Abstract
Background
Aspirin reduces the incidence of conventional adenomas driven by APC mutation and thus colorectal cancer. The effect of aspirin on the ~20% of colorectal cancers arising via BRAF mutation is yet to be established.
Methods
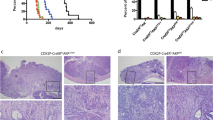
BrafV637E/+;Villin-CreERT2/+ mice were allocated to a control (n = 86) or aspirin-supplemented (n = 83) diet. After 14 months the incidence of murine serrated lesions, carcinoma and distant metastases were measured by histological examination. RNA was extracted from carcinomas from each cohort and subjected to sequencing to identify differentially expressed genes and molecular pathways.
Results
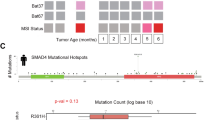
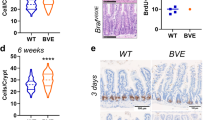
Aspirin did not reduce the incidence of murine serrated lesions or carcinoma when compared to control, however, did significantly reduce lesion size (P = 0.0042). Among the mice with carcinoma there was a significant reduction in the incidence of distant metastasis with aspirin treatment (RR 0.69, 95% CI 0.48–0.90, P = 0.0134). Key pathways underlying metastasis of carcinoma cells include NOTCH, FGFR and PI3K signalling, were significantly downregulated in carcinomas sampled from mice on an aspirin-supplemented diet.
Conclusions
Aspirin reduces the incidence of metastatic Braf mutant carcinoma, although this is not due to a reduction in primary disease. The reduction in metastasis could be attributed to a delay or prevention of molecular changes within the primary site driving metastatic growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A. & Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Zauber, A. G., Winawer, S. J., O’Brien, M. J., Lansdorp-Vogelaar, I., van Ballegooijen, M., Hankey, B. F. et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N. Engl. J. Med. 366, 687–696 (2012).
Shaukat, A., Mongin, S. J., Geisser, M. S., Lederle, F. A., Bond, J. H., Mandel, J. S. et al. Long-term mortality after screening for colorectal cancer. N. Engl. J. Med. 369, 1106–1114 (2013).
Baron, J. A., Cole, B. F., Sandler, R. S., Haile, R. W., Ahnen, D., Bresalier, R. et al. A randomized trial of aspirin to prevent colorectal adenomas. N. Engl. J. Med. 348, 891–899 (2003).
Sandler, R. S., Halabi, S., Baron, J. A., Budinger, S., Paskett, E., Keresztes, R. et al. A randomized trial of aspirin to prevent colorectal adenomas in patients with previous colorectal cancer. N. Engl. J. Med. 348, 883–890 (2003).
Logan, R. F. A., Grainge, M. J., Shepherd, V. C., Armitage, N. C. & Muir, K. R. Aspirin and folic acid for the prevention of recurrent colorectal adenomas. Gastroenterology 134, 29–38 (2008).
Rothwell, P. M., Wilson, M., Elwin, C.-E., Norrving, B., Algra, A., Warlow, C. P. et al. Long-term effect of aspirin on colorectal cancer incidence and mortality: 20-year follow-up of five randomised trials. Lancet 376, 1741–1750 (2010).
Burn, J., Gerdes, A.-M., Macrae, F., Mecklin, J.-P., Moeslein, G., Olschwang, S. et al. Long-term effect of aspirin on cancer risk in carriers of hereditary colorectal cancer: an analysis from the CAPP2 randomised controlled trial. Lancet 378, 2081–2087 (2012).
Pai, R. K., Bettington, M., Srivastava, A. & Rosty, C. An update on the morphology and molecular pathology of serrated colorectal polyps and associated carcinomas. Mod. Pathol. 32, 1390–1415 (2019).
Leggett, B. & Whitehall, V. Role of the serrated pathway in colorectal cancer pathogenesis. Gastroenterology 138, 2088–2100 (2010).
Snover, D. C. Update on the serrated pathway to colorectal carcinoma. Hum. Pathol. 42, 1–10 (2011).
Spring, K. J., Zhao, Z. Z., Karamatic, R., Walsh, M. D., Whitehall, V. L. J., Pike, T. et al. High prevalence of sessile serrated adenomas with BRAF mutations: a prospective study of Patients Undergoing Colonoscopy. Gastroenterology 131, 1400–1407 (2006).
Hinoue, T., Weisenberger, D. J., Pan, F., Campan, M., Kim, M., Young, J. et al. Analysis of the association between CIMP and BRAF(V600E) in colorectal cancer by DNA methylation profiling. PLoS ONE 4, e8357 (2009).
