Abstract
Introduction
In patients with metastatic colorectal cancer (mCRC), baseline circulating tumour DNA (ctDNA) variant allele fraction (VAF) might serve as a surrogate of disease burden and should be evaluated in comparison with CEA and RECIST-defined sum of target lesions.
Methods
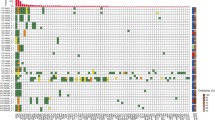
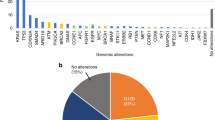
In this pre-planned analysis of the VALENTINO trial, we included patients with RAS wild-type mCRC receiving upfront FOLFOX/panitumumab with available baseline liquid biopsy. CtDNA was analysed by means of a 14-gene NGS panel. For each patient, the gene with the highest VAF in ctDNA was selected.
Results
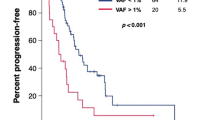
The final cohort included 135 patients. The median VAF was 12.6% (IQR: 2.0–45.2%). Higher VAF was observed in patients with liver metastases and with synchronous metastases presentation. Patients with high VAF had poorer median OS compared to those with low VAF (21.8 vs 36.5 months; HR: 1.82, 95%CI: 1.20–2.76; p = 0.005). VAF outperformed baseline CEA and target lesion diameter in the prognostic stratification and remained significantly correlated with OS (p = 0.003) in a multivariate model. VAF was not significantly correlated with dimensional response and PFS.
Conclusion
CtDNA measured by VAF is prognostic in patients with RAS wild-type mCRC. Response and PFS after an anti-EGFR-based first-line strategy are independent from initial tumour burden.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Code availability
Codes can be made available upon request to the corresponding author.
References
Antoniotti C, Pietrantonio F, Corallo S, De Braud F, Falcone A, Cremolini C. Circulating tumor DNA analysis in colorectal cancer: from dream to reality. JCO Precis Oncol. 2019;3:1–14.
Bettegowda C, Sausen M, Leary RJ, Kinde I, Wang Y, Agrawal N, et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci Transl Med. 2014;6:224ra24.
Manca P, Corallo S, Busico A, Lonardi S, Corti F, Antoniotti C, et al. The added value of baseline circulating tumor DNA profiling in patients with molecularly hyperselected, left-sided metastatic colorectal cancer. Clin Cancer Res. 2021;27:2505–14.
Randon G, Yaeger R, Hechtman JF, Manca P, Fucà G, Walch H, et al. EGFR amplification in metastatic colorectal cancer. J Natl Cancer Inst. 2021:djab069.
Normanno N, Esposito Abate R, Lambiase M, Forgione L, Cardone C, Iannaccone A, et al. RAS testing of liquid biopsy correlates with the outcome of metastatic colorectal cancer patients treated with first-line FOLFIRI plus cetuximab in the CAPRI-GOIM trial. Ann Oncol. 2018;29:112–8.
Siravegna G, Lazzari L, Crisafulli G, Sartore-Bianchi A, Mussolin B, Cassingena A, et al. Radiologic and genomic evolution of individual metastases during HER2 blockade in colorectal cancer. Cancer Cell. 2018;34:148.e7–62.e7.
Cremolini C, Rossini D, Dell’Aquila E, Lonardi S, Conca E, Del Re M, et al. Rechallenge for patients with RAS and BRAF wild-type metastatic colorectal cancer with acquired resistance to first-line cetuximab and irinotecan: a phase 2 single-arm clinical trial. JAMA Oncol. 2019;5:343–50.
Janku F, Zhang S, Waters J, Liu L, Huang HJ, Subbiah V, et al. Development and validation of an ultradeep next-generation sequencing assay for testing of plasma cell-free DNA from patients with advanced cancer. Clin Cancer Res. 2017;23:5648–56.
Hsu HC, Lapke N, Wang CW, Lin PY, You JF, Yeh CY, et al. Targeted sequencing of circulating tumor DNA to monitor genetic variants and therapeutic response in metastatic colorectal cancer. Mol Cancer Ther. 2018;17:2238–47.
Tie J, Kinde I, Wang Y, Wong HL, Roebert J, Christie M, et al. Circulating tumor DNA as an early marker of therapeutic response in patients with metastatic colorectal cancer. Ann Oncol. 2015;26:1715–22.
Compton CC, Fielding LP, Burgart LJ, Conley B, Cooper HS, Hamilton SR, et al. Prognostic factors in colorectal cancer. College of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med. 2000;124:979–94.
Pietrantonio F, Morano F, Corallo S, Miceli R, Lonardi S, Raimondi A, et al. Maintenance therapy with panitumumab alone vs panitumumab plus fluorouracil-leucovorin in patients with RAS wild-type metastatic colorectal cancer: a phase 2 randomized clinical trial. JAMA Oncol. 2019;5:1268–75.
Manca P, Corallo S, Randon G, Lonardi S, Cremolini C, Rimassa L, et al. Impact of early tumor shrinkage and depth of response on the outcomes of panitumumab-based maintenance in patients with RAS wild-type metastatic colorectal cancer. Eur J Cancer. 2021;144:31–40.
