Abstract
Trastuzumab is commonly used in the treatment of human epidermal growth factor receptor-2 positive (HER-2+) breast cancer, but its efficacy is often limited by the emergence of chemoresistance. Recent studies indicate that exosomes act as vehicles for exchange of genetic cargo between heterogeneous populations of tumor cells, engendering a transmitted drug resistance for cancer development and progression. However, the specific contribution of breast cancer-derived exosomes is poorly understood. In this study, publicly available expression profiling data from breast cancer and bioinformatics analyses were used to screen potential miRNAs in trastuzumab resistance. A series of gain- or loss-functional assays were performed to define the function of miR-567 and ATG5 in trastuzumab resistance and autophagy, both in vitro and in vivo. Our results showed that miR-567 was significantly decreased in trastuzumab-resistant patients compared with responding patients. Moreover, miR-567 was also downregulated in trastuzumab-resistant cells compared with parental cells. Overexpression of miR-567 reversed chemoresistance, whereas silence of miR-567 induced trastuzumab resistance, both in vitro and in vivo. In addition, enhanced miR-567 could be packaged into exosomes, incorporated into receipt cells, suppressing autophagy and reversed chemoresistance by targeting ATG5. To conclude, exosomal miR-567 plays a key role in reversing trastuzumab resistance via regulating autophagy, indicating it may be a promising therapeutic target and prognostic indicator for breast cancer patients.
Similar content being viewed by others
Introduction
Breast cancer is one of the most frequent cancers and is the second leading cause of cancer death among women1. The human epidermal growth factor receptor-2 (HER-2), overexpressed in ~20% of breast cancers2, predicts both reduced overall survival and shorter time to relapse in patients with breast cancer3. Currently, trastuzumab, a humanized monoclonal antibody targeting extracellular region of HER-2, has become the alternative choice in the treatment of HER-2-positive breast cancer4. However, only a fraction of metastatic patients responds to trastuzumab and ~60% develop resistance after initial response5.
A series of reports confirmed that cell-secreted exosomes can disseminate or reverse the resistance ability of tumor cells6,7,8, affecting prognosis. The exosome is a nanoscale membrane vesicle with a diameter of 30–150 nm, which participates in intercellular communication by delivering lipids and nucleic acids to recipient cells9. Cell-secreted exosomes mediate crosstalk between neighboring cells or transport to anatomically distal tissues where they send signals or messages to particular recipient cells.
MicroRNAs (miRNAs) are small noncoding RNAs that control gene expression at the posttranscriptional level10. As a vital regulator of numerous cell biological processes, numerous miRNAs have been shown to be involved in tumor progression and response to therapy11. Evidence is mounting that numerous miRNAs are involved in regulating drug resistance, especially in colorectal cancer12. miRNAs incorporated into exosomes are protected by bilateral membrane structures, thus reducing the degradation of miRNAs and facilitating cell-to-cell communication13. Research has shown that exosomal miRNAs participate in the processes of chemical resistance and may be shipped to the premetastatic niche in a variety of tumor types14.
Mounting evidence has demonstrated that anti-cancer therapies, including the cytotoxic chemotherapy, can induce cyto-protective autophagy in most cancer cells15,16. Briefly, autophagy is a highly conserved cellular process during evolution, which is induced by diverse pathologies and cellular stresses containing nutrient deprivation, endoplasmic reticulum stress, and hypoxia17. Autophagy has also been involved in cancer resistance to multiple chemotherapeutic drugs, including cisplatin18, paclitaxel19, and gemcitabine20. Autophagy plays a vital role in regulating colorectal cancer chemoresistance, blocking of which will be developed as a promising therapy strategy for colorectal cancer treatment21. By modulating key autophagy-related protein expression, miRNAs has an effect on regulating autophagy. More generally, it has attracted substantial attention that the contribution of modulation of autophagy mediated by miRNAs in cancer therapy and drug resistance.
In this study, we revealed the essential role of miR-567 in inhibiting autophagy and increasing chemosensitivity to trastuzumab in breast cancer. Moreover, we provided evidence showing that miR-567 could target a key autophagy-related protein: autophagy-related 5 (ATG5), which is associated with carcinogenesis. Finally, we proved that miR-567 delivery via exosomes increases the sensitivity of breast cancer cells to trastuzumab.
Materials and methods
Breast cancer RNA-expression data retrieval and analysis
Microarray datasets from the Gene Expression Omnibus (GEO) database were used to test miR-567 differential expression. Raw microarray data were downloaded from GEO datasets GSE104076. Then, the raw microarray data were normalized and z-score-transformed using RMA Express (http://www.rmaexpress.bmbolstad.com/). RNA-Sequencing data (from The Cancer Genome Atlas, TCGA) of miRNAs of breast cancer were from TANRIC (http://ibl.mdanderson.org/tanric/_design/basic/index.html)22.
Patient samples
Overall, 60 HER-2-positive patients, who received trastuzumab treatment, and 38 HER-2-negative patients were enrolled in this study, to ensure adequate power. Cancerous tissues were collected from January 2013 to August 2015 and were snap-frozen in liquid nitrogen instantly at −80 °C. The 60 HER-2-positive patients receiving trastuzumab treatment were divided into responding (30 patients) and non-responding (30 patients) according to the Immune-related Response Evaluation Criteria In Solid Tumors23. The patients were pathologically confirmed and clinical characteristics were shown in Supplementary Table S1. Patients who received radiotherapy and chemotherapy before surgical treatment were excluded. Meanwhile, the general clinical information and detailed pathological records were collected. Written-informed consent was obtained from all patients and the study protocol was approved by the Research Scientific Ethics Committee of The First Affiliated Hospital of Zhengzhou University, The Second Affiliated Hospital of Chongqing Medical University, and Hainan General Hospital.
Cell culture and treatment
Human HER-2-positive breast cancer cell lines SKBR-3 and BT474 were purchased from American Type Culture Collection (Manassas, USA) and maintained in Dulbecco’s modified Eagle’s medium (HyClone Lab., Inc., Logan, UT) supplemented with 10% fetal bovine serum (FBS) (Sigma-Aldrich, St. Louis, MO, USA), 100 U/ml penicillin, and 100 μg/ml streptomycin (Life Technologies, Grand Island, NY, USA) in humidified air at 37 °C with 5% CO2. The cell lines were authenticated by short tandem repeat profiling. Trastuzumab (Herceptin) was purchased from Roche (Basel, Switzerland) and was used by dissolving in phosphate-buffered saline (PBS). The SKBR-3 and BT474 cells resistant to trastuzumab treatment (named as SKBR-3-TR and BT474-TR, respectively) were built by establishing xenografts followed by four courses of trastuzumab treatment as previously described24.
