Abstract
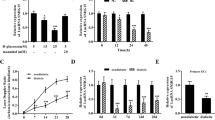
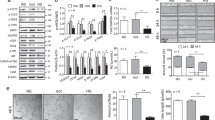
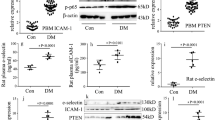
Endothelial dysfunction is a common complication of diabetes mellitus (DM) and contributes to the high incidence and mortality of cardiovascular and cerebrovascular diseases. Aberrant epigenetic regulation under diabetic conditions, including histone modifications, DNA methylation, and non-coding RNAs (ncRNAs) play key roles in the initiation and progression of diabetic vascular complications. ASH2L, a H3K4me3 regulator, triggers genetic transcription, which is critical for physiological and pathogenic processes. In this study we investigated the role of ASH2L in mediating diabetic endothelial dysfunction. We showed that ASH2L expression was significantly elevated in vascular tissues from diabetic db/db mice and in rat aortic endothelial cells (RAECs) treated with high glucose medium (11 and 22 mM). Knockdown of ASH2L in RAECs markedly inhibited the deteriorating effects of high glucose, characterized by reduced oxidative stress and inflammatory responses. Deletion of endothelial ASH2L in db/db mice by injection of an adeno-associated virus (AAV)-endothelial specific system carrying shRNA against Ash2l (AAV-shAsh2l) restored the impaired endothelium-dependent relaxations, and ameliorated DM-induced vascular dysfunction. We revealed that ASH2L expression activated reductase STEAP4 transcription in vitro and in vivo, which consequently elevated Cu(I) transportation into ECs by the copper transporter CTR1. Excess copper produced by STEAP4-mediated copper uptake triggered oxidative stress and inflammatory responses, resulting in endothelial dysfunction. Our results demonstrate that hyperglycemia triggered ASH2L-STEAP4 axis contributes to diabetic endothelial dysfunction by modulating copper uptake into ECs and highlight the therapeutic potential of blocking the endothelial ASH2L in the pathogenesis of diabetic vascular complications.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Kaur R, Kaur M, Singh J. Endothelial dysfunction and platelet hyperactivity in type 2 diabetes mellitus: molecular insights and therapeutic strategies. Cardiovasc Diabetol. 2018;17:121. https://doi.org/10.1186/s12933-018-0763-3.
Zhang T, Li H, Ouyang C, Cao G, Gao J, Wu J, et al. Liver kinase B1 inhibits smooth muscle calcification via high mobility group box 1. Redox Biol. 2021;38:101828. https://doi.org/10.1016/j.redox.2020.101828.
Niu C, Chen Z, Kim KT, Sun J, Xue M, Chen G, et al. Metformin alleviates hyperglycemia-induced endothelial impairment by downregulating autophagy via the Hedgehog pathway. Autophagy. 2019;15:843–70. https://doi.org/10.1080/15548627.2019.1569913.
Xu S, Ilyas I, Little PJ, Li H, Kamato D, Zheng X, et al. Endothelial dysfunction in atherosclerotic cardiovascular diseases and beyond: from mechanism to pharmacotherapies. Pharmacol Rev. 2021;73:924–67. https://doi.org/10.1124/pharmrev.120.000096.
Zhang Y, Liu J, Tian XY, Wong WT, Chen Y, Wang L, et al. Inhibition of bone morphogenic protein 4 restores endothelial function in db/db diabetic mice. Arterioscler Thromb Vasc Biol. 2014;34:152–9. https://doi.org/10.1161/ATVBAHA.113.302696.
Tang R, Xu J, Zhang B, Liu J, Liang C, Hua J, et al. Ferroptosis, necroptosis, and pyroptosis in anticancer immunity. J Hematol Oncol. 2020;13:110. https://doi.org/10.1186/s13045-020-00946-7.
Feng X, Yang W, Huang L, Cheng H, Ge X, Zan G, et al. Causal effect of genetically determined blood copper concentrations on multiple diseases: a Mendelian randomization and phenome-wide association study. Phenomics. 2022;2:242–53. https://doi.org/10.1007/s43657-022-00052-3.
Kang YJ. Copper and homocysteine in cardiovascular diseases. Pharmacol Ther. 2011;129:321–31. https://doi.org/10.1016/j.pharmthera.2010.11.004.
Yin R, Wang H, Li C, Wang L, Lai S, Yang X, et al. Induction of apoptosis and autosis in cardiomyocytes by the combination of homocysteine and copper via NOX-mediated p62 expression. Cell Death Discov. 2022;8:75. https://doi.org/10.1038/s41420-022-00870-4.
