Abstract
Observational studies suggest that posttraumatic stress disorder (PTSD) increases risk for various autoimmune diseases. Insights into shared biology and causal relationships between these diseases may inform intervention approaches to PTSD and co-morbid autoimmune conditions. We investigated the shared genetic contributions and causal relationships between PTSD, 18 autoimmune diseases, and 3 immune/inflammatory biomarkers. Univariate MiXeR was used to contrast the genetic architectures of phenotypes. Genetic correlations were estimated using linkage disequilibrium score regression. Bi-directional, two-sample Mendelian randomization (MR) was performed using independent, genome-wide significant single nucleotide polymorphisms; inverse variance weighted and weighted median MR estimates were evaluated. Sensitivity analyses for uncorrelated (MR PRESSO) and correlated horizontal pleiotropy (CAUSE) were also performed. PTSD was considerably more polygenic (10,863 influential variants) than autoimmune diseases (median 255 influential variants). However, PTSD evidenced significant genetic correlation with nine autoimmune diseases and three inflammatory biomarkers. PTSD had putative causal effects on autoimmune thyroid disease (p = 0.00009) and C-reactive protein (CRP) (p = 4.3 × 10−7). Inferences were not substantially altered by sensitivity analyses. Additionally, the PTSD-autoimmune thyroid disease association remained significant in multivariable MR analysis adjusted for genetically predicted inflammatory biomarkers as potential mechanistic pathway variables. No autoimmune disease had a significant causal effect on PTSD (all p values > 0.05). Although causal effect models were supported for associations of PTSD with CRP, shared pleiotropy was adequate to explain a putative causal effect of CRP on PTSD (p = 0.18). In summary, our results suggest a significant genetic overlap between PTSD, autoimmune diseases, and biomarkers of inflammation. PTSD has a putative causal effect on autoimmune thyroid disease, consistent with existing epidemiologic evidence. A previously reported causal effect of CRP on PTSD is potentially confounded by shared genetics. Together, results highlight the nuanced links between PTSD, autoimmune disorders, and associated inflammatory signatures, and suggest the importance of targeting related pathways to protect against disease and disability.
Similar content being viewed by others
Introduction
Posttraumatic stress disorder (PTSD), characterized by intrusive memories of traumatic experiences, avoidance of trauma-related stimuli, negative changes in thinking and mood, and high levels of arousal, is a debilitating psychiatric illness that can develop in response to exposure to trauma [1]. The burden of PTSD on individuals and society is extensive [2], with numerous emotional, interpersonal, and socioeconomic consequences [3,4,5]. Moreover, PTSD is also associated with numerous physical health outcomes [6], notably including a variety of health conditions and diseases related to immune dysregulation and inflammation [7, 8]. Longitudinal research has demonstrated PTSD to be associated with a heightened incidence of autoimmune diseases, including diseases of the endocrine, skin, nervous, digestive systems, connective tissues, inflammatory arthritis, and vasculitis [7,8,9,10,11,12,13,14]. Shared biological mechanisms related to immune dysregulation are thought to be a primary factor linking PTSD and autoimmune diseases, as studies of peripheral inflammatory biomarkers, the epigenome, transcriptome, and common genetic variation all support the notion of immune dysregulation being involved in PTSD pathophysiology [15,16,17].
While longitudinal observational studies suggest that PTSD precedes the development of autoimmune diseases, causal relationships have yet to be delineated [17, 18]. Insights into whether PTSD and a variety of autoimmune diseases are causally related may enhance biological understanding of PTSD and its sequelae, as well as inform intervention approaches for PTSD and co-morbid autoimmune diseases [17, 19, 20]. In addition to directly assessing the relationship between PTSD and autoimmune diseases, it is also beneficial to evaluate the relationship between PTSD and biomarkers of inflammation, which can be the first non-specific sign of an autoimmune disorder that prompts further evaluation [21]. Moreover, as inflammation underlies PTSD and autoimmune disease, it is of interest to determine whether inflammation confounds or mediates their relationship. Therefore, multiple causal questions need to be investigated: (1) if PTSD gives rise to autoimmune diseases or vice versa; (2) if this relationship is bi-directional; (3) if shared underlying pathology is a common cause (confounder) between PTSD and autoimmune disease; (4) if inflammation mediates or confounds the relationship between PTSD and autoimmune disease.
Two-sample Mendelian randomization (MR) [22] analyses can be utilized to test these questions. MR uses genetic variants as instrumental variables (i.e., presumedly unconfounded stand-in variables) for the phenotypes of interest, leveraging the random assignment of alleles at conception to produce an analysis akin to a randomized experiment. Prior MR analyses identified putative causal associations between psychiatric disorders and autoimmune diseases, but none have been identified for PTSD thus far [23, 24]. This may be due to lack of power: the instrumental variables selected were significant SNPs identified by large-scale genome-wide association studies (GWAS) [18], yet only a few significant loci were identified by prior PTSD GWAS [25]. However, with respect to the context of PTSD and inflammation, an MR study of PTSD and the inflammatory biomarker C-reactive protein (CRP) found significant evidence of a bidirectional causal association [26].
In this study, we expanded upon prior efforts by comprehensively investigating the shared genetic contributions and causal relationships between PTSD and a range of autoimmune diseases using well-powered GWAS summary statistics, including from a substantially larger PTSD GWAS [27] than previously utilized [25]. We contrasted the genetic architectures of PTSD, 18 autoimmune diseases, and 3 non-specific immune/inflammatory biomarkers, and investigated their genetic overlap. We used bidirectional two-sample MR to test causal hypotheses for PTSD, autoimmune disease, and immune/inflammatory biomarkers. We also conducted several sensitivity analyses to evaluate the robustness of our findings given the potential confounding effects of horizontal pleiotropy and reverse causation. Finally, for autoimmune diseases with significant MR findings, we applied multivariable MR analysis that adjusted for immune/inflammatory biomarkers. Under the assumption that inflammatory biomarkers serve as measures of systemic inflammation [28], the difference between multivariable and standard MR estimates would indicate the contribution of a general inflammatory signature (either as a mediator or confounder) to the relationship between PTSD and the autoimmune disease.
