Abstract
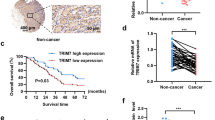
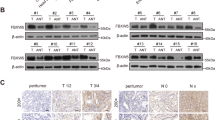
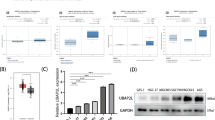
Tripartite motif 17 (TRIM17) belongs to a subfamily of the RING-type E3 ubiquitin ligases, and regulates several cellular processes and pathological conditions including cancer. However, its potential function in gastric cancer (GC) remains obscure. Here, we have found TRIM17 mRNA and protein levels are both upregulated in human GC compared with normal specimens, and TRIM17 upregulation indicates poor survival for GC patients. Functionally, TRIM17 was found to act as an oncogene by promoting the proliferation and survival of GC cell lines AGS and HGC-27. Mechanistically, TRIM17 acts to interact with BAX and promote its ubiquitination and proteasomal degradation, leading to a deficiency in BAX-dependent apoptosis in GC cells in the absence and presence of apoptosis stimuli. Moreover, TRIM17 and BAX expression levels are inversely correlated in human GC specimens. Our data thus suggest TRIM17 contributes to gastric cancer survival through regulating BAX protein stability and antagonizing apoptosis, which provides a promising therapeutic target for GC treatment and a biomarker for prognosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The TGCA data in Fig. 1A and supplementary Fig. S1A-B were downloaded from http://xena.ucsc.edu/public/. The Kaplan-Meier survival analysis in Figs. 1B and 7C was downloaded from https://www.kmplot.com. The GSEA data in Fig. 3A and supplementary Fig. S3A were retrieved from http://www.gsea-msigdb.org/gsea/downloads.jsp. All of the original immunoblots were provided as Supplementary material. Other raw data that support the findings of this study can be made available upon reasonable request.
References
World health organization, international agency for research on cancer 2023. GLOBOCAN 2020. Accessed January 2, 2023. gco.iarc.fr.
Joshi SS, Badgwell BD. Current treatment and recent progress in gastric cancer. CA Cancer J Clin. 2021;71:264–79.
Selim JH, Shaheen S, Sheu WC, Hsueh CT. Targeted and novel therapy in advanced gastric cancer. Exp Hematol Oncol. 2019;8:25.
Hatakeyama S. TRIM family proteins: roles in autophagy, immunity, and carcinogenesis. Trends Biochem Sci. 2017;42:297–311.
Hatakeyama S. TRIM proteins and cancer. Nat Rev Cancer. 2011;11:792–804.
Venuto S, Merla G. E3 ubiquitin ligase TRIM proteins, cell cycle and mitosis. Cells 2019;8:510.
Basu SM, Kozoriz A, Desagher S, Lassot I. To ubiquitinate or not to ubiquitinate: TRIM17 in cell life and death. Cells 2021;10:1235.
Endo H, Ikeda K, Urano T, Horie-Inoue K, Inoue S. Terf/TRIM17 Stimulates degradation of kinetochore protein ZWINT and regulates cell proliferation. J Biochem. 2012;151:139–44.
Lionnard L, Duc P, Brennan MS, Kueh AJ, Pal M, Guardia F, et al. TRIM17 and TRIM28 antagonistically regulate the ubiquitination and anti-apoptotic activity of BCL2A1. Cell Death Differ. 2019;26:902–17.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011;144:646–74.
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, et al. Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 2001;292:727–30.
Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9:47–59.
Rampino N, Yamamoto H, Ionov Y, Li Y, Sawai H, Reed JC, et al. Somatic frameshift mutations in the BAX gene in colon cancers of the microsatellite mutator phenotype. Science 1997;275:967–9.
Nunes CT, Miners KL, Dolton G, Pepper C, Fegan C, Mason MD, et al. A novel tumor antigen derived from enhanced degradation of Bax protein in human cancers. Cancer Res. 2011;71:5435–44.
Liu Z, Ding Y, Ye N, Wild C, Chen H, Zhou J. Direct activation of Bax protein for cancer therapy. Med Res Rev. 2016;36:313–41.
Li B, Dou QP. Bax degradation by the ubiquitin/proteasome-dependent pathway: involvement in tumor survival and progression. Proc Natl Acad Sci USA. 2000;97:3850–5.
Johnson BN, Berger AK, Cortese GP, Lavoie MJ. The ubiquitin E3 ligase parkin regulates the proapoptotic function of Bax. Proc Natl Acad Sci USA. 2012;109:6283–8.
Charan RA, Johnson BN, Zaganelli S, Nardozzi JD, LaVoie MJ. Inhibition of apoptotic Bax translocation to the mitochondria is a central function of parkin. Cell Death Dis. 2014;5:e1313.
Cakir Z, Funk K, Lauterwasser J, Todt F, Zerbes RM, Oelgeklaus A, et al. Parkin promotes proteasomal degradation of misregulated BAX. J Cell Sci. 2017;130:2903–13.
Benard G, Neutzner A, Peng G, Wang C, Livak F, Youle RJ, et al. IBRDC2, an IBR-type E3 ubiquitin ligase, is a regulatory factor for Bax and apoptosis activation. EMBO J. 2010;29:1458–71.
Yuan Y, Wang YY, Liu X, Luo B, Zhang L, Zheng F, et al. KPC1 alleviates hypoxia/reoxygenation-induced apoptosis in rat cardiomyocyte cells though BAX degradation. J Cell Physiol. 2019;234:22921–34.
Agrawal SG, Liu FT, Wiseman C, Shirali S, Liu H, Lillington D, et al. Increased proteasomal degradation of Bax is a common feature of poor prognosis chronic lymphocytic leukemia. Blood 2008;111:2790–6.
