Abstract
Granulocyte colony-stimulating factor (G-CSF) is used in a majority of healthy donors to obtain peripheral blood stem cells for allogeneic stem cell transplantation. Since high levels of G-CSF activates endothelial cells and can induce a pro-coagulatory state, and fuelled by case reports of cardiovascular events in donors, some concerns have been raised about a potential for an increased risk of cardiovascular events for the donors after donation. We studied the incidence of cardiovascular disease following stem cell donation in a Swedish national register based cohort of 1098 peripheral blood stem cell donors between 1998 and 2016. The primary objective was to evaluate if the incidence of cardiovascular disease was increased for donors treated with G-CSF. The incidence of any new cardiovascular disease was 6.0 cases per 1000 person years, with a median follow up of 9.8 years. The incidence did not exceed that of age- sex- and residency-matched population controls (hazard ratio 0.90, 95% confidence interval (CI) 0.76–1.07, p-value 0.23), bone marrow donors, or non-donating siblings. Long-term cardiovascular disease incidence was not increased in this national register based study of peripheral blood stem cell donors treated with G-CSF.
Similar content being viewed by others
Introduction
During the last 20 years G-CSF (granulocyte-colony stimulating factor) has been used for blood stem cell mobilisation in healthy blood stem cell donors.
Endogenous G-CSF works as a regulator of myeloid progenitor cell proliferation and maturation into mature neutrophils, mainly mediated through effects on the transmembrane G-CSF receptor. The G-CSF receptor is found on both hematopoietic stem cells and myeloid progenitors, as well as mature cells, such as neutrophilic granulocytes, monocytes and lymphocytes. The receptor is also expressed in a variety of non-hematopoietic cells, including cardiovascular, neuronal, endothelial and placental cells [1, 2].
G-CSF and haematopoietic stem cells have been suggested to play a role in the development of atherosclerosis, and reports of severe cardiovascular adverse events has led to a concern that G-CSF treatment of healthy donors would increase the risk of cardiovascular events [3, 4]. The short-term health risks associated with G-CSF treatment and donation have been well described and are generally considered acceptable [5,6,7,8], although Halter et al., in a large retrospective study, reported a higher number of cases (n = 7 vs. n = 1) of pulmonary embolism and/or deep venous thrombosis in peripheral blood stem cell donors (PBSC) donors than in bone marrow (BM) donors [4].
Less is known about medium to long-term effects on the incidence of cardiovascular disease (CVD).
Aim
To investigate if the incidence of cardiovascular disease in Swedish peripheral blood stem cell donors is increased after treatment with G-CSF.
Methods
Combining data from several national Swedish population based registers, we present a prospective register-based cohort study of the incidence of new cardiovascular disease after peripheral blood stem cell donation.
Data collection
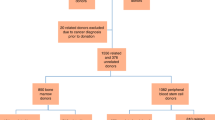
A cohort of related (donating to a sibling, child or parent) haematopoietic stem cell donors between 1977 and 2014 was collected from all Swedish centres for allogeneic stem cell transplantation (Fig. 1). In total, 1576 first time related donors were identified with complete records of personal national identification number, stem cell source, and donation date. For 19 additional donors not all necessary information could not be verified and they were excluded from the study.
An additional 376 unrelated Swedish donors previously included in the Nordic Register for Haematopoietic Stem cell Donors (NRHSD), donating between 1998 and 2014, were also included in the study, making the number of peripheral blood stem cell donors 1098 [8].
A database was created by linking data from Swedish national population based registers; The Swedish Multi-Generation Register, The Swedish Patient Register, and The Swedish Cause-of-death register, containing data on all incident diagnoses of cardiovascular disease for the 1098 peripheral blood stem cell donors (PBSC), 1062 siblings and 854 bone marrow donors. For each PBSC donor, five age-, sex- and county of residence-matched controls were assigned from the general Swedish population, 5495 in total. The linkage was performed at the Swedish National Board for Health and Welfare using personal identity numbers, which were then removed before delivery of the datasets for statistical analyses.