Methods
GWAS summary data collection and curation
We searched for GWAS of all phenotypes that were featured in a pair of recent, comprehensive studies of the relationship between PTSD and autoimmune diseases [8] and related immune/inflammatory biomarkers [29]. In total, 46 different phenotypes were considered (40 autoimmune diseases and 6 biomarkers) for investigation (Supplementary Table 1). From March 14–21, 2023, we searched the NHGRI-EBI GWAS Catalog [30] and Google Scholar for GWAS of these phenotypes. Search criteria included [phenotype name] + GWAS. GWAS had been performed in 41 of the 46 phenotypes, making for 51 results retrieved (sometimes multiple GWAS had been performed for a given phenotype). When multiple GWAS were available for a given phenotype, we selected for investigation the GWAS with the largest number of case samples; it was generally that these GWAS were more recent and had included all samples from smaller previously reported GWAS of the same phenotype. Single nucleotide polymorphism (SNP)-level summary statistics were obtained from online publicly available data repositories or, if not available online, by request from study authors. Study authors were contacted for participation. If not provided a response within 1 month, authors were contacted a second time. Phenotypes were excluded if authors did not respond. Upon acquisition of GWAS summary data, to be included in our analysis, the data needed to contain rsID, the allele coded as the risk allele and the allele coded as the non-risk allele, effect sizes, corresponding standard errors, and p values. For statistical power reasons, only GWAS with at least one significant SNP instrument were included in our analysis. To prevent confounding due to ancestral differences, we only included GWAS of individuals of European ancestry. After these criteria were applied, 21 phenotypes were deemed suitable for analyses in this investigation (Supplementary Fig. 1). A complete list of GWAS identified for usage, along with inclusion/exclusion information, are provided in Supplementary Table 1.
PGC-PTSD GWAS
The PGC-PTSD Freeze 3 European ancestry GWAS contains N = 1,222,882 participants (137,136 cases and 1,085,746 controls) from 88 studies [27]. Studies included civilian and military populations. PTSD was assessed with clinician-administered or self-report instruments or via ICD code derivation. Genotyping was conducted on Illumina or Affymetrix arrays. Standard quality control procedures were applied to genotype data. All datasets were imputed based on a population-suitable reference panel. GWAS were performed within European ancestry participants, adjusting for 5–10 principal components calculated within-sample. Sample size-weighted meta-analysis of GWAS summary data was conducted in METAL. Batches of contributing studies were compared for genetic overlap, where genetic correlation (rg) > 0.8 between all tested pairs, indicating excellent genetic overlap, despite heterogeneity in populations, genotyping, and methods of PTSD assessment. The rg between male and female subsets was 0.95, thus sex-stratified analyses were not evaluated here.
Phenotypes under investigation
After curating data, our phenotype collection included PTSD (obtained from the PGC-PTSD Freeze 3 GWAS [27]); 3 inflammatory/immune biomarkers: CRP [31], interleukin-6 (IL-6) [32], and white blood cell count (WBC) [33]; and 19 autoimmune diseases: primary adrenal insufficiency (Addison’s disease) [34], autoimmune thyroid disease [35], celiac disease [36], Crohn’s disease [37], eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome) [38], mucocutaneous lymph node syndrome (Kawasaki disease) [39], multiple sclerosis [40], myasthenia gravis [41], neuromyelitis optica spectrum disorder [42], pernicious anemia [43], primary biliary cholangitis [44], psoriasis [45], rheumatoid arthritis [46], systemic lupus erythematosus [47], systemic sclerosis [48], type 1 diabetes [49], ulcerative colitis [50], and vitiligo [51] (Supplementary Table 1).
Heritability and genetic overlap analyses
SNP based heritability (h2SNP) and rg of phenotypes were estimated using linkage disequilibrium (LD) score regression (LDSC) [52]. LD scores calculated within 1000 Genomes Phase 3 European populations [53] were used for the input. Analyses were limited to HapMap 3 SNPs. For genetic correlation analyses, the major histocompatibility complex (MHC) region was excluded (hg19 coordinates: chromosome 6: 26–34 million base pairs). For binary traits, h2SNP is reported on the liability scale adjusted to the population prevalence. h2SNP is proportional to the product of two subcomponents: the proportion of non-null SNPs (polygenicity) and variance of effect sizes of non-null SNPs (discoverability). By estimating these subcomponents for each phenotype, it is possible to identify nuances that further clarify their genetic architectures, such as whether heritability is the result of a few variants with strong effects or many variants with weak effects. We used univariate MiXeR [54] (version 1.3) to estimate the polygenicity and discoverability of all phenotypes. Using these results, the polygenic overlap between phenotypes was estimated using bivariate MiXeR [55]. We used the default settings and the supplied 1000 Genomes European ancestry LD reference panel. For interpretability, rather than directly reporting polygenicity outputs from MiXeR, we reported the number of influential variants necessary to explain 90% of h2SNP (calculated as polygenicity ✕ constant).
MR analyses
The TwoSampleMR R package [56] was used to perform two-sample bidirectional MR. Effect allele coding was harmonized across phenotypes using the harmonise_data function. Strand ambiguous SNPs were excluded. To avoid weak instrument bias, genetic instruments were constructed using genome-wide significant SNPs. Genome-wide significant SNPs were LD clumped (r2 ≤ 0.001 in 1000 Genomes Phase 3 European data [53], over a 10 megabase window) to ensure independence. SNPs within two highly pleiotropic regions, the MHC region [57] (hg19 coordinates: Chromosome 6, 28,477,797–33,448,354 base pairs) and 17q21.31 region inversion (hg19 coordinates: Chromosome 17, 40,928,986–42,139,672 base pairs) were excluded, with a 3 megabase buffer added to ensure markers in LD were also removed. The primary MR analysis was conducted using the inverse variance weighted (IVW) estimator with multiplicative random effects. Additional MR analysis was performed using weighted median (WM) [58] estimators. Pearson correlations between IVW and WM estimates were estimated. To account for the multiple testing burden, Bonferroni correction was applied within each estimator, such that significance was declared if p < 0.05/21. As in prior research [28], multivariable MR analysis was performed using CRP, IL-6, and WBC as covariates to statistically control for non-specific markers of systemic inflammation that could confound or mediate associations of PTSD with autoimmune disease, using the MendelianRandomization R package [59]. These multivariable MR analyses were conducted for autoimmune disease phenotypes with significant MR findings. Owing to instrumental variable loss due to incomplete summary data overlap, immune/inflammatory biomarkers were modeled one at a time rather than together in a joint model.
MR sensitivity analyses
Several sensitivity analyses were conducted to assess the robustness of findings. A substantial fraction of the heritability of autoimmune diseases is explained by a limited set of risk variation with large effect sizes. This lends to the possibility of reverse causation if these variants are also included in genetically predicted PTSD. Accordingly, we performed a sensitivity analysis where regions that were genome-wide significant in autoimmune GWAS (±3 megabases) were removed from genetically predicted PTSD. MR PRESSO [60] was used to identify heterogeneity (global test) and outliers (outlier test), and to determine if the outlier-adjusted IVW estimate was significantly different from the unadjusted. To evaluate if our associations could be explained by correlated horizontal pleiotropy (defined as genes influencing a third factor, which in turn has pleiotropic effects on the exposure and outcome), we used the CAUSE method [61]. This method fits a series of nested models: a “null” model where only uncorrelated horizontal pleiotropy (defined as direct effects of genes on the outcome with net zero effect) is modeled (parameter q), a “sharing” model where an additional parameter (parameter eta) is fit to account for correlated horizontal pleiotropy, and a “causal” model where a causal effect parameter (parameter gamma) is fit in addition to the sharing parameter. To test the hypothesis that a causal model explained the relationship better than a sharing model, the causal and sharing model fits were compared using the difference in the expected log pointwise posterior density. Specifically, if the causal model fits better than the sharing model, this implies that the additional complexity needed to model a causal effect is justified, and thus is evidence that data are consistent with a causal effect. If, however, there is not significant evidence that the causal model fits better than the sharing model, this implies that shared pleiotropy alone is sufficient to explain the observed association. To account for potential biases related to sample overlap [62], we repeated IVW MR analysis of significant findings with the UKBB sample data removed from the PTSD GWAS.
