Abstract
Cancer cachexia has long been perceived as a nutritional syndrome. However, nutritional interventions have continued to be ineffective. With the recent recognition of the importance of systemic inflammation in the definition of this syndrome and treatment, has the time come to consider whether this syndrome is primarily a manifestation of systemic inflammation with the consequent implications for future treatment?
Similar content being viewed by others
Introduction
Cancer cachexia is universally recognised to be associated with poor outcomes [1]. In the last decade, since the international consensus on the definition of cancer cachexia [2], there has been increasing research interest in the field. Indeed, a search of PubMed.gov using the term ‘cancer cachexia’, returns 216 published articles in 2011, compared with 457 articles published in 2020 [3].
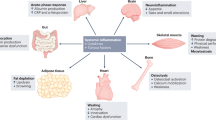
The definition of cancer cachexia has evolved over this time, with the development of robust criteria for diagnosis [4]. Specifically, systemic inflammation has slowly moved to the forefront of the definition, diagnosis and treatment of cancer cachexia. While the basis of the systemic inflammatory response (SIR) in cancer patients is not clear, it is thought to be the non-specific host response to tumour hypoxia/necrosis and/or local tissue damage [5]. The host response to a noxious stimulus such as cancer include changes in neuroendocrine metabolism, haematopoietic changes and production of acute-phase proteins by the liver, such as C-reactive protein (CRP), which initiates and/or sustains the SIR [6]. Conversely, a drop in serum albumin is also observed with rising CRP [7]. Given that the SIR is a key component of the cancer cachexia, CRP and albumin have been combined in scores including the Glasgow Prognostic Score (GPS) and modified GPS (mGPS) to objectively define cachexia [5, 7].
The following article examines the trend of increased recognition of the importance of systemic inflammation in the definitions of cancer cachexia over time and diagnostic frameworks proposed. Furthermore, how the shift to viewing cancer cachexia as a SIR syndrome has and will inform on treatment and clinical trials [8].
The role of inflammation in defining cachexia
To date, there have been numerous definitions of cancer cachexia proposed within the literature [9] (see Table 1). One of the first arose in 2006, in the ESPEN Guidelines on Enteral Nutrition: Non-surgical oncology. Arends and colleagues defined cancer cachexia as activation of “systemic pro-inflammatory processes in response to the tumour”, with “metabolic derangements include insulin resistance, increased lipolysis and high normal or increased lipid oxidation with loss of body fat, increased protein turnover with loss of muscle mass and an increase in production of acute-phase proteins” [10]. This definition was supported by the work of Fearon et al. who found that weight loss alone did not identify the full effect of cachexia on physical function and was not a prognostic variable. As such, they proposed a three-factor assessment (weight loss, reduced food intake and systemic inflammation), to identify patients at risk of adverse outcomes [11].
In 2008, the cachexia consensus conference defined cancer cachexia as “a complex metabolic syndrome associated with underlying illness and characterised by loss of muscle with or without loss of fat mass”. Evans et al. also came to the conclusion that “the prominent clinical feature of cachexia is weight loss in adults (corrected for fluid retention) or growth failure in children (excluding endocrine disorders)” and that “Anorexia, inflammation, insulin resistance and increased muscle protein breakdown are frequently associated with wasting disease” [12]. This proposed definition was supported by the Special Interest Group (SIG), part European Society for Clinical Nutrition and Metabolism (ESPEN). Furthermore, Muscaritoli et al. specifically noted that “inflammation does play a pivotal role in the pathogenesis of cachexia and its presence allows for cachexia identification” [13]. Given that systemic inflammation was a key component of the syndrome of cancer cachexia, scores including the Glasgow Prognostic Score (GPS, combination of CRP and albumin) were subsequently developed to objectively define cachexia [5, 7]. This led to Grey and co-workers terming this combination laboratory cachexia [14].