Cell transfection
miRNA mimics, inhibitors, and negative controls (NC or NC inhibitors) were purchased from GenePharma (Shanghai, China). To overexpress or knock down the expression level of miR-567 or ATG5, gene-specific overexpression plasmids (FulenGen, Guangzhou, China) or small interfering RNAs (GenePharma) were transfected into cells. Lipofectamine 3000 (Invitrogen, CAT. L3000008, Carlsbad, CA, USA) was used for transient transfection with a final concentration of 100 nM for in vitro assays. miRNA mimics, inhibitors, and NCs were synthesized and loaded into lentivirus, which were purchased from Shanghai Genechem Co., Ltd, for in vivo assays. The sequences of silencing sequences were shown in Supplementary Table S2.
RNA extraction and quantitative RT-PCR
Total RNA was extracted and reverse transcribed to cDNA using a miRcute miRNA Isolation Kit and a miRcute Plus miRNA First-Strand cDNA Synthesis Kit (Tiangen Biotech, CAT. hz130911–251, China) following standard protocols. PCR was performed on a Biosystems 7500 Fast Real-Time PCR System (Applied Biosystems, USA). The relative expression level of miR-567 was normalized to that of U6 and the expression of ATG5 mRNA was normalized to that of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). miRNAs in exosomes were normalized using cel-miR-39 (Applied Biosystem, CA, USA) as an external control. The primer sequences were provided in Supplementary Table S2.
Luciferase reporter assay
Luciferase vectors were purchased from Genescript (Nanjing, China). Briefly, for miRNA-binding site assays, luciferase reporter gene plasmids harboring the wild-type or mutant-type 3′-untranslated region (3′-UTR) of ATG5 were constructed as previously described24. We co-transfected SKBR-3-TR cells with luciferase vectors and small RNA oligo expression plasmid (Ambion, Carlsbad, CA, USA). Luciferase activity was measured using a luciferase assay kit (CAT. E1910, Promega, USA).
Cell viability assay
The ability of cell viability was detected by CCK8 (Dojindo, CAT. CK04, Kumamoto, Japan) according to the manufacturer’s instructions. A total of 5000 cells with corresponding treatment were seeded onto 96-well culture plates. At a specific time point, CCK8 reagent was used for incubation for 2 h at 37 °C. Then, the absorbance at 450 nm was determined by the Infinite M200 spectrophotometer (Tecan, Switzerland).
TUNEL assay
Breast cancer cells or tissues were fixed with paraformaldehyde for 30 min on ice. Then, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) kit was used as per the manufacturer’s instructions (Roche, DNA Fragmentation Imaging Kit, CAT. 6432344001). DAPI (4′,6-diamidino-2-phenylindole) was used for nuclear staining. TUNEL-positive areas were quantified under an Olympus FSX100 microscope (Olympus, Tokyo, Japan).
Exosome isolation and characterization
Exosomes were isolated from the supernatant of CRC cells. Cells were maintained in medium containing 10% exosome-depleted FBS (Vivacell, Shanghai, China). Briefly, cell supernatants (collected after 48 h) were differentially centrifuged at 500 × g and 3000 × g for 10 min to remove cellular debris. Then, the supernatant was filtered through a 0.22 µm filter (Millipore) and centrifuged at 120,000 × g for 2 h at 4 °C. Exosomes were resuspended in PBS. Size distribution of exosomes were analyzed by Zetasizer (Zetasizer Nano ZS, Malvin Co. UK). Exosomes were irradiated with a laser and their motion (under Brownian motion) was recorded. A 10 s sample video was analyzed with nanoparticle tracking analysis (NTA) software (version 2.3, Nano-sight). Exosomes were observed by transmission electron microscopy (TEM; H-7650, Hitachi, Japan).
Exosome labeling and electron microscopy
Exosomes were stained with PKH26 membrane dye (Sigma, CAT. MIDI26–1KT). After culturing with the labeled exosomes for 3 h, the cells were fixed and stained with Hoechst. The cellular uptake of exosomes was observed on a Leica TCS-SP5 LSM electron microscope (JEM-1220, JEOL, Ltd, Japan). For the in vitro experiments, 1 × 105 receptor cells were co-cultured with 10 mg of exosomes.
Western blotting analysis
Western blotting analysis was carried out following standard protocols. The primary rabbit antibodies used were as follows: TSG101 (1:1000, Abcam, ab125011), HSP70 (1:1000, Abcam, ab2787), ATG5 (1:1000, Abcam, ab228668), and GAPDH (1:5000, Abcam, ab9485). After incubation with the goat anti-rabbit secondary antibody (1:5000, Abcam, ab205718, USA), the protein band was visualized with super chemiluminescent reagent (Millipore, CAT. WBKLS0050, MA, USA) using a Bio-Rad ChemiDoc XRS system (Bio-Rad, CA, USA).
In vivo nude mouse model
Tumor xenografts were established with male BALB/c nude mice (4–6 weeks old), which were purchased from Model Animal Research Center of Nanjing University (Nanjing, China). Blinding grouping was used and mice were randomly divided into four groups (n = 4 in each group): SKBR-3-TR/control + trastuzumab, SKBR-3-TR/miR-567 + trastuzumab, SKBR-3/control + trastuzumab, and SKBR-3/miR-567 inhibitor sponge + trastuzumab. Equal numbers of SKBR-3-TR or SKBR-3 cells (5 × 106) were subcutaneously injected into each mouse to establish the breast cancer xenograft model. One week later, mice received an intraperitoneal injection of PBS or trastuzumab (3 mg/kg) once every 2 days for 20 days. On day 28, the animals were killed and tumors were removed.
The exclusion criteria for removing the mice before finishing the study were approved. For our experiments, we had the following exclusion criteria: (1) immobilization/disability because of tumor size or tumor volume more than 2 × 103 mm3; (2) breathing difficulties; (3) no contact reactions; and (4) signs of infection. All the procedures were performed on the basis of the guidelines of the Institutional Review Board of The First Affiliated Hospital of Zhengzhou University (Henan, China).