The Swedish Multi-Generation Register
The identity of all the donors’ siblings was obtained from The Multi-Generation Register, Statistics Sweden. The register is derived from Swedish population statistics and include information about the identity of parents, siblings and children for Swedish residents born after 1931 and resident in Sweden at some point in time from 1961 and onwards [9].
The Swedish Patient Register
The Patient Register includes diagnoses in ICD-format from admissions to hospital since 1965, and specialised out-patient care since 2001 [10]. The patient register was used as the primary source for diagnoses of cardiovascular disease, supplemented with data the Swedish Cause-of-death Register for those without a relevant cardiovascular disease diagnosis before time of death.
The Swedish Cause-of-death Register
Since 1952, dates and causes of death are recorded in the Causes of Death Register [11].
Classification of cardiovascular disease
Cases of cardiovascular disease was classified according to ICD-8, ICD-9 or ICD-10 depending on year of diagnosis, Supplementary Appendix 1.
Statistical analysis
For data analysis, statistical software SAS version 9.4 for Windows (SAS Institute, Cary, NC) was used.
Cardiovascular disease incidences for donors and their comparison groups were modelled by multivariable Cox regression, using the SAS procedure PHREG [12, 13], with age at donation and sex (male/female) included as variables.
To adjust for potential time trends in diagnosis of cardiovascular disease, and because bone marrow donors were more likely to have donated during the use of ICD-code classification ICD-8 and ICD-9, year of donation was also included as a potential confounding factor in the models comparing disease incidence between PBSC and BM donors.
For each disease category, individuals with a diagnosis of the examined disease prior to the time of donation were excluded from that analysis.
Relative risks, compared to population controls, siblings and bone marrow donors, were estimated as hazard ratios with 95% Cis, and the results are presented both as crude number of events, event rates per 1000 person-years of follow up, and in models adjusted for confounding factors.
Mortality for PBSC donors and population controls were calculated using data from the Cause of death register. All-cause mortality and CVD-specific mortality was compared in separate regression models. Two different models were analysed; one with age at donation and sex as possible confounders, and one with the addition of a weighted Charlson Comorbidity Index variable, calculated according to Ludvigsson et al. [14].
Ethical review
The study was approved by the regional ethical vetting board of Stockholm, 98-259, and Uppsala, 2016-497.
Results
Donor characteristics
1576 related and 376 unrelated first time donors were included in the study.
Population based controls for PBSC donors were well matched for sex, age, and highest education level reached, Table 1.
Choice of donation method was associated with age, with 95% of donors less than 18 years donating bone marrow, while 91% of donors of age above 60, and all above 70 years, donated PBSC.
Bone marrow donation was used exclusively until the last years of the 1990s. A gradual increase in the proportion of PBSC donation is seen in the decades after introduction of G-CSF. The proportion of PBSC donors had increased to around 75% during the last years of the study (2010–2014).
Donor cardiovascular disease incidence
Compared with population based controls
Of 1098 PBSC donors, 60 donors (5.5%) had a diagnoses of cardiovascular disease prior to donation, as detailed in Supplementary Appendix 2. Among the remaining 1038 PBSC donors without any cardiovascular disease before donation, a new diagnosis of cardiovascular disease was recorded for 167 donors (16.5%), with a median follow up of 9.2 years (min: 0–max 21.2). The donors event rate of any new cardiovascular disease, 18.1 cases per 1000 person-years, was not different from that of matched controls (event rate 19.2 cases per 1000 person-years, hazard ratio 0.89 (95% CI 0.75–1.06, p-value 0.19).
Similarly, there was no risk difference seen between donors and controls for any specific cardiovascular disease category, although a general non-significant trend towards lower risk for donors can be noted, Table 2.
Compared with bone marrow donors
The new cardiovascular disease incidence after donation for 1038 PBSC donors was not different from that of 831 bone marrow donors, (hazard ratio 0.90, 95% CI 0.63–1.27, p-value 0.90) in a model adjusted for age, sex, year of donation and year of diagnosis, Table 3. Median follow-up time for bone marrow donors was 18.3 years (0.3–38.3 years).
