Abstract
Background
The study aimed to examine the significance of insulin receptor (INSR) expression in predicting resistance to axitinib in clear cell renal cell carcinoma (ccRCC).
Methods
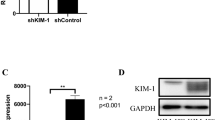
Clinicopathological data were collected from 36 consecutive patients with metastatic RCC who received axitinib. Thirty-three primary tumours were obtained for immunohistochemistry. Patient-derived xenograft (PDX) models were created by transplanting primary tumours into immunodeficient mice, establishing axitinib-resistant PDX models. RCC cell lines were co-cultured with human renal glomerular endothelial cells (HGECs) treated with siRNA of INSR (HGEC-siINSR). Gene expression alteration was analysed using microarray.
Results
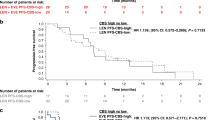
The patients with low INSR expression who received axitinib had a poorer outcome. Multivariate analysis showed that INSR expression was the independent predictor of progression-free survival. INSR expression decreased in axitinib-resistant PDX tumours. RCC cell lines showed upregulated interferon responses and highly increased interferon-β levels by co-culturing with HGEC-siINSR. HGECs showed decreased INSR and increased interferon-β after axitinib administration. RCC cell lines co-cultured with HGEC-siINSR showed high programmed death-ligand 1 (PD-L1) expression, which increased after interferon-β administration.
Conclusions
Decreased INSR in RCC could be a biomarker to predict axitinib resistance. Regarding the resistant mechanism, vascular endothelial cells with decreased INSR in RCC may secrete interferon-β and induce PD-L1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data sets analysed during this study are available from the corresponding author upon reasonable request. Publicly available data sets were extracted from The Cancer Genome Atlas (TCGA) (https://portal.gdc.cancer.gov/) and cBioportal (https://www.cbioportal.org/).
References
Motzer RJ, Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S, et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015;373:1803–13.
Motzer RJ, Tannir NM, McDermott DF, Aren Frontera O, Melichar B, Choueiri TK, et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378:1277–90.
Rini BI, Plimack ER, Stus V, Gafanov R, Hawkins R, Nosov D, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1116–27.
Choueiri TK, Powles T, Burotto M, Escudier B, Bourlon MT, Zurawski B, et al. Nivolumab plus cabozantinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2021;384:829–41.
Motzer R, Alekseev B, Rha SY, Porta C, Eto M, Powles T, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289–300.
Motzer RJ, Penkov K, Haanen J, Rini B, Albiges L, Campbell MT, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103–15.
Aeppli S, Schmaus M, Eisen T, Escudier B, Grünwald V, Larkin J, et al. First-line treatment of metastatic clear cell renal cell carcinoma: a decision-making analysis among experts. ESMO Open. 2021;6:100030.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356:125–34.
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115–24.
Rini BI, Escudier B, Tomczak P, Kaprin A, Szczylik C, Hutson TE, et al. Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (axis): a randomised phase 3 trial. Lancet. 2011;378:1931–9.
Sternberg CN, Davis ID, Mardiak J, Szczylik C, Lee E, Wagstaff J, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase iii trial. J Clin Oncol. 2010;28:1061–8.
Heng DY, Xie W, Regan MM, Harshman LC, Bjarnason GA, Vaishampayan UN, et al. External validation and comparison with other models of the International Metastatic Renal-Cell Carcinoma Database Consortium Prognostic Model: a population-based study. Lancet Oncol. 2013;14:141–8.
Lindskog M, Wahlgren T, Sandin R, Kowalski J, Jakobsson M, Lundstam S, et al. Overall survival in swedish patients with renal cell carcinoma treated in the period 2002 to 2012: Update of the rencomp study with subgroup analysis of the synchronous metastatic and elderly populations. Urol Oncol. 2017;35:541.e515–e522.
Busch J, Seidel C, Erber B, Issever AS, Hinz S, Kempkensteffen C, et al. Retrospective comparison of triple-sequence therapies in metastatic renal cell carcinoma. Eur Urol. 2013;64:62–70.
Heng DY, Mackenzie MJ, Vaishampayan UN, Bjarnason GA, Knox JJ, Tan MH, et al. Primary anti-vascular endothelial growth factor (vegf)-refractory metastatic renal cell carcinoma: clinical characteristics, risk factors, and subsequent therapy. Ann Oncol. 2012;23:1549–55.
Garcia-Donas J, Leandro-Garcia LJ, Gonzalez Del Alba A, Morente M, Alemany I, Esteban E, et al. Prospective study assessing hypoxia-related proteins as markers for the outcome of treatment with sunitinib in advanced clear-cell renal cell carcinoma. Ann Oncol. 2013;24:2409–14.
Takahashi M, Rhodes DR, Furge KA, Kanayama H, Kagawa S, Haab BB, et al. Gene expression profiling of clear cell renal cell carcinoma: gene identification and prognostic classification. Proc Natl Acad Sci USA 2001;98:9754–9.
Dandona P, Aljada A, Bandyopadhyay A. Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol. 2004;25:4–7.
