ_0007312 promotes third-generation epidermal growth factor receptor-tyrosine kinase inhibitor resistance through pyroptosis and apoptosis via the MiR-764/MAPK1 axis in lung adenocarcinoma cells. J Cancer. 2022;13:2798–809.
Zhang T, Wu DM, Luo PW, Liu T, Han R, Deng SH, et al. CircNEIL3 mediates pyroptosis to influence lung adenocarcinoma radiotherapy by upregulating PIF1 through miR-1184 inhibition. Cell Death Dis. 2022;13:167.
Gong W, Xu J, Wang Y, Min Q, Chen X, Zhang W, et al. Nuclear genome-derived circular RNA circPUM1 localizes in mitochondria and regulates oxidative phosphorylation in esophageal squamous cell carcinoma. Signal Transduct Target Ther. 2022;7:40.
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell. 2017;171:273–85.
Zhang Y, Shi J, Liu X, Feng L, Gong Z, Koppula P, et al. BAP1 links metabolic regulation of ferroptosis to tumour suppression. Nat Cell Biol. 2018;20:1181–92.
Farooqi AA, Kapanova G, Kalmakhanov S, Kussainov AZ, Datkhayeva Z. Regulation of ferroptosis by non-coding RNAs: mechanistic insights. J Pharm Exp Ther. 2023;384:20–27.
Liu B, Ma H, Liu X, Xing W. CircSCN8A suppresses malignant progression and induces ferroptosis in non-small cell lung cancer by regulating miR-1290/ACSL4 axis. Cell Cycle. 2022;22:758–76.
Wang L, Wu S, He H, Ai K, Xu R, Zhang L, et al. CircRNA-ST6GALNAC6 increases the sensitivity of bladder cancer cells to erastin-induced ferroptosis by regulating the HSPB1/P38 axis. Lab Invest. 2022;102:1323–34.
Zhang X, Xu Y, Ma L, Yu K, Niu Y, Xu X, et al. Essential roles of exosome and circRNA_101093 on ferroptosis desensitization in lung adenocarcinoma. Cancer Commun (Lond). 2022;42:287–313.
Pęczek P, Gajda M, Rutkowski K, Fudalej M, Deptała A, Badowska-Kozakiewicz AM. Cancer-associated inflammation: pathophysiology and clinical significance. J Cancer Res Clin Oncol. 2023;149:2657–72.
Munn LL. Cancer and inflammation. Wiley Interdiscip Rev Syst Biol Med. 2017;9. https://doi.org/10.1002/wsbm.1370.
Qadir J, Wen SY, Yuan H, Yang BB. CircRNAs regulate the crosstalk between inflammation and tumorigenesis: the bilateral association and molecular mechanisms. Mol Ther. 2023;31:1514–32.
Zhao H, Wu L, Yan G, Chen Y, Zhou M, Wu Y, et al. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduct Target Ther. 2021;6:263.
Wang S, Feng X, Wang Y, Li Q, Li X. Dysregulation of tumour microenvironment driven by circ-TPGS2/miR-7/TRAF6/NF-κB axis facilitates breast cancer cell motility. Autoimmunity. 2021;54:284–93.
Liu T, Ye P, Ye Y, Lu S, Han B. Circular RNA hsa_circRNA_002178 silencing retards breast cancer progression via microRNA-328-3p-mediated inhibition of COL1A1. J Cell Mol Med. 2020;24:2189–201.
Wan D, Wang S, Xu Z, Zan X, Liu F, Han Y, et al. PRKAR2A-derived circular RNAs promote the malignant transformation of colitis and distinguish patients with colitis-associated colorectal cancer. Clin Transl Med. 2022;12:e683.
Liu J, Zhang Q, Yang D, Xie F, Wang Z. The role of long non-coding RNAs in angiogenesis and anti-angiogenic therapy resistance in cancer. Mol Ther Nucleic Acids. 2022;28:397–407.
Teleanu RI, Chircov C, Grumezescu AM, Teleanu DM. Tumor angiogenesis and anti-angiogenic strategies for cancer treatment. J Clin Med. 2019;9:84.
Lugano R, Ramachandran M, Dimberg A. Tumor angiogenesis: causes, consequences, challenges and opportunities. Cell Mol Life Sci. 2020;77:1745–70.