Immunohistochemistry analysis and scoring methods
Paraffin-embedded sections of tumor tissues from nude mice were placed in an incubator maintained at 60 °C for 2 h and then immersed. Different concentrations of ethanol (including 70%, 85%, 95%, 100%) and deionized water were used to hydrate these slices, then these slices were immersed in citrate buffer solution (0.01 mol/L, pH 6.0) and heated, keeping the temperature between 95 °C and 100 °C for 30 min. After washing with PBS, it was incubated with 0.5% Triton X-100 for 30 min. This fraction was then stained using the biotin-streptavidin horseradish peroxidase detection system (ZSGB, CAT. PV-9003, China). These slices were incubated with primary rabbit antibody targeting Ki67 (1:200, ab15580, Abcam) overnight at 4 °C and the presence of brown chromogen in the nuclear staining indicates positive immunoreactivity.
The immunostaining intensity of each sample was graded as negative = 0, weak = 1, moderate = 2, or strong = 3. The proportion of positively staining cells was assessed as the percentage. The score was then calculated as the intensity score multiplied by the percentage of cells stained (score = intensity × % of positive cells). Images were visualized using a Nikon ECLIPSE Ti (Fukasawa, Japan) microscope system and processed with Nikon software.
Statistical analysis
All experiments were performed in triplicate. Statistics were presented as mean ± SD. Comparison between two groups were analyzed using the two-sided Student’s t-test. One-way analysis of variance was used for the comparison of multiple groups (>2) and variance similar between the groups that are being statistically compared. Fisher’s exact test was performed to evaluate the difference of proportions between different groups. Statistical analyses were performed using GraphPad Prism (v5.01, GraphPad Software, Inc., San Diego, CA, USA). P < 0.05 was considered to indicate a statistically significant difference.
Results
Identification of miR-567 by analyzing breast cancer RNA-expression profiling data
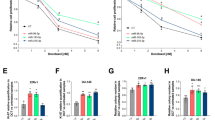
Raw microarray data were downloaded from GEO dataset, GSE104076 (n = 8), which showed the global expression data from trastuzumab-resistant cells and -sensitive cells. To obtain differentially expressed miRNAs, signal data were normalized and z-score transformed. The top 20 dysregulated miRNAs were shown in Fig. 1a, b. Interestingly, we identified a clear downregulation of miR-567 in trastuzumab-resistant cells (Fig. 1c). Previous studies demonstrated that miR-567 may serve as a tumor suppressor in gastric cancer and breast cancer25,26. To confirm the functional role of miR-567 in breast cancer tumorigenesis and trastuzumab resistance, we determined its expression in breast cancer specimens. Quantitative reverse-transcriptase PCR (qRT-PCR) showed that miR-567 was downregulated in 38 breast cancer tissues in contrast to paired non-tumor tissues (Fig. 1d). By using two trastuzumab-resistant cell lines SKBR-3-TR and BT474-TR, which we previously established24, we detected miR-567 expression. Figure 1e showed that miR-567 was downregulated in trastuzumab-resistant breast cancer cells compared with parental cancer cells. Moreover, miR-567 expression was highest in normal breast epithelial cells, MCF-10A (Fig. 1e). In addition, we detected the expression of miR-567 in 60 breast cancer patients who received trastuzumab-based chemotherapy. miR-567 level was lower in 30 non-responding patients when compared with 30 responding patients (Fig. 1f). The proportion of patients responding to trastuzumab treatment was higher in high miR-567 expression group than that in low-miR-567-expressing group (Fig. 1g). We also verified a decreased expression of miR-567 in breast cancer patients in contrast to healthy population by analyzing data from TCGA datasets, including 1384 breast cancer patients and 297 healthy controls (Fig. 1h), and higher miR-567 expression was associated with better survival rate in contrast to breast cancer patients with low miR-567 patients (median value used as cutoff value) (Fig. 1i). The above data suggest that miR-567 may be closely associated with trastuzumab resistance of breast cancer patients.
a, b Raw microarray data were downloaded from GEO, GSE104076 (n = 8). Then, the raw data were normalized and the top 20 dysregulated miRNAs in these GEO datasets are shown in a and b. c The expression of miR-567 in trastuzumab-resistant cells or -sensitive cells was shown by analyzing the GEO datasets. d qRT-PCR was performed to assess the expression level of miR-567 in breast cancer cells, **P < 0.01. e miR-567 level was determined in breast cancer patients responding to trastuzumab treatment or not, *P < 0.05. f miR-567 was downregulated in breast cancer tissues compared with normal tissues, *P < 0.05. g The proportion of patients responding to trastuzumab therapy in high miR-567 group and low-miR-567 group was shown. h miR-567 expression in breast cancer tissues compared with noncancerous tissues analyzed using the TCGA database, **P < 0.01. i High miR-567 expression levels correlated with worse overall survival in breast cancer patients by analyzing the TCGA data.
MiR-567 reverses the trastuzumab resistance of breast cancer cells
Based on the accumulated data, we sought to examine the effect of miR-567 on breast cancer cell chemoresistance in vitro. We inhibited miR-567 expression in trastuzumab-sensitive cells and overexpressed miR-567 in trastuzumab-resistant cells, respectively (Fig. 2a, b). Subsequently, the growth curves showed that miR-567 inhibitor increased the IC50 of trastuzumab in trastuzumab-sensitive cells. On the contrary, the IC50 of trastuzumab concomitantly decreased in miR-567-overexpressing cells (Fig. 2c, d). Next, by performing 5-ethynyl-2’-deoxyuridine assay, we found that overexpression of miR-567 could enhance the effect of trastuzumab on inhibiting cell proliferation, whereas the miR-567 inhibitor had contrasting effects (Fig. 2e, f). To further investigate the role of miR-567 in trastuzumab-induced cell death, we performed TUNEL analysis. The results showed that the overexpression of miR-567 could accelerate cell death of SKBR-3-TR cells, whereas silence of miR-567 decreased the cell death of SKBR-3 cells (Fig. 2g, h). Thus, miR-567 may reverse the chemoresistance of breast cancer cells to trastuzumab.
a, b miR-567 was silenced (a) or overexpressed (b) by transfection of respective vectors, **P < 0.01 compared with control groups. c Silence of miR-567 induced resistance of trastuzumab in parental cancer cells, *P < 0.05, **P < 0.01. d Increased miR-567 increased chemosensitivity of trastuzumab-resistant cells, *P < 0.05. e, f EdU staining showed that overexpression of miR-567 suppressed cell proliferation (e), whereas knockdown of miR-567 promoted cell proliferation (f); scale bars: 10 μm, *P < 0.05 compared with control groups. g, h TUNEL assay revealed that miR-567 induced cell apoptosis (g), whereas silence of miR-567 inhibited apoptosis (h); scale bars: 10 μm, P < 0.05 compared with control groups.