Compared with non-donating siblings
A separate comparison was made between sibling donors and their non-donating siblings. Only sibling PBSC donors with at least one non-transplant-recipient sibling, alive at the time of the donation, were included in the analyses, in total 694 sibling donors and 1222 siblings. Out of 649 donors without any diagnosis of cardiovascular disease at donation, 107 (16.5%) received a new diagnosis of cardiovascular disease during follow up, compared with 209 (19.6%) of 1091 siblings.
The PBSC donors did not have an increased risk for new cardiovascular disease when compared to their non-donating siblings (hazard ratio 0.86, 95% CI 0.68–1.09, p-value 0.21), Table 4.
Donor mortality
Mortality from any cause was registered for 32 of 1098 PBSC donors (2.9%) during follow up, compared to 240 of 5495 population controls (4.4%). All-cause mortality was lower for donors than controls (hazard ratio 0.65, 95% CI 0.45–0.94, p-value 0.02), in a regression model including sex and age at donation. If a weighted Charlson Comorbidity Index-variable (CCI) was included in the model, as an adjustment for potential different baseline risks of cardiovascular disease between donors and controls, this difference was no longer significant (hazard ratio 0.70, 95% CI 0.49–1.02, p-value 0.06).
Similarly, CVD-specific mortality was lower for donors (1.5%) than for controls (2.4%) (hazard ratio 0.59, 95% CI 0.35–0.99, p-value 0.04), in the model including sex and age, but no longer significantly so when the CCI variable was included, (hazard ratio 0.65, 95% CI 0.39–1.09, p-value 0.10).
There was no donor mortality registered within the first 12 months after donation. The first registered case occurred after 1.8 years, in a related donor aged 61 at time of donation, dying from pancreatic cancer.
Mortality causes for all deceased PBSC donors can be found in Supplementary Appendix 3.
Discussion
The number of allogeneic transplantations have been increasing steadily over the last 30 years, with an increase in older recipients, and a gradual increase of donations from both sibling and haplo-identical related donors [15, 16]. An expected consequence of this is the increasing use of older donors, presumably with a higher degree of risk factors for cardiovascular disease.
Stem cell donation is generally safe, and associated with low risk of short-term morbidity. However, since the donors are healthy volunteers, all measures must be taken to avoid negative consequences, both at donation and in a longer perspective. Cardiovascular risk has in this setting been high-lighted in international guidelines [17].
In our analyses, the overall risk of cardiovascular disease after PBSC donation reassuringly does not seem to be increased, compared to population controls, bone marrow donors or the donors´ siblings, even among donors followed for a median of almost ten years. There is even an indication that the risk of new cardiovascular disease is slightly lower for PBSC donors compared to age, sex and county-of-residence matched population controls (hazard ratios ranging from 0.72 to 0.98), likely due to unadjusted differences in cardiovascular disease risk factors. The precision of the individual estimates are however fairly low, as seen in Table 2.
The same signal of a possibly lower cardiovascular risk for PBSC donors is not as evident in the comparisons with bone marrow donors or the donors´ siblings. One explanation could be that remaining confounding factors, not accounted for in our models, are differently distributed between comparison groups, combined with a lower statistical power for these analyses due to the smaller study base.
All-cause and CVD-specific mortality in our study seems to be 30–40% reduced compared to age, sex and residency-matched control, even after adjusting for Charlson Comorbidity Index calculated for diagnoses prior to donation. We believe this to be caused by remaining unmeasured confounders not accounted for in this study.
The cardiovascular disease risk after donation have been previously studied using different study designs than in our study. In a large retrospective study by Halter et al., four fatalities from cardiovascular causes occurred within 30 days after donation in PBSC donors, three cardiac arrests and one subarachnoid haemorrhage [4]. The overall number of serious adverse cardiovascular events, also including one case of supraventricular tachycardia, two myocardial infarctions and one subdural haematoma, were low and were judged to be comparable to that of BM donors.
Martino et al. reported that 23% of donors (58/276) were diagnosed with cardiovascular disease during a median follow-up of 7.8 years, similar to the numbers found in our study [18]. The most frequent diagnosis in this study was hypertension in 48 (17%) patients, while 3.6% had other diagnoses, including 4 strokes, 3 acute myocardial infarctions, and single cases of aortic occlusion, pulmonary embolism, and cardiac valvulopathy. The incidence of cardiovascular disease was estimated to be similar to that expected in the general population, although no presentation of the power of this analysis was included, nor any data on loss to follow up.
