Abstract
Background
Sarcomas are rare cancers of high heterogeneity. Health-Related Quality of Life (HRQoL) has been shown to be a prognostic factor for survival in other cancer entities but it is unclear whether this applies to sarcoma patients.
Patients and methods
HRQoL was prospectively assessed in adult sarcoma patients from 2017 to 2020 in 39 German recruiting sites using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30). Vital status was ascertained over the course of 1 year. HRQoL domains were analysed by multivariable cox-regressions including clinical and socio-economic risk factors.
Results
Of 1102 patients, 126 (11.4%) died during follow-up. The hazard ratio (HR) for global health was 0.73 per 10-point increase (95% confidence interval (CI) 0.64–0.85). HR for the HRQoL-summary score was 0.74 (CI 0.64–0.85) and for physical functioning 0.82 (CI 0.74–0.89). There was also evidence that fatigue (HR 1.17, CI 1.10–1.25), appetite loss (HR 1.15, CI 1.09–1.21) and pain (HR 1.14, CI 1.08–1.20) are prognostic factors for survival.
Conclusion
Our study adds sarcoma-specific evidence to the existing data about cancer survival in general. Clinicians and care-givers should be aware of the relations between HRQoL and survival probability and include HRQoL in routine assessment.
Similar content being viewed by others
Introduction
Sarcomas are rare cancers with about 7000 reported new cases per year in Germany [1] and an incidence of around 5 per 100 000 per year in Europe [2]. These tumours are heterogeneous and can be grouped into many histological subtypes [3], which appear almost everywhere in the body. Sarcoma therapy is based on complex and divergent treatment algorithms [4] and relative survival of patients varies. The European rare cancer project published the relative 1- and 5-year survival rates for the years 2000–2002 showing that after 1 year 75% of soft tissue sarcoma patients, 84% of bone sarcoma and 84% of GIST patients are alive. These rates dropped to 58%, 62% and 68%, respectively, after 5 years [2]. There was a great variation in the 5-year survival rates of different subtypes, ranging from 94% in skin sarcomas to only 11% in sarcomas of the heart.
In the last decades, the investigation of Health-Related Quality of Life (HRQoL) domains gained attention as potential prognostic factors for survival in oncology. A recent systematic review and meta-analysis investigated patient-reported outcomes (PRO) across 138 studies published between 2013 and 2018 that included around 160,000 cancer patients in total [5]. In 120 of these studies, at least one PRO was reported to be prognostic for overall survival. The European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) was most often used for PRO measurement. The physical functioning scale of the EORTC QLQ-C30 was the most frequently reported independent prognostic PRO with a pooled hazard ratio of 0.88 per 10-point increase, while appetite loss and fatigue ranked second and third. Similarly, a population-based study from 2020 of 7000 patients reported that the EORTC QLQ-C30 summary score (compared to global health and physical functioning) had highest predictability of survival (0.77 per 10-point increase) [6] in 12 cancer sites combined. However, this study also reported that the analysed PROs were not of prognostic value for every investigated cancer site. Lastly, a meta-analysis of phase-II or -III randomised controlled trials showed that in 41 of 44 studies at least one HRQoL domain was significantly associated with overall survival [7]. The most commonly evaluated factors were physical functioning, global health and pain; caution is, however, necessary, as this meta-analysis also noted a lack of methodological standards in the reporting of results.
Two larger studies of HRQoL of sarcoma patients were published in 2020 [8, 9]; however, data on the relationship between HRQoL and survival in adult sarcoma patients were scarce. Another study has investigated HRQoL in pediatric osteosarcoma patients [10] and several studies have evaluated patients with advanced cancers and included smaller groups of sarcoma patients populations that were not evaluated separately [11,12,13,14]. Given that there is considerable evidence for the association between HRQoL domains and survival in several cancers, we here investigated the following open questions for sarcoma patients:
-
(1)
We hypothesised that global health, summary score, physical functioning, appetite loss, fatigue and pain are significantly associated with overall survival in sarcoma patients.
-
(2)
As there is no data yet on the relation between several other HRQoL domains assessed by the EORTC QLQ-C30 and survival, we additionally explored the relation of other available HRQoL domains and survival.
