Abstract
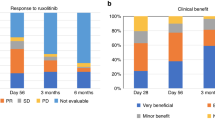
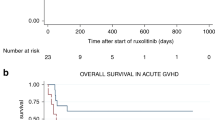
Acute and chronic steroid-refractory graft-versus-host disease (srGVHD) is a life-threatening complication of allogeneic stem cell transplantation. There are a number of reports on case series describing efficacy of ruxolitinib in both acute and chronic srGVHD. We conducted a prospective study (NCT02997280) in 75 patients with srGVHD (32 acute, 43 chronic, 41 adults, and 34 children). Patients with chronic GVHD had severe disease in 83% of cases, and acute GVHD patients had grade III–IV disease in 66% of cases. The overall response rate (ORR) was 75% (95% CI 57–89%) in acute GVHD and 81% (95% CI 67–92%) in chronic. Overall survival was 59% (95% CI 49–74%) in acute group and 85% (95% CI 70–93%). The major risk factors for lower survival were grade III–IV gastrointestinal involvement (29% vs 93%, p = 0.0001) in acute form and high disease risk score in chronic (65% vs 90%, p = 0.038). Toxicity was predominantly hematologic with 79% and 44% of grade III–IV neutropenia in acute and chronic groups, respectively. There was no difference between adults and children in terms of ORR (p = 0.31, p = 0.35), survival (p = 0.44, p = 0.12) and toxicity (p > 0.93). The study demonstrated that ruxolitinib is an effective option in acute and chronic srGVHD and can be used both in adults and children.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Greinix HT, Eikema DJ, Koster L, Penack O, Yakoub-Agha I, Montoto S, et al. Incidence of acute graft-versus-host disease and survival after allogeneic hematopoietic cell transplantation over time: a study from the Transplant Complications and Chronic Malignancies Working Party of the EBMT. Blood. 2018;132(Suppl 1):2120. https://doi.org/10.1182/blood-2018-99-111764.
Martin PJ, Rizzo JD, Wingard JR, Ballen K, Curtin PT, Cutler C, et al. First- and second-line systemic treatment of acute graft-versus-host disease: recommendations of the American Society of Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2012;18:1150–63.
Sarantopoulos S, Cardones AR, Sullivan KM. How I treat refractory chronic graft-versus-host disease. Blood. 2019;133:1191–1200.
Miklos D, Cutler CS, Arora M, Waller EK, Jagasia M, Pusic I, et al. Ibrutinib for chronic graft-versus-host disease after failure of prior therapy. Blood. 2017;130:2243–50.
Jagasia M, Zeiser R, Arbushites M, Delaite P, Gadbaw B, Bubnoff NV. Ruxolitinib for the treatment of patients with steroid-refractory GVHD: an introduction to the REACH trials. Immunotherapy. 2018;10:391–402.
Verstovsek S, Mesa RA, Gotlib J, Gupta V, DiPersio JF, Catalano JV, et al. COMFORT-I investigators. Long-term treatment with ruxolitinib for patients with myelofibrosis: 5-year update from the randomized, double-blind, placebo-controlled, phase 3 COMFORT-I trial. J Hematol Oncol. 2017;10:55.
Heine A, Held SA, Daecke SN, Wallner S, Yajnanarayana SP, Kurts C, et al. The JAK-inhibitor ruxolitinib impairs dendritic cell function in vitro and in vivo. Blood. 2013;122:1192–202.
Spoerl S, Mathew NR, Bscheider M, Schmitt-Graeff A, Chen S, Mueller T, et al. Activity of therapeutic JAK 1/2 blockade in graft-versus-host disease. Blood. 2014;123:3832–42.
Zeiser R, Burchert A, Lengerke C, et al. Ruxolitinib in corticosteroid-refractory graft-versus-host disease after allogeneic stem cell transplantation: a multicenter survey. Leukemia. 2015;29:2062–8.
Ruutu T, Gratwohl A, de Witte T, Afanasyev B, Apperley J, Bacigalupo A, et al. Prophylaxis and treatment of GVHD: EBMT-ELN working group recommendations for a standardized practice. Bone Marrow Transplant. 2014;49:168–73.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–8.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant. 2005;11:945–56.
