Abstract
This study aimed to address the prognostic impact of center experience based on the data of 7821 adults with acute myeloid leukemia who underwent allogeneic hematopoietic cell transplantation (HCT) from 2010 to 2019 in Japan, where medical care was provided within a uniform healthcare system. Center experience was defined based on the number of allogeneic HCTs performed for any indication during the study period, by which centers were divided into low-, intermediate-, and high-volume centers. After adjusting for known confounding factors, the risk of overall mortality was lowest for the high-volume centers and highest for the low-volume centers, with the difference between the center categories attributed primarily to the risk of relapse. Patients transplanted at high-volume centers had higher risks of acute and chronic graft-versus-host diseases but without an increased risk of non-relapse mortality (NRM). These findings reveal the presence of a center effect in allogeneic HCT conducted during the past decade in Japan, highlighting the difference in relapse based on center experience. The weaker effect on NRM compared with that on relapse suggests that the transplantation care quality is becoming equalized across the country.
Similar content being viewed by others
Introduction
Allogeneic hematopoietic cell transplantation (HCT) is an effective therapy that minimizes the risk of relapse in patients with acute myeloid leukemia (AML), but in exchange for a substantial risk of non-relapse mortality (NRM) [1, 2]. Given the complex and specialized nature of the modality, it is expected that allogeneic HCT outcomes could be affected by treating centers’ experience. This concept, referred to as “the center effect,” has been investigated; however, the results are conflicting [3,4,5,6,7,8,9,10,11,12,13,14,15,16]. When interpreting the existing literature, it is worth noting that most of the previous studies analyzed heterogeneous patient populations with various diseases based on relatively older data.
The situation surrounding allogeneic HCT is changing. Owing to the rapid progress toward an information society, it has become much easier to obtain medical information by means other than one’s own experience, which may act to reduce the center effect. On the other hand, the growing diversification of transplantation procedures may act to increase the center effect. Therefore, it is worthwhile to reevaluate the center effect in this era of major changes in transplantation practice. In this study, we analyzed the center effect using the Japanese nationwide registry data of patients with AML who underwent allogeneic HCT during the past decade from 2010 to 2019 under a uniform healthcare system.
Methods
Study population
All data were collected using the Transplant Registry Unified Management Program sponsored by the Japanese Society for Transplantation and Cellular Therapy and the Japanese Data Center for Hematopoietic Cell Transplantation [17]. This registration program covers >99% of the more than 300 transplantation centers nationwide [18].
Patients aged ≥16 years with AML (except for acute promyelocytic leukemia) were eligible for the analysis if they underwent their first allogeneic HCT during first complete remission (CR), second CR, or non-CR between 2010 and 2019 from a matched related or unrelated donor, a haploidentical related donor, or single-unit umbilical cord blood (UCB) containing a total nucleated cell dose of ≥2.0 × 107/kg of the recipient’s body weight with at least a 4/6 match at the antigen level for human leukocyte antigen (HLA)-A, HLA-B, and HLA-DR [19].
This study was approved by the Institutional Review Board of the Aichi Cancer Center. The patients provided informed consent for the use of their clinical information for research.
Definitions
Center experience was defined based on the number of allogeneic HCTs performed for any indication during the study period, by which centers were first divided into four groups using quartiles. Preliminary analysis showed no significant difference between the second and third quartile groups in terms of overall survival (OS), relapse, or NRM (data not shown); thus, these two groups were combined, leading to the adoption of the following three categories: low-volume (median [range], 65 [1–93] cases per decade), intermediate-volume (median [range], 170 [94–316] cases per decade), and high-volume (median [range], 389 [328–1028] cases per decade) centers for subsequent analyses.
CR was determined when the following conditions were fulfilled: the presence of <5% of blasts in the bone marrow, absence of leukemic blasts in the peripheral blood and extramedullary sites, and recovery of blood counts [20]. Relapse was defined as the loss of CR, whereas NRM was defined as death without relapse. For the analysis of relapse, patients who had never achieved CR after transplantation were categorized as relapse cases at time zero. The Eastern Cooperative Oncology Group performance status [21], HCT-specific comorbidity index (HCT-CI) [22], conditioning intensity [23], and cytogenetic risk [24] were defined based on published criteria. Neutrophil and platelet engraftments were defined as the first of three consecutive measures with neutrophil and platelet counts of ≥0.5 × 109/L and ≥2.0 × 109/L, respectively [18]. Acute and chronic graft-versus-host disease (GVHD) were diagnosed and graded according to the standard criteria [25, 26].
