Abstract
Background
Evidence is limited on inflammation-related dietary patterns and mortality in ovarian cancer survivors.
Methods
We examined the associations between pre- and post-diagnosis dietary patterns, including change in diet from before to after diagnosis, and mortality among 1003 ovarian cancer survivors in two prospective cohort studies. Dietary pattern scores for empirical dietary inflammatory pattern (EDIP) and Alternative Healthy Eating Index (AHEI) were calculated based on food frequency questionnaires. We used Cox proportional hazard models to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) for ovarian cancer-specific and all-cause mortality.
Results
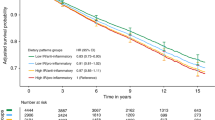
Pre-diagnosis EDIP score and AHEI were not associated with mortality. Among non-high grade serous cases, a higher post-diagnosis EDIP score was associated with increased risk of all-cause mortality (HR5th vs 1st quintile = 1.95, 95% CI = 1.04–3.67, p-trend = 0.06). Compared to survivors consuming a low EDIP score diet before and after diagnosis, high post-diagnosis EDIP was associated with increased risk of ovarian cancer specific mortality (pre-to-post diagnosis low/high, HR = 1.38, 95% CI = 0.99–1.92; high/high HR = 1.58, 95% CI = 1.09–2.30) and all-cause mortality (low/high HR = 1.44, 95% CI = 1.06–1.95; high/high HR = 1.55, 95% CI = 1.10–2.19).
Conclusion
Consuming a more inflammatory dietary pattern post-diagnosis was associated with increased mortality in ovarian cancer survivors, suggesting limiting the inflammatory potential of diet post-diagnosis could lead to enhanced survivorship.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The data generated in this study are not publicly available but are available upon request. Further information including the procedures to obtain and access data from the Nurses’ Health Studies is described at https://www.nurseshealthstudy.org/researchers (contact email: nhsaccess@channing.harvard.edu).
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clinicians. 2018;68:394–424.
Lheureux S, Braunstein M, Oza AM. Epithelial ovarian cancer: evolution of management in the era of precision medicine. CA: A Cancer J Clinicians. 2019;69:280–304.
Yi M, Li T, Niu M, Luo S, Chu Q, Wu K. Epidemiological trends of women’s cancers from 1990 to 2019 at the global, regional, and national levels: a population-based study. Biomark Res. 2021;9:55–55.
American Cancer Society. Cancer Facts & Figures 2021. Atlanta: American Cancer Society; 2021.
Ness RB, Cottreau C. Possible role of ovarian epithelial inflammation in ovarian cancer. JNCI 1999;91:1459–67.
Poole EM, Lee IM, Ridker PM, Buring JE, Hankinson SE, Tworoger SS. A prospective study of circulating C-reactive protein, interleukin-6, and tumor necrosis factor α receptor 2 levels and risk of ovarian cancer. Am J Epidemiol. 2013;178:1256–64.
Peres LC, Mallen AR, Townsend MK, Poole EM, Trabert B, Allen NE, et al. High levels of C-reactive protein are associated with an increased risk of ovarian cancer: results from the ovarian cancer cohort consortium. Cancer Res. 2019;79:5442–51.
Savant, SS, Sriramkumar, S, O’Hagan, HM. The role of inflammation and inflammatory mediators in the development, progression, metastasis, and chemoresistance of epithelial ovarian cancer. Cancers (Basel). 2018;10.
Hefler LA, Concin N, Hofstetter G, Marth C, Mustea A, Sehouli J, et al. Serum C-reactive protein as independent prognostic variable in patients with ovarian cancer. Clin Cancer Res. 2008;14:710–4.
Yang Z, Gu JH, Guo CS, Li XH, Yang WC. Preoperative neutrophil-to-lymphocyte ratio is a predictor of survival of epithelial ovarian cancer: a systematic review and meta-analysis of observational studies. Oncotarget.2017;8:46414–24.
Gao XP, Liu YH, Liu ZY, Wang LJ, Jing CX, Zhu S, et al. Pretreatment lymphocyte-to-monocyte ratio as a predictor of survival among patients with ovarian cancer: a meta-analysis. Cancer Manag Res. 2019;11:1907–20.
Galland L. Diet and inflammation. Nutr Clin Pr. 2010;25:634–40.
Thomson CA, Crane TE, Wertheim BC, Neuhouser ML, Li W, Snetselaar LG, et al. Diet quality and survival after ovarian cancer: results from the Women’s Health Initiative. J Natl Cancer Inst. 2014;106.
Peres LC, Hebert JR, Qin B, Guertin KA, Bandera EV, Shivappa N, et al. Prediagnostic proinflammatory dietary potential is associated with all-cause mortality among African-American women with high-grade serous ovarian carcinoma. J Nutr. 2019;149:1606–16.