Results
After filtering criteria, we assembled a collection of GWAS summary statistics for PTSD, 18 autoimmune diseases, and 3 biomarkers (Supplementary Table 1 and Supplementary Fig. 1). The PTSD GWAS included 1,222,822 participants (including 137,136 PTSD cases). The median autoimmune disease GWAS sample size was 38,078, and ranged from 1459 (neuromyelitis optica spectrum disorder) to 755,406 (autoimmune thyroid disease). The biomarker GWAS sample sizes were 52,654 (IL-6), 363,000 (CRP), and 562,243 (WBC).
Genetic architecture of PTSD and immune-related phenotypes
The h2SNP of PTSD was 5.3% (on the observed scale). Estimates of h2SNP varied across autoimmune diseases, ranging from 3.5% (systemic sclerosis) to 27% (Kawasaki disease) (Fig. 1a). The h2SNP estimates of the three biomarkers were 5.3% (IL-6), 14.3% (CRP), and 17.5% (WBC). We separated h2SNP into its polygenicity and discoverability subcomponents (Fig. 1b and Supplementary Table 2). PTSD was the most polygenic (N causal SNPs = 10,863, SE = 377) and least discoverable phenotype. Across autoimmune diseases, the estimated number of causal SNPs ranged from 18 (Addison’s disease) to 721 (autoimmune thyroid disease). For the three biomarkers, the estimated number of causal SNPs were 127 (IL-6), 934 (CRP), and 2187 (WBC).
a SNP based heritability of assessed phenotypes. SNP based heritability estimates for autoimmune diseases are provided on the liability scale, assuming disease specific prevalence. Phenotypes are colored by category. Black bars ends indicate 95% confidence intervals. Confidence intervals extending beyond the plot range are indicated with arrows. b Polygenicity and discoverability components of heritability. The x-axis depicts the number of influential variants necessary to explain 90% of SNP based heritability (polygenicity ✕ constant). The y-axis depicts the discoverability of the phenotype. Circle sizes indicate relative SNP based heritability values. NMO Neuromyelitis Optica Spectrum Disorder, EGPA Eosinophilic granulomatosis with polyangiitis.
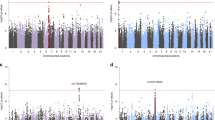
Previous reports [23] of psychiatric disorders and autoimmune diseases have used genetic correlation models to quantify their genetic overlap, and thus we first used this same approach to examine their genetic association. Under a genetic correlation model, PTSD was significantly positively correlated with nine autoimmune diseases and all three biomarkers after Bonferroni correction (Fig. 2 and Supplementary Table 3). We also examined expanded models of genetic overlap between PTSD and all phenotypes (Bivariate MiXeR). These models suggested that for 10 phenotypes examined, genetic overlap was more complex than what could be summarized by rg alone (AIC values > 0). Most often, this meant that a substantial fraction of autoimmune disease/biomarker variants were influential on PTSD, and that the rg estimated only among the reduced set of predicted shared variants was stronger than rg estimated from all variants (Supplementary Table 4).
Genetic correlations (rg) are indicated by circles that are drawn along the x axis. Phenotypes are colored by domain. Hollow circles indicate SNP based heritability (h2SNP) z-score <4 in the immune-related phenotype GWAS (rg estimates may be unreliable). The dotted vertical bar indicates the point of zero correlation. EGPA eosinophilic granulomatosis with polyangiitis.
Causal associations between PTSD and immune-related phenotypes
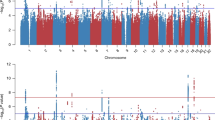
Genetically predicted PTSD (gPTSD) was estimated using 62 LD-independent genome-wide significant risk loci. Under the primary IVW analysis method, gPTSD was significantly positively associated with risk of autoimmune thyroid disease (beta = 0.150, SE = 0.018, p = 0.0001) and CRP (beta = 0.090, SE = 0.018, p = 4.3 × 10−7) after Bonferroni correction (Fig. 3 and Table 1). Under the WM analysis, gPTSD was significantly associated with elevated levels of CRP (beta WM = 0.071, SE = 0.015, p = 1.4 × 10−6) and WBC (beta = 0.036, SE = 0.011, p = 0.002). gPTSD effect size estimates were broadly similar between IVW and WM methods, with a 91% correlation between them. Several phenotypes (Crohn’s disease, ulcerative colitis, rheumatoid arthritis, multiple sclerosis, vitiligo, CRP, and WBC) indicated significant heterogeneity of SNP instruments (IVW heterogeneity statistic p < 0.05/21), indicative of horizontal pleiotropy. Similarly, the global test in MR PRESSO indicated the presence of outliers for these phenotypes. However, the gPTSD effect estimates were not significantly different for any phenotype after MR PRESSO outlier removal (all MR PRESSO distortion test p > 0.05; Supplementary Table 5).
Causal effects of PTSD on immune-related phenotypes, as estimated by the genetically determined PTSD instrument (gPTSD), are indicated by circles that are drawn along the x axis. Phenotypes are colored by domain. Hollow circles indicate non-significance (p > 0.0025). The dotted vertical bar indicates the point of zero effect. Confidence intervals extending beyond the plot range are indicated with arrows. EGPA eosinophilic granulomatosis with polyangiitis.
As a sensitivity analysis for reverse causation, any regions that were significant in the target phenotype GWAS (±3 megabases) were not used as SNP instruments in gPTSD. The gPTSD associations with autoimmune thyroid disease (N SNP instruments = 52, IVW beta = 0.122, SE = 0.037, p = 0.001) and CRP (N SNP instruments = 40, IVW beta = 0.084, SE = 0.015, p = 1 × 10−7) remained significant. We also performed MR in the opposite causal effect direction, examining the association of genetically predicted phenotypes with PTSD (Supplementary Table 6). We did not identify any significant associations.
As a sensitivity analysis for sample overlap, we removed the UK BioBank data from the PTSD GWAS, as it represented the main source of sample overlap. The IVW effect estimates of PTSD on autoimmune thyroid disease (N = 38 instruments, beta = 0.11, SE = 0.04, p = 0.01) and CRP (N = 38 instruments; beta = 0.069, SE = 0.02, p = 0.001) were comparable in magnitude to the original estimates.