Wilmes LJ, Pallavicini MG, Fleming LM, Gibbs J, Wang D, Li KL, et al. Ag-013736, a novel inhibitor of vegf receptor tyrosine kinases, inhibits breast cancer growth and decreases vascular permeability as detected by dynamic contrast-enhanced magnetic resonance imaging. Magn Reson Imaging. 2007;25:319–27.
Kirihara Y, Takechi M, Kurosaki K, Kobayashi Y, Saito Y, Takeuchi T. Anesthetic effects of a three-drugs mixture–comparison of administrative routes and antagonistic effects of atipamezole in mice. Exp Anim. 2015;64:39–47.
Thompson RH, Kuntz SM, Leibovich BC, Dong H, Lohse CM, Webster WS, et al. Tumor b7-h1 is associated with poor prognosis in renal cell carcinoma patients with long-term follow-up. Cancer Res. 2006;66:3381–5.
Thompson RH, Dong H, Kwon ED. Implications of b7-h1 expression in clear cell carcinoma of the kidney for prognostication and therapy. Clin Cancer Res. 2007;13:709s–15s.
Xu F, Xu L, Wang Q, An G, Feng G, Liu F. Clinicopathological and prognostic value of programmed death ligand-1 (pd-l1) in renal cell carcinoma: a meta-analysis. Int J Clin Exp Med. 2015;8:14595–603.
Lee SH, Hu W, Matulay JT, Silva MV, Owczarek TB, Kim K, et al. Tumor evolution and drug response in patient-derived organoid models of bladder cancer. Cell. 2018;173:515.e7–28.e7.
Rini BI. Metastatic renal cell carcinoma: many treatment options, one patient. J Clin Oncol. 2009;27:3225–34.
Seo GJ, Yang A, Tan B, Kim S, Liang Q, Choi Y, et al. Akt kinase-mediated checkpoint of cGAS DNA sensing pathway. Cell Rep. 2015;13:440–9.
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, et al. Tumor-associated b7-h1 promotes t-cell apoptosis: a potential mechanism of immune evasion. Nat Med. 2002;8:793–800.
Curiel TJ, Wei S, Dong H, Alvarez X, Cheng P, Mottram P, et al. Blockade of b7-h1 improves myeloid dendritic cell-mediated antitumor immunity. Nat Med. 2003;9:562–7.
Gerlinger M, Rowan AJ, Horswell S, Math M, Larkin J, Endesfelder D, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366:883–92.
Osterman CK, Rose TL. A systematic review of systemic treatment options for advanced non-clear cell renal cell carcinoma. Kidney Cancer. 2020;4:15–27.
Choueiri TK, Escudier B, Powles T, Mainwaring PN, Rini BI, Donskov F, et al. Cabozantinib versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015;373:1814–23.
Motzer RJ, Hutson TE, Ren M, Dutcus C, Larkin J. Independent assessment of lenvatinib plus everolimus in patients with metastatic renal cell carcinoma. Lancet Oncol. 2016;17:e4–5.
Zou S, Tong Q, Liu B, Huang W, Tian Y, Fu X. Targeting stat3 in cancer immunotherapy. Mol Cancer. 2020;19:145.
Funding
Funding was provided by a research grant from Novartis Pharma (grant numbers not applicable) and MSD (grant numbers not applicable).
Author information
Authors and Affiliations
Contributions
M Takahashi planned the study, collected the patients’ data and tumour samples, obtained informed consent from the patients, analysed the results, and wrote the manuscript. KD planned and performed the experiments, collected tumour samples, obtained informed consent, analysed the results, and helped write the manuscript. TF planned the experiments, collected tumour samples, obtained informed consent, and helped write the manuscript. YF performed the experiments and helped write the manuscript. YB and HU performed immunohistochemical staining and evaluation. MK performed the bioinformatic analysis for the microarray data. T-OD performed immunohistochemical staining and animal experiments. YS, RT, YU, M Tsuda, YK, KY, and YY collected tumour samples, obtained informed consents, and helped write the manuscript. HK supervised the study as a whole and helped write the manuscript.
Corresponding author
Ethics declarations
Competing interests
M Takahashi received honoraria from Pfizer Inc. and a research grant from Novartis Pharma and MSD. HK received honoraria from Pfizer Inc. Other authors have no conflict of interest.
Ethics approval and consent to participate
The study was approved by the ethical review board of Tokushima University Hospital (permission number 709) and conducted according to the ethical standards of the Declaration of Helsinki. The ethical review board of Tokushima University Hospital (permission number: 1841) and the Institutional Animal Care and Use Committee (Permission number: T2019-56) of Tokushima University approved the animal experiments. All of the animal experiments were performed according to guidelines of the animal facility at the University of Tokushima and conform to Fundamental Guidelines for Proper Conduct of Animal Experiments and Related Activities in Academic Research Institutions under the jurisdiction of the Ministry of Education, Culture, Sports, Science and Technology in Japan.
Consent for publication
Written informed consent for data usage and publication was obtained from all patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takahashi, M., Daizumoto, K., Fukawa, T. et al. Insulin receptor expression to predict resistance to axitinib and elucidation of the underlying molecular mechanism in metastatic renal cell carcinoma. Br J Cancer 129, 521–530 (2023). https://doi.org/10.1038/s41416-023-02325-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-023-02325-8