Statistical analysis
The probability of OS was estimated using the Kaplan–Meier estimator, and differences between groups were analyzed using the log-rank test [27]. Multivariate analyses were performed using the Cox proportional hazards model [28]. All covariates listed in Table 1 were included in the final multivariate model considering their potentially confounding effects. Relapse and NRM were mutually competing events, and their incidence was estimated using the cumulative incidence estimator [29]. Engraftment and GVHD were also assessed by accommodating the competing risks. The Fine–Gray model was used for the univariate and multivariate analyses in a competing risk setting [30].
Results
Patients
A total of 7821 patients were eligible for the analysis, with 1833 (23%), 3884 (50%), and 2104 (27%) patients being classified into the low-, intermediate-, and high-volume center groups, respectively. The median follow-up for survivors was 4.3 (interquartile range [IQR], 2.3–6.6) years. Table 1 shows the characteristics of the patients in each center category. Patients who underwent transplantation at high-volume centers were characterized by older age, worse HCT-CI, and a higher frequency of non-CR at the time of transplantation. In addition, there was a notable imbalance regarding donor type; matched related donors accounted for 34%, 24%, and 19% in the low-, intermediate-, and high-volume centers, whereas UCB accounted for 32%, 45%, and 49%, respectively.
OS, relapse, and NRM
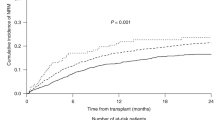
Figure 1 shows the OS, relapse, and NRM according to the center category. The 4-year probabilities of OS were 58% (95% confidence interval [CI], 55–61%), 63% (95% CI, 61–65%), and 66% (95% CI, 63–69%) for patients who underwent transplantation during CR at low-, intermediate-, and high-volume centers, respectively (P < 0.001, Fig. 1a). Their respective cumulative incidences of relapse were 23% (95% CI, 21–26%), 18% (95% CI, 17–20%), and 19% (95% CI, 17–22%) (P = 0.016, Fig. 1b), and their respective cumulative incidences of NRM were 22% (95% CI, 19–25%), 21% (95% CI, 19–23%), and 19% (95% CI, 17–22%) (P = 0.333, Fig. 1c). The 4-year probabilities of OS were 22% (95% CI, 19–25%), 29% (95% CI, 26–31%), and 29% (95% CI, 26–31%) for patients who underwent transplantation during non-CR at low-, intermediate-, and high-volume centers, respectively (P = 0.008, Fig. 1d). Their respective cumulative incidences of relapse were 56% (95% CI, 52–59%), 49% (95% CI, 47–52%), and 42% (95% CI, 39–45%) (P < 0.001, Fig. 1e), and their respective cumulative incidences of NRM were 27% (95% CI, 23–30%), 26% (95% CI, 24–28%), and 33% (95% CI, 30–36%) (P < 0.001, Fig. 1f).
Patients who underwent transplantation at low-, intermediate-, and high-volume centers are compared. a Overall survival (OS), (b) cumulative incidence of relapse, and (c) and cumulative incidence of non-relapse mortality (NRM) in patients with acute myeloid leukemia (AML) who underwent allogeneic hematopoietic cell transplantation (HCT) during complete remission (CR). d OS, (e) cumulative incidence of relapse, and (f) cumulative incidence of NRM in patients with AML who underwent allogeneic HCT during non-CR.