MiR-567 promotes chemosensitivity of trastuzumab treatment in vivo
To assess the effect of miR-567 in trastuzumab-induced tumor suppression in vivo, we stably transfected SKBR-3-TR cells with lentivirus overexpressing miR-567 or with lentivirus expressing miR-NC. In addition, we also stably transfected SKBR-3 cells with lentivirus expressing miR-567 inhibitor or NC inhibitor. In the xenograft mouse models, SKBR-3-TR cells and SKBR-3 cells were subcutaneously transplanted into nude mice and then received treatment with trastuzumab, as shown in Fig. 3a. The results showed that SKBR-3-TR cells stably overexpressing miR-567 were more sensitive to trastuzumab therapy than the control group (Fig. 3b). In contrast, inhibition of miR-567 in SKBR-3 cells weakened the effect of trastuzumab on inhibiting tumor growth (Fig. 3c). In addition, miR-567-overexpressing xenograft tumors showed a significantly reduced Ki67-positive areas (Fig. 3d), whereas tumors infected with miR-567 inhibitor exhibited an increased percentage of Ki67-positive area (Fig. 3e). Altogether, these results strongly indicate that miR-567 reverses therapeutic resistance in vivo.
a A schematic outline of the experimental design. b Effect of miR-567 silence on transplanted tumor growth from SKBR-3 cells. Representative images of tumors as indicated in left panel and tumor growth as indicated in right panel, *P < 0.05. c Effect of miR-567 overexpression on transplanted tumor growth from SKBR-3-TR cells. Representative images of tumors as indicated in the left panel and tumor growth as indicated in the right panel, *P < 0.05. d Representative images of tumor samples derived from SKBR-3 cells that were stained with Ki67 by immunohistochemistry (IHC, left panel) and quantified (right panel); scale bars: 50 μm, **P < 0.01 compared with control inhibitor. e Representative images of tumor samples derived from SKBR-3-TR cells that were stained with Ki67 by IHC (left panel) and quantified (right panel); scale bars: 50 μm, **P < 0.01 compared with control mimics.
MiR-567 inhibits the trastuzumab-induced autophagy
Considering that autophagy can protect cancer cells from cytotoxic drugs, we explored whether autophagy participated in chemoresistance to trastuzumab in breast cancer. Thus, we measured the LC3 and p62 levels, which are the widely used markers of autophagy27,28. Remarkably, trastuzumab-resistant cells showed higher LC3-II and lower p62 protein levels than the corresponding parental cells, suggesting that autophagic flux was induced when chemoresistance occurred (Fig. 4a). Consistently, trastuzumab-resistant cells showed increased formation of LC3 puncta (Fig. 4b) and autophagosomes (Fig. 4c). To verify whether autophagy is essential for trastuzumab resistance, we treated trastuzumab-resistant cells with chloroquine (CQ), which is a well-known inhibitor for autophagy29. Our results showed that CQ treatment suppressed autophagy and reversed the resistant status (Fig. 4d, e).
a Western blotting showed an increased autophagy activity in trastuzumab-resistant cells compared with parental cells, as evidenced by a higher LC3-II and lower p62 protein levels. b After transfected with GFP-LC3 plasmid for 48 h, green fluorescent LC3 puncta were observed via confocal microscopy; scale bars: 10 μm, **P < 0.01 compared with parental cells. c Autophagosomes were observed by transmission electron microscopy (TEM). Representative images are shown and autophagosomes per cell were quantified; scale bars: 10 μm, **P < 0.01 compared with parental cells. d Western blotting was performed in SKBR-3-TR and BT474-TR cells treated with or without 30 μM CQ. e Cell viability was detected in cells treated with or without CQ, **P < 0.01. f The expression levels of LC3 and p62 were measured by western blotting in resistant cells overexpressed with miR-567. g The expression levels of LC3 and p62 were measured by western blotting in parental cells silenced with miR-567. h Breast cancer cells were transfected with the GFP-LC3 plasmid and small RNA oligos. Then, green fluorescent LC3 puncta were observed; scale bars: 10 μm, **P < 0.01 compared with controls. i Autophagosomes were detected via TEM in cells treated with small RNA oligos; scale bars: 10 μm; **P < 0.01 compared with control.
We next investigated the role of miR-567 in autophagic activity. Overexpression of miR-567 resulted in a reduced level of LC3-II and an enhanced level of p62 in SKBR-3-TR and BT474-TR cells (Fig. 4f). In contrast, in parental cells with miR-567 suppression, the level of LC3-II was increased, whereas the level of p62 was decreased (Fig. 4g). Overexpression of miR-567 in SKBR-3-TR cells has diminished the numbers of LC3 puncta, whereas suppression of miR-567 led to an increase in the number of LC3 puncta in SKBR-3 cells (Fig. 4h). In addition, upregulation of miR-567 blocked the formation of autophagosomes and inhibition of miR-567 enhanced the formation of autophagosomes (Fig. 4i). Collectively, our data indicate that trastuzumab resistance induced enhanced autophagy activity and inhibition of miR-567 reversed autophagy, thereby inducing enhanced chemosensitivity.