In a small study by Cavallaro et al. of 95 PBSC donors, with a median follow up of 3.3 years, one stroke and one case of angina was reported. No comparison to expected rates in the population was made [19].
In a much larger study by Pulsipher at al., including 6788 PBSC donors with a fairly short median follow up of 3 years, the incidence of thrombosis, including stroke, did not differ between PBSC and BM donors. However,the analysis did not adjust for age- and sex- differences between donor groups, and did not report any clear definition of what diagnosis were included, nor absolute numbers of events [7].
Added knowledge from this study
This study provides an estimate of the long-term incidence rate of cardiovascular disease after donation, in a relatively large national cohort of 1098 healthy PBSC donors. The median follow up of 9.8 years for cardiovascular disease in peripheral blood stem cell donors is, according to our knowledge, the longest so far reported. Outcome measures are clearly defined by ICD-codes from validated national registers, with a very low loss to follow up. The incidence rate for cardiovascular disease is compared to that of both matched population controls as well as to that of BM donors and the donors´ siblings, increasing the reliability of the results.
Limitations to our study
One limitation to this study is a lack of data regarding the distribution of a majority of known risk factors for cardiovascular disease, such as smoking, obesity, physical activity, that may be different between donors and controls. Those accepted for stem cell donation are likely to be healthier and have fewer cardiovascular disease risk factors than the general population, beyond what can be fully adjusted for in our data, possibly leading to a lower sensitivity to detect an eventual risk increase from G-CSF. Another limitation is that only Swedish donors were included in the study, possibly affecting the generalisability of our results. Even though the cardiovascular disease burden in Sweden is fairly similar to that of most Western European, North and South American countries, the cardiovascular disease burden is considerably lower than that seen in many countries [20] performing stem cell transplantations globally.
We could further only study first-time cardiovascular events following donation, due to the nature of the registered data, and not the effect of G-CSF on donors who had records of cardiovascular disease before donation. This latter small group of donors would be of special interest to follow, as this group today might be more likely to be advised against donating due to worries about negative effects of G-CSF. As well, limitations imposed by study size and the nature of available data, did not allow us to investigate the cardiovascular disease risk separately for several specific donor groups of relevance, such as related/unrelated donors, younger/older donors, or those with pre-existing cardiovascular disease, and this should be considered when interpreting our results.
Conclusion
In this national, register based cohort study of 1098 Swedish PBSC donors followed for a median of 9.8 years, we found no evidence of an increased risk of new cardiovascular disease after donation.
We conclude that concerns about long-term cardiovascular risk, based on today’s knowledge and appropriate donor selection, should not hinder stem cell collection with G-CSF in healthy volunteers.
Data availability
The data in our study results are not publicly available due to restrictions according to national data protection legislation. Datasets analysed during the current study are available from the corresponding author upon reasonable request.
References
van de Geijn GJ, Aarts LH, Erkeland SJ, Prasher JM, Touw IP. Granulocyte colony-stimulating factor and its receptor in normal hematopoietic cell development and myeloid disease. Rev Physiol Biochem Pharm. 2003;149:53–71.
Touw IP, Geijn G-JMVD. Granulocyte colony-stimulating factor and its receptor in normal myeloid cell development, leukemia and related blood cell disorders. Front Biosci-Landmark. 2007;12:800–15.
Lang JK, Cimato TR. Cholesterol and hematopoietic stem cells: inflammatory mediators of atherosclerosis. Stem Cells Transl Med. 2014;3:549–52.
Halter J, Kodera Y, Ispizua AU, Greinix HT, Schmitz N, Favre G, et al. Severe events in donors after allogeneic hematopoietic stem cell donation. Haematologica. 2009;94:94–101.
Pulsipher MA, Chitphakdithai P, Miller JP, Logan BR, King RJ, Rizzo JD, et al. Adverse events among 2408 unrelated donors of peripheral blood stem cells: results of a prospective trial from the National Marrow Donor Program. Blood. 2009;113:3604–11.
