Abstract
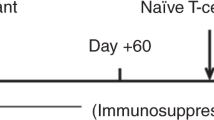
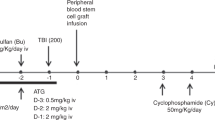
Depletion of αβ T cells from the graft prevents graft-vs.-host disease (GVHD) and improves outcome of HSCT from haploidentical donors. In a randomized trial, we aimed to evaluate the safety and efficacy of low-dose memory (CD45RA-depleted) donor lymphocytes (mDLI) after HSCT with αβ T-cell depletion. A cohort of 149 children was enrolled, 76 were randomized to receive scheduled mDLI and 73 received standard care. Conditioning was based on either 12 Gy total body irradiation or treosulfan. Rabbit antithymocyte globulin was replaced by tocilizumab and abatacept. Primary end points were the incidence of acute GVHD grades II–IV and the incidence of cytomegalovirus (CMV) viremia. The incidence of grades II–IV aGVHD was 14% in the experimental arm and 12% in the control arm, p—0.8. The incidence of CMV viremia was 45% in the experimental arm and 55% in the control arm, p—0.4. Overall, in the total cohort 2-year NRM was 2%, cumulative incidence of relapse was 25%, event-free survival 71%, and overall survival 80%, without difference between the study arms. Memory DLI was associated with improved recovery of CMV-specific T-cell responses in a subcohort of CMV IgG seropositive recipients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Maschan M, Shelikhova L, Ilushina M, Kurnikova E, Boyakova E, Balashov D, et al. TCR-alpha/beta and CD19 depletion and treosulfan-based conditioning regimen in unrelated and haploidentical transplantation in children with acute myeloid leukemia. Bone Marrow Transplant. 2016;51:668–74. https://doi.org/10.1038/bmt.2015.343.
Balashov D, Shcherbina A, Maschan M, Trakhtman P, Skvortsova Y, Shelikhova L, et al. Single-center experience of unrelated and haploidentical stem cell transplantation with TCRalphabeta and CD19 depletion in children with primary immunodeficiency syndromes. Biol Blood Marrow Transplant. 2015;21:1955–62. https://doi.org/10.1016/j.bbmt.2015.07.008.
Bertaina A, Merli P, Rutella S, Pagliara D, Bernardo ME, Masetti R, et al. HLA-haploidentical stem cell transplantation after removal of alphabeta+ T and B cells in children with nonmalignant disorders. Blood. 2014;124:822–6. https://doi.org/10.1182/blood-2014-03-563817.
Locatelli F, Merli P, Pagliara D, Li Pira G, Falco M, Pende D, et al. Outcome of children with acute leukemia given HLA-haploidentical HSCT after alphabeta T-cell and B-cell depletion. Blood. 2017;130:677–85. https://doi.org/10.1182/blood-2017-04-779769.
Jacoby E, Varda-Bloom N, Goldstein G, Hutt D, Churi C, Vernitsky H, et al. Comparison of two cytoreductive regimens for alphabeta-T-cell-depleted haploidentical HSCT in pediatric malignancies: improved engraftment and outcome with TBI-based regimen. Pediatr Blood Cancer. 2018;65. https://doi.org/10.1002/pbc.26839.
Lang P, Feuchtinger T, Teltschik HM, Schwinger W, Schlegel P, Pfeiffer M, et al. Improved immune recovery after transplantation of TCRalphabeta/CD19-depleted allografts from haploidentical donors in pediatric patients. Bone Marrow Transplant. 2015;50 Suppl 2:S6–10. https://doi.org/10.1038/bmt.2015.87.
Handgretinger R. New approaches to graft engineering for haploidentical bone marrow transplantation. Semin Oncol. 2012;39:664–73. https://doi.org/10.1053/j.seminoncol.2012.09.007.
Im HJ, Koh KN, Suh JK, Lee SW, Choi ES, Jang S, et al. Haploidentical HCT using an alphabeta T-cell-depleted graft with targeted alphabeta(+) cells by add-back after a reduced intensity preparative regimen containing low-dose TBI. Bone Marrow Transplant. 2016;51:1217–22. https://doi.org/10.1038/bmt.2016.114.
Zvyagin IV, Mamedov IZ, Tatarinova OV, Komech EA, Kurnikova EE, Boyakova EV, et al. Tracking T-cell immune reconstitution after TCRalphabeta/CD19-depleted hematopoietic cells transplantation in children. Leukemia. 2017;31:1145–53. https://doi.org/10.1038/leu.2016.321.
Maschan M, Blagov S, Shelikhova L, Shekhovtsova Z, Balashov D, Starichkova J, et al. Low-dose donor memory T-cell infusion after TCR alpha/beta depleted unrelated and haploidentical transplantation: results of a pilot trial. Bone Marrow Transpl. 2018;53:264–73. https://doi.org/10.1038/s41409-017-0035-y.
Blagov S, Zvyagin IV, Shelikhova L, Khismatullina R, Balashov D, Komech E, et al. T-cell tracking, safety, and effect of low-dose donor memory T-cell infusions after alphabeta T cell-depleted hematopoietic stem cell transplantation. Bone Marrow Transplant. 2020; https://doi.org/10.1038/s41409-020-01128-2.