Kambara, T., Simms, L. A., Whitehall, V. L. J., Spring, K. J., Wynter, C. V. A., Walsh, M. D. et al. BRAF mutation is associated with DNA methylation in serrated polyps and cancers of the colorectum. Gut 53, 1137–1144 (2004).
Bettington, M., Walker, N., Rosty, C., Brown, I., Clouston, A., McKeone, D. et al. Clinicopathological and molecular features of sessile serrated adenomas with dysplasia or carcinoma. Gut 66, 97–106 (2017).
Bailie, L., Loughrey, M. B. & Coleman, H. G. Lifestyle risk factors for serrated colorectal polyps: a systematic review and meta-analysis. Gastroenterology 152, 92–104 (2017).
Wallace, K., Grau, M. V., Ahnen, D., Snover, D. C., Robertson, D. J., Mahnke, D. et al. The association of lifestyle and dietary factors with the risk for serrated polyps of the colorectum. Cancer Epidemiol. Biomark. Prev. 18, 2310–2317 (2009).
Bouwens, M. W. E., Winkens, B., Rondagh, E. J. A., Driessen, A. L., Riedl, R. G., Masclee, A. A. M. et al. Simple clinical risk score identifies patients with serrated polyps in routine practice. Cancer Prev. Res. 6, 855–863 (2013).
Nishihara, R., Lochhead, P., Kuchiba, A., Jung, S., Yamauchi, M., Liao, X. et al. Aspirin use and risk of colorectal cancer according to BRAF mutation status. JAMA 309, 2563–2571 (2013).
Bond, C. E., Liu, C., Kawamata, F., McKeone, D. M., Fernando, W., Jamieson, S. et al. Oncogenic BRAF mutation induces DNA methylation changes in a murine model for human serrated colorectal neoplasia. Epigenetics 13, 40–48 (2018).
Mahmoud, N. N., Dannenberg, A. J., Mestre, J., Bilinski, R. T., Churchill, M. R., Martucci, C. et al. Aspirin prevents tumors in a murine model of familial adenomatous polyposis. Surgery 124, 225–231 (1998).
Barnes, C. J. & Lee, M. Chemoprevention of spontaneous intestinal adenomas in the adenomatous polyposis coli Min mouse model with aspirin. Gastroenterology 114, 873–877 (1998).
Reagan-Shaw, S., Nihal, M. & Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 22, 659–661 (2008).
Rad, R., Cadiñanos, J., Rad, L., Varela, I., Strong, A., Kriegl, L. et al. A genetic progression model of BrafV600E-induced intestinal tumorigenesis reveals targets for therapeutic intervention. Cancer Cell 24, 15–29 (2013).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal https://doi.org/10.14806/ej.17.1.200 (2011).
Dobin, A., Davis, C. A., Schlesinger, F., Drenkow, J., Zaleski, C., Jha, S. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2012).
DeLuca, D. S., Levin, J. Z., Sivachenko, A., Fennell, T., Nazaire, M.-D., Williams, C. et al. RNA-SeQC: RNA-seq metrics for quality control and process optimization. Bioinformatics 28, 1530–1532 (2012).
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12, 323 (2011).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Hänzelmann, S., Castelo, R. & Guinney, J. GSVA: gene set variation analysis for microarray and RNA-Seq data. BMC Bioinformatics 14, 7 (2013).
Aravind, S., Pablo, T., Vamsi, K. M., Sayan, M., Benjamin, L. E., Michael, A. G. et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Sturm, G., Finotello, F., Petitprez, F., Zhang, J. D., Baumbach, J., Fridman, W. H. et al. Comprehensive evaluation of transcriptome-based cell-type quantification methods for immuno-oncology. Bioinformatics 35, i436–i445 (2019).
Lannagan, T. R. M., Lee, Y. K., Wang, T., Roper, J., Bettington, M. L., Fennell, L. et al. Genetic editing of colonic organoids provides a molecularly distinct and orthotopic preclinical model of serrated carcinogenesis. Gut 68, 684–692 (2019).
Kane, A. M., Fennell, L. J., Liu, C., Borowsky, J., McKeone, D. M., Bond, C. E. et al. Alterations in signaling pathways that accompany spontaneous transition to malignancy in a mouse model of BRAF mutant microsatellite stable colorectal cancer. Neoplasia 22, 120–128 (2020).
Benamouzig, R., Uzzan, B., Deyra, J., Martin, A., Girard, B., Little, J. et al. Prevention by daily soluble aspirin of colorectal adenoma recurrence: 4-year results of the APACC randomised trial. Gut 61, 255–261 (2012).
Burn, J., Bishop, D. T., Mecklin, J.-P., Macrae, F., Möslein, G., Olschwang, S. et al. Effect of aspirin or resistant starch on colorectal neoplasia in the lynch syndrome. N. Engl. J. Med. 359, 2567–2578 (2008).