Nunes KZ, Fioresi M, Marques VB, Vassallo DV. Acute copper overload induces vascular dysfunction in aortic rings due to endothelial oxidative stress and increased nitric oxide production. J Toxicol Environ Health A. 2018;81:218–28. https://doi.org/10.1080/15287394.2018.1437490.
Zhang H, Yan C, Yang Z, Zhang W, Niu Y, Li X, et al. Alterations of serum trace elements in patients with type 2 diabetes. J Trace Elem Med Biol. 2017;40:91–6. https://doi.org/10.1016/j.jtemb.2016.12.017.
Li P, Yin J, Zhu Y, Li S, Chen S, Sun T, et al. Association between plasma concentration of copper and gestational diabetes mellitus. Clin Nutr. 2019;38:2922–7. https://doi.org/10.1016/j.clnu.2018.12.032.
Yang F, Pei R, Zhang Z, Liao J, Yu W, Qiao N, et al. Copper induces oxidative stress and apoptosis through mitochondria-mediated pathway in chicken hepatocytes. Toxicol Vitr. 2019;54:310–6. https://doi.org/10.1016/j.tiv.2018.10.017.
Jiang C, Wu B, Xue M, Lin J, Hu Z, Nie X, Cai G. Inflammation accelerates copper-mediated cytotoxicity through induction of six-transmembrane epithelial antigens of prostate 4 expression. Immunol Cell Biol. 2021;99:392–402. https://doi.org/10.1111/imcb.12427.
Scarl RT, Lawrence CM, Gordon HM, Nunemaker CS. STEAP4: its emerging role in metabolism and homeostasis of cellular iron and copper. J Endocrinol. 2017;234:R123–R134. https://doi.org/10.1530/JOE-16-0594.
Liao Y, Zhao J, Bulek K, Tang F, Chen X, Cai G, et al. Inflammation mobilizes copper metabolism to promote colon tumorigenesis via an IL-17-STEAP4-XIAP axis. Nat Commun. 2020;11:900. https://doi.org/10.1038/s41467-020-14698-y.
Tsai P-H, Chien Y, Wang M-L, Hsu C-H, Laurent B, Chou S-J, et al. Ash2l interacts with Oct4-stemness circuitry to promote super-enhancer-driven pluripotency network. Nucleic Acids Res. 2019;47:10115–33. https://doi.org/10.1093/nar/gkz801.
Takahashi Y, Westfield GH, Oleskie AN, Trievel RC, Shilatifard A, Skiniotis G. Structural analysis of the core COMPASS family of histone H3K4 methylases from yeast to human. Proc Natl Acad Sci USA. 2011;108:20526–31. https://doi.org/10.1073/pnas.1109360108.
Steward MM, Lee J-S, O’Donovan A, Wyatt M, Bernstein BE, Shilatifard A. Molecular regulation of H3K4 trimethylation by ASH2L, a shared subunit of MLL complexes. Nat Struct Mol Biol. 2006;13:852–4. https://doi.org/10.1038/nsmb1131.
Wu Y-J, Ko B-S, Liang S-M, Lu Y-J, Jan Y-J, Jiang S-S, et al. ZNF479 downregulates metallothionein-1 expression by regulating ASH2L and DNMT1 in hepatocellular carcinoma. Cell Death Dis. 2019;10:408. https://doi.org/10.1038/s41419-019-1651-9.
Zeng K, Wu Y, Wang C, Wang S, Sun H, Zou R, et al. ASH2L is involved in promotion of endometrial cancer progression via upregulation of PAX2 transcription. Cancer Sci. 2020;111:2062–77. https://doi.org/10.1111/cas.14413.
Han P, Gao D, Zhang W, Liu S, Yang S, Li X. Puerarin suppresses high glucose-induced MCP-1 expression via modulating histone methylation in cultured endothelial cells. Life Sci. 2015;130:103–7. https://doi.org/10.1016/j.lfs.2015.02.022.
Yang D, Xiao C, Long F, Wu W, Huang M, Qu L, et al. Fra-1 plays a critical role in angiotensin II-induced vascular senescence. FASEB J. 2019;33:7603–14. https://doi.org/10.1096/fj.201801671RRRR.
Wu W, Wang J, Xiao C, Su Z, Su H, Zhong W, et al. SMYD2-mediated TRAF2 methylation promotes the NF-κB signaling pathways in inflammatory diseases. Clin Transl Med. 2021;11:e591. https://doi.org/10.1002/ctm2.591.