Jiang S, Fu R, Shi J, Wu H, Mai J, Hua X, et al. CircRNA-mediated regulation of angiogenesis: a new chapter in cancer biology. Front Oncol. 2021;11:553706.
Zhong Z, Huang M, Lv M, He Y, Duan C, Zhang L, et al. Circular RNA MYLK as a competing endogenous RNA promotes bladder cancer progression through modulating VEGFA/VEGFR2 signaling pathway. Cancer Lett. 2017;403:305–17.
Guo Y, Guo Y, Chen C, Fan D, Wu X, Zhao L, et al. Circ3823 contributes to growth, metastasis and angiogenesis of colorectal cancer: involvement of miR-30c-5p/TCF7 axis. Mol Cancer. 2021;20:93.
Li Y, Zheng F, Xiao X, Xie F, Tao D, Huang C, et al. CircHIPK3 sponges miR-558 to suppress heparanase expression in bladder cancer cells. EMBO Rep. 2017;18:1646–59.
Chen Y, Li Z, Zhang M, Wang B, Ye J, Zhang Y, et al. Circ-ASH2L promotes tumor progression by sponging miR-34a to regulate Notch1 in pancreatic ductal adenocarcinoma. J Exp Clin Cancer Res. 2019;38:466.
Xu Y, Leng K, Yao Y, Kang P, Liao G, Han Y, et al. A circular RNA, cholangiocarcinoma-associated circular RNA 1, contributes to cholangiocarcinoma progression, induces angiogenesis, and disrupts vascular endothelial barriers. Hepatology. 2021;73:1419–35.
Xie M, Yu T, Jing X, Ma L, Fan Y, Yang F, et al. Exosomal circSHKBP1 promotes gastric cancer progression via regulating the miR-582-3p/HUR/VEGF axis and suppressing HSP90 degradation. Mol Cancer. 2020;19:112.
Jin MZ, Jin WL. The updated landscape of tumor microenvironment and drug repurposing. Signal Transduct Target Ther. 2020;5:166.
Balkwill FR, Capasso M, Hagemann T. The tumor microenvironment at a glance. J Cell Sci. 2012;125:5591–6.
Todoric J, Karin M. The fire within: cell-autonomous mechanisms in inflammation-driven cancer. Cancer Cell. 2019;35:714–20.
Deng S, Clowers MJ, Velasco WV, Ramos-Castaneda M, Moghaddam SJ. Understanding the complexity of the tumor microenvironment in K-ras mutant lung cancer: finding an alternative path to prevention and treatment. Front Oncol. 2020;9:1556.
Zhang Q, Wang W, Zhou Q, Chen C, Yuan W, Liu J, et al. Roles of circRNAs in the tumour microenvironment. Mol Cancer. 2020;19:14.
Xie T, Fu DJ, Li ZM, Lv DJ, Song XL, Yu YZ, et al. CircSMARCC1 facilitates tumor progression by disrupting the crosstalk between prostate cancer cells and tumor-associated macrophages via miR-1322/CCL20/CCR6 signaling [published correction appears in Mol Cancer. 2023 Oct 23;22(1):173]. Mol Cancer 2022;21:173.
Pan Z, Zhao R, Li B, Qi Y, Qiu W, Guo Q, et al. EWSR1-induced circNEIL3 promotes glioma progression and exosome-mediated macrophage immunosuppressive polarization via stabilizing IGF2BP3. Mol Cancer. 2022;21:16.
Hsu PP, Sabatini DM. Cancer cell metabolism: Warburg and beyond. Cell. 2008;134:703–7.
Faubert B, Solmonson A, DeBerardinis RJ. Metabolic reprogramming and cancer progression. Science. 2020;368:eaaw5473.
Yu T, Wang Y, Fan Y, Fang N, Wang T, Xu T, et al. CircRNAs in cancer metabolism: a review. J Hematol Oncol. 2019;12:90.
Shen S, Yao T, Xu Y, Zhang D, Fan S, Ma J. CircECE1 activates energy metabolism in osteosarcoma by stabilizing c-Myc. Mol Cancer. 2020;19:151.
Zhang X, Wang S, Wang H, Cao J, Huang X, Chen Z, et al. Circular RNA circNRIP1 acts as a microRNA-149-5p sponge to promote gastric cancer progression via the AKT1/mTOR pathway. Mol Cancer. 2019;18:20.