Inflammation as a diagnostic criterion of cachexia
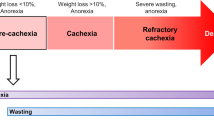
In the 2011 international consensus, Fearon and colleagues outlined the “agreed diagnostic criterion for cachexia as weight loss greater than 5%, or weight loss greater than 2% in individuals already showing depletion according to current bodyweight and height (body mass index [BMI] <20 kg/m2) or skeletal muscle mass (sarcopenia). An agreement was made that the cachexia syndrome can develop progressively through various stages—pre-cachexia to cachexia to refractory cachexia” [2]. They noted that although C-reactive protein (CRP), the prototypical marker of systemic inflammation, had prognostic value in these patients, cachexia could occur in the absence of systemic inflammation. These Fearon criteria have been taken into the BMI/weight loss grade framework that has been shown to have prognostic value [15] and to be associated with quality of life in patients with advanced cancer [16]. However, uncertainty remains around the existence of cancer cachexia in the absence of systemic inflammation, a recognised hallmark of cancer [17].
Where the Evans and the Fearon criteria have been prospectively compared, it was concluded that “This study presents a correlation with prognosis in favour of Evans et al.” definition as a tool for cachexia diagnosis. This means that weight loss and BMI decline are both key factors in patients with cancer leading to cachexia but less decisive as stated by Fearon et al. Instead, extra factors gain importance in order to predict survival, such as chronic inflammation, anaemia, protein depletion, reduced food intake, fatigue, decreased muscle strength and lean tissue depletion” [18]. This conclusion is in accord with subsequent attempts to define cachexia [19, 20] and in recent guidelines [21]. In particular, the Global Leadership Initiative on Malnutrition (GLIM) criteria proposed by Cederholm et al. in a consensus report defined cancer cachexia as chronic disease-related malnutrition with inflammation and the three phenotypic criteria identified were involuntary weight loss, low BMI, low muscle mass and the tumour aetiologic criteria were reduced food intake or assimilation, and inflammation or disease burden [4]. Indeed, Laird et al., in ~2500 patients, proposed that the combination of Eastern Cooperative Oncology Group Performance Status (ECOG-PS) and mGPS effectively stratified quality of life and survival in patients with advanced cancer [22, 23]. More recently, Martin et al. reported, in a cohort of almost 5000 patients that weight loss is largely determined by dietary intake and systemic inflammation (as evidenced by CRP [24]). They concluded that “Modelling weight loss as the dependent variable is an approach that can help to identify clinical features and biomarkers associated with weight loss. Here, we identify criterion values for food intake impairment and CRP that may improve the diagnosis and classification of cancer-associated cachexia” [24]. With reference to the other GLIM phenotypic criteria, low muscle mass, a consistent association with the systemic inflammatory response has been reported [25] and interestingly tumour burden appears to be a less important aetiologic factor [26].
The prognostic value of modulating the systemic inflammatory response in the treatment of cancer cachexia
The role of systemic inflammation in cancer cachexia is highlighted in the definition of Evans et al. and Cederholm et al. [4, 12]. Therefore, nutritional supplementation or therapeutic modification of skeletal muscle mass alone in patients with cancer, whom demonstrate signs of systemic inflammation may be futile [27]. This is consistent with the findings Merker and co-workers [28], who found that nutritional support was not effective in cancer and non-cancer patients with high-grade systemic inflammation (CRP >100 m/l), in a secondary analysis of a randomised clinical trial conducted across eight Swiss hospitals (n = 1950 with a CRP measurement). Indeed, the significance of the systemic inflammatory response to outcomes in patients with cancer is well recognised [29,30,31,32,33], with McAllister and Weinberg noting measures of the systemic inflammatory response (such as the GPS and neutrophil: lymphocyte ratio (NLR) “would seem to represent the tip of a far larger iceberg” [34].
The developments in the last decade now allow us to think differently about the syndrome of cancer cachexia. This was foretold by MacDonald nearly a decade ago “Particularly in the more aggressive tumour types (e.g. pancreas and lung), the future of patients with elevated mGPS scores (systemic inflammation) is so grim that they should be given pre-cachexia status and offered multimodal therapy which may delay the onset of cachexia and/or death [35]”. On this background, our approach to the management of cachexia has evolved [36]. Specifically, the rationale for multimodal therapy, such as that utilised in the MENAC (Multimodal-Exercise, Nutrition and Anti-inflammatory medication for Cachexia) studies [37, 38].