Influence of third variables on putative causal effect estimates
Systemic inflammation may link PTSD to autoimmune disease. To explore this pathway, we performed multivariable MR analysis that adjusted for genetically predicted CRP, IL-6, and WBC as surrogate measures of systemic inflammation. In each multivariable model, inference of the association between gPTSD and autoimmune thyroid disease was not substantially altered (CRP adjusted gPTSD IVW beta = 0.162, SE = 0.047, p = 0.0006; IL-6 adjusted gPTSD IVW beta = 0.174, SE = 0.043, p = 5.2 × 10−5; WBC adjusted gPTSD IVW beta = 0.163, SE = 0.044, p = 0.0002) compared to the unadjusted gPTSD estimate.
It is also possible that significant MR associations were confounded by an unmeasured third factor with causal effects on both PTSD and the target phenotypes (i.e., correlated horizontal pleiotropy). To determine whether there was evidence for causation beyond what could be accounted for by correlated horizontal pleiotropy, we compared nested competing models. Results were considered consistent with a causal effect if the model with a causal effect parameter (causal model) provided a significantly better fit than the reduced model fit with only a shared effect parameter for correlated horizontal pleiotropy (sharing model). Our analyses supported causal effects of PTSD on autoimmune thyroid disease (beta = 0.11, 95% CI = [0.07–0.14]; causal versus sharing model p = 2.6 × 10−3) and CRP (beta = 0.05, 95% CI = [0.04–0.07]; causal versus sharing model p = 8.5 × 10−8) (Supplementary Table 7). We note that the causal effect estimates of these models were comparable in magnitude (overlapping 95% CIs) to the IVW estimates. In testing for the effects of genetically predicted phenotypes on PTSD (Supplementary Table 8), the causal model of CRP on PTSD did not substantially improve fit over the sharing model (causal versus sharing model p = 0.18), thus indicating no strong evidence of a causal effect.
Discussion
In this most comprehensive study to date of potential causal relationships between PTSD and autoimmune disease based on their genetic underpinnings, an initial screening of their genetic overlap indicated broadly different genetic architectures. Relative to PTSD, a greater proportion of the phenotypic variation in numerous autoimmune diseases is accounted for by common genetic variation. Moreover, relatively few variants (from a few dozen to a few hundred) explain the majority of h2SNP in autoimmune diseases, whereas relatively more variants (~10,000) are needed to explain the h2SNP of PTSD. Despite these differences, we have identified shared genetic variation between PTSD and autoimmune diseases. Our results would suggest that this overlap is more nuanced than what is indicated by genetic correlation, such that a substantial fraction of variation influential to autoimmune diseases also influences PTSD. Thus, our results support hypotheses that shared underlying biology contributes to their epidemiologic associations [7,8,9,10,11,12,13,14]. We expect that detailed interrogation of shared loci [63] and systems [64] between PTSD and autoimmune diseases will provide better insights into their shared biology. Indeed, a recent familial coaggregation study [65] has identified five shared functional modules (potential molecular complexes [66]) between PTSD and autoimmune diseases, including signaling by G proteins/G protein complex receptors, an essential component of immune response [67].
We also leveraged genetic associations from GWAS to investigate the causal relationship between PTSD and autoimmune disease, as it will help determine clinical relevance of the recognition and treatment of PTSD in individuals living with primary disorders of the immune system [19] and provide directions for future mechanistic research. Our results support a putative causal effect of PTSD on autoimmune thyroid disease, corroborating findings from large prospective cohort studies suggesting that PTSD precedes the development of autoimmune thyroid disease [7,8,9]. In suggesting that the relationship is causal, our results strongly support the notion that clinical attention should be paid to thyroid health in those with PTSD symptoms [68]. However, given the broad phenotyping used for autoimmune thyroid disease, it is unclear whether our findings relate to specific types of autoimmune-related thyroid dysfunction (i.e., Grave’s or Hashimoto’s). Further research is needed to explore this question as more nuanced genetic instruments are generated. In terms of potential genetically regulated mechanisms linking PTSD to autoimmune thyroid disease, we considered the shared genetic signal between PTSD and inflammatory biomarkers that we have identified. However, adjusting for inflammatory biomarkers in multivariate MR did not substantially influence the positive association of PTSD with autoimmune thyroid disease. Thus, from our data alone, it is unclear what factors drive the putative causal association between PTSD and autoimmune thyroid disease. Prior studies have suggested some shared biological pathways between these conditions, including “signaling by G proteins/GPCRs,” “cilium assembly,” and “membrane trafficking” [65]. Additionally, our findings do not provide insight into PTSD’s potential pathophysiologic alterations on thyroid function through other mechanisms, such as through metabolic alterations [69] or effects on the central nervous system [70], making these future points of investigation.
We speculate on whether the influence of PTSD on autoimmune disease risk is mediated by the mechanisms considered to be highly relevant to PTSD pathophysiology. PTSD is associated with alterations in the hypothalamic-pituitary-adrenal (HPA)-axis [71], which plays a key role in stress response and also regulates the immune system [72]. Indeed, glucocorticoids released by activation of the HPA axis regulate a range of immune-related genes, including the expression of inflammatory cytokines [15]. In the context of our findings, dysregulation of the HPA-axis in PTSD may interfere with regulation of immune function by the HPA-axis, thereby leading to excessive inflammation [15]. In fact, impaired HPA responsiveness, such as found in PTSD, has demonstrated associations with other autoimmune and inflammatory diseases [73]. It is also important to note that certain aspects of HPA-axis dysregulation result from trauma exposure rather than PTSD [74]. This prompts the question of whether trauma exposure itself (or trauma of a particular type or timing [75]) is a driving force behind our observed associations. In considering other mediating mechanisms, Song et al. speculated that trauma-related lifestyle changes may influence autoimmune disease risk [8]. Ultimately, the clinical implications of our findings would be greatly enhanced if risk pathways were delineated. This delineation will inform whether the most relevant modalities to reduce the elevated risk of autoimmune disease development involve the treatment of PTSD itself, early interventions to reduce trauma exposure, or alterations to trauma-related lifestyle changes. One possible approach for a future investigation leveraging genomic data to estimate the contribution of these factors would be to conduct network MR analyses [76].
In assessing hypotheses that PTSD produces an inflammatory state [15], we observed a putative causal effect of PTSD on CRP. Contrary to our expectation of a bidirectional association based on prior work [26], we found no causal influence of immune/inflammatory biomarkers on PTSD. Rather, our results suggest that the previously reported causal effect of CRP on PTSD [26] was confounded by a (unmeasured) genetically determined factor. At first glance, this seems to conflict with existing epidemiologic evidence that elevated CRP precedes PTSD development [77]. However, we note that a genetically determined confounder could lead to elevated CRP before the development of PTSD. Thus, our results only argue against the hypothesized mechanistic role of CRP in PTSD [78], and do not weigh against hypotheses that inflammation causally influences PTSD [79]. With the increasing availability of genetic data, as with autoimmune disease, locus- and systems-level interrogations may help further validate this hypothesis or elucidate alternative mechanisms. Indeed, recent genomic studies converge to support the notion that there is systemic immune dysregulation in PTSD and importantly, highlight specific genes and pathways [16]. These will make excellent candidates to explore in future genomic studies of the overlap of PTSD, inflammation, and autoimmune disease.