Wang S, Zhang Y, Cai Q, Ma M, Jin LY, Weng M, et al. Circular RNA FOXP1 promotes tumor progression and Warburg effect in gallbladder cancer by regulating PKLR expression. Mol Cancer. 2019;18:145.
Zhu J, Huang Q, Liu S, Peng X, Xue J, Feng T, et al. Construction of a Novel LncRNA signature related to genomic instability to predict the prognosis and immune activity of patients with hepatocellular carcinoma. Front Immunol. 2022;13:856186.
Petermann E, Lan L, Zou L. Sources, resolution and physiological relevance of R-loops and RNA-DNA hybrids. Nat Rev Mol Cell Biol. 2022;23:521–40.
Conn VM, Gabryelska M, Toubia J. Circular RNAs drive oncogenic chromosomal translocations within the MLL recombinome in leukemia. Cancer Cell. 2023;41:1309–.e10.
Tang X, Guo M, Ding P. BUB1B and circBUB1B_544aa aggravate multiple myeloma malignancy through evoking chromosomal instability. Signal Transduct Target Ther. 2021;6:361.
Gu C, Wang W, Tang X, Xu T, Zhang Y, Guo M, et al. CHEK1 and circCHEK1_246aa evoke chromosomal instability and induce bone lesion formation in multiple myeloma. Mol Cancer. 2021;20:84.
Jiang W, Pan S, Chen X, Wang ZW, Zhu X. The role of lncRNAs and circRNAs in the PD-1/PD-L1 pathway in cancer immunotherapy. Mol Cancer. 2021;20:116.
Yang J, Jia Y, Wang B, Yang S, Du K, Luo Y, et al. Circular RNA CHST15 sponges miR-155-5p and miR-194-5p to promote the immune escape of lung cancer cells mediated by PD-L1. Front Oncol. 2021;11:595609.
Hong W, Xue M, Jiang J, Zhang Y, Gao X. Circular RNA circ-CPA4/ let-7 miRNA/PD-L1 axis regulates cell growth, stemness, drug resistance and immune evasion in non-small cell lung cancer (NSCLC). J Exp Clin Cancer Res. 2020;39:149.
Li L, Zhang Q, Lian K. Circular RNA circ_0000284 plays an oncogenic role in the progression of non-small cell lung cancer through the miR-377-3p-mediated PD-L1 promotion. Cancer Cell Int. 2020;20:247.
Li J, Xu J, Wu G, Ren Y, Wang X, Zhang Q. Circular RNA hsa_circ_0068252 functions in cisplatin resistance and immune response via miR-1304-5p/PD-L1 axis in non-small cell lung cancer. Chemotherapy. 2022;67:223–33.
Zhang N, Fan J, Deng Z. CircFOXK2 enhances tumorigenesis and immune evasion in non-small cell lung cancer by miR-485-5p/PD-L1 axis. Anticancer Drugs. 2022;33:437–47.
Xu G, Zhang P, Liang H, Xu Y, Shen J, Wang W, et al. Circular RNA hsa_circ_0003288 induces EMT and invasion by regulating hsa_circ_0003288/miR-145/PD-L1 axis in hepatocellular carcinoma. Cancer Cell Int. 2021;21:212.
Fu X, Sun G, Tu S, Fang K, Xiong Y, Tu Y, et al. Hsa_circ_0046523 mediates an immunosuppressive tumor microenvironment by regulating MiR-148a-3p/PD-L1 axis in pancreatic cancer. Front Oncol. 2022;12:877376.
Tanaka E, Miyakawa Y, Kishikawa T, Seimiya T, Iwata T, Funato K, et al. Expression of circular RNA CDR1-AS in colon cancer cells increases cell surface PD-L1 protein levels. Oncol Rep. 2019;42:1459–66.
Xu YJ, Zhao JM, Gao C, Ni XF, Wang W, Hu WW, et al. Hsa_circ_0136666 activates Treg-mediated immune escape of colorectal cancer via miR-497/PD-L1 pathway. Cell Signal. 2021;86:110095.
Yang K, Zhang J, Bao C. Exosomal circEIF3K from cancer-associated fibroblast promotes colorectal cancer (CRC) progression via miR-214/PD-L1 axis. BMC Cancer. 2021;21:933.