Triplett BM, Muller B, Kang G, Li Y, Cross SJ, Moen J, et al. Selective T-cell depletion targeting CD45RA reduces viremia and enhances early T-cell recovery compared with CD3-targeted T-cell depletion. Transplant Infect Dis. 2018;20. https://doi.org/10.1111/tid.12823.
Laberko A, Bogoyavlenskaya A, Shelikhova L, Shekhovtsova Z, Balashov D, Voronin K, et al. Risk factors for and the clinical impact of cytomegalovirus and Epstein-Barr virus infections in pediatric recipients of TCR-alpha/beta- and CD19-depleted grafts. Biol Blood Marrow Transplant. 2017;23:483–90. https://doi.org/10.1016/j.bbmt.2016.12.635.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant. 2005;11:945–56. https://doi.org/10.1016/j.bbmt.2005.09.004.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–8.
Kennedy GA, Varelias A, Vuckovic S, Le Texier L, Gartlan KH, Zhang P, et al. Addition of interleukin-6 inhibition with tocilizumab to standard graft-versus-host disease prophylaxis after allogeneic stem-cell transplantation: a phase 1/2 trial. Lancet Oncol. 2014;15:1451–9. https://doi.org/10.1016/S1470-2045(14)71017-4.
Jaiswal SR, Bhakuni P, Zaman S, Bansal S, Bharadwaj P, Bhargava S, et al. T cell costimulation blockade promotes transplantation tolerance in combination with sirolimus and post-transplantation cyclophosphamide for haploidentical transplantation in children with severe aplastic anemia. Transplant Immunol. 2017;43–44:54–9. https://doi.org/10.1016/j.trim.2017.07.004.
Shelikhova L, Ilushina M, Shekhovtsova Z, Shasheleva D, Khismatullina R, Kurnikova E, et al. Alphabeta T cell-depleted haploidentical hematopoietic stem cell transplantation without antithymocyte globulin in children with chemorefractory acute myelogenous leukemia. Biol Blood Marrow Transplant. 2019;25:e179–82. https://doi.org/10.1016/j.bbmt.2019.01.023.
Shelikhova L, Glushkova S, Nikolaev R, Dunaikina M, Shekhovtsova Z, Blagov S, et al. Serotherapy-free regimen improves serotherapy-free regimen improves non-relapse mortality and immune recovery among the recipients of αβ T-cell-depleted haploidentical grafts: retrospective study in childhood leukemia. J Transplant Cell Ther. 2021. https://doi.org/10.1016/j.jtct.2021.01.010.
Arcese W, De Angelis G, Cerretti R. Granulocyte-mobilized bone marrow. Curr Opin Hematol. 2012;19:448–53. https://doi.org/10.1097/MOH.0b013e32835903ab.
Tumino N, Besi F, Di Pace AL, Mariotti FR, Merli P, Li Pira G, et al. PMN-MDSC are a new target to rescue graft-versus-leukemia activity of NK cells in haplo-HSC transplantation. Leukemia. 2020;34:932–7. https://doi.org/10.1038/s41375-019-0585-7.
Acknowledgements
The authors would wish to thank “Podari zhizn” charitable foundation and “Science for children” endowment foundation for constant support of research in the field of hematopoietic stem cell transplantation and clinical care of children in the Department of Hematopoietic Stem Cell Transplantation of the Dmitriy Rogachev National Medical Research Center of Pediatric Hematology, Oncology and Immunology.
Funding
This work was partially supported by Ministry of Health of Russia via approbation protocols 2015-25-4 and 2018-45-7. MM, AM, and EO were partly supported by the grant of the Ministry of Science and Higher Education of the Russian Federation No. 075-15-2020-807.
Author information
Authors and Affiliations
Contributions
MM, LS, MD, ZZ, SG, and RN contributed equally to this work. MM designed the study, analyzed data, and wrote the paper. LS and ZZ designed the study, collected and analyzed data, and wrote the paper. MD, DP, SG, RN, SB, and RK collected and analyzed data and wrote the paper. DB, EK, YM, AK, and EO collected data and reviewed the paper. NM, DL, GN, and AM contributed to study design and reviewed the paper.
Corresponding author
Ethics declarations
Conflict of interest
MM received lecturer’s fee from Miltenyi Biotec, all other authors declare no competing interests in relation to the current paper.
Ethical approval
The trial was approved by the local ethics committee and IRB of the Dmitriy Rogachev National Medical Center of Pediatric Hematology, Oncology and Immunology and registered as NCT02942173 at clinicaltrials.gov.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Dunaikina, M., Zhekhovtsova, Z., Shelikhova, L. et al. Safety and efficacy of the low-dose memory (CD45RA-depleted) donor lymphocyte infusion in recipients of αβ T cell-depleted haploidentical grafts: results of a prospective randomized trial in high-risk childhood leukemia. Bone Marrow Transplant 56, 1614–1624 (2021). https://doi.org/10.1038/s41409-021-01232-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01232-x
This article is cited by
-
Ex-Vivo TCR αβ and CD19 Depleted Haploidentical Stem Cell Transplantation with CD45RO Memory T Cell Addback for CARD11 Deficiency
Indian Journal of Hematology and Blood Transfusion (2024)
-
Automatic generation of alloreactivity-reduced donor lymphocytes and hematopoietic stem cells from the same mobilized apheresis product
Journal of Translational Medicine (2023)