Despite advances in our understanding of the molecular and clinical pathogenesis of cachexia, the basis of the consistent association between weight loss, sarcopenia and the systemic inflammatory response (as evidenced by CRP) is not altogether clear. Inflammation is a recognised driver of the loss of skeletal muscle mass in patients with cancer [39, 40]. Specifically, an increased production of pro-inflammatory cytokines including the interleukins 1 and 6 [41, 42]. Furthermore, it has long been recognised that at a tissue and cellular level, that such pro-inflammatory cytokines have a profound impact on cellular and tissue metabolism and therefore provide a rational therapeutic target [43]. While the use of anti-inflammatory agents in cancer patients was once daunting [44], the experience of corticosteroids and anti-IL-6 agents inhibitors in other areas of routine clinical care of systemic inflammation [45], in cancer immunotherapy [46] and recently in COVID-19 [47] has informed how these agents might be efficaciously used. However, further study will be required to address how we down-regulate the systemic inflammatory response in patients with cancer.
Discussion
Over the last decade or so, systemic inflammation has progressively moved to the forefront of the definition and diagnosis of cancer cachexia. The work of Vanhoutte in carrying out a direct comparison between the Fearon and Evans criteria showed clearly the additional prognostic role of a number of criteria, in particular systemic inflammation. However, the more extensive Evans criteria made assessment more complex and therefore pre-cachectic patients and the opportunity for early intervention could be missed. Indeed, it is of interest that some of the Evans criteria such as anaemia and low muscle mass in cancer patients are now recognised to be associated with systemic inflammation and therefore these criteria may be simplified [25, 48].
More recently, the GLIM criteria have included systemic inflammation as an aetiologic factor and introduced the concept that cachexia in patients with cancer is “disease-related malnutrition with inflammation. However, this concept may also miss the pre-cachectic patients and the opportunity for early intervention. Therefore, a more useful approach going forward may be that of MacDonald (2012) who proposed “Particularly in the more aggressive tumour types (e.g. pancreas and lung), the future of patients with elevated mGPS scores is so grim that they should be given pre-cachexia status and offered multimodal therapy which may delay the onset of cachexia and/or death [35].” In accord with the above perhaps a more useful interpretation of cancer cachexia is “disease-related inflammation with malnutrition” since this would emphasise the pre-cachectic state and the opportunity for early intervention.
From the above-detailed pathophysiology and criteria of cancer-related cachexia there are a number of potential implications for daily clinical practice. These include:
-
1.
Monitoring on a routine clinical basis of markers of systemic inflammation such as the mGPS [19, 21]. This will identify patients at risk of cachexia although with no weight loss and good performance status [49, 50]. It may be in the future with the increased recognition of inflammation in the determination of nutritional status that there will be a more detailed assessment of the host inflammatory response including pro-inflammatory cytokines such as IL-1, TNF alpha and IL-6.
-
2.
Treatment of patients with pre-cachexia/cachexia, together with standard oncological care, with a multimodal approach including ant-inflammatory medication, nutrition, exercise and symptom control [38]. However, further clinical work is required to determine the optimal modal treatment both individually and in combination. In particular, the use anti-inflammatory medications in patients with cancer is increasingly being examined. For example, Elwood and co-workers have recently reported that, in a systematic review and meta-analysis including ~250,000 patients, that there was a considerable body of evidence suggestive of about a 20% reduction in mortality in patients with cancer who take aspirin, and the benefit appears not to be restricted to one or a few cancers. They concluded that Aspirin deserves serious consideration as an adjuvant treatment for cancer [51].
Conclusion
In summary, based on the present evidence, the role of systemic inflammation in cancer cachexia is exigent. Therefore, we feel the time has now come to consider cancer cachexia as primarily a SIR syndrome and herald a new era in the treatment of this life-shortening condition.