Morano F, Corallo S, Lonardi S, Raimondi A, Cremolini C, Rimassa L, et al. Negative hyperselection of patients with RAS and BRAF wild-type metastatic colorectal cancer who received panitumumab-based maintenance therapy. J Clin Oncol. 2019;37:3099–110.
R Core Team. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing, 2019.
Kopetz S, Grothey A, Yaeger R, Van Cutsem E, Desai J, Yoshino T, et al. Encorafenib, binimetinib, and cetuximab in BRAF V600E-mutated colorectal cancer. N. Engl J Med. 2019;381:1632–43.
El Messaoudi S, Mouliere F, Du Manoir S, Bascoul-Mollevi C, Gillet B, Nouaille M, et al. Circulating DNA as a strong multimarker prognostic tool for metastatic colorectal cancer patient management care. Clin Cancer Res. 2016;22:3067–77.
Elez E, Chianese C, Sanz-García E, Martinelli E, Noguerido A, Mancuso FM, et al. Impact of circulating tumor DNA mutant allele fraction on prognosis in RAS-mutant metastatic colorectal cancer. Mol Oncol. 2019;13:1827–35.
Peeters M, Price T, Boedigheimer M, Kim TW, Ruff P, Gibbs P, et al. Evaluation of emergent mutations in circulating cell-free DNA and clinical outcomes in patients with metastatic colorectal cancer treated with panitumumab in the ASPECCT Study. Clin Cancer Res. 2019;25:1216–25.
Spindler KG, Appelt AL, Pallisgaard N, Andersen RF, Jakobsen A. KRAS-mutated plasma DNA as predictor of outcome from irinotecan monotherapy in metastatic colorectal cancer. Br J Cancer. 2013;109:3067–72.
Bidard FC, Kiavue N, Ychou M, Cabel L, Stern MH, Madic J, et al. Circulating tumor cells and circulating tumor DNA detection in potentially resectable metastatic colorectal cancer: a prospective ancillary study to the Unicancer Prodige-14 Trial. Cells. 2019;8:516.
Sefrioui D, Beaussire L, Gillibert A, Blanchard F, Toure E, Bazille C, et al. CEA, CA19-9, circulating DNA and circulating tumour cell kinetics in patients treated for metastatic colorectal cancer (mCRC). Br J Cancer. 2021;125:725–33.
Tie J, Cohen JD, Wang Y, Christie M, Simons K, Lee M, et al. Circulating tumor DNA analyses as markers of recurrence risk and benefit of adjuvant therapy for stage III colon cancer. JAMA Oncol. 2019;5:1710–7.
Lin JK, Lin PC, Lin CH, Jiang JK, Yang SH, Liang WY, et al. Clinical relevance of alterations in quantity and quality of plasma DNA in colorectal cancer patients: based on the mutation spectra detected in primary tumors. Ann Surg Oncol. 2014;21:S680–6.
Jones RP, Pugh SA, Graham J, Primrose JN, Barriuso J. Circulating tumour DNA as a biomarker in resectable and irresectable stage IV colorectal cancer; a systematic review and meta-analysis. Eur J Cancer. 2021;144:368–81.
Parikh AR, Mojtahed A, Schneider JL, Kanter K, Van Seventer EE, Isobel J Fetter, et al. Serial ctDNA monitoring to predict response to systemic therapy in metastatic gastrointestinal cancers. Clin Cancer Res. 2020;26:1877–85.
Aranda E, Viéitez JM, Gómez-España A, Gil Calle S, Salud-Salvia A, Begoña Graña, et al. FOLFOXIRI plus bevacizumab versus FOLFOX plus bevacizumab for patients with metastatic colorectal cancer and ≥3 circulating tumour cells: the randomised phase III VISNÚ-1 trial. ESMO Open. 2020;5:e000944.
Borelli B, Moretto R, Lonardi S, Bonetti A, Antoniotti C, Pietrantonio F, et al. TRIPLETE: a randomised phase III study of modified FOLFOXIRI plus panitumumab versus mFOLFOX6 plus panitumumab as initial therapy for patients with unresectable RAS and BRAF wild-type metastatic colorectal cancer. ESMO Open. 2018;3:e000403.
Cremolini C, Antoniotti C, Rossini D, Lonardi S, Loupakis F, Pietrantonio F, et al. Upfront FOLFOXIRI plus bevacizumab and reintroduction after progression versus mFOLFOX6 plus bevacizumab followed by FOLFIRI plus bevacizumab in the treatment of patients with metastatic colorectal cancer (TRIBE2): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2020;21:497–507.
Author information
Authors and Affiliations
Contributions
PM and FP designed the work, interpreted the results and drafted the original version of the manuscript. FP conceived the work. All authors acquired data, revised the manuscript, approved the final version and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Institutional review board approval was obtained from all participating Centres and all patients provided written informed consent. The study was conducted in accordance with the Declaration of Helsinki.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manca, P., Corallo, S., Lonardi, S. et al. Variant allele frequency in baseline circulating tumour DNA to measure tumour burden and to stratify outcomes in patients with RAS wild-type metastatic colorectal cancer: a translational objective of the Valentino study. Br J Cancer 126, 449–455 (2022). https://doi.org/10.1038/s41416-021-01591-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01591-8