Identification of ATG5 as a direct target of miR-567
We further investigated the target gene involved in mediating the effect of miR-567 on modulating autophagy by using miRDB (http://mirdb.org/) and verified ATG5 was targeted by miR-567 in 3′-UTR area (Fig. 5a). By performing luciferase reporter assay, we identified a significant reduction in luciferase activity when ectopic miR-567 was expressed in SKBR-3-TR and BT474-TR cells (Fig. 5b). When the binding site of miR-567 was mutated, miR-567 had no influence on the luciferase activity. Furthermore, western blotting showed that ATG5 expression was upregulated in trastuzumab-resistant cells compared with parental cells (Fig. 5c). Overexpression of miR-567 inhibited the expression of ATG5 in trastuzumab-resistant cells (Fig. 5d), whereas inhibition of miR-567 increased the ATG5 level in trastuzumab-sensitive cells at both the transcript and protein levels (Fig. 5e). Furthermore, we measured the ATG5 levels in the above-mentioned 60 breast cancer tissues and found that the ATG5 level was upregulated in trastuzumab-resistant patient than in the trastuzumab-responding patients (Fig. 5f). The Spearman’s rank correlation analysis revealed that the expression level of ATG5 was significantly negatively related to the level of miR-567 (Fig. 5g). In sum, these results demonstrate that miR-567 posttranscriptionally regulates the expression of ATG5.
a Schematic of the hypothetical duplexes formed by miR-567 and the 3′-UTR of ATG5. b Relative luciferase activity in cells transfected with the miR-567 mimic were determined, *P < 0.05, **P < 0.01 compared with control mimics. c ATG5 expression was shown via western blotting in trastuzumab-resistant or parental cells. d, e ATG5 transcript and protein levels were detected by qRT-PCR and western blotting, respectively; P < 0.01 compared with control groups. f ATG5 mRNA level was identified in primary breast cancer tissues from patients responding or resistant to trastuzumab treatment. g Spearman’s correlation test was performed to verify the association between miR-567 and ATG5 mRNA expression. *P < 0.05, **P < 0.01.
MiR-567 reverses trastuzumab resistance by inhibiting ATG5 and autophagy
Based on our confirmation of effect of autophagy in trastuzumab resistance, we hypothesized that miR-567 reverses trastuzumab resistance by attenuating autophagic activity through inhibiting ATG5. First, we investigated whether ATG5 is involved in autophagic activity induced by miR-567 in breast cancer cells. Silencing of ATG5 attenuated the effect of the miR-567 inhibitor on the protein levels of ATG5, p62, and LC3-II (Fig. 6a), and the formation of LC3 puncta (Fig. 6b). On the other hand, increased ATG5 level diminished the inhibitory effect of miR-567 on the protein levels of ATG5, p62, and LC3-II (Fig. 6c), and the LC3 puncta formation (Fig. 6d).
a Western blotting showed that silence of ATG5 reversed the miR-567 inhibition-induced upregulation of LC3 and downregulation of p62 in SKBR-3-TR cells. b Knockdown of ATG5 abrogated the miR-567 inhibitor-induced LC3 puncta formation in SKBR-3-TR cells, **P < 0.01 compared with miR-567 inhibitor + si-NC group. c Western blotting showed that increased ATG5 reversed the miR-567 mimics-induced suppression of LC3 and increase of p62 in SKBR-3-TR cells. d Overexpression of ATG5 reversed the miR-567 mimics-induced LC3 puncta suppression in SKBR-3 cells, **P < 0.01 compared with miR-567 + p-NC group. e CCK8 assay showed that knockdown of ATG5 significantly reversed trastuzumab resistance, **P < 0.01. f Enhanced ATG5 level induced chemoresistance to trastuzumab of SKBR-3 and BT474 cells, *P < 0.05, **P < 0.01. g, h Knockdown of ATG5 reversed the miR-567 inhibitor-caused chemoresistance (g), whereas overexpression of ATG5 abrogated the miR-567 mimics-induced chemosensitivity (h).
Next, we sought to prove ATG5 is essential for miR-567-regulated trastuzumab resistance. By performing a series of gain- or loss-functional experiments, we found that inhibition of ATG5 (si-ATG5) attenuated chemoresistance of trastuzumab-resistant cells (Fig. 6e). In contrast, overexpression of ATG5 (plasmid-ATG5, p-ATG5) in sensitive cells promoted drug resistance (Fig. 6f). Moreover, suppression of ATG5 abolished the trastuzumab resistance induced by miR-567 inhibitor in sensitive cells (Fig. 6g), whereas enhanced ATG5 abrogated the miR-567-induced chemosensitivity of resistant cells (Fig. 6h).
Extracellular miR-567 reverses trastuzumab resistance via incorporating into exosomes
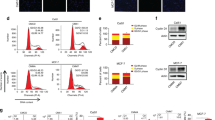
To demonstrate whether extracellular miR-567 reversed trastuzumab resistance via incorporating into exosomes, we detected the existence pattern of extracellular miR-567. miR-567 level in culture medium was unchanged upon treatment with RNase but significantly decreased when treated with RNase and Triton simultaneously (Fig. 7a), proving that miR-567 was wrapped with membrane instead of being released directly. To confirm this hypothesis, we isolated exosomes from culture medium. The representative micrograph taken by TEM showed vesicles with round or oval membrane (Fig. 7b). NTA analysis revealed that the size of exosomes mostly ranges from 30 nm to 200 nm in diameter (Fig. 7c). Western blotting assay further verified that the exosome proteins, TSG101 and HSP70, were enriched in exosomes but not in cell extracts (Fig. 7d). In addition, the expression of exosomal miR-567 levels were almost equal to that in extracellular miR-567 levels (Fig. 7e), suggesting that exosome was the main carrier for extracellular miR-567. Figure 7f showed that exosomal miR-567 level was mostly high in culture medium from MCF-10A cells and mostly low in that from trastuzumab-resistant cells.
a Extracellular miR-567 was degraded by treatment with RNAse A and Triton simultaneously, **P < 0.01 compared with RNase A group. b TEM scanning showed the exosome images released by SKBR-3-TR and BT474-TR cells; scale bars: 200 nm. c Size distribution of exosomes were analyzed by Zetasizer. d Exosomal protein markers (TSG101 and HSP70) detection by western blotting from purified exosomes and exosome-depleted cell extracts. e qRT-PCR analysis of miR-567 expression in culture medium and exosomes. f qRT-PCR analysis showed that exosomal miR-567 was downregulated in trastuzumab-resistant cells in contrast to sensitive cells, *P < 0.05.