In contrasting our findings with epidemiologic studies [7,8,9] of PTSD and autoimmune disease, these studies identified a wide spectrum of disease associations that we did not identify here. The effect sizes reported for these conditions varied, but altogether were relatively modest (hazard ratios and relative risks of ~2 or less). Thus, null association in our investigation may reflect power limitations of our analyses, rather than evidence that epidemiologic associations were confounded. In regard to power, of all autoimmune disease GWAS we examined, the autoimmune thyroid disease GWAS had the largest sample size and was thus likely the most powered. Similarly, a previous familial genetic and polygenic risk score based investigation of stress related disorders and autoimmune diseases [65] identified a significant association of PTSD with autoimmune thyroid disease, but yielded less conclusive evidence for other individual autoimmune diseases. The investigators hypothesized that the autoimmune thyroid disease association was identified due to its higher prevalence (and thus statistical power) relative to other autoimmune diseases, rather than effects specific to autoimmune thyroid disease [65]. Therefore, we speculate that significant associations with other autoimmune diseases may come to light as larger GWAS of autoimmune diseases are conducted.
Another factor that may have influenced our findings is the heterogeneity existing within autoimmune diseases [80]. If cases included in autoimmune disease GWAS are heterogeneous, effect estimates will not accurately capture risk, and this will in turn harm the reliability of MR. It will be useful to parse out whether PTSD influences risk of only particular subtypes of an autoimmune disease, as this can help identify more specific shared pathways. In regard to heterogeneity, we highlight how the age of onset of autoimmune disease reflects etiologic heterogeneity [81]. In the epidemiologic study by Song et al. [8], PTSD preceded the development of autoimmune disorders in adults that typically have an early age of onset (e.g., type 1 diabetes [81]). Thus, lack of corroborating findings here may reflect the need to specifically only use GWAS of adult-onset cases of autoimmune diseases in MR analyses. If it is the case that PTSD influences risk particularly in adult forms of autoimmune disease, this prompts the discussion of whether it will be important to consider PTSD status in the screening, management, and treatment of these diseases.
An additional notable challenge specific to conducting well-powered MR in this context is the usage of the HLA region as an instrumental variable. This region is highly relevant to many autoimmune diseases [82]. However, we excluded this region because of its high degree of pleiotropy and complicated LD structure, likely sacrificing statistical power as a consequence. Very detailed colocalization analysis [83] would need to be performed prior to integrating this region into PTSD MR analyses. Indeed, given its relevance to PTSD [84], the investigation of the HLA region should be a focal point of future research into the genetic overlap of PTSD and autoimmune diseases.
This study did not evaluate if effects observed for PTSD are specific to this psychiatric disorder. Autoimmune diseases are associated with other psychiatric disorders [23], which raises the possibility that shared variation between psychiatric disorders (general psychopathology) drives the observed associations. Individuals with psychiatric co-morbidities along with PTSD have elevated risk of developing autoimmune diseases relative to those with just PTSD [8], further suggesting the influence of other forms of psychopathology on autoimmune disease development. We also note the wide symptom overlap of PTSD with other psychiatric conditions, promoting the possibility that the observed associations arose from shared symptoms, rather than PTSD-specific symptomatology. Moreover, our definition of PTSD in the PGC-PTSD GWAS included participants with non-clinical assessments of PTSD, thus increasing the potential for misclassification generating non-PTSD-specific results. In considering mediating mechanisms such as HPA-axis dysregulation, we note how alterations in the HPA-axis are not specific to PTSD, and are present in other trauma-relevant mental disorders, e.g., bipolar disorder, borderline personality disorder [85], and depression (albeit with differences in manifestation [86]). In defense of the relevance of PTSD, epidemiologic evidence suggests that individuals with PTSD are at significantly higher risk of autoimmune disease than those afflicted by other mental health disorders [9]. We propose that future MR analyses of more homogenous PTSD symptom dimensions may help to distinguish risk. Namely, it would be interesting to see whether autoimmune disease risk is particularly elevated by the re-experiencing symptoms that are a hallmark of PTSD rather than symptoms that are shared with other forms of psychopathology [87].
While we performed several sensitivity analyses to assess the validity of our MR analyses, we highlight some potential limitations. First, the high polygenicity and low discoverability of PTSD pose challenges to the selection of valid SNP instruments for MR. Namely, the pathways by which implicated SNPs influence PTSD risk are not obvious, and thus it is not clear whether these SNPs could have confounding horizontal pleiotropic effects on autoimmune disease. Indeed, our results indicate heterogeneity across instruments for many autoimmune diseases, suggesting horizontal pleiotropy, albeit adjusting for this did not substantially alter results. In the future, the MR framework [88] could be used to help understand the causal pathways of risk SNPs, leading to the best possible selection of SNP instruments for PTSD. Second, there was potential for sample overlap between PTSD and a subset of the autoimmune GWAS (such as data originating from widely-used sources such as the UK BioBank), which can potentially bias two-sample MR analyses due to weak instrument bias. Recent work demonstrates that many two sample MR approaches can successfully be used for one-sample MR [89], a scenario of complete sample overlap where weak instrument bias is a primary concern. Importantly, our investigation used genome-wide significant variants from large GWAS, which demonstrate strong, replicable associations. Thus, our results are less likely to be influenced by the weak instrument bias that arises from using underpowered GWAS. Last, associations of PTSD with autoimmune and thyroid disease and CRP were still significant when we removed the UKBB sample.
In summary, shared genetics between PTSD and autoimmune diseases may underlie, in part, their epidemiologic associations. We also observed a putative causal effect of PTSD on autoimmune thyroid disease and CRP, consistent with epidemiologic evidence of stress- and trauma-related disorders predicting elevated systemic inflammation and onset of autoimmune disease [7,8,9, 85]. Together, these results highlight the nuanced links between PTSD, autoimmune disorders, and associated inflammatory signatures, and suggest the importance of targeting related pathways to protect against disease and disability.
Data availability
URLs for GWAS summary data used in this manuscript are provided in Supplementary Table 1.
Code availability
Analysis code is available on GitHub (https://github.com/nievergeltlab/PTSD_MR).
References
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th edition. Arlington, VA, USA: American Psychiatric Association; 2013.
Atwoli L, Stein DJ, Koenen KC, McLaughlin KA. Epidemiology of posttraumatic stress disorder: prevalence, correlates and consequences. Curr Opin Psychiatry. 2015;28:307–11.
Sareen J. Posttraumatic stress disorder in adults: impact, comorbidity, risk factors, and treatment. Can J Psychiatry. 2014;59:460–7.
Brunello N, Davidson JR, Deahl M, Kessler RC, Mendlewicz J, Racagni G, et al. Posttraumatic stress disorder: diagnosis and epidemiology, comorbidity and social consequences, biology and treatment. Neuropsychobiology. 2001;43:150–62.
McCrone P, Knapp M, Cawkill P. Posttraumatic stress disorder (PTSD) in the Armed Forces: health economic considerations. J Trauma Stress. 2003;16:519–22.