The results of the multivariate analysis on OS, relapse, and NRM are shown in Table 2. Using the high-volume centers as a reference, the risk of overall mortality was higher for the low- (hazard ratio [HR], 1.31; 95% CI, 1.20–1.44; P < 0.001) and intermediate-volume centers (HR, 1.11; 95% CI, 1.03–1.20; P = 0.008). Patients who underwent transplantation at low- and intermediate-volume centers showed a higher risk of relapse than those treated at high-volume centers (HR, 1.31; 95% CI, 1.16–1.48; P < 0.001 for the low-volume centers; and HR, 1.18; 95% CI, 1.07–1.31; P = 0.001, for the intermediate-volume centers). On the other hand, no difference was found in the risk of NRM between the low- and high-volume centers (HR, 1.08; 95% CI, 0.94–1.23; P = 0.278) or between the intermediate- and high-volume centers (HR, 0.90; 95% CI, 0.81–1.01; P = 0.070). Compared to the intermediate-volume centers, the low-volume centers were associated with higher risks of overall mortality (HR, 1.18; 95% CI, 1.09–1.28; P < 0.001) and NRM (HR, 1.19; 95% CI, 1.05–1.35; P = 0.005), and a trend toward a higher risk of relapse (HR, 1.10; 95% CI, 0.99–1.23; P = 0.062). Table 3 summarizes adjusted effects of the center volume on overall mortality, relapse, and NRM for each donor group. The center effect was most pronounced in UCB transplantation (UCBT), whereas it was not seen in haploidentical related HCT (Haplo-HCT).
Engraftment and GVHD
The adjusted effects of the center volume on engraftment and GVHD are shown in Supplementary Table 1. The likelihood of neutrophil engraftment was different only between the low- and high-volume centers (HR, 0.91; 95% CI, 0.85–0.98; P = 0.010); the differences in the likelihood of platelet engraftment were significant only between the intermediate- and high-volume centers (HR, 1.09; 95% CI, 1.02–1.17; P = 0.007). Compared to patients in the high-volume centers, those in the low-volume centers had lower risks of grade 2–4 acute GVHD (HR, 0.80; 95% CI, 0.71–0.90; P < 0.001), grade 3–4 acute GVHD (HR, 0.76; 95% CI, 0.62–0.92; P = 0.006), overall chronic GVHD (HR, 0.74; 95% CI, 0.66–0.84; P < 0.001), and extensive chronic GVHD (HR, 0.85; 95% CI, 0.72–1.00; P = 0.048), and those in the intermediate-volume centers had lower risks of grade 2–4 acute GVHD (HR, 0.91; 95% CI, 0.83–0.99; P = 0.032), overall chronic GVHD (HR, 0.68; 95% CI, 0.61–0.75; P < 0.001), and extensive chronic GVHD (HR, 0.76; 95% CI, 0.66–0.87; P < 0.0001). The difference between patients transplanted at low- and intermediate-volume centers was detected only in terms of grade 2–4 acute GVHD (HR, 0.88; 95% CI, 0.80–0.98; P = 0.019).
UCBT cohort
We next restricted our analysis to 3381 patients who underwent UCBT by dividing centers into four groups based on the number of UCBTs performed during the study period. The second and third quartile groups were combined because no difference was found in OS, relapse, or NRM (data not shown), and three categories of low-volume (n = 801, 24%), intermediate-volume (n = 1628, 48%), and high-volume centers (n = 952, 28%) were generated, with the numbers of UCBT performed during the study period of 10 years ranging 1–34 (median, 21), 35–117 (median, 79), and 121–794 (median, 204) cases, respectively. Consistent with the overall cohort, patients who underwent transplantation at high-volume centers were characterized by older age, worse HCT-CI, and a higher frequency of non-CR at transplantation (Table 4). Figure 2 shows the OS, relapse, and NRM according to the center category, and Table 5 presents the results of the multivariate analysis on OS, relapse, and NRM. The risk of overall mortality was higher for the low-volume centers (HR, 1.58; 95% CI, 1.38–1.82; P < 0.001) and the intermediate-volume centers (HR, 1.28; 95% CI, 1.13–1.44; P < 0.001) than the high-volume centers. Patients who underwent transplantation at low- and intermediate-volume centers had a higher risk of relapse than those treated at high-volume centers (HR, 1.66; 95% CI, 1.37–2.02; P < 0.001 for the low-volume centers; and HR, 1.54; 95% CI, 1.31–1.82; P < 0.001, for the intermediate-volume centers). There was no difference in the risk of NRM between the low- and high-volume centers (HR, 1.11; 95% CI, 0.93–1.34; P = 0.253) or