Then, we investigated whether the miR-567-contained exosomes could be taken up by receipt cells via using two prolonged stages. First, we isolated exosomes from MCF-10A cells and labeled with PKH26 dye followed by incubation with SKBR-3-TR and BT474-TR cells for 48 h. Figure 8a showed a strong red signal in receipt cells, indicating that the exosomes were taken up by receipt cells. Second, we examined whether these exosomes could deliver miR-567 to recipient cells. By extracting cytoplasm RNA from receipt cells followed by qRT-PCR assay, we verified a significantly increased miR-567 level in recipient cells upon incubation with exosomes from MCF-10A cells; however, this influence was eliminated when incubated with exosomes from miR-567-knockdown MCF-10A cells (Fig. 8b), suggesting that the miR-567-contained exosomes can be taken by recipient cells.
a Intercellular trafficking of exosomes among different cell lines by isolated exosomes labeled with PKH26 dye; scale bars: 10 μm. b Treatment with exosomes derived from MCF-10A cells increased miR-567 level; however, this increase was eliminated when MCF-10A cells were silenced with miR-567, *P < 0.05. c Western blotting showed that autophagy was induced in cells treated with MCF-10A-derived exosomes. d CCK8 assay showed that MCF-10A-derived exosomes induced chemosensitivity in SKBR-3-TR and BT474-TR cells, *P < 0.05. e Nano-sight particle tracking analysis of the size distributions and number of exosomes from SKBR-3-TR cells treated with nSMase, GW4869; **P < 0.01 compared with DMSO group. f CCK8 assay showed that incubation with exosomes from MCF-10A cells treated with GW4869 failed to reverse trastuzumab resistance to recipient cells. g CCK8 assay verified that silence of miR-567 inhibited the ability of co-cultured parental cells to acquire chemo-response. h A scheme of the proposed mechanisms. High expressed miR-567 in MCF-10A cells was secreted via packaging into exosomes. Then, the extracellular miR-567 was incorporated into recipient cells, suppressed autophagy, and reversed chemoresistance via suppressing ATG5 expression.
We further examined whether exosome-transferred miR-567 could reverse the resistant phenotype of recipient cells. Western blotting results showed that MCF-10A-derived exosomes suppressed autophagy (Fig. 8c). Consistently, trastuzumab-resistant cells incubated with MCF-10A-derived exosomes exhibited increased sensitivity to trastuzumab treatment (Fig. 8d). To explore whether exosomes played a critical role in this effect, we blocked exosome production through the pharmacological inhibition of neutral sphingomyelinase-2 (nSMase) with GW4869 (Fig. 8e). As shown in Fig. 8f, incubation with culture medium from MCF-10A cells treated with GW4869 failed to reverse trastuzumab resistance of recipient cells. More importantly, incubation with culture medium from MCF-10A cells silenced miR-567 also failed to reverse trastuzumab resistance of recipient cells (Fig. 8g). Altogether, we proved that extracellular miR-567 reverses resistance of trastuzumab-resistant cells by packaging into exosomes.
Discussion
Although surgery combined with targeted therapy has dramatically prolonged the lifespan, the prognosis of advanced breast cancer patients remains poor. In addition, due to the target gene diversity and tissue-type specificity of miRNAs, the specific regulatory functions of miRNAs in chemoresistance have yet to be fully delineated30. In the current study, utilizing publicly available miRNA expression profiling data of breast cancer and integrating analyses of TCGA data, we screened and identified miR-567 as the dysregulated one during trastuzumab resistance. Our experimental analysis showed that enhanced miR-567 suppressed autophagy, thereby reversing trastuzumab resistance of breast cancer, whereas knockdown of miR-567 induced resistance to trastuzumab treatment. ATG5 was verified as a functional target of miR-567 during autophagy and chemoresistance. More importantly, we proved that extracellular miR-567 could reverse trastuzumab resistance of recipient cells (Fig. 8h).
Even more effective therapies using trastuzumab emtansine are now available; unfortunately, resistance to trastuzumab and other therapies that target the HER-2 pathway still occurs and some patients do not benefit from trastuzumab-based drug regimen31. In addition, the identification of patients that will benefit from HER-2-directed therapies is greatly hindered by a lack of biomarkers for predicting therapeutic response32. This is despite the extensive retrospective analysis of phase III clinical trial data for the potential roles of related receptor tyrosine kinases (i.e., EGFR, HER-3, IGF-1R), ligands (i.e., EGF, TGFα), or phosphatidylinositol 3-kinase (PI3K) pathway alterations. The expression of HER-2 itself is currently the only biomarker to guide treatment decisions within this HER-2-positive patient cohort33. Therefore, numerous studies have been devoted to identifying the potential pathways and predictive biomarkers34,35,36.
Acting as tumor suppressor genes or oncogenes, miRNAs are involved in many cellular processes such as differentiation, proliferation, metastasis, and chemoresistance37. miR-567 was previously reported to target FGF5 and inhibit cell proliferation, migration, and invasion in osteosarcoma38. In addition, miR-567 acts as a tumor suppressor gene and suppresses gastric cancer tumorigenesis and chemoresistance via a PIK3AP1-PI3K/AKT-c-Myc feedback loop25. Importantly, miR-567 was shown to inhibit the carcinogenesis of breast cancer26. However, the biological role of miR-567 and its molecular mechanism underlying trastuzumab resistance still remains unknown. In this study, miR-567 was selected based on the differential expression between trastuzumab-resistant and -sensitive cells from GEO datasets. qRT-PCR followed by experimental validation confirmed that miR-567 was inhibited in trastuzumab-resistant cells and enhanced miR-567 could reverse trastuzumab resistance, suggesting a crucial role in trastuzumab resistance of breast cancer.
Autophagy is known to play roles in maintaining the survival of tumor cells under a variety of adverse conditions, including nutrient deficiency, chemotherapy, and radiation treatment39. Accumulating evidence suggests that regulating the autophagic activity could enhance the action of many antitumor agents, including oxaliplatin, cisplatin, doxorubicin, and 5-fluorouracil. Thus, autophagy has been proposed as a potential drug target to reverse drug resistance. Previous reports have shown that a series of miRNAs regulate the drug resistance by modulating autophagy40,41,42. Notably, we revealed that trastuzumab-resistant cells showed increased autophagic activity compared with that in the corresponding parental cells. Moreover, overexpression of miR-567 inhibited LC3-I to LC3-II conversion, GFP-LC3 accumulation, and autophagosome synthesis. Therefore, miR-567 may serve as a vital autophagy-regulating miRNA by blocking autophagy to promote cell sensitivity to trastuzumab.