Pietrzak RH, Goldstein RB, Southwick SM, Grant BF. Physical health conditions associated with posttraumatic stress disorder in U.S. older adults: results from wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. J Am Geriatr Soc. 2012;60:296–303.
Bookwalter DB, Roenfeldt KA, LeardMann CA, Kong SY, Riddle MS, Rull RP. Posttraumatic stress disorder and risk of selected autoimmune diseases among US military personnel. BMC Psychiatry. 2020;20:23.
Song H, Fang F, Tomasson G, Arnberg FK, Mataix-Cols D, Fernández de la Cruz L, et al. Association of stress-related disorders with subsequent autoimmune disease. JAMA. 2018;319:2388–400.
O’Donovan A, Cohen BE, Seal KH, Bertenthal D, Margaretten M, Nishimi K, et al. Elevated risk for autoimmune disorders in iraq and afghanistan veterans with posttraumatic stress disorder. Biol Psychiatry. 2015;77:365–74.
Case SM, Feldman CH, Guan H, Stevens E, Kubzansky LD, Koenen KC, et al. Posttraumatic stress disorder and risk of systemic lupus erythematosus among medicaid recipients. Arthritis Care Res. 2023;75:174–9.
Roberts AL, Malspeis S, Kubzansky LD, Feldman CH, Chang SC, Koenen KC, et al. Association of trauma and posttraumatic stress disorder with incident systemic lupus erythematosus in a longitudinal cohort of women. Arthritis Rheumatol. 2017;69:2162–9.
Lee YC, Agnew-Blais J, Malspeis S, Keyes K, Costenbader K, Kubzansky LD, et al. Post-traumatic stress disorder and risk for incident rheumatoid arthritis. Arthritis Care Res. 2016;68:292–8.
Miller-Archie SA, Izmirly PM, Berman JR, Brite J, Walker DJ, Dasilva RC, et al. Systemic autoimmune disease among adults exposed to the September 11, 2001 terrorist attack. Arthritis Rheumatol. 2020;72:849–59.
Hsu TW, Bai YM, Tsai SJ, Chen TJ, Chen MH, Liang CS. Risk of autoimmune diseases after post-traumatic stress disorder: a nationwide cohort study. Eur Arch Psychiatry Clin Neurosci. 2023.
Katrinli S, Oliveira NCS, Felger JC, Michopoulos V, Smith AK. The role of the immune system in posttraumatic stress disorder. Transl Psychiatry. 2022;12:313.
Núñez-Rios DL, Martínez-Magaña JJ, Nagamatsu ST, Andrade-Brito DE, Forero DA, Orozco-Castaño CA, et al. Central and peripheral immune dysregulation in posttraumatic stress disorder: convergent multi-omics evidence. Biomedicines. 2022;10:1107.
Hori H, Kim Y. Inflammation and post-traumatic stress disorder. Psychiatry Clin Neurosci. 2019;73:143–53.
Sumner JA, Nishimi KM, Koenen KC, Roberts AL, Kubzansky LD. Posttraumatic stress disorder and inflammation: untangling issues of bidirectionality. Biol Psychiatry. 2020;87:885–97.
Neigh GN, Ali FF. Co-morbidity of PTSD and immune system dysfunction: opportunities for treatment. Curr Opin Pharmacol. 2016;29:104–10.
Gill JM, Saligan L, Woods S, Page G. PTSD is associated with an excess of inflammatory immune activities. Perspect Psychiatr Care. 2009;45:262–77.
Castro C, Gourley M. Diagnostic testing and interpretation of tests for autoimmunity. J Allergy Clin Immunol. 2010;125:S238–47.
Sekula P, Del Greco MF, Pattaro C, Köttgen A. Mendelian randomization as an approach to assess causality using observational data. J Am Soc Nephrol. 2016;27:3253.
Tylee DS, Lee YK, Wendt FR, Pathak GA, Levey DF, De Angelis F, et al. An atlas of genetic correlations and genetically informed associations linking psychiatric and immune-related phenotypes. JAMA Psychiatry. 2022;79:667–76.
Saccaro LF, Gasparini S, Rutigliano G. Applications of Mendelian randomization in psychiatry: a comprehensive systematic review. Psychiatr Genet. 2022;32:199–213.
Nievergelt CM, Maihofer AX, Klengel T, Atkinson EG, Chen C-Y, Choi KW, et al. International meta-analysis of PTSD genome-wide association studies identifies sex- and ancestry-specific genetic risk loci. Nat Commun. 2019;10:4558.
Muniz Carvalho C, Wendt FR, Maihofer AX, Stein DJ, Stein MB, Sumner JA, et al. Dissecting the genetic association of C-reactive protein with PTSD, traumatic events, and social support. Neuropsychopharmacol. 2021;46:1071–77.
Nievergelt CM, Maihofer AX, Atkinson EG, Chen CY, Choi KW, Coleman JR, et al. Discovery of 95 PTSD loci provides insight into genetic architecture and neurobiology of trauma and stress-related disorders. medRxiv. 2023. https://doi.org/10.1101/2023.08.31.23294915.
Perry BI, Burgess S, Jones HJ, Zammit S, Upthegrove R, Mason AM, et al. The potential shared role of inflammation in insulin resistance and schizophrenia: a bidirectional two-sample Mendelian randomization study. PLoS Med. 2021;18:e1003455.
Yang J-J, Jiang W. Immune biomarkers alterations in post-traumatic stress disorder: a systematic review and meta-analysis. J Affect Disord. 2020;268:39–46.
Sollis E, Mosaku A, Abid A, Buniello A, Cerezo M, Gil L, et al. The NHGRI-EBI GWAS Catalog: knowledgebase and deposition resource. Nucleic Acids Res. 2023;51:D977–85.
Sinnott-Armstrong N, Tanigawa Y, Amar D, Mars N, Benner C, Aguirre M, et al. Genetics of 35 blood and urine biomarkers in the UK Biobank. Nat Genet. 2021;53:185–94.
Ahluwalia TS, Prins BP, Abdollahi M, Armstrong NJ, Aslibekyan S, Bain L, et al. Genome-wide association study of circulating interleukin 6 levels identifies novel loci. Hum Mol Genet. 2021;30:393–409.
Chen M-H, Raffield LM, Mousas A, Sakaue S, Huffman JE, Moscati A, et al. Trans-ethnic and ancestry-specific blood-cell genetics in 746,667 individuals from 5 global populations. Cell. 2020;182:1198–213.e14.
Eriksson D, Røyrvik EC, Aranda-Guillén M, Berger AH, Landegren N, Artaza H, et al. GWAS for autoimmune Addison’s disease identifies multiple risk loci and highlights AIRE in disease susceptibility. Nat Commun. 2021;12:959.
Saevarsdottir S, Olafsdottir TA, Ivarsdottir EV, Halldorsson GH, Gunnarsdottir K, Sigurdsson A, et al. FLT3 stop mutation increases FLT3 ligand level and risk of autoimmune thyroid disease. Nature. 2020;584:619–23.