Nagle CM, Ibiebele T, Shivappa N, Hébert JR, DeFazio A, Webb PM. The association between the inflammatory potential of diet and risk of developing, and survival following, a diagnosis of ovarian cancer. Eur J Nutr. 2019;58:1747–56.
Al Ramadhani RM, Nagle CM, Ibiebele TI, Grant P, Friedlander M, DeFazio A, et al. Pre- and post-diagnosis diet quality and ovarian cancer survival. Cancer Epidemiol Biomark Prev. 2021;30:229–32.
Tabung FK, Smith-Warner SA, Chavarro JE, Wu K, Fuchs CS, Hu FB, et al. Development and validation of an empirical dietary inflammatory index. J Nutr. 2016;146:1560–70.
Li SX, Hodge AM, MacInnis RJ, Bassett JK, Ueland PM, Midttun Ø, et al. Inflammation-related marker profiling of dietary patterns and all-cause mortality in the Melbourne Collaborative Cohort Study. J Nutr. 2021;151:2908–16.
Colditz GA, Hankinson SE. The Nurses’ Health Study: lifestyle and health among women. Nat Rev Cancer. 2005;5:388–96.
Rockhill B, Willett WC, Hunter DJ, Manson JE, Hankinson SE, Spiegelman D, et al. Physical activity and breast cancer risk in a cohort of young women. J Natl Cancer Inst. 1998;90:1155–60.
Yuan C, Spiegelman D, Rimm EB, Rosner BA, Stampfer MJ, Barnett JB, et al. Validity of a dietary questionnaire assessed by comparison with multiple weighed dietary records or 24-hour recalls. Am J Epidemiol. 2017;185:570–84.
Stampfer MJ, Willett WC, Speizer FE, Dysert DC, Lipnick R, Rosner B, et al. Test of the national death index. Am J Epidemiol. 1984;119:837–9.
Tabung FK, Smith-Warner SA, Chavarro JE, Fung TT, Hu FB, Willett WC, et al. An empirical dietary inflammatory pattern score enhances prediction of circulating inflammatory biomarkers in adults. J Nutr. 2017;147:1567–77.
Chiuve SE, Fung TT, Rimm EB, Hu FB, McCullough ML, Wang M, et al. Alternative dietary indices both strongly predict risk of chronic disease. J Nutr. 2012;142:1009–18.
Rhee JJ, Mattei J, Hughes MD, Hu FB, Willett WC. Dietary diabetes risk reduction score, race and ethnicity, and risk of type 2 diabetes in women. Diabetes Care. 2015;38:596–603.
Merritt MA, Rice MS, Barnard ME, Hankinson SE, Matulonis UA, Poole EM, et al. Pre-diagnosis and post-diagnosis use of common analgesics and ovarian cancer prognosis (NHS/NHSII): a cohort study. Lancet Oncol. 2018;19:1107–16.
Zhong X, Guo L, Zhang L, Li Y, He R, Cheng G. Inflammatory potential of diet and risk of cardiovascular disease or mortality: a meta-analysis. Sci Rep. 2017;7:6367.
Deng FE, Shivappa N, Tang Y, Mann JR, Hebert JR. Association between diet-related inflammation, all-cause, all-cancer, and cardiovascular disease mortality, with special focus on prediabetics: findings from NHANES III. Eur J Nutr. 2017;56:1085–93.
Garcia-Arellano A, Martínez-González MA, Ramallal R, Salas-Salvadó J, Hébert JR, Corella D, et al. Dietary inflammatory index and all-cause mortality in large cohorts: The SUN and PREDIMED studies. Clin Nutr. 2019;38:1221–31.
Tabung FK, Steck SE, Liese AD, Zhang J, Ma Y, Caan B, et al. Association between dietary inflammatory potential and breast cancer incidence and death: results from the Women’s Health Initiative. Br J Cancer. 2016;114:1277–85.
Zucchetto A, Gini A, Shivappa N, Hébert JR, Stocco C, Dal Maso L, et al. Dietary inflammatory index and prostate cancer survival. Int J Cancer. 2016;139:2398–404.
Yuan C, Morales-Oyarvide V, Khalaf N, Perez K, Tabung FK, Ho GYF, et al. Prediagnostic inflammation and pancreatic cancer survival. J Natl Cancer Inst. 2021;113:1186–93.
Playdon MC, Nagle CM, Ibiebele TI, Ferrucci LM, Protani MM, Carter J, et al. Pre-diagnosis diet and survival after a diagnosis of ovarian cancer. Br J Cancer. 2017;116:1627–37.
Nagle CM, Purdie DM, Webb PM, Green A, Harvey PW, Bain CJ. Dietary influences on survival after ovarian cancer. Int J Cancer. 2003;106:264–9.