Data availability
Raw data will be made available on request to the senior author (DCM).
References
Baracos VE, Martin L, Korc M, Guttridge DC, Fearon KCH. Cancer-associated cachexia. Nat Rev Dis Prim. 2018;4:17105.
Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12:489–95.
Medicine NLo. Cancer Cachexia. NIH; 2022 [cited 2022 March]. Available from: https://pubmed.ncbi.nlm.nih.gov/?term=cancer+cachexia.
Cederholm T, Jensen GL, Correia M, Gonzalez MC, Fukushima R, Higashiguchi T, et al. GLIM criteria for the diagnosis of malnutrition—a consensus report from the global clinical nutrition community. Clin Nutr. 2019;38:1–9.
McMillan DC. Systemic inflammation, nutritional status and survival in patients with cancer. Curr Opin Clin Nutr Metab Care. 2009;12:223–6.
Gabay C, Kushner I. Acute-phase proteins and other systemic responses to inflammation. N. Engl J Med. 1999;340:448–54.
McMillan DC. An inflammation-based prognostic score and its role in the nutrition-based management of patients with cancer. Proc Nutr Soc. 2008;67:257–62.
Zimmers TA, Fishel ML, Bonetto A. STAT3 in the systemic inflammation of cancer cachexia. Semin Cell Dev Biol. 2016;54:28–41.
Fox KM, Brooks JM, Gandra SR, Markus R, Chiou CF. Estimation of cachexia among cancer patients based on four definitions. J Oncol. 2009;2009:693458.
Arends J, Bodoky G, Bozzetti F, Fearon K, Muscaritoli M, Selga G, et al. ESPEN guidelines on enteral nutrition: non-surgical oncology. Clin Nutr. 2006;25:245–59.
Fearon KC, Voss AC, Hustead DS. Definition of cancer cachexia: effect of weight loss, reduced food intake, and systemic inflammation on functional status and prognosis. Am J Clin Nutr. 2006;83:1345–50.
Evans WJ, Morley JE, Argilés J, Bales C, Baracos V, Guttridge D, et al. Cachexia: a new definition. Clin Nutr. 2008;27:793–9.
Muscaritoli M, Anker SD, Argilés J, Aversa Z, Bauer JM, Biolo G, et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: joint document elaborated by Special Interest Groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin Nutr. 2010;29:154–9.
Gray S, Axelsson B. The prevalence of deranged C-reactive protein and albumin in patients with incurable cancer approaching death. PLoS ONE. 2018;13:e0193693.
Martin L, Senesse P, Gioulbasanis I, Antoun S, Bozzetti F, Deans C, et al. Diagnostic criteria for the classification of cancer-associated weight loss. J Clin Oncol. 2015;33:90–9.
Daly L, Dolan R, Power D, Ní Bhuachalla É, Sim W, Fallon M, et al. The relationship between the BMI-adjusted weight loss grading system and quality of life in patients with incurable cancer. J Cachexia Sarcopenia Muscle. 2020;11:160–8.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70.
Vanhoutte G, van de Wiel M, Wouters K, Sels M, Bartolomeeussen L, De Keersmaecker S, et al. Cachexia in cancer: what is in the definition? BMJ Open Gastroenterol. 2016;3:e000097.
Arends J, Baracos V, Bertz H, Bozzetti F, Calder PC, Deutz NEP, et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin Nutr. 2017;36:1187–96.
Cederholm T, Barazzoni R, Austin P, Ballmer P, Biolo G, Bischoff SC, et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin Nutr. 2017;36:49–64.
Arends J, Strasser F, Gonella S, Solheim TS, Madeddu C, Ravasco P, et al. Cancer cachexia in adult patients: ESMO Clinical Practice Guidelines(☆). ESMO Open. 2021;6:100092.
Laird BJ, Fallon M, Hjermstad MJ, Tuck S, Kaasa S, Klepstad P, et al. Quality of life in patients with advanced cancer: differential association with performance status and systemic inflammatory response. J Clin Oncol. 2016;34:2769–75.