Hölig K, Kramer M, Kroschinsky F, Bornhäuser M, Mengling T, Schmidt AH, et al. Safety and efficacy of hematopoietic stem cell collection from mobilized peripheral blood in unrelated volunteers: 12 years of single-center experience in 3928 donors. Blood. 2009;114:3757–63.
Pulsipher MA, Chitphakdithai P, Logan BR, Navarro WH, Levine JE, Miller JP, et al. Lower risk for serious adverse events and no increased risk for cancer after PBSC vs BM donation. Blood. 2014;123:3655–63.
Pahnke S, Larfors G, Axdorph-Nygell U, Fischer-Nielsen A, Haastrup E, Heldal D, et al. Short-term side effects and attitudes towards second donation: A comparison of related and unrelated haematopoietic stem cell donors. J Clin Apher. 2018 Jun;33(3):226–235
Ekbom, A. (2011). The Swedish Multi-generation Register. In: Dillner, J. (eds) Methods in Biobanking. Methods in Molecular Biology, vol 675. Humana Press, Totowa, NJ.
Ludvigsson JF, Andersson E, Ekbom A, Feychting M, Kim JL, Reuterwall C, et al. External review and validation of the Swedish national inpatient register. BMC Public Health. 2011;11:450.
Causes of death. The National Board of Health and Welfare. Stockholm. 2010;2011:258.
SAS_Institute_Inc. PHREG procedure: SAS Institute Inc; 2021. https://documentation.sas.com/doc/en/pgmsascdc/9.4_3.4/statug/statug_phreg_references.htm#statug_phregcox_d72.
Cox DR. Regression models and life-tables. J R Stat Soc Ser B. 1972;34:187–202.
Ludvigsson JF, Appelros P, Askling J, Byberg L, Carrero JJ, Ekström AM, et al. Adaptation of the Charlson Comorbidity Index for Register-Based Research in Sweden. Clin Epidemiol. 2021;13:21–41.
Passweg JR, Baldomero H, Chabannon C, Basak GW, de la Cámara R, Corbacioglu S, et al. Hematopoietic cell transplantation and cellular therapy survey of the EBMT: monitoring of activities and trends over 30 years. Bone Marrow Transplant. 2021;56:1651–64.
D’Souza A, Fretham C, Lee SJ, Arora M, Brunner J, Chhabra S, et al. Current Use of and Trends in Hematopoietic Cell Transplantation in the United States. Biol Blood Marrow Transplant. 2020;26:e177–82.
World Marrow Donor Association, Donor Medical Suitability Recommendations. 2022. https://share.wmda.info/display/DMSR/WMDA+Donor+Medical+Suitability+Recommendations+Main+page.
Martino M, Bonizzoni E, Moscato T, Recchia AG, Fedele R, Gallo GA, et al. Mobilization of hematopoietic stem cells with lenograstim in healthy donors: efficacy and safety analysis according to donor age. Biol Blood Marrow Transpl. 2015;21:881–8.
Cavallaro AM, Lilleby K, Majolino I, Storb R, Appelbaum FR, Rowley SD, et al. Three to six year follow-up of normal donors who received recombinant human granulocyte colony-stimulating factor. Bone Marrow Transpl. 2000;25:85–9.
Vaduganathan M, Mensah GA, Turco JV, Fuster V, Roth GA. The Global Burden of Cardiovascular Diseases and Risk. J Am Coll Cardiol. 2022;80:2361–71.
Acknowledgements
We would like to thank all the participating centres for registering donors. The study has been supported by funding from the Nordic Cancer Union, the Swedish Cancer Society and the Uppsala-Örebro Regional Research Council.
Funding
Open access funding provided by Uppsala University.
Author information
Authors and Affiliations
Contributions
SP, HH and GL designed the study. SP performed the statistical analysis with assistance from GL. SP wrote the article. All authors collected data, gave input to the article, and approved of the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pahnke, S., Hägglund, H. & Larfors, G. Incidence of cardiovascular disease in healthy Swedish peripheral blood stem cell donors – a nationwide study. Bone Marrow Transplant 59, 403–408 (2024). https://doi.org/10.1038/s41409-023-02196-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-023-02196-w