Dolecek TA, McCarthy BJ, Joslin CE, Peterson CE, Kim S, Freels SA, et al. Prediagnosis food patterns are associated with length of survival from epithelial ovarian cancer. J Am Diet Assoc. 2010;110:369–82.
Peres LC, Cushing-Haugen KL, Köbel M, Harris HR, Berchuck A, Rossing MA, et al. Invasive epithelial ovarian cancer survival by histotype and disease stage. J Natl Cancer Inst. 2019;111:60–8.
Kurman RJ, Shih Ie,M. Molecular pathogenesis and extraovarian origin of epithelial ovarian cancer-shifting the paradigm. Hum Pathol. 2011;42:918–31.
Wang T, Townsend MK, Simmons V, Terry KL, Matulonis UA, Tworoger SS. Prediagnosis and postdiagnosis smoking and survival following diagnosis with ovarian cancer. Int J Cancer. 2020;147:736–46.
Jeon SY, Go RE, Heo JR, Kim CW, Hwang KA, Choi KC. Effects of cigarette smoke extracts on the progression and metastasis of human ovarian cancer cells via regulating epithelial-mesenchymal transition. Reprod Toxicol. 2016;65:1–10.
Kelemen LE, Warren GW, Koziak JM, Köbel M, Steed H. Smoking may modify the association between neoadjuvant chemotherapy and survival from ovarian cancer. Gynecol Oncol. 2016;140:124–30.
Müezzinler A, Mons U, Gellert C, Schöttker B, Jansen E, Kee F, et al. Smoking and all-cause mortality in older adults: results from the CHANCES Consortium. Am J Prev Med. 2015;49:e53–63.
Maunsell E, Drolet M, Brisson J, Robert J, Deschênes L. Dietary change after breast cancer: extent, predictors, and relation with psychological distress. J Clin Oncol. 2002;20:1017–25.
Salminen EK, Lagström HK, Heikkilä S, Salminen S. Does breast cancer change patients’ dietary habits? Eur J Clin Nutr. 2000;54:844–8.
Harris HR, Bergkvist L, Wolk A. Folate intake and breast cancer mortality in a cohort of Swedish women. Breast Cancer Res Treat. 2012;132:243–50.
Tabung FK, Huang T, Giovannucci EL, Smith-Warner SA, Tworoger SS, Poole EM. The inflammatory potential of diet and ovarian cancer risk: results from two prospective cohort studies. Br J Cancer. 2017;117:907–11.
Lau TS, Chan LK, Wong EC, Hui CW, Sneddon K, Cheung TH, et al. A loop of cancer-stroma-cancer interaction promotes peritoneal metastasis of ovarian cancer via TNFα-TGFα-EGFR. Oncogene. 2017;36:3576–87.
Kulbe H, Hagemann T, Szlosarek PW, Balkwill FR, Wilson JL. The inflammatory cytokine tumor necrosis factor-alpha regulates chemokine receptor expression on ovarian cancer cells. Cancer Res. 2005;65:10355–62.
Wang Y, Niu XL, Qu Y, Wu J, Zhu YQ, Sun WJ, et al. Autocrine production of interleukin-6 confers cisplatin and paclitaxel resistance in ovarian cancer cells. Cancer Lett. 2010;295:110–23.
Acknowledgements
We would like to thank the following state cancer registries for their help: AL, AZ, AR, CA, CO, CT, DE, FL, GA, ID, IL, IN, IA, KY, LA, ME, MD, MA, MI, NE, NH, NJ, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, VA, WA, WY. The authors assume full responsibility for analyses and interpretation of these data.
Funding
This work was supported in part by the National Institutes of Health (UM1 CA186107, P01 CA87969, U01 CA176726). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. NS was supported by the Rivkin Scientific Scholars Award and Department of Defense W81XWH2110320.
Author information
Authors and Affiliations
Contributions
NS, KLT, SST, HRH contributed to the study concept and design. NS conducted the statistical analysis and drafted the initial manuscript. All authors contributed to the data interpretation and provided critical revisions of the final manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
SST received a grant from BMS that is unrelated to this work. The authors declare no competing interest.
Ethics approval and consent to participate
The study protocol was approved by the institutional review boards of the Brigham and Women’s Hospital and Harvard T.H. Chan School of Public Health, and those of participating registries as required. Completion of the questionnaire implied informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41416_2022_1901_MOESM1_ESM.docx
Supplementary Table1, Supplementary Table2, Supplementary Table3, Supplementary Table4, Supplementary Table5, Supplementary Table6, Supplementary Table7, Supplementary Table8
Rights and permissions
About this article
Cite this article
Sasamoto, N., Wang, T., Townsend, M.K. et al. Pre-diagnosis and post-diagnosis dietary patterns and survival in women with ovarian cancer. Br J Cancer 127, 1097–1105 (2022). https://doi.org/10.1038/s41416-022-01901-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-022-01901-8