between the intermediate- and high-volume centers (HR, 0.93; 95% CI, 0.80–1.09; P = 0.398). Compared to the intermediate-volume centers, the low-volume centers were associated with a higher risk of overall mortality (HR, 1.24; 95% CI, 1.09–1.40; P < 0.001) and a trend for a higher risk of NRM (HR, 1.19; 95% CI, 1.00–1.42; P = 0.054), but the risk of relapse was not different (HR, 1.08; 95% CI, 0.91–1.27; P = 0.381). The adjusted effects of the center category on engraftment and GVHD are shown in Supplementary Table 2. The high-volume centers had a higher likelihood of neutrophil engraftment than the low- or intermediate-volume centers (P = 0.003 and P = 0.014, respectively), and a higher likelihood of platelet engraftment than the low-volume centers (P = 0.017). As for GVHD, the high-volume centers showed higher risks for grade 2–4 acute GVHD, grade 3–4 acute GVHD, and overall chronic GVHD (P < 0.001 each, compared with the low- or intermediate-volume centers). The risk of extensive chronic GVHD did not differ between the low- and high-volume centers (P = 0.657) or between the intermediate- and high-volume centers (P = 0.336). The difference between patients transplanted at low- versus intermediate-volume centers was significant only in terms of grade 2–4 acute GVHD (HR, 0.78; 95% CI, 0.66–0.93; P = 0.005) and grade 3–4 acute GVHD (HR, 0.63; 95% CI, 0.45–0.87; P = 0.005).
Patients who underwent transplantation at low-, intermediate-, and high-volume centers are compared. a Overall survival (OS), (b) cumulative incidence of relapse, and (c) cumulative incidence of non-relapse mortality (NRM) in patients with acute myeloid leukemia (AML) who underwent UCBT during complete remission (CR). d OS, (e) cumulative incidence of relapse, and (f) cumulative incidence of NRM in patients with AML who underwent UCBT during non-CR.
Discussion
To date, several studies have investigated the center effect in allogeneic HCT; some have demonstrated the survival advantage for patients who undergo transplantation at high-volume centers [3,4,5, 8, 10,11,12,13, 15], whereas others have not [6, 7, 14]. Moreover, only a fraction of these studies have evaluated the center effect on relapse and NRM, with mixed results [3, 4, 8, 10, 11, 15]. The major limitation in the existing literature is patient heterogeneity in terms of disease and healthcare environment. In addition, most of the published studies are based on relatively old data, raising questions about the applicability of the results to current practice. With these considerations in mind, we evaluated the center effect with a focus on adults with AML who underwent allogeneic HCT during the past decade in Japan where medical care was provided within a uniform healthcare system.
Our study underscores the difference in patient characteristics according to the center volume. Patients transplanted at high-volume centers were older, had more comorbidities, and were more frequently in non-CR at the time of transplantation, indicating that the high-volume centers were more likely to undertake challenging cases. After adjustment for known confounding factors, the risk of overall mortality was lowest for the high-volume centers and highest for the low-volume centers, with the difference in overall mortality being attributed primarily to the difference in relapse rather than NRM. The lack of difference in NRM between the intermediate- and high-volume centers and between the low- and high-volume centers suggests that the transplantation care quality is becoming equalized among centers in recent years. On the other hand, a strong center effect was demonstrated in terms of relapse. As expected, disease-related factors, such as disease status and cytogenetic risk, were greatly associated with the risk of relapse. Thus, the observation that patients who underwent transplantation at high-volume centers included more patients who did so while not in CR merits special attention. In this respect, the difference in the relapse incidence according to the center category was significant in both patients with and without CR at the time of transplantation and the multivariate analysis adjusting for these disease-related factors corroborates the finding of the center effect on relapse.