The molecular mechanism of autophagy involves several conserved autophagy-related genes and these genes have multiple functions in various physiological contexts. Among these genes, ATG5 protein in a conjugated form with ATG12 and ATG8 (LC3) are involved in the early stages of autophagosome formation43. Especially, ATG5 is important for development, cell differentiation and maintenance of homeostasis, including early neonatal starvation44. In our study, we demonstrated that miR-567 regulates the expression of ATG5 at the posttranscriptional level. Moreover, we confirmed the effect of ATG5 in miR-567-mediated inhibition of autophagy and interference of trastuzumab resistance.
After having verified the functional role and regulatory mechanism of miR-567 in trastuzumab resistance, we ask how miR-567 disseminates the anti-resistant function. As expected, we identified that extracellular miR-567 could reverse trastuzumab resistance through incorporating it into exosomes. Exosomes are a new means of intercellular information exchange that have aroused great research interest45. Long neglected in research, exosomes were deemed non-functional cellular components to be discarded. However, it has been gradually revealed that exosomes are an important tool for the exchange of intercellular information and material46. Extracellular exosomes may spread or reverse drug resistance among heterogeneous populations of cancer cells and ultimately influencing treatment efficiency of many cancer types47. However, the precise regulatory mechanism of how exosomes influence the tumor microenvironment is largely unclear. By using a two-step validation method, we verified that extracellular miR-567 could reverse trastuzumab resistance of recipient cells by incorporating into exosomes and further transferring into recipient cells through a cell membrane fusion way.
Conclusion
In conclusion, we reported the crucial role of miR-567 in trastuzumab resistance of breast cancer by directly suppressing ATG5 expression and thereby inhibiting autophagy. More importantly, miR-567 abrogates trastuzumab resistance among heterogeneous populations of breast cancer cells via incorporating into exosomes. Our discovery will not only increase our knowledge of noncoding RNA-regulated therapeutic effect in cancer but also help develop more efficient strategies to reverse chemoresistance.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
10 November 2023
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1038/s41419-023-06254-5
References
Scott, L. C., Mobley, L. R., Kuo, T. M. & Il’yasova, D. Update on triple-negative breast cancer disparities for the United States: A population-based study from the United States Cancer Statistics database, 2010 through 2014. Cancer 125, 3412–3417. https://doi.org/10.1002/cncr.32207 (2019).
Ziaeemehr, A. et al. Association of the involvement of axillary lymph nodes in HER-2/neu overexpression in patients with breast cancer. Breast J. 25, 537–538 (2019).
Giannone, G. et al. What is the best pharmacotherapeutic strategy for HER-2 positive breast cancer? Expert Opin. Pharmacother. 20, 5–9 (2019).
Daniels, B. et al. Long-term survival in trastuzumab-treated patients with HER2-positive metastatic breast cancer: real-world outcomes and treatment patterns in a whole-of-population Australian cohort (2001–2016). Breast Cancer Res. Treat. 171, 151–159 (2018).
Adamczyk, A. et al. Relationship between HER2 gene status and selected potential biological features related to trastuzumab resistance and its influence on survival of breast cancer patients undergoing trastuzumab adjuvant treatment. OncoTargets Ther. 11, 4525–4535 (2018).
Boelens, M. C. et al. Exosome transfer from stromal to breast cancer cells regulates therapy resistance pathways. Cell 159, 499–513 (2014).
Qu, L. et al. Exosome-transmitted lncARSR promotes Sunitinib resistance in renal cancer by acting as a competing endogenous RNA. Cancer cell 29, 653–668 (2016).
Stone, L. Kidney cancer: exosome transmission of sunitinib resistance. Nat. Rev. Urol. 13, 297 (2016).
Petersen, K. E. et al. A review of exosome separation techniques and characterization of B16-F10 mouse melanoma exosomes with AF4-UV-MALS-DLS-TEM. Anal. Bioanal. Chem. 406, 7855–7866 (2014).
Liu, C. H. et al. miRNAs in patients with non-alcoholic fatty liver disease: A systematic review and meta-analysis. J. Hepatol. 69, 1335–1348 (2018).
Yu, Y. & Chai, J. The function of miRNAs and their potential as therapeutic targets in burn-induced insulin resistance (review). Int. J. Mol. Med. 35, 305–310 (2015).
Fesler, A. et al. Autophagy regulated by miRNAs in colorectal cancer progression and resistance. Cancer Transl. Med. 3, 96–100 (2017).
Li, J. et al. GPC1 exosome and its regulatory miRNAs are specific markers for the detection and target therapy of colorectal cancer. J. Cell. Mol. Med. 21, 838–847 (2017).
Feng, Y. et al. Exosome-derived miRNAs as predictive biomarkers for diffuse large B-cell lymphoma chemotherapy resistance. Epigenomics 11, 35–51 (2019).
Deng, S. et al. Targeting autophagy using natural compounds for cancer prevention and therapy. Cancer 125, 1228–1246 (2019).
Zheng, K., He, Z., Kitazato, K. & Wang, Y. Selective autophagy regulates cell cycle in cancer therapy. Theranostics 9, 104–125 (2019).
Ueno, T. et al. Differential involvement of autophagy and apoptosis in response to chemoendocrine and endocrine therapy in breast cancer: JBCRG-07TR. Int. J. Mol. Sci. 20, pii: E984 (2019).
Zhou, F. et al. Down-regulation of OGT promotes cisplatin resistance by inducing autophagy in ovarian cancer. Theranostics 8, 5200–5212 (2018).
Zhan, Y. et al. The novel autophagy inhibitor alpha-hederin promoted paclitaxel cytotoxicity by increasing reactive oxygen species accumulation in non-small cell lung cancer cells. Int. J. Mol. Sci. 19, https://doi.org/10.3390/ijms19103221 (2018).
Ma, T. et al. USP9X inhibition improves gemcitabine sensitivity in pancreatic cancer by inhibiting autophagy. Cancer Lett. 436, 129–138 (2018).
Yu, T. et al. Fusobacterium nucleatum promotes chemoresistance to colorectal cancer by modulating autophagy. Cell 170, 548–563. e516 (2017).
Li, J. et al. TANRIC: an interactive open platform to explore the function of lncRNAs in cancer. Cancer Res. 75, 3728–3737 (2015).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Dong, H. et al. Activation of LncRNA TINCR by H3K27 acetylation promotes Trastuzumab resistance and epithelial-mesenchymal transition by targeting MicroRNA-125b in breast cancer. Mol. Cancer 18, 3 (2019).