Martin PJ, Bachier CR, Klingemann HG, McCarthy PL, Szabolcs P, Uberti JP, et al. Endpoints for clinical trials testing treatment of acute graft-versus-host disease: a joint statement. Biol Blood Marrow Transplant. 2009;15:777–84.
Lee SJ, Wolff D, Kitko C, Koreth J, Inamoto Y, Jagasia M, et al. Measuring therapeutic response in chronic graft-versus-host disease. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: IV. The 2014 Response Criteria Working Group report. Biol Blood Marrow Transplant. 2015;21:984–99.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71.
Veeraraghavan S, Thappali S, Viswanadha S, Chennupati S, Nalla S, Golla M, et al. Simultaneous quantification of ruxolitinib and nilotinib in rat plasma by LC-MS/MS: application to a pharmacokinetic study. J Pharm Biomed Anal. 2014;94:125–31.
Escamilla Gómez V, García-Gutiérrez V, López Corral L, García Cadenas I, Pérez Martínez A, Márquez Malaver FJ, et al. Ruxolitinib in refractory acute and chronic graft-versus-host disease: a multicenter survey study. Bone Marrow Transplant. 2019. https://doi.org/10.1038/s41409-019-0731-x.
Steinbach G, Hockenbery DM, Huls G, Furlong T, Myerson D, Loeb KR, et al. Pilot study of lithium to restore intestinal barrier function in severe graft-versus-host disease. PLoS ONE. 2017;12:e0183284.
Sadowska-Klasa A, Piekarska A, Prejzner W, Sadowska-Klasa A, Piekarska A, Prejzner W, et al. Colonization with multidrug-resistant bacteria increases the risk of complications and a fatal outcome after allogeneic hematopoietic cell transplantation. Ann Hematol. 2018;97:509–17.
Goloshchapov OV, Kucher MA, Moiseev IS, Afanasyev BV. Fecal microbiota transplantation in patients after allogeneichematopoietic stem cell transplantation. Cell Ther Transplant. 2017;6:20–29.
Ferrara JLM, Chaudhry MS. GVHD: biology matters. Blood Adv. 2018;2:3411–7.
Markey KA, MacDonald KP, Hill GR. The biology of graft-versus-host disease: experimental systems instructing clinical practice. Blood. 2014;124:354–62.
Leveque-El Mouttie L, Koyama M, Le Texier L, Markey KA, Cheong M, Kuns RD, et al. Corruption of dendritic cell antigen presentation during acute GVHD leads to regulatory T-cell failure and chronic GVHD. Blood. 2016;128:794–804.
Dertschnig S, Hauri-Hohl MM, Vollmer M, Holländer GA, Krenger W. Impaired thymic expression of tissue-restricted antigens licenses the de novo generation of autoreactive CD4+ T cells in acute GVHD. Blood. 2015;125:2720–3.
Allen JL, Fore MS, Wooten J, Roehrs PA, Bhuiya NS, Hoffert T, et al. B cells from patients with chronic GVHD are activated and primed for survival via BAFF-mediated pathways. Blood. 2012;120:2529–36.
MacMillan ML, Robin M, Harris AC, DeFor TE, Martin PJ, Alousi A, et al. A refined risk score for acute graft-versus-host disease that predicts response to initial therapy, survival, and transplant-related mortality. Biol Blood Marrow Transplant. 2015;21:761–7.
Wysham NG, Sullivan DR, Allada G. An opportunistic infection associated with ruxolitinib, a novel janus kinase 1,2 inhibitor. Chest. 2013;143:1478–9.
Afram G, Watz E, Remberger M, Nygell UA, Sundin M, Hägglund H, et al. Higher response rates in patients with severe chronic skin graft-versus-host disease treated with extracorporeal photopheresis. Cent Eur J Immunol. 2019;44:84–91.