Dubois PCA, Trynka G, Franke L, Hunt KA, Romanos J, Curtotti A, et al. Multiple common variants for celiac disease influencing immune gene expression. Nat Genet. 2010;42:295–302.
Liu JZ, van Sommeren S, Huang H, Ng SC, Alberts R, Takahashi A, et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat Genet. 2015;47:979–86.
Lyons PA, Peters JE, Alberici F, Liley J, Coulson RMR, Astle W, et al. Genome-wide association study of eosinophilic granulomatosis with polyangiitis reveals genomic loci stratified by ANCA status. Nat Commun. 2019;10:5120.
Hoggart C, Shimizu C, Galassini R, Wright VJ, Shailes H, Bellos E, et al. Identification of novel locus associated with coronary artery aneurysms and validation of loci for susceptibility to Kawasaki disease. Eur J Hum Genet. 2021;29:1734–44.
International Multiple Sclerosis Genetics Consortium. Multiple sclerosis genomic map implicates peripheral immune cells and microglia in susceptibility. Science. 2019;365:eaav7188.
Chia R, Saez-Atienzar S, Murphy N, Chiò A, Blauwendraat C, Roda RH, et al. Identification of genetic risk loci and prioritization of genes and pathways for myasthenia gravis: a genome-wide association study. Proc Natl Acad Sci USA. 2022;119:e2108672119.
Estrada K, Whelan CW, Zhao F, Bronson P, Handsaker RE, Sun C, et al. A whole-genome sequence study identifies genetic risk factors for neuromyelitis optica. Nat Commun. 2018;9:1929.
Laisk T, Lepamets M, Koel M, Abner E, Metspalu A, Nelis M, et al. Genome-wide association study identifies five risk loci for pernicious anemia. Nat Commun. 2021;12:3761.
Cordell HJ, Han Y, Mells GF, Li Y, Hirschfield GM, Greene CS, et al. International genome-wide meta-analysis identifies new primary biliary cirrhosis risk loci and targetable pathogenic pathways. Nat Commun. 2015;6:8019.
Sakaue S, Kanai M, Tanigawa Y, Karjalainen J, Kurki M, Koshiba S, et al. A cross-population atlas of genetic associations for 220 human phenotypes. Nat Genet. 2021;53:1415–24.
Okada Y, Wu D, Trynka G, Raj T, Terao C, Ikari K, et al. Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature. 2014;506:376–81.
Langefeld CD, Ainsworth HC, Graham DSC, Kelly JA, Comeau ME, Marion MC, et al. Transancestral mapping and genetic load in systemic lupus erythematosus. Nat Commun. 2017;8:16021.
López-Isac E, Acosta-Herrera M, Kerick M, Assassi S, Satpathy AT, Granja J, et al. GWAS for systemic sclerosis identifies multiple risk loci and highlights fibrotic and vasculopathy pathways. Nat Commun. 2019;10:4955.
Chiou J, Geusz RJ, Okino M-L, Han JY, Miller M, Melton R, et al. Interpreting type 1 diabetes risk with genetics and single-cell epigenomics. Nature. 2021;594:398–402.
Auton A, Abecasis GR, Altshuler DM, Durbin RM, Abecasis GR, Bentley DR, et al. A global reference for human genetic variation. Nature. 2015;526:68–74.
Jin Y, Andersen G, Yorgov D, Ferrara TM, Ben S, Brownson KM, et al. Genome-wide association studies of autoimmune vitiligo identify 23 new risk loci and highlight key pathways and regulatory variants. Nat Genet. 2016;48:1418–24.
Bulik-Sullivan BK, Loh P-R, Finucane HK, Ripke S, Yang J, Patterson N, et al. LD score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat Genet. 2015;47:291–5.
Clarke L, Zheng-Bradley X, Smith R, Kulesha E, Xiao C, Toneva I, et al. The 1000 Genomes Project: data management and community access. Nat Methods. 2012;9:459–62.
Holland D, Frei O, Desikan R, Fan C-C, Shadrin AA, Smeland OB, et al. Beyond SNP heritability: polygenicity and discoverability of phenotypes estimated with a univariate Gaussian mixture model. PLoS Genet. 2020;16:e1008612.
Frei O, Holland D, Smeland OB, Shadrin AA, Fan CC, Maeland S, et al. Bivariate causal mixture model quantifies polygenic overlap between complex traits beyond genetic correlation. Nat Commun. 2019;10:2417.
Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, Baird D, et al. The MR-Base platform supports systematic causal inference across the human phenome. eLife. 2018;7:e34408.
O’Connor LJ, Price AL. Distinguishing genetic correlation from causation across 52 diseases and complex traits. Nat Genet. 2018;50:1728–34.
Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent estimation in Mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol. 2016;40:304–14.
Yavorska OO, Burgess S. MendelianRandomization: an R package for performing Mendelian randomization analyses using summarized data. Int J Epidemiol. 2017;46:1734–9.
Verbanck M, Chen C-Y, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50:693–8.
Morrison J, Knoblauch N, Marcus JH, Stephens M, He X. Mendelian randomization accounting for correlated and uncorrelated pleiotropic effects using genome-wide summary statistics. Nat Genet. 2020;52:740–7.
Burgess S, Davies NM, Thompson SG. Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol. 2016;40:597–608.
Lincoln MR, Connally N, Axisa P-P, Gasperi C, Mitrovic M, Heel DV, et al. Joint analysis reveals shared autoimmune disease associations and identifies common mechanisms. medRxiv. 2021. https://doi.org/10.1101/2021.05.13.21257044.
Gokuladhas S, Schierding W, Golovina E, Fadason T, O’Sullivan J. Unravelling the shared genetic mechanisms underlying 18 autoimmune diseases using a systems approach. Front Immunol. 2021;12:693142.
Zeng Y, Suo C, Yao S, Lu D, Larsson H, D’Onofrio BM, et al. Genetic associations between stress-related disorders and autoimmune disease. Am J Psychiatry. 2023;180:294–304.
Bader GD, Hogue CW. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinform. 2003;4:2.
Lämmermann T, Kastenmüller W. Concepts of GPCR-controlled navigation in the immune system. Immunol Rev. 2019;289:205–31.
Jung SJ, Kang JH, Roberts AL, Nishimi K, Chen Q, Sumner JA, et al. Posttraumatic stress disorder and incidence of thyroid dysfunction in women. Psychol Med. 2019;49:2551–60.
Wang S, Mason J. Elevations of serum T3 levels and their association with symptoms in World War II veterans with combat-related posttraumatic stress disorder: replication of findings in vietnam combat veterans. Psychosom Med. 1999;61:131–8.
Toloza FJK, Mao Y, Menon LP, George G, Borikar M, Erwin PJ, et al. Association of thyroid function with posttraumatic stress disorder: a systematic review and meta-analysis. Endocr Pract. 2020;26:1173–85.
