Abstract
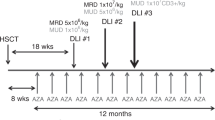
Prophylactic donor lymphocyte infusions (DLI) are used to augment post-transplant immune recovery to reduce both infectious complications and disease recurrence. Preclinical studies implicate the naive T-cell subset as the primary driver of graft-versus-host disease (GvHD). In this phase I dose escalation study, we assessed the safety of a DLI that was depleted of CD45RA+ naive T cells. Sixteen adult patients received a prophylactic DLI at a median of 113 days (range 76–280 days) following an HLA-identical, non-myeloablative allogeneic hematopoietic stem cell transplantation. Three patients each received the naive T-cell depleted DLI with a CD3+ dose of 1 × 105/kg, 1 × 106/kg, and 5 × 106/kg. The maximum dose of 1 × 107/kg was expanded to 7 patients. No dose-limiting grade III/IV acute GvHD or adverse events attributable to the DLI were observed at any dose level. One patient developed grade 2 acute GvHD of skin and upper intestines, and another developed moderate chronic GvHD of the lungs following the DLI. With a median follow-up of 2.8 years, 2-year progression-free and overall survival is 50.0% and 68.8%, respectively. In conclusion, these data suggest that a DLI that has been depleted of CD45RA+ naive T cells is feasible and carries a low risk of acute or chronic GvHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Scott BL, Pasquini MC, Logan BR, Wu J, Devine SM, Porter DL, et al. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J Clin Oncol. 2017;35:1154–61.
Barrett AJ, Mavroudis D, Tisdale JR, Molldrem J, Clave E, Dunbar CE, et al. T cell-depleted bone marrow transplantation and delayed T cell add-back to control acute GVHD and conserve a graft-versus-leukemia effect. Bone Marrow Transplant. 1998;21:543–51.
Bar M, Sandmaier BM, Inamoto Y, Bruno B, Hari P, Chauncey T, et al. Donor lymphocyte infusion for relapsed hematological malignancies after allogeneic hematopoietic cell transplantation: prognostic relevance of the initial CD3+ T cell dose. Biol Blood Marrow Transplant. 2013;19:949–57.
Chen BJ, Deoliveira D, Cui X, Le NT, Son J, Whitesides JF, et al. Inability of memory T cells to induce graft-versus-host disease is a result of an abortive alloresponse. Blood 2007;109:3115–23.
Chen BJ, Cui X, Sempowski GD, Liu C, Chao NJ. Transfer of allogeneic CD62L- memory T cells without graft-versus-host disease. Blood 2004;103:1534–41.
Anderson BE, McNiff J, Yan J, Doyle H, Mamula M, Shlomchik MJ, et al. Memory CD4+ T cells do not induce graft-versus-host disease. J Clin Investig. 2003;112:101–8.
Xystrakis E, Bernard I, Dejean AS, Alsaati T, Druet P, Saoudi A, et al. Alloreactive CD4 T lymphocytes responsible for acute and chronic graft-versus-host disease are contained within the CD45RChigh but not the CD45RClow subset. Eur J Immunol. 2004;34:408–17.
Dutt S, Tseng D, Ermann J, George TI, Liu YP, Davis CR, et al. Naive and memory T cells induce different types of graft-versus-host disease. J Immunol. 2007;179(November):6547–54.
Muffly L, Sheehan K, Armstrong R, Jensen K, Tate K, Rezvani AR, et al. Infusion of donor-derived CD8+ memory T cells for relapse following allogeneic hematopoietic cell transplantation. Blood Adv. 2018;2(March):681–90.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, et al. Thomas ED Clinical manifestations of graft-versus-host disease in human recipients of marrow from HLA-matched sibling donors. Transplantation 1974;18:295–304.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National institutes of health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 diagnosis and staging working group report. Biol Blood Marrow Transplant. 2015;21:389–401.
Armand P, Gibson CJ, Cutler C, Ho VT, Koreth J, Alyea EP, et al. A disease risk index for patients undergoing allogeneic stem cell transplantation. Blood. 2012;120(July):905–13.
Bleakley M, Heimfeld S, Loeb KR, Jones LA, Chaney C, Seropian S, et al. Outcomes of acute leukemia patients transplanted with naive T cell–depleted stem cell grafts. J Clin Invest. 2015;125:2677–89.
Shook DR, Triplett BM, Eldridge PW, Kang G, Srinivasan A, Leung W. Haploidentical stem cell transplantation augmented by CD45RA negative lymphocytes provides rapid engraftment and excellent tolerability. Pediatr Blood Cancer. 2015;62:666–73.
Triplett BM, Muller B, Kang G, Li Y, Cross SJ, Moen J, et al. Selective T-cell depletion targeting CD45RA reduces viremia and enhances early T-cell recovery compared with CD3-targeted T-cell depletion. Transplant Infectious Dis. 2018;20.
Rizzieri DA, Dev P, Long GD, Gasparetto C, Sullivan KM, Horwitz M, et al. Response and toxicity of donor lymphocyte infusions following T cell depleted non-myeloablative allogeneic hematopoietic stem cell transplantation from 3–6/6 HLA matched donors. Bone Marrow Transplant. 2009;43:327–33.
Acknowledgements
Ko Maung is supported by NIH T32HL007057 grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maung, K.K., Chen, B.J., Barak, I. et al. Phase I dose escalation study of naive T-cell depleted donor lymphocyte infusion following allogeneic stem cell transplantation. Bone Marrow Transplant 56, 137–143 (2021). https://doi.org/10.1038/s41409-020-0991-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-020-0991-5
This article is cited by
-
Automatic generation of alloreactivity-reduced donor lymphocytes and hematopoietic stem cells from the same mobilized apheresis product
Journal of Translational Medicine (2023)
-
The graft-versus-leukemia effect of prophylactic donor lymphocyte infusions after allogeneic stem cell transplantation is equally effective in relapse prevention but safer compared to spontaneous graft-versus-host disease
Annals of Hematology (2023)