Greenspan, E. J., Nichols, F. C. & Rosenberg, D. W. Molecular alterations associated with sulindac-resistant colon tumors in ApcMin/+ mice. Cancer Prev. Res. (Philos.) 3, 1187–1197 (2010).
Yang, K., Fan, K., Kurihara, N., Shinozaki, H., Rigas, B., Augenlicht, L. et al. Regional response leading to tumorigenesis after sulindac in small and large intestine of mice with Apc mutations. Carcinogenesis 24, 605–611 (2003).
Murakami, T., Akazawa, Y., Yatagai, N., Hiromoto, T., Sasahara, N., Saito, T. et al. Molecular characterization of sessile serrated adenoma/polyps with dysplasia/carcinoma based on immunohistochemistry, next-generation sequencing, and microsatellite instability testing: a case series study. Diagn. Pathol. 13, 88 (2018).
Lochhead, P., Kuchiba, A., Imamura, Y., Liao, X., Yamauchi, M., Nishihara, R. et al. Microsatellite instability and BRAF mutation testing in colorectal cancer prognostication. J. Natl Cancer Inst. 105, 1151–1156 (2013).
Rothwell, P. M., Wilson, M., Price, J. F., Belch, J. F., Meade, T. W. & Mehta, Z. Effect of daily aspirin on risk of cancer metastasis: a study of incident cancers during randomised controlled trials. Lancet 379, 1591–1601 (2012).
Rothwell, P. M., Fowkes, F. G. R., Belch, J. F. F., Ogawa, H., Warlow, C. P. & Meade, T. W. Effect of daily aspirin on long-term risk of death due to cancer: analysis of individual patient data from randomised trials. Lancet 377, 31–41 (2011).
Liao, X., Lochhead, P., Nishihara, R., Morikawa, T., Kuchiba, A., Yamauchi, M. et al. Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N. Engl. J. Med. 367, 1596–1606 (2012).
Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 16, 582–598 (2016).
Liu, R., Li, J., Xie, K., Zhang, T., Lei, Y., Chen, Y. et al. FGFR4 promotes stroma-induced epithelial-to-mesenchymal transition in colorectal cancer. Cancer Res. 73, 5926–5935 (2013).
Matsuda, Y., Ishiwata, T., Yamahatsu, K., Kawahara, K., Hagio, M., Peng, W.-X. et al. Overexpressed fibroblast growth factor receptor 2 in the invasive front of colorectal cancer: a potential therapeutic target in colorectal cancer. Cancer Lett. 309, 209–219 (2011).
Jackstadt, R., van Hooff, S. R., Leach, J. D., Cortes-Lavaud, X., Lohuis, J. O., Ridgway, R. A. et al. Epithelial NOTCH signaling rewires the tumor microenvironment of colorectal cancer to drive poor-prognosis subtypes and metastasis. Cancer Cell 36, 319–336.e317 (2019).
Chan, A. T., Ogino, S. & Fuchs, C. S. Aspirin use and survival after diagnosis of colorectal cancer. JAMA 302, 649–658 (2009).
Li, P., Wu, H., Zhang, H., Shi, Y., Xu, J., Ye, Y. et al. Aspirin use after diagnosis but not prediagnosis improves established colorectal cancer survival: a meta-analysis. Gut 64, 1419–1425 (2015).
Acknowledgements
We gratefully acknowledge the generous support of the Garry Whyte Sea Angel PAF.
Author information
Authors and Affiliations
Contributions
A.K., P.P., G.Y., B.L. and V.W. conceptualised and designed the study. A.K., C.L., L.F., D.M. and C.B. contributed to methodology and experiments. A.K. and L.F. analysed data. A.K. drafted manuscript. B.L. and V.W. supervised the study.
Corresponding author
Ethics declarations
Ethics approval
All studies were approved by the QIMR Berghofer Animal Ethics Committee (P2178).
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Funding information
Funding for this research was provided by a Cancer Council Queensland grant (grant number 1100701). This work was supported by Cancer Council Queensland, National Health and Medical Research Council of Australia.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kane, A.M., Liu, C., Fennell, L.J. et al. Aspirin reduces the incidence of metastasis in a pre-clinical study of Braf mutant serrated colorectal neoplasia. Br J Cancer 124, 1820–1827 (2021). https://doi.org/10.1038/s41416-021-01339-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01339-4
This article is cited by
-
Role of Combination Treatment of Aspirin and Zinc in DMH-DSS-induced Colon Inflammation, Oxidative Stress and Tumour Progression in Male BALB/c Mice
Biological Trace Element Research (2023)