-
(3)
In the absence of established standards, we used different measurement levels of the prognostic factors studied: PROs were analysed as continuous variables, as quartiles, and in dichotomised form (thresholds).
Patients and methods
The prospective PROSa-cohort study (Burden and Medical Care of Sarcoma in Germany: Nationwide Cohort Study Focusing on Modifiable Determinants of Patient-Reported Outcome Measures in Sarcoma Patients) (www.uniklinikum-dresden.de/prosastudie) was conducted nationwide from 09/2017 to 05/2020 in 39 study centers in Germany (NCT03521531; ClinicalTrials.gov). PROSa gathered information on a range of patient-reported outcomes (for example, HRQoL and distress) at baseline, as well as after 6 (t1) and after 12 months (t2), clinical data (such as diagnosis and treatment), as well as structural data of the participating study centers (for example, certifications and numbers of treated patients). Patients who were mentally or linguistically unable to complete questionnaires were excluded. For the present analysis, data of adult patients with histologically proven sarcoma of any entity were analysed. We analysed only participants with HRQoL data at baseline and information on survival.
Eligible patients were asked to participate at the referral centers during visits and sometimes by phone or letter. Participation required informed consent. The study was approved by the local ethics committees of the Technical University of Dresden (EK1790422017) and of the participating centers. HRQoL-data and sociodemographic data were sent by the participants to the study center by mail or online. Clinical information was submitted online by the participating centers using documentation forms. Data collection was performed using REDCap [15]. More detailed information on study design and participation have previously been published [8, 16].
Measurement of Health-Related Quality of Life and follow-up
HRQoL was measured at baseline according to the EORTC QLQ-C30 [17]. This instrument measures in units from 0 to 100 global health/ QoL as well as 5 functioning and 9 symptom domains, where high values indicate better HRQoL (functioning domains) and higher symptom burden (symptom domains), respectively. The functioning and the symptom domains were aggregated in the EORTC QLQ-C30 summary score. [18] Potentially confounding clinical and sociodemographic variables were measured at baseline (see below).
Patients were followed up after study inclusion for 1 year. If no data on vital status was available at t2 (follow-up time), we considered them as lost to follow-up and censored them at the time of the last available data point.
Statistics
Number of events and censoring are presented in Table 1. Continuous model variables were evaluated by mean and standard deviation (SD) if normally distributed and by median and interquartile range (IQR) if not. Categorical variables were presented with absolute and relative frequencies. All variables were stratified by vital status at last presentation (Table 2).
Multivariable cox-regressions were fitted to test for differences in survival between different HRQoL levels. HRQoL domains were evaluated in continuous form (model 1). Additionally we analysed them as quartiles (model 2) and in dichotomous form (model 3), using the thresholds of Giesinger et al. [19]. These indicate the proportion of patients with clinical important symptoms and limitations in the HRQoL domains. Proportional hazard assumption was tested using log minus log plots.
We considered global health, summary score, physical functioning, appetite loss, fatigue and pain as likely prognostic factors of survival. For those, results of model 2 were displayed as survival curves [20]. The other domains of the EORTC QLQ-C30 were evaluated exploratively (Table 3).
To adjust for potential confounding, we included socio-economic as well as clinical variables in the models, namely (variable values are shown in Table 2) sex, age at baseline, employment status at baseline, school education, sarcoma type, tumour site, grading at diagnosis, tumour size at diagnosis, time since diagnosis, tumour recurrence until baseline, metastasis until baseline, disease status at baseline, comorbidities at baseline and received treatments until baseline (surgery, chemotherapy, radiotherapy).
Statistical analyses were performed with SPSS V.27 (IBM Corporation, Armonk, New York, USA).
Results
Participation and sample description
After excluding patients without HRQoL or without follow-up data, 1102 patients could be included in the analysis (Table 1). Of those, 126 died until t2 and 28 were censored before t2. Patients were almost at gender parity with nearly half (49%) of analysed patients being female. The mean age of all patients was 56.2 years and 44% were employed at baseline. Seventy percent of analysed patients had a soft tissue sarcoma, 18% a bone sarcoma and 12% a GIST. Forty-eight percent of patients had an extremity sarcoma (Table 2). Forty-four percent of patients were in complete remission while 15% had progressive disease. Thirty-two percent were metastasised (Table 2).