Zhang, F. et al. A miR-567-PIK3AP1-PI3K/AKT-c-Myc feedback loop regulates tumour growth and chemoresistance in gastric cancer. EBioMedicine 44, 311–321 (2019).
Bertoli, G. et al. MicroRNA-567 dysregulation contributes to carcinogenesis of breast cancer, targeting tumor cell proliferation, and migration. Breast Cancer Res. Treat. 161, 605–616 (2017).
Pankiv, S. et al. p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J. Biol. Chem. 282, 24131–24145 (2007).
Shvets, E. & Elazar, Z. Autophagy-independent incorporation of GFP-LC3 into protein aggregates is dependent on its interaction with p62/SQSTM1. Autophagy 4, 1054–1056 (2008).
Mauthe, M. et al. Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion. Autophagy 14, 1435–1455 (2018).
Kanlikilicer, P. et al. Exosomal miRNA confers chemo resistance via targeting Cav1/p-gp/M2-type macrophage axis in ovarian cancer. EBioMedicine 38, 100–112 (2018).
Verma, S. et al. Trastuzumab emtansine for HER2-positive advanced breast cancer. N. Engl. J. Med. 367, 1783–1791 (2012).
Perez, E. A. et al. Relationship between tumor biomarkers and efficacy in MARIANNE, a phase III study of trastuzumab emtansine +/− pertuzumab versus trastuzumab plus taxane in HER2-positive advanced breast cancer. BMC Cancer 19, 517 (2019).
Shetty, P. et al. Annexin A2 and its downstream IL-6 and HB-EGF as secretory biomarkers in the differential diagnosis of Her-2 negative breast cancer. Ann. Clin. Biochem. 54, 463–471 (2017).
Kim, S. B. et al. Relationship between tumor biomarkers and efficacy in TH3RESA, a phase III study of trastuzumab emtansine (T-DM1) vs. treatment of physician’s choice in previously treated HER2-positive advanced breast cancer. Int. J. Cancer 139, 2336–2342 (2016).
Montemurro, F. et al. Potential biomarkers of long-term benefit from single-agent trastuzumab or lapatinib in HER2-positive metastatic breast cancer. Mol. Oncol. 8, 20–26 (2014).
Ponde, N. et al. Cardiac biomarkers for early detection and prediction of trastuzumab and/or lapatinib-induced cardiotoxicity in patients with HER2-positive early-stage breast cancer: a NeoALTTO sub-study (BIG 1-06). Breast Cancer Res. Treat. 168, 631–638 (2018).
Binenbaum, Y. et al. Transfer of miRNA in macrophage-derived exosomes induces drug resistance in pancreatic adenocarcinoma. Cancer Res. 78, 5287–5299 (2018).
Liu, D. et al. MicroRNA-567 inhibits cell proliferation, migration and invasion by targeting FGF5 in osteosarcoma. EXCLI J. 17, 102–112 (2018).
Degenhardt, K. et al. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell 10, 51–64 (2006).
Fu, X. T. et al. MicroRNA-30a suppresses autophagy-mediated anoikis resistance and metastasis in hepatocellular carcinoma. Cancer Lett. 412, 108–117 (2018).
Ren, W. W. et al. MicroRNA-125b reverses oxaliplatin resistance in hepatocellular carcinoma by negatively regulating EVA1A mediated autophagy. Cell Death Dis. 9, 547 (2018).
Xiong, J. et al. MicroRNA-410-3p attenuates gemcitabine resistance in pancreatic ductal adenocarcinoma by inhibiting HMGB1-mediated autophagy. Oncotarget 8, 107500–107512 (2017).
Le Bars, R., Marion, J., Le Borgne, R., Satiat-Jeunemaitre, B. & Bianchi, M. W. ATG5 defines a phagophore domain connected to the endoplasmic reticulum during autophagosome formation in plants. Nat. Commun. 5, 4121 (2014).
Rai, S. et al. The ATG5-binding and coiled coil domains of ATG16L1 maintain autophagy and tissue homeostasis in mice independently of the WD domain required for LC3-associated phagocytosis. Autophagy 15, 599–612 (2019).
Sun, Y. & Liu, J. Potential of cancer cell-derived exosomes in clinical application: a review of recent research advances. Clin. Ther. 36, 863–872 (2014).
Azmi, A. S., Bao, B. & Sarkar, F. H. Exosomes in cancer development, metastasis, and drug resistance: a comprehensive review. Cancer Metastasis Rev. 32, 623–642 (2013).
Phinney, D. G. & Pittenger, M. F. Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells 35, 851–858 (2017).
Acknowledgements
We thank Professor Manran Liu, The Key Laboratory of Laboratory Medical Diagnostics, Chinese Ministry of Education, Chongqing Medical University, Chongqing 400016, China, for critical technical support. This study is supported by National Science Foundation of China (81960475, 81601726, 81702557).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This study was approved by Research Scientific Ethics Committee of the First Affiliated Hospital of Zhengzhou University, The Second Affiliated Hospital of Chongqing Medical University, and Hainan General Hospital. All participants signed informed consent prior to using the tissues and serum samples for scientific research.
Consent for publication
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by B. Zhivotovsky
This article has been retracted. Please see the retraction notice for more detail:https://doi.org/10.1038/s41419-023-06254-5
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, M., Hu, J., Lu, P. et al. RETRACTED ARTICLE: Exosome-transmitted miR-567 reverses trastuzumab resistance by inhibiting ATG5 in breast cancer. Cell Death Dis 11, 43 (2020). https://doi.org/10.1038/s41419-020-2250-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-020-2250-5
This article is cited by
-
Drug resistance in breast cancer is based on the mechanism of exocrine non-coding RNA
Discover Oncology (2024)
-
The importance of social media and brand trust to promote tourism brands: case study in Ho Chi Minh City
Quality & Quantity (2024)
-
Genetic variations in tumor-suppressor miRNA-encoding genes and their target genes: focus on breast cancer development and possible therapeutic strategies
Clinical and Translational Oncology (2023)
-
Global research trends in tumor stem cell-derived exosomes and tumor microenvironment: visualization biology analysis
Journal of Cancer Research and Clinical Oncology (2023)
-
Relevance and mechanism of STAT3/miR-221-3p/Fascin-1 axis in EGFR TKI resistance of triple-negative breast cancer
Molecular and Cellular Biochemistry (2023)