Liu FT, Agrawal SG, Gribben JG, Ye H, Du MQ, Newland AC, et al. Bortezomib blocks Bax degradation in malignant B cells during treatment with TRAIL. Blood 2008;111:2797–805.
Magiera MM, Mora S, Mojsa B, Robbins I, Lassot I, Desagher S. Trim17-mediated ubiquitination and degradation of Mcl-1 initiate apoptosis in neurons. Cell Death Differ. 2013;20:281–92.
Grice GL, Nathan JA. The recognition of ubiquitinated proteins by the proteasome. Cell Mol Life Sci. 2016;73:3497–506.
Sun D, Wu R, Zheng J, Li P, Yu L. Polyubiquitin chain-induced p62 phase separation drives autophagic cargo segregation. Cell Res. 2018;28:405–15.
Peng R, Zhu J, Deng S, Shi H, Xu S, Wu H, et al. Targeting BAX ubiquitin-binding sites reveals that BAX activation is essential for its ubiquitin-dependent degradation. J Cell Biochem. 2020;121:2802–10.
Mohamad Anuar NN, Nor Hisam NS, Liew SL, Ugusman A. Clinical review: navitoclax as a pro-apoptotic and anti-fibrotic agent. Front Pharm. 2020;11:564108.
Manoochehri M, Karbasi A, Bandehpour M, Kazemi B. Down-regulation of BAX gene during carcinogenesis and acquisition of resistance to 5-FU in colorectal cancer. Pathol Oncol Res. 2014;20:301–7.
Sharifi S, Barar J, Hejazi MS, Samadi N. Roles of the Bcl-2/Bax ratio, caspase-8 and 9 in resistance of breast cancer cells to paclitaxel. Asian Pac J Cancer Prev. 2014;15:8617–22.
Wu Y, Xing D, Liu L, Gao B. Regulation of Bax activation and apoptotic response to UV irradiation by p53 transcription-dependent and -independent pathways. Cancer Lett. 2008;271:231–9.
Mandell MA, Jain A, Arko MJ, Chauha S, Kimura T, Dinkins C, et al. TRIM proteins regulate authphagy and can target autophagic substrates by direct recognition. Dev Cell. 2014;30:394–409.
Mandell MA, Jain A, Kumar S, Castleman MJ, Anwar T, Eskelinen EL, et al. TRIM17 contributes to autophagy of midbodies while activety sparing other targets from degradation. J Cell Sci. 2016;129:3562–73.
Zhong T, Zhang J, Liu X, Li H TRIM17-mediated ubiquitination and degradation of RBM38 promotes cisplatin resistance in non-small cell lung cancer. Cell Oncol (Dordr). 2023;https://doi.org/10.1007/s13402-023-00825-6.
Wei Y, Zhang L, Wang C, Li Z, Luo M, Xie G, et al. Anti-apoptotic protein BCL-XL as a therapeutic vulnerability in gastric cancer. Anim Model Exp Med. 2023;6:245–54.
Nor Hisam NS, Ugusman A, Rajab NF, Ahmad MF, Fenech M, Liew SL, et al. Combination therapy of navitoclax with chemotherapeutic agents in solid tumors and blood cancer: a review of current evidence. Pharmaceutics 2021;13:1353.
Lassot I, Robbins I, Kristiansen M, Rahmeh R, Jaudon F, Magiera MM, et al. Trim17, a novel E3 ubiquitin-ligase, initiates neuronal apoptosis. Cell Death Differ. 2010;17:1928–41.
Mojsa B, Mora S, Bossowski JP, Lassot I, Desagher S. Control of neuronal apoptosis by reciprocal regulation of NFATc3 and Trim17. Cell Death Differ. 2015;22:274–86.
Sha MQ, Zhao XL, Li L, Li LH, Li Y, Dong TG, et al. EZH2 mediates lidamycin-induced cellular senescence through regulating p21 expression in human colon cancer cells. Cell Death Dis. 2016;7:e2486.
Shen J, Li P, Shao X, Yang Y, Liu XJ, Feng M, et al. The E3 ligase RING1 targets p53 for degradation and promotes cancer cell proliferation and survival. Cancer Res. 2018;78:359–71.
Acknowledgements
This project is sponsored by the National Natural Science Foundation of China (Nos. 82002966; 82073241), the CAMS Innovation Fund for Medical Sciences (Nos. 2021-I2M-1-030; 2022-I2M-2-002; 2022-I2M-1-016) and the National Key Research and Development Program (2022YFC2804700).
Author information
Authors and Affiliations
Contributions
ZW, JS, and HY designed the experiments; JS and HY performed most of the animal, functional, and biochemical experiments; HY performed bioinformatical analyses and tissue IHC staining; XQ, YC, LZ, and JL(in) performed some animal and biochemical experiments; JL(ang), QY and ZW assisted in the analysis of some experiments and interpreted the data; ZW, JS and HY drafted the manuscript; ZW conceived and supervised the project. All authors have reviewed and approved the submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
All animal experiments were approved by the Ethics Committee of the Institute of Medicinal Biotechnology, Peking Union Medical College and Chinese Academy of Medical Sciences in China, and conducted in accordance with the regulations and operational procedures of experimental animal management.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, J., Yang, H., Qiao, X. et al. The E3 ubiquitin ligase TRIM17 promotes gastric cancer survival and progression via controlling BAX stability and antagonizing apoptosis. Cell Death Differ 30, 2322–2335 (2023). https://doi.org/10.1038/s41418-023-01221-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41418-023-01221-1
This article is cited by
-
The histone methyltransferase ASH1L protects against bone loss by inhibiting osteoclastogenesis
Cell Death & Differentiation (2024)