Determining the exact reason for the lower relapse risk observed in high-volume centers is challenging due to the retrospective nature of the present registry-based study. However, it can be assumed that the availability of advanced diagnostic technologies and novel targeted therapies in these centers might have contributed to the reduction in relapse [31,32,33]. Moreover, higher risk for acute and chronic GVHD in patients who underwent transplantation at high-volume centers suggests that GVHD policies might differ across the centers. It is widely recognized that GVHD is accompanied by the graft-versus-leukemia (GVL) effect, which plays a beneficial role in reducing relapse in patients undergoing allogeneic HCT [34,35,36]. Although the strategy to allow the development of GVHD can increase NRM, it is noteworthy that NRM was not higher in the high-volume centers.
The subgroup analysis by donor source showed that the center effect was most pronounced in patients who underwent UCBT. Even when the center effect for UCBT recipients was analyzed separately based on the number of UCBTs performed during the study period, the results remained unchanged. A recent study showed that the GVL effect was more potent in patients who underwent UCBT than those who underwent allogeneic bone marrow transplantation (BMT) or peripheral blood stem cell transplantation (PBSCT) [37], lending support to the hypothesis that an occurrence of GVHD is more beneficial in UCBT than in BMT/PBSCT. By contrast, NRM was not affected by the center category in our UCBT cohort. UCBT has been performed actively in Japan, accounting for one-third of all allogeneic HCTs [38], and it is conceivable that equalization of the transplantation techniques among centers has been achieved in not only BMT/PBSCT but also UCBT. In our study, the center effect was not seen in Haplo-HCT. This could be explained at least partly by the small sample size; however, another potential explanation could be that the characteristic of Haplo-HCT to suppress GVHD extensively in a straightforward way may have provided little room to modulate the GVL effect. In the secondary analysis of the Blood and Marrow Transplant Clinical Trials Network 1101 study, in which patients were randomized to either UCBT or Haplo-HCT, centers experienced in UCBT (>10 UCBTs per year) had similar outcomes with UCBT and Haplo-HCT, whereas those experienced in Haplo-HCT (>10 Haplo-HCTs but ≤10 UCBTs per year) had worse outcomes with UCBT than Haplo-HCT because of a higher risk of relapse [16], which is in keeping with our findings.
Because of the retrospective nature of the study, our results suffer from inherent biases, such as patient selection, treatment heterogeneity, and the presence of unmeasured factors. In addition, we used the number of allogeneic HCTs as an indicator of center experience, whereas several studies have shown that the prognostic relevance of other center factors, such as patient-per-physician ratio [7], accreditation status [9], and program duration [11], which were not taken into consideration in our study. While acknowledging these potential limitations, our analysis was strengthened by a homogeneous patient population, namely, adults with AML who underwent allogeneic HCT under a uniform healthcare system. In this regard, however, consideration should be given to the fact that our findings were exclusively based on Japanese patient data; thus, it is premature to apply our conclusions to other patient populations.
In summary, this study reveals the presence of the center effect in allogeneic HCT performed during the past decade, 2010–2019, in Japan. The difference in overall mortality according to the center volume resulted primarily from the difference in relapse, and this may be derived from centers’ attitude toward the allowance of GVHD. The weaker effect of center experience on NRM compared with that on relapse suggests the advancement in the transplantation care quality across the country.
Data availability
The data of this study are not publicly available due to ethical restrictions that it exceeds the scope of the recipient/donor’s consent for research use in the registry. Data may be available from the corresponding author upon reasonable request and with permission of the JSTCT/JDCHCT.
References
Dholaria B, Savani BN, Hamilton BK, Oran B, Liu HD, Tallman MS, et al. Hematopoietic cell transplantation in the treatment of newly diagnosed adult acute myeloid leukemia: an evidence-based review from the American Society of Transplantation and Cellular Therapy. Transpl Cell Ther. 2021;27:6–20.
Yanada M. The evolving concept of indications for allogeneic hematopoietic cell transplantation during first complete remission of acute myeloid leukemia. Bone Marrow Transpl. 2021;56:1257–65.
Horowitz MM, Przepiorka D, Champlin RE, Gale RP, Gratwohl A, Herzig RH, et al. Should HLA-identical sibling bone marrow transplants for leukemia be restricted to large centers? Blood. 1992;79:2771–4.