Zhang XL, Xu LL, Wang F. Hsa_circ_0020397 regulates colorectal cancer cell viability, apoptosis and invasion by promoting the expression of the miR-138 targets TERT and PD-L1. Cell Biol Int. 2017;41:1056–64.
Zhang DJ, Fu ZM, Guo YY, Guo F, Wan YN, Guan GF. Circ_0000052/miR-382-3p axis induces PD-L1 expression and regulates cell proliferation and immune evasion in head and neck squamous cell carcinoma. J Cell Mol Med. 2023;27:113–26.
Huang L, Ma J, Cui M. Circular RNA hsa_circ_0001598 promotes programmed death-ligand-1-mediated immune escape and trastuzumab resistance via sponging miR-1184 in breast cancer cells. Immunol Res. 2021;69:558–67.
Liu Q, You B, Meng J, Huang CP, Dong G, Wang R, et al. Targeting the androgen receptor to enhance NK cell killing efficacy in bladder cancer by modulating ADAR2/circ_0001005/PD-L1 signaling. Cancer Gene Ther. 2022;29:1988–2000.
Dong L, Huang J, Gao X, Du J, Wang Y, Zhao L. CircPCBP2 promotes the stemness and chemoresistance of DLBCL via targeting miR-33a/b to disinhibit PD-L1. Cancer Sci. 2022;113:2888–903.
Gao C, Xu YJ, Qi L, Bao YF, Zhang L, Zheng L. CircRNA VIM silence synergizes with sevoflurane to inhibit immune escape and multiple oncogenic activities of esophageal cancer by simultaneously regulating miR-124/PD-L1 axis. Cell Biol Toxicol. 2022;38:825–45.
Zhang PF, Pei X, Li KS, Jin LN, Wang F, Wu J, et al. Circular RNA circFGFR1 promotes progression and anti-PD-1 resistance by sponging miR-381-3p in non-small cell lung cancer cells. Mol Cancer. 2019;18:179.
Niu R, Li D, Chen J, Zhao W. Circ_0014235 confers Gefitinib resistance and malignant behaviors in non-small cell lung cancer resistant to Gefitinib by governing the miR-146b-5p/YAP/PD-L1 pathway. Cell Cycle. 2022;21:86–100.
Liu Z, Wang T, She Y, Wu K, Gu S, Li L, et al. N6-methyladenosine-modified circIGF2BP3 inhibits CD8+ T-cell responses to facilitate tumor immune evasion by promoting the deubiquitination of PD-L1 in non-small cell lung cancer. Mol Cancer. 2021;20:105.
Tian Q, Wu T, Zhang X, Xu K, Yin X, Wang X, et al. Immunomodulatory functions of the circ_001678/miRNA-326/ZEB1 axis in non-small cell lung cancer via the regulation of PD-1/PD-L1 pathway. Hum Mol Genet. 2022;31:4094–106.
Li C, Zhang J, Yang X, Hu C, Chu T, Zhong R, et al. hsa_circ_0003222 accelerates stemness and progression of non-small cell lung cancer by sponging miR-527. Cell Death Dis. 2021;12:807.
Ye R, Lu X, Liu J, Duan Q, Xiao J, Duan X, et al. CircSOD2 contributes to tumor progression, immune evasion and anti-PD-1 resistance in hepatocellular carcinoma by targeting miR-497-5p/ANXA11 axis. Biochem Genet. 2023;61:597–614.
Ge J, Wang J, Xiong F, Jiang X, Zhu K, Wang Y, et al. Epstein-Barr virus-encoded circular RNA CircBART2.2 promotes immune escape of nasopharyngeal carcinoma by regulating PD-L1. Cancer Res. 2021;81:5074–88.
Sun J, Zhang H, Wei W, Xiao X, Huang C, Wang L, et al. Regulation of CD8+ T cells infiltration and immunotherapy by circMGA/HNRNPL complex in bladder cancer. Oncogene. 2023;42:1247–62.
Lei J, Zhu J, Hui B, Jia C, Yan X, Jiang T, et al. Circ-HSP90A expedites cell growth, stemness, and immune evasion in non-small cell lung cancer by regulating STAT3 signaling and PD-1/PD-L1 checkpoint. Cancer Immunol Immunother. 2023;72:101–24.