We then tested if there are observable differences in survival between different HRQoL levels and evaluated HRQoL domains in continuous form (model 1), as quartiles (model 2) and in dichotomous (model 3) form.
Model 1—HRQoL as continuous scale
In the multivariable models, global health had the biggest impact on survival with a hazard ratio (HR) of 0.73 per 10-point increase (95% confidence interval (95% CI) 0.64–0.85). The HR for the summary score was 0.74 (95% CI 0.64–0.85) and for physical functioning 0.82 (95% CI 0.74–0.89). With the exception of cognitive functioning, other functioning scales showed significant results as well (Table 3). As hypothesised, fatigue (HR 1.17, 95% CI 1.10–1.25), appetite loss (HR 1.15, 95% CI 1.09–1.21) and pain (HR 1.14, 95% CI 1.08–1.20) were significant prognostic factors for survival. Furthermore, nausea/vomiting, dyspnoea, constipation and financial difficulties reached significance among the symptom scales (Table 3).
Model 2—HRQoL as quartiles
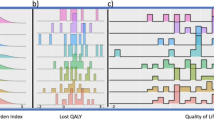
When comparing the least affected quartile of patients (1. Qu) with the most affected quartile (4. Qu), we observed the highest risk of mortality associated with global health (HR 8.29, 95% CI 3.13–22.00, Fig. 1b), followed by the summary score (HR 3.65, 95% CI 1.73–7.69, Fig. 1a) and physical functioning (HR 2.89, 1.52–5.51, Fig. 1c). Further functioning scales showed significant results as well (Table 3). As hypothesised, among the symptom scales fatigue (HR 4.21, 95% CI 1.98–8.94, Fig. 1d), appetite loss (HR 3.43, 95% CI 1.71–6.88, Fig. 1f), and pain (HR 2.99, 95% CI 1.72–5.19, Fig. 1e) were significant prognostic factors for survival. Among the other symptoms scales results for nausea/ vomiting, dyspnea and constipation reached significance comparing the first with the fourth quartile. (Table 3, Fig. 1)
Survival curves for summary score (a), global health (b), physical functioning (c), fatigue (d), pain (e), appetite loss (f). N = 1102. Events = 126. Variables in the model: sex, age at baseline, employment status at baseline, school education, sarcoma type, tumour site, grading at diagnosis, tumour size at diagnosis, time since diagnosis, tumour recurrence until baseline, metastasis until baseline, disease status at baseline, comorbidities, surgery, chemotherapy, radiotherapy until baseline.
Model 3—thresholds
No validated thresholds for summary score and for global health are available. Patients with clinical important restrictions in physical functioning showed a higher risk for mortality (HR 2.35, 95%-CI 1.43–3.87). Significant hazard ratios could also be observed in role, cognitive and social functioning (Table 3). As hypothesised, fatigue (HR 2.58, 95% CI 1.64–4.06), appetite loss (2.68, 95% CI 1.76–4.07) and pain (2.02, 95% CI 1.30–3.15) were significant prognostic factors for survival from the symptom scale. With the exception of insomnia and diarrhea, there was also evidence for significance in all other symptom scales. (Table 3)
Discussion
Results in context
Our results demonstrate that HRQoL domains are an independent prognostic factor for survival in sarcoma patients. The domains global health, physical functioning, fatigue, pain and appetite loss as well as the summary score have been previously associated with survival in cancer patients in general [5,6,7] and reached significance in the continuous, quartile and threshold models in our analysis. Additionally, we found significant associations between survival and the exploratively analysed HRQoL domains. Dyspnoea, nausea/ vomiting and social functioning showed significant associations in all three models, while in contrast insomnia and diarrhea were not associated with survival in any of the three models. The observed associations between dyspnoea, nausea/ vomiting and social functioning are noteworthy as they are less frequently discussed in the literature.