Wang Y, Gao L, Li Z, Ma X. MicroRNA-301a-3p promotes diabetic retinopathy via regulation of six-transmembrane epithelial antigen of prostate 4. Inflamm Res. 2021;70:445–57. https://doi.org/10.1007/s00011-020-01431-0.
Chen X, Huang Z, Zhou B, Wang H, Jia G, Liu G, Zhao H. STEAP4 and insulin resistance. Endocrine. 2014;47:372–9. https://doi.org/10.1007/s12020-014-0230-1.
Sharp PA. Ctr1 and its role in body copper homeostasis. Int J Biochem Cell Biol. 2003;35:288–91. https://doi.org/10.1016/s1357-2725(02)00134-6.
Li H, Xia N, Hasselwander S, Daiber A. Resveratrol and vascular function. Int J Mol Sci. 2019. https://doi.org/10.3390/ijms20092155.
Lee Y-T, Ayoub A, Park S-H, Sha L, Xu J, Mao F, et al. Mechanism for DPY30 and ASH2L intrinsically disordered regions to modulate the MLL/SET1 activity on chromatin. Nat Commun. 2021;12:2953. https://doi.org/10.1038/s41467-021-23268-9.
Qi J, Huo L, Zhu YT, Zhu Y-J. Absent, small or homeotic 2-like protein (ASH2L) enhances the transcription of the estrogen receptor α gene through GATA-binding protein 3 (GATA3). J Biol Chem. 2014;289:31373–81. https://doi.org/10.1074/jbc.M114.579839.
Takizawa F, Mizutani S, Ogawa Y, Sawada N. Glucose-independent persistence of PAI-1 gene expression and H3K4 tri-methylation in type 1 diabetic mouse endothelium: implication in metabolic memory. Biochem Biophys Res Commun. 2013;433:66–72. https://doi.org/10.1016/j.bbrc.2013.02.064.
Oosterheert W, van Bezouwen LS, Rodenburg RNP, Granneman J, Förster F, Mattevi A, Gros P. Cryo-EM structures of human STEAP4 reveal mechanism of iron(III) reduction. Nat Commun. 2018;9:4337. https://doi.org/10.1038/s41467-018-06817-7.
Chuang C-T, Guh J-Y, Lu C-Y, Wang Y-T, Chen H-C, Chuang L-Y. Steap4 attenuates high glucose and S100B-induced effects in mesangial cells. J Cell Mol Med. 2015;19:1234–44. https://doi.org/10.1111/jcmm.12472.
Kan H, Zhang K, Mao A, Geng L, Gao M, Feng L, et al. Single-cell transcriptome analysis reveals cellular heterogeneity in the ascending aortas of normal and high-fat diet-fed mice. Exp Mol Med. 2021;53:1379–89. https://doi.org/10.1038/s12276-021-00671-2.
Tsvetkov P, Coy S, Petrova B, Dreishpoon M, Verma A, Abdusamad M, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375:1254–61. https://doi.org/10.1126/science.abf0529.
Shimada K, Reznik E, Stokes ME, Krishnamoorthy L, Bos PH, Song Y, et al. Copper-binding small molecule induces oxidative stress and cell-cycle arrest in glioblastoma-patient-derived cells. Cell Chem Biol. 2018;25:585–94.e7. https://doi.org/10.1016/j.chembiol.2018.02.010.
Zeng C, Duan F, Hu J, Luo B, Huang B, Lou X, et al. NLRP3 inflammasome-mediated pyroptosis contributes to the pathogenesis of non-ischemic dilated cardiomyopathy. Redox Biol. 2020;34:101523. https://doi.org/10.1016/j.redox.2020.101523.
Członkowska A, Litwin T, Dusek P, Ferenci P, Lutsenko S, Medici V, et al. Wilson disease. Nat Rev Dis Prim. 2018;4:21. https://doi.org/10.1038/s41572-018-0018-3.
Acknowledgements
This work was supported by Shanghai Municipal Science and Technology Major Project (Grant No. 2017SHZDZX01). We would like to thank Prof. Hui-ru Tang (Human Phenome Institute, Fudan University) for help with the ICP-MS experiments.
Author information
Authors and Affiliations
Contributions
WZ designed the study, performed initial experiments, analyzed data, and wrote the manuscript. YJD performed experiments and helped edit the manuscript. CH, YHL, and CXX performed some experiments. XHL and JC conceived, designed, and supervised all experiments, and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhong, W., Dong, Yj., Hong, C. et al. ASH2L upregulation contributes to diabetic endothelial dysfunction in mice through STEAP4-mediated copper uptake. Acta Pharmacol Sin 45, 558–569 (2024). https://doi.org/10.1038/s41401-023-01174-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-023-01174-8