Greinix HT, van Besien K, Elmaagacli AH, Hillen U, Grigg A, Knobler R, et al. UVADEX Chronic GVHD Study Group. Progressive improvement in cutaneous and extracutaneous chronic graft-versus-host disease after a 24-week course of extracorporeal photopheresis-results of a crossover randomized study. Biol Blood Marrow Transplant. 2011;17:1775–82.
Hamilton BK, Storer BE, Wood WA, Pidala JA, Cutler CS, Martin PJ, et al. Disability related to chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2019. pii: S1083-8791(19)30676-7. https://doi.org/10.1016/j.bbmt.2019.10.019.
Khoury HJ, Langston AA, Kota VK, Wilkinson JA, Pusic I, Jillella A, et al. Ruxolitinib: a steroid sparing agent in chronic graft-versus-host disease. Bone Marrow Transplant. 2018;53:826–31.
Holland HK, Wingard JR, Beschorner WE, Saral R, Santos GW. Bronchiolitis obliterans in bone marrow transplantation and its relationship to chronic graft-v-host disease and low serum IgG. Blood. 1988;72:621–7.
Koc S, Leisenring W, Flowers ME, Anasetti C, Deeg HJ, Nash RA. Therapy for chronic graft-versus-host disease: a randomized trial comparing cyclosporine plus prednisone versus prednisone alone. Blood. 2002;100:48–51.
Ayuk F, Veit R, Zabelina T, Bussmann L, Christopeit M, Alchalby H. Prognostic factors for survival of patients with newly diagnosed chronic GVHD according to NIH criteria. Ann Hematol. 2015;94:1727–32.
González Vicent M, Molina B, González de Pablo J, González de Pablo J, Castillo A, Díaz MÁ. Ruxolitinib treatment for steroid refractory acute and chronic graft vs host disease in children: Clinical and immunological results. Am J Hematol. 2019;94:319–26.
Khandelwal P, Teusink-Cross A, Davies SM, Nelson AS, Dandoy CE, El-Bietar J, et al. Ruxolitinib as salvage therapy in steroid-refractory acute graft-versus-host disease in pediatric hematopoietic stem cell transplant patients. Biol Blood Marrow Transplant. 2017;23:1122–7.
Dominietto A, Raiola AM, van Lint MT, Lamparelli T, Gualandi F, Berisso G, et al. Factors influencing haematological recovery after allogeneic haemopoietic stem cell transplants: graft-versus-host disease, donor type, cytomegalovirus infections and cell dose. Br J Haematol. 2001;112:219–27.
Acknowledgements
The study was supported clinical approbation program No.2016–29–1 by the Ministry of Health of Russian Federation. The laboratory assays were performed with the support of Russian Science Fund, grant No 17–75–20145. We thank Valerii Beklenischev for managing the frozen samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
ISM received consultancy and lecturer honoraria from Novartis. EVM received lecturer honoraria from Novartis. The other authors have no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Moiseev, I.S., Morozova, E.V., Bykova, T.A. et al. Long-term outcomes of ruxolitinib therapy in steroid-refractory graft-versus-host disease in children and adults. Bone Marrow Transplant 55, 1379–1387 (2020). https://doi.org/10.1038/s41409-020-0834-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-020-0834-4
This article is cited by
-
Ruxolitinib for the treatment of acute and chronic graft-versus-host disease in children: a systematic review and individual patient data meta-analysis
Bone Marrow Transplantation (2024)
-
Extracorporeal photopheresis in acute and chronic steroid‑refractory graft-versus-host disease: an evolving treatment landscape
Leukemia (2022)
-
Ruxolitinib in the management of steroid-resistant/-dependent acute and chronic graft-versus-host disease: results of routine practice in an academic centre
Annals of Hematology (2022)
-
Effect of ruxolitinib on the oral mucosa of patients with steroid-refractory chronic Graft-versus-Host disease and oral involvement
Clinical Oral Investigations (2022)
-
The JAK2 inhibitor TG101209 exhibits anti-tumor and chemotherapeutic sensitizing effects on Burkitt lymphoma cells by inhibiting the JAK2/STAT3/c-MYB signaling axis
Cell Death Discovery (2021)