A key purpose of HRQoL domains in clinical settings is to measure aspects of disease burden that are not fully captured by assessing factors like disease stage, comorbidities or performance status (PS) alone. At present, the relation between PS and physical functioning, and which kind of measurement is more appropriate for different purposes, is still being discussed [21, 22]. PS und physical functioning could be considered as evaluations of the same complex status but from different perspectives and evidence suggests that some subjective self-reported toxicities may be missed by the examining physician [23]. Al-Rashdan et al. recently showed that PS und physical functioning were similarly predictive for overall survival [24]. Our analysis demonstrated that global health and the C30 summary score each had greater effect than physical functioning on predicting patient outcomes. This indicates that measuring physical functioning alone might not suffice to completely assess how disease severity is linked with survival from a patient’s perspective. The C30 summary score, which comprises all domains of the EORTC except financial problems, could alternatively provide a more comprehensive view on how survival and disease severity are linked. It is noteworthy that the generic global health domain with only two questions, on general life quality and health status, reached a similar effect size as the summary score.
At present, we are not able to answer the question if there are sarcoma-specific associations between HRQoL and survival. This is partly due to the fact that studies comparing individual cancer entities across all QoL domains with respect to survival are still lacking. Sarcomas are a highly heterogenous group of diseases with different QoL profiles [9]. An evaluation of the HRQoL of our PROSa-cohort previously revealed particular burdens in social and role functioning [8]. These may contribute to the observed association between social functioning and survival in this analysis.
In our study, continuous, quartiles and dichotomous measurement levels reached similar results. To our knowledge, there are no evaluated standards for the most appropriate form of measurement level—should a continuous scale be used or should the population be divided into groups (and if so in how many)? It is possible that no one size fits all solution exists and that domain-specific solutions have to be found. According to our results for social functioning and fatigue domains a ‘continuous’ presentation appears most appropriate due to its linear relationship; for others like global health, dyspnoea and appetite loss, we observed that a categorical approach with one or two thresholds appears useful. To give to examples: We found the most pronounced differences between the patient quartile with the best and the patient quartile with the worst global health score. With regard to the summary score, there seem to be no differences regarding survival in the least affected two quartiles of the population. Survival probabilities differed from the third quartile onward. However, as our study was not designed to evaluate thresholds but addressed the preceding question, whether there are associations between HRQoL and survival at all, it would be an overreach to derive cut-off values from these observations. In order to do that, a number of questions would need to be discussed, which are beyond the scope of this paper. For example, it should be clarified to what extent one should ask about the clinical relevance of the observed HR and whether different purposes (patient communication, medical interventions) require different thresholds.
Strengths and limitations
To date the PROSa study is one of the largest studies on HRQoL in sarcoma patients worldwide. In this analysis, we demonstrated an association between HRQoL and survival in sarcoma patients. Patients from 39 hospitals and medical offices were included. The participating centers comprehensively represent the aspects of sarcoma treatment in Germany and have a large network of referring institutions [16].
The study potentially may be subject to a selection bias as the majority of our patients were recruited in university hospitals and/or specialised centers. Selection biases are also possible at the patient level with a possible sick survivor bias, as healthy survivors are less likely to frequent recruiting study centers over time. Another bias may be sociodemographic selection, which is a factor in any observational study. These factors, however, in our opinion will not profoundly impact or influence the group comparisons made.
The possibility of undetected systematic confounding is inherent in any observational study. We measured a broad variety of potentially confounding variables. We were not able to include performance status (PS) in our analysis as it is not routinely collected at hospital visits. We would not consider PS as a confounding variable, but to compare the effect size of the measurements, inclusion of PS would have been beneficial. We did not include time-dependent variables in our analysis. Time-dependent variables would have resulted in somewhat more precise results, but at the same time would have further restricted the time horizon of the prognosis.