Laird BJ, Kaasa S, McMillan DC, Fallon MT, Hjermstad MJ, Fayers P, et al. Prognostic factors in patients with advanced cancer: a comparison of clinicopathological factors and the development of an inflammation-based prognostic system. Clin Cancer Res. 2013;19:5456–64.
Martin L, Muscaritoli M, Bourdel-Marchasson I, Kubrak C, Laird B, Gagnon B, et al. Diagnostic criteria for cancer cachexia: reduced food intake and inflammation predict weight loss and survival in an international, multi-cohort analysis. J Cachexia Sarcopenia Muscle. 2021;12:1189–202.
Abbass T, Dolan RD, Laird BJ, McMillan DC. The relationship between imaging-based body composition analysis and the systemic inflammatory response in patients with cancer: a systematic review. Cancers. 2019;11:1304.
McGovern J, Dolan RD, Horgan PG, Laird BJ, McMillan DC. Computed tomography-defined low skeletal muscle index and density in cancer patients: observations from a systematic review. J Cachexia Sarcopenia Muscle. 2021;12:1408–17.
Temel JS, Abernethy AP, Currow DC, Friend J, Duus EM, Yan Y, et al. Anamorelin in patients with non-small-cell lung cancer and cachexia (ROMANA 1 and ROMANA 2): results from two randomised, double-blind, phase 3 trials. Lancet Oncol. 2016;17:519–31.
Merker M, Felder M, Gueissaz L, Bolliger R, Tribolet P, Kägi-Braun N, et al. Association of baseline inflammation with effectiveness of nutritional support among patients with disease-related malnutrition: a secondary analysis of a randomized clinical trial. JAMA Netw Open. 2020;3:e200663.
Diakos CI, Charles KA, McMillan DC, Clarke SJ. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15:e493–503.
Roxburgh CS, McMillan DC. Cancer and systemic inflammation: treat the tumour and treat the host. Br J Cancer. 2014;110:1409–12.
Dolan RD, Lim J, McSorley ST, Horgan PG, McMillan DC. The role of the systemic inflammatory response in predicting outcomes in patients with operable cancer: Systematic review and meta-analysis. Sci Rep. 2017;7:16717.
Dolan RD, McSorley ST, Horgan PG, Laird B, McMillan DC. The role of the systemic inflammatory response in predicting outcomes in patients with advanced inoperable cancer: Systematic review and meta-analysis. Crit Rev Oncol Hematol. 2017;116:134–46.
Dolan RD, Laird BJA, Horgan PG, McMillan DC. The prognostic value of the systemic inflammatory response in randomised clinical trials in cancer: a systematic review. Crit Rev Oncol Hematol. 2018;132:130–7.
McAllister SS, Weinberg RA. The tumour-induced systemic environment as a critical regulator of cancer progression and metastasis. Nat Cell Biol. 2014;16:717–27.
MacDonald N. Terminology in cancer cachexia: importance and status. Curr Opin Clin Nutr Metab Care. 2012;15:220–5.
Del Fabbro E. Combination therapy in cachexia. Ann Palliat Med. 2019;8:59–66.
Solheim TS, Laird BJA, Balstad TR, Stene GB, Bye A, Johns N, et al. A randomized phase II feasibility trial of a multimodal intervention for the management of cachexia in lung and pancreatic cancer. J Cachexia Sarcopenia Muscle. 2017;8:778–88.
Solheim TS, Laird BJA, Balstad TR, Bye A, Stene G, Baracos V, et al. Cancer cachexia: rationale for the MENAC (multimodal-exercise, nutrition and anti-inflammatory medication for cachexia) trial. BMJ Support Palliat Care. 2018;8:258–65.
Fearon KC, Glass DJ, Guttridge DC. Cancer cachexia: mediators, signaling, and metabolic pathways. Cell Metab. 2012;16:153–66.
MacDonald N. Cancer cachexia and targeting chronic inflammation: a unified approach to cancer treatment and palliative/supportive care. J Supportive Oncol. 2007;5:157–62.