Frassoni F, Labopin M, Powles R, Mary JY, Arcese W, Bacigalupo A, et al. Effect of centre on outcome of bone-marrow transplantation for acute myeloid leukaemia. acute leukaemia working party of the European Group for Blood and Marrow Transplantation. Lancet. 2000;355:1393–8.
Matsuo K, Hamajima N, Morishima Y, Harada M. Hospital capacity and post-transplant survival after allogeneic bone marrow transplantation: analysis of data from the Japan Society for Hematopoietic Cell Transplantation. Bone Marrow Transpl. 2000;26:1061–7.
Mesnil F, Jouet JP, Tuppin P, Vernant JP, Golmard JL. Evaluation of centre and period effects in allogeneic haematopoietic stem cell transplantation in France. Bone Marrow Transpl. 2004;34:645–51.
Loberiza FR Jr, Zhang MJ, Lee SJ, Klein JP, LeMaistre CF, Serna DS, et al. Association of transplant center and physician factors on mortality after hematopoietic stem cell transplantation in the United States. Blood. 2005;105:2979–87.
Klingebiel T, Cornish J, Labopin M, Locatelli F, Darbyshire P, Handgretinger R, et al. Results and factors influencing outcome after fully haploidentical hematopoietic stem cell transplantation in children with very high-risk acute lymphoblastic leukemia: impact of center size: an analysis on behalf of the Acute Leukemia and Pediatric Disease Working Parties of the European Blood and Marrow Transplant group. Blood. 2010;115:3437–46.
Gratwohl A, Brand R, Niederwieser D, Baldomero H, Chabannon C, Cornelissen J, et al. Introduction of a quality management system and outcome after hematopoietic stem-cell transplantation. J Clin Oncol. 2011;29:1980–6.
Giebel S, Labopin M, Mohty M, Mufti GJ, Niederwieser D, Cornelissen JJ, et al. The impact of center experience on results of reduced intensity: allogeneic hematopoietic SCT for AML. An analysis from the acute leukemia working party of the EBMT. Bone Marrow Transpl. 2013;48:238–42.
Gratwohl A, Sureda A, Baldomero H, Gratwohl M, Dreger P, Kroger N, et al. Economics and outcome after hematopoietic stem cell transplantation: a retrospective cohort study. EBioMedicine. 2015;2:2101–9.
Terakura S, Atsuta Y, Tsukada N, Kobayashi T, Tanaka M, Kanda J, et al. Comparison of outcomes of 8/8 and 7/8 allele-matched unrelated bone marrow transplantation and single-unit cord blood transplantation in adults with acute leukemia. Biol Blood Marrow Transpl. 2016;22:330–8.
Shouval R, Ruggeri A, Labopin M, Mohty M, Sanz G, Michel G, et al. An integrative scoring system for survival prediction following umbilical cord blood transplantation in acute leukemia. Clin Cancer Res. 2017;23:6478–86.
Rashidi A, Hamadani M, Zhang MJ, Wang HL, Abdel-Azim H, Aljurf M, et al. Outcomes of haploidentical vs matched sibling transplantation for acute myeloid leukemia in first complete remission. Blood Adv. 2019;3:1826–36.
Kanda J, Hayashi H, Ruggeri A, Kimura F, Volt F, Takahashi S, et al. Prognostic factors for adult single cord blood transplantation among European and Japanese populations: the Eurocord/ALWP-EBMT and JSHCT/JDCHCT collaborative study. Leukemia. 2020;34:128–37.
Brunstein CG, O’Donnell PV, Logan B, Dawson P, Costa L, Cutler C, et al. Impact of center experience with donor type on outcomes: a secondary analysis, blood and marrow transplant clinical trials network 1101Open for Accrual June 2012Open for Accrual June 2012. Transpl Cell Ther. 2022;28:406.e1–06.e6.
Arai Y, Konuma T, Yanada M. Hematopoietic cell transplantation in adults with acute myeloid leukemia: a review of the results from various nationwide registry studies in Japan. Blood Cell Ther. 2021;4:35–43.
Atsuta Y. Introduction of transplant registry unified management program 2 (TRUMP2): scripts for TRUMP data analyses, part I (variables other than HLA-related data). Int J Hematol. 2016;103:3–10.