Conclusion
HRQoL domains are independent prognostic factors for survival in sarcoma patients. All hypothesised HRQoL domains, namely global health, summary score, physical functioning, fatigue, pain and appetite loss were significantly associated with survival. Our analysis therefore adds disease-specific evidences to the already existing data reported for cancer patients in general. This opens the possibility for further studies that can firmly establish the potentially domain-specific relationship between HRQoL and survival probability. It would benefit patients if clinicians and care-givers would monitor HRQoL domains in patients on a regular basis. Future studies should evaluate whether specific interventions to improve HRQoL domains might have a positive influence on patient survival.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ressing M, Wardelmann E, Hohenberger P, Jakob J, Kasper B, Emrich K, et al. Strengthening health data on a rare and heterogeneous disease: sarcoma incidence and histological subtypes in Germany. BMC Public Health. 2018;18:235.
Stiller CA, Trama A, Serraino D, Rossi S, Navarro C, Chirlaque MD, et al. Descriptive epidemiology of sarcomas in Europe: Report from the RARECARE project. Eur J Cancer. 2013;49:684–95.
Fletcher CDM, World Health Organization, International Agency for Research on Cancer, editors. WHO classification of tumours of soft tissue and bone. 4th edn. Lyon: IARC Press; 2013. 468 pp.
Casali PG, Abecassis N, Bauer S, Biagini R, Bielack S, Bonvalot S, et al. Soft tissue and visceral sarcomas: ESMO–EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv51–67.
Efficace F, Collins GS, Cottone F, Giesinger JM, Sommer K, Anota A, et al. Patient-reported outcomes as independent prognostic factors for survival in oncology: systematic review and meta-analysis. Value Health. 2021;24:250–67.
Husson O, de Rooij BH, Kieffer J, Oerlemans S, Mols F, Aaronson NK, et al. The EORTC QLQ-C30 summary score as prognostic factor for survival of patients with cancer in the ‘Real-World’: results from the population-based PROFILES registry. Oncologist. 2020;25:e722–32.
Mierzynska J, Piccinin C, Pe M, Martinelli F, Gotay C, Coens C, et al. Prognostic value of patient-reported outcomes from international randomised clinical trials on cancer: a systematic review. Lancet Oncol. 2019;20:e685–98.
Eichler M, Hentschel L, Richter S, Hohenberger P, Kasper B, Andreou D, et al. The Health-Related Quality of Life of sarcoma patients and survivors in Germany—cross-sectional results of a Nationwide Observational Study (PROSa). Cancers. 2020;12:3590.
van Eck I, den Hollander D, Desar IME, Soomers VLMN, van de Sande MAJ, de Haan JJ, et al. Unraveling the heterogeneity of sarcoma survivors’ Health-Related Quality of Life regarding primary sarcoma location: results from the SURVSARC study. Cancers. 2020;12:3083.
Bishop MW, Advani SM, Villarroel M, Billups CA, Navid F, Rivera C, et al. Health-Related Quality of Life and survival outcomes of pediatric patients with nonmetastatic osteosarcoma treated in countries with different resources. J Glob Oncol. 2018;4:1–11.
Choi D, Fox Z, Albert T, Arts M, Balabaud L, Bunger C, et al. Prediction of quality of life and survival after surgery for symptomatic spinal metastases: a multicenter cohort study to determine suitability for surgical treatment. Neurosurgery. 2015;77:698–708.
Habboush Y, Shannon RP, Niazi SK, Hollant L, Single M, Gaines K, et al. Patient-reported distress and survival among patients receiving definitive radiation therapy. Adv Radiat Oncol. 2017;2:211–9.
Kypriotakis G, Vidrine DJ, Francis LE, Rose JH. The longitudinal relationship between quality of life and survival in advanced stage cancer. Psycho Oncol. 2016;25:225–31.
Paiva CE, Paiva BSR. Prevalence, predictors, and prognostic impact of fatigue among Brazilian outpatients with advanced cancers. Support Care Cancer. 2013;21:1053–60.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81.
Eichler M, Andreou D, Golcher H, Hentschel L, Richter S, Hohenberger P, et al. Utilization of interdisciplinary tumor boards for sarcoma care in Germany: results from the PROSa study. Oncol Res Treat. 2021;44:301–12.
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for research and treatment of cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365–76.