Patton R, Paval DR, McDonald JJ, Brown D, Gallagher IJ, Skipworth RJE, et al. Relationship between cytokines and symptoms in people with incurable cancer: a systematic review. Crit Rev Oncol/hematol. 2021;159:103222.
Dolan RD, Laird BJA, Klepstad P, Kaasa S, Horgan PG, Paulsen Ø, et al. An exploratory study examining the relationship between performance status and systemic inflammation frameworks and cytokine profiles in patients with advanced cancer. Medicine. 2019;98:e17019.
Hotamisligil GS. Foundations of immunometabolism and implications for metabolic health and disease. Immunity. 2017;47:406–20.
Solheim TS, Fearon KC, Blum D, Kaasa S. Non-steroidal anti-inflammatory treatment in cancer cachexia: a systematic literature review. Acta Oncol. 2013;52:6–17.
McSorley ST, Horgan PG, McMillan DC. The impact of preoperative corticosteroids on the systemic inflammatory response and postoperative complications following surgery for gastrointestinal cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol. 2016;101:139–50.
Burdett N, Desai J. New biomarkers for checkpoint inhibitor therapy. ESMO Open. 2020;5:e000597.
Angriman F, Ferreyro BL, Burry L, Fan E, Ferguson ND, Husain S, et al. Interleukin-6 receptor blockade in patients with COVID-19: placing clinical trials into context. Lancet Respir Med. 2021;9:655–64.
McSorley ST, Talwar D, McMillan DC. Comment on the biomarkers reflecting inflammation and nutritional determinants of anemia (BRINDA) project. Am J Clin Nutr. 2018;108:204–5.
Dolan RD, Daly L, Sim WMJ, Fallon M, Ryan A, McMillan DC, et al. Comparison of the prognostic value of ECOG-PS, mGPS and BMI/WL: implications for a clinically important framework in the assessment and treatment of advanced cancer. Clin Nutr. 2020;39:2889–95.
Abbass T, Dolan RD, MacLeod N, Horgan PG, Laird BJ, McMillan DC. Comparison of the prognostic value of MUST, ECOG-PS, mGPS and CT derived body composition analysis in patients with advanced lung cancer. Clin Nutr ESPEN. 2020;40:349–56.
Elwood PC, Morgan G, Delon C, Protty M, Galante J, Pickering J, et al. Aspirin and cancer survival: a systematic review and meta-analyses of 118 observational studies of aspirin and 18 cancers. Ecancermedicalscience. 2021;15:1258.
Funding
None.
Author information
Authors and Affiliations
Contributions
JM and DCM wrote the paper and analysed the data. RDD, RJS and BJL aided in conceptualisation, reviewing, and writing of the paper. DCM had primary responsibility for the final content.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The following review was conducted in accordance with the declaration of Helsinki. Ethical approval was not required for this study as this was a systematic review of published data.
Consent to publish
All authors consent to publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McGovern, J., Dolan, R.D., Skipworth, R.J. et al. Cancer cachexia: a nutritional or a systemic inflammatory syndrome?. Br J Cancer 127, 379–382 (2022). https://doi.org/10.1038/s41416-022-01826-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-022-01826-2
This article is cited by
-
Relevance of blood tumor markers in inpatients with significant involuntary weight loss and elevated levels of inflammation biomarkers
BMC Cancer (2024)
-
Prognostic significance of pretreatment albumin–bilirubin (ALBI) grade and platelet–albumin–bilirubin (PALBI) grade in patients with small cell lung cancer
Scientific Reports (2024)
-
Nomograms based on the lymphocyte–albumin–neutrophil ratio (LANR) for predicting the prognosis of nasopharyngeal carcinoma patients after definitive radiotherapy
Scientific Reports (2024)
-
Cancer cachexia and chronic inflammation: an unbreakable bond
British Journal of Cancer (2023)
-
Decreased liver B vitamin-related enzymes as a metabolic hallmark of cancer cachexia
Nature Communications (2023)