Luo YH, Yang YP, Chien CS, Yarmishyn AA, Adekunle Ishola A, Chien Y, et al. Circular RNA hsa_circ_0000190 facilitates the tumorigenesis and immune evasion by upregulating the expression of soluble PD-L1 in non-small-cell lung cancer. Int J Mol Sci. 2021;23:64.
Zhao R, Ni J, Lu S, Jiang S, You L, Liu H, et al. CircUBAP2-mediated competing endogenous RNA network modulates tumorigenesis in pancreatic adenocarcinoma. Aging (Albany NY). 2019;11:8484–501.
Liu L, Liao R, Wu Z, Du C, You Y, Que K, et al. Hepatic stellate cell exosome-derived circWDR25 promotes the progression of hepatocellular carcinoma via the miRNA-4474-3P-ALOX-15 and EMT axes. Biosci Trends. 2022;16:267–81.
Liu Z, Zheng N, Li J, Li C, Zheng D, Jiang X, et al. N6-methyladenosine-modified circular RNA QSOX1 promotes colorectal cancer resistance to anti-CTLA-4 therapy through induction of intratumoral regulatory T cells. Drug Resist Updat. 2022;65:100886.
Zhang PF, Gao C, Huang XY, Lu JC, Guo XJ, Shi GM, et al. Cancer cell-derived exosomal circUHRF1 induces natural killer cell exhaustion and may cause resistance to anti-PD1 therapy in hepatocellular carcinoma. Mol Cancer. 2020;19:110.
Oronsky B, Carter C, Reid T, Brinkhaus F, Knox SJ. Just eat it: a review of CD47 and SIRP-α antagonism. Semin Oncol. 2020;47:117–24.
Fang W, Mu J, Yang Y, Liu L. CircRERE confers the resistance of multiple myeloma to bortezomib depending on the regulation of CD47 by exerting the sponge effect on miR-152-3p. J Bone Oncol. 2021;30:100381.
Zhang H, Wang X, Li Y, Bai Y, Li Q, Wang S, et al. The hsa_circ_0000276-ceRNA regulatory network and immune infiltration in cervical cancer. BMC Cancer. 2023;23:222.
Kato S, Goodman A, Walavalkar V, Barkauskas DA, Sharabi A, Kurzrock R. Hyperprogressors after Immunotherapy: analysis of genomic alterations associated with accelerated growth rate. Clin Cancer Res. 2017;23:4242–50.
Lin A, Wei T, Meng H, Luo P, Zhang J. Role of the dynamic tumor microenvironment in controversies regarding immune checkpoint inhibitors for the treatment of non-small cell lung cancer (NSCLC) with EGFR mutations. Mol Cancer. 2019;18:139.
Kamada T, Togashi Y, Tay C, Ha D, Sasaki A, Nakamura Y, et al. PD-1+ regulatory T cells amplified by PD-1 blockade promote hyperprogression of cancer. Proc Natl Acad Sci USA. 2019;116:9999–10008.
Lo Russo G, Moro M, Sommariva M, Cancila V, Boeri M, Centonze G, et al. Antibody-Fc/FcR interaction on macrophages as a mechanism for hyperprogressive disease in non-small cell lung cancer subsequent to PD-1/PD-L1 blockade. Clin Cancer Res. 2019;25:989–99.
Li M, Wang Y, Wu P, Zhang S, Gong Z, Liao Q, et al. Application prospect of circular RNA-based neoantigen vaccine in tumor immunotherapy. Cancer Lett. 2023;563:216190.
Niu D, Wu Y, Lian J. Circular RNA vaccine in disease prevention and treatment. Signal Transduct Target Ther. 2023;8:341.
Funding
This work was supported by National Nature Science Foundation of China (82002577); Natural Science Foundation of Hebei Province (H2023206289); Precision Medicine United Fund Key Program of Hebei Natural Science Foundation (H2021206070).
Author information
Authors and Affiliations
Contributions
LJM and LHL designed the concept and outline of this review. LJM searched the literature and wrote the original manuscript. LJM, HTW, JXW, PAD, and JCH prepared the figures and tables. MXS and LHL revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Hans-Uwe Simon
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Meng, L., Wu, H., Wu, J. et al. Mechanisms of immune checkpoint inhibitors: insights into the regulation of circular RNAS involved in cancer hallmarks. Cell Death Dis 15, 3 (2024). https://doi.org/10.1038/s41419-023-06389-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-023-06389-5