Giesinger JM, Kieffer JM, Fayers PM, Groenvold M, Petersen MAA, Scott NW, et al. Replication and validation of higher order models demonstrated that a summary score for the EORTC QLQ-C30 is robust. J Clin Epidemiol. 2016;69:79–88.
Giesinger JM, Loth FLC, Aaronson NK, Arraras JI, Caocci G, Efficace F, et al. Thresholds for clinical importance were established to improve interpretation of the EORTC QLQ-C30 in clinical practice and research. J Clin Epidemiol. 2020;118:1–8.
Hernán MA. The hazards of hazard ratios. Epidemiology. 2010;21:13–5.
Efficace F, Rosti G, Aaronson N, Cottone F, Angelucci E, Molica S, et al. Patient-versus physician-reporting of symptoms and health status in chronic myeloid leukemia. Haematologica. 2014;99:788–93.
Atkinson TM, Dueck AC, Satele DV, Thanarajasingam G, Lafky JM, Sloan JA, et al. Clinician vs patient reporting of baseline and postbaseline symptoms for adverse event assessment in cancer clinical trials. JAMA Oncol. 2020;6:437.
Di Maio M, Gallo C, Leighl NB, Piccirillo MC, Daniele G, Nuzzo F, et al. Symptomatic toxicities experienced during anticancer treatment: agreement between patient and physician reporting in three randomized trials. J Clin Oncol. 2015;33:910–5.
Al-Rashdan A, Sutradhar R, Nazeri-Rad N, Yao C, Barbera L. Comparing the ability of physician-reported versus patient-reported performance status to predict survival in a population-based cohort of newly diagnosed cancer patients. clinical oncology [Internet]. 2021. https://www.sciencedirect.com/science/article/pii/S093665552100008X.
Funding
The PROSa study was funded by the German Cancer Aid (No. 111713). MB and HKJ received support by the German Cancer Aid, Mildred-Scheel Nachwuchszentrum P2. Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
ME wrote the article and analysed the data. ME, MKS and LH developed questionnaires and study design. JS and MKS developed the conception of the study and supervised with MB the work throughout the whole study. EW supervised development of inclusion criteria. KA supervised the study from a patient’s perspective. ME and SS developed the statistical analysis plan for this paper. HKJ did scientific editing and developed visualisations. SR, PH, BK, DA, DP, JJ, RG, SF, VH, SAZB, VIG, JW, KDS and MS were responsible for the recruitment of patients or recruited patients directly. All authors have revised the manuscript critically and approved the published version.
Corresponding author
Ethics declarations
Competing interests
SS received lecture fees from Lilly, BMS, Boehringer-Ingelheim and Pfizer, all outside of this work. LH received fees from SERVIER, outside of this work. DA received lecture fees from Lilly and Implantcast, all outside of this work. DP received fees for consulting services from Lilly, PharmaMar, Roche and fees for lectures from Lilly, PharmaMar, all outside of this work. JJ received fees from Lilly and Boehringer-Ingelheim, all outside of this work. SF received reimbursements for congress attendance fees from PharmaMar, all outside of this work. VIG received reimbursements of congress attendance fees and travel and accommodation expenses from Novartis and Lilly, all outside this work. SAZB received reimbursements of congress attendance fees and travel and accommodation expenses from Lilly, Pfizer und Gilead, all outside this work. JS received consulting fees from Novartis, Sanofi, ALK, and Lilly, all outside of this work. MKS received research funding from PharmaMar and Novartis, all outside of this work. ME, SR, PH, BK, RG, EW, KA, VH, KDS, HKJ, JW and MB declare no competing interests.
Ethics
This study was approved by the ethics committee of the Technical University of Dresden (AZ: EK1790422017) and the ethics committees of the participating centers and it was conducted in accordance with the Declaration of Helsinki. The study participants gave written informed consent. This study is registered under NCT03521531 on ClinicalTrials.gov.
Consent for publication
Not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Eichler, M., Singer, S., Hentschel, L. et al. The association of Health-Related Quality of Life and 1-year-survival in sarcoma patients—results of a Nationwide Observational Study (PROSa). Br J Cancer 126, 1346–1354 (2022). https://doi.org/10.1038/s41416-022-01702-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-022-01702-z