Yanada M, Konuma T, Kuwatsuka Y, Kondo T, Kawata T, Takahashi S, et al. Unit selection for umbilical cord blood transplantation for adults with acute myeloid leukemia in complete remission: a Japanese experience. Bone Marrow Transpl. 2019;54:1789–98.
Dohner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Buchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424–47.
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–55.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Yanada M, Harada K, Shimomura Y, Arai Y, Konuma T. Conditioning regimens for allogeneic hematopoietic cell transplantation in acute myeloid leukemia: Real-world data from the Japanese registry studies. Front Oncol. 2022;12:1050633.
Yanada M, Mori J, Aoki J, Harada K, Mizuno S, Uchida N, et al. Effect of cytogenetic risk status on outcomes for patients with acute myeloid leukemia undergoing various types of allogeneic hematopoietic cell transplantation: an analysis of 7812 patients. Leuk Lymphoma. 2018;59:601–09.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transpl. 1995;15:825–8.
Sullivan KM, Agura E, Anasetti C, Appelbaum F, Badger C, Bearman S, et al. Chronic graft-versus-host disease and other late complications of bone marrow transplantation. Semin Hematol. 1991;28:250–9.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81.
Cox DR. Regression models and life-tables. J R Stat Soc B. 1972;34:187–220.
Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med. 1999;18:695–706.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Short NJ, Ravandi F. How close are we to incorporating measurable residual disease into clinical practice for acute myeloid leukemia? Haematologica. 2019;104:1532–41.
Spyridonidis A. How I treat measurable (minimal) residual disease in acute leukemia after allogeneic hematopoietic cell transplantation. Blood. 2020;135:1639–49.
Mohty R, El Hamed R, Brissot E, Bazarbachi A, Mohty M. New drugs before, during, and after hematopoietic stem cell transplantation for patients with acute myeloid leukemia. Haematologica. 2023;108:321–41.
Weiden PL, Flournoy N, Thomas ED, Prentice R, Fefer A, Buckner CD, et al. Antileukemic effect of graft-versus-host disease in human recipients of allogeneic-marrow grafts. N. Engl J Med. 1979;300:1068–73.
Horowitz MM, Gale RP, Sondel PM, Goldman JM, Kersey J, Kolb HJ, et al. Graft-versus-leukemia reactions after bone marrow transplantation. Blood. 1990;75:555–62.
Stern M, de Wreede LC, Brand R, van Biezen A, Dreger P, Mohty M, et al. Sensitivity of hematological malignancies to graft-versus-host effects: an EBMT megafile analysis. Leukemia. 2014;28:2235–40.
Konuma T, Kanda J, Kuwatsuka Y, Yanada M, Kondo T, Hirabayashi S, et al. Differential effect of graft-versus-host disease on survival in acute leukemia according to donor type. Clin Cancer Res. 2021;27:4825–35.
Yanada M, Takami A, Yamasaki S, Arai Y, Konuma T, Uchida N, et al. Allogeneic hematopoietic cell transplantation for adults with acute myeloid leukemia conducted in Japan during the past quarter century. Ann Hematol. 2020;99:1351–60.
Acknowledgements
This work was supported in part by a grant from the Aichi Cancer Research Foundation, grant number: 2023-1-7 (MY), and a grant from the Practical Research Project for Allergic Diseases and Immunology (Research Technology of Medical Transplantation) from the Japan Agency for Medical Research and Development (AMED) (grant no. 19ek0510023h0003) (YA).
Author information
Authors and Affiliations
Contributions
MY designed the study, analyzed data, interpreted the results, and drafted the paper; SY, YK, K Kawamura, and TK interpreted results and edited the paper; TF, TI, YH, HG, K Kato, FI, AS, M Onizuka, KM, YI, AY, M Ohbiki, KT, YA, and JK contributed to data management, interpreted the results, and edited the paper; and all authors approved the final version of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yanada, M., Yano, S., Kuwatsuka, Y. et al. The effect of center experience on allogeneic hematopoietic cell transplantation outcomes in acute myeloid leukemia. Bone Marrow Transplant 59, 541–549 (2024). https://doi.org/10.1038/s41409-024-02222-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-024-02222-5