Dunlop BW, Wong A. The hypothalamic-pituitary-adrenal axis in PTSD: pathophysiology and treatment interventions. Prog Neuropsychopharmacol Biol Psychiatry. 2019;89:361–79.
Smith SM, Vale WW. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci. 2006;8:383–95.
Silverman MN, Sternberg EM. Glucocorticoid regulation of inflammation and its functional correlates: from HPA axis to glucocorticoid receptor dysfunction. Ann N Y Acad Sci. 2012;1261:55–63.
Morris MC, Compas BE, Garber J. Relations among posttraumatic stress disorder, comorbid major depression, and HPA function: a systematic review and meta-analysis. Clin Psychol Rev. 2012;32:301–15.
Dube SR, Fairweather D, Pearson WS, Felitti VJ, Anda RF, Croft JB. Cumulative childhood stress and autoimmune diseases in adults. Psychosom Med. 2009;71:243–50.
Burgess S, Daniel RM, Butterworth AS, Thompson SG. Network Mendelian randomization: using genetic variants as instrumental variables to investigate mediation in causal pathways. Int J Epidemiol. 2015;44:484–95.
Eraly SA, Nievergelt CM, Maihofer AX, Barkauskas DA, Biswas N, Agorastos A, et al. Assessment of plasma C-reactive protein as a biomarker of posttraumatic stress disorder risk. JAMA Psychiatry. 2014;71:423–31.
Friend SF, Nachnani R, Powell SB, Risbrough VB. C-Reactive Protein: marker of risk for post-traumatic stress disorder and its potential for a mechanistic role in trauma response and recovery. Eur J Neurosci. 2022;55:2297–310.
Michopoulos V, Rothbaum AO, Jovanovic T, Almli LM, Bradley B, Rothbaum BO, et al. Association of CRP genetic variation and CRP level with elevated PTSD symptoms and physiological responses in a civilian population with high levels of trauma. Am J Psychiatry. 2014;172:353–62.
Cho JH, Feldman M. Heterogeneity of autoimmune diseases: pathophysiologic insights from genetics and implications for new therapies. Nat Med. 2015;21:730–8.
Amador-Patarroyo MJ, Rodriguez-Rodriguez A, Montoya-Ortiz G. How does age at onset influence the outcome of autoimmune diseases? Autoimmune Dis. 2012;2012:251730.
Gough SC, Simmonds MJ. The HLA region and autoimmune disease: associations and mechanisms of action. Curr Genomics. 2007;8:453–65.
Zuber V, Grinberg NF, Gill D, Manipur I, Slob EAW, Patel A, et al. Combining evidence from Mendelian randomization and colocalization: review and comparison of approaches. Am J Hum Genet. 2022;109:767–82.
Katrinli S, Smith AK. Immune system regulation and role of the human leukocyte antigen in posttraumatic stress disorder. Neurobiol Stress. 2021;15:100366.
Saccaro LF, Schilliger Z, Dayer A, Perroud N, Piguet C. Inflammation, anxiety, and stress in bipolar disorder and borderline personality disorder: a narrative review. Neurosci Biobehav Rev. 2021;127:184–92.
D’Elia ATD, Juruena MF, Coimbra BM, Mello MF, Mello AF. Posttraumatic stress disorder (PTSD) and depression severity in sexually assaulted women: hypothalamic-pituitary-adrenal (HPA) axis alterations. BMC Psychiatry. 2021;21:174.
Zoellner LA, Pruitt LD, Farach FJ, Jun JJ. Understanding heterogeneity in PTSD: fear, dysphoria, and distress. Depress Anxiety. 2014;31:97–106.
Cho Y, Haycock PC, Sanderson E, Gaunt TR, Zheng J, Morris AP, et al. Exploiting horizontal pleiotropy to search for causal pathways within a Mendelian randomization framework. Nat Commun. 2020;11:1010.
Minelli C, Del Greco MF, van der Plaat DA, Bowden J, Sheehan NA, Thompson J. The use of two-sample methods for Mendelian randomization analyses on single large datasets. Int J Epidemiol. 2021;50:1651–9.
Acknowledgements
This work was supported by the National Institute of Mental Health/U.S. Army Medical Research and Development Command (Grant No. R01MH106595 [to CMN, MBS, KJR, and KCK]), and National Institutes of Health (Grant No. 5U01MH109539 [to the Psychiatric Genomics Consortium]). Financial support for the PTSD PGC was provided by Cohen Veterans Bioscience, Stanley Center for Psychiatric Research at the Broad Institute, and One Mind. This research has been conducted using the UK Biobank resource under application number 41209. This work would not have been possible without the contributions of the investigators who comprise the PGC-PTSD working group, and especially the more than 1,307,247 research participants worldwide who shared their life experiences and biological samples with PGC-PTSD investigators. For making GWAS summary data available to us, we thank the Psychiatric Genomics Consortium (PGC) for PTSD, Natalia V. Rivera of the Karolinska Institutet, Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) consortium, International Multiple Sclerosis Genetics Consortium, as well as all the researchers from all other studies mentioned in this article. The members of the Psychiatric Genomics Consortium Working Group are Caroline M. Nievergelt, Adam X. Maihofer, Elizabeth G. Atkinson, Chia-Yen Chen, Karmel W. Choi, Jonathan R. I. Coleman, Nikolaos P. Daskalakis, Laramie E. Duncan, Renato Polimanti, Cindy Aaronson, Ananda B. Amstadter, Soren B. Andersen, Ole A. Andreassen, Paul A. Arbisi, Allison E. Ashley-Koch, S. Bryn Austin, Esmina Avdibegoviç, Dragan Babic, Silviu-Alin Bacanu, Dewleen G. Baker, Anthony Batzler, Jean C. Beckham, Sintia Belangero, Corina Benjet, Carisa Bergner, Linda M. Bierer, Joanna M. Biernacka, Laura J. Bierut, Jonathan I. Bisson, Marco P. Boks, Elizabeth A. Bolger, Amber Brandolino, Gerome Breen, Rodrigo Affonseca Bressan, Richard A. Bryant, Angela C. Bustamante, Jonas Bybjerg-Grauholm, Marie Bækvad-Hansen, Anders D. Børglum, Sigrid Børte, Leah Cahn, Joseph R. Calabrese, Jose Miguel Caldas-de-Almeida, Chris Chatzinakos, Sheraz Cheema, Sean A. P. Clouston, LucÍa Colodro-Conde, Brandon J. Coombes, Carlos S. Cruz-Fuentes, Anders M. Dale, Shareefa Dalvie, Lea K. Davis, Jürgen Deckert, Douglas L. Delahanty, Michelle F. Dennis, Terri de Roon-Cassini, Frank Desarnaud, Christopher P. DiPietro, Seth G. Disner, Anna R. Docherty, Katharina Domschke, Grete Dyb, Alma Dzubur Kulenovic, Howard J. Edenberg, Alexandra Evans, Chiara Fabbri, Negar Fani, Lindsay A. Farrer, Adriana Feder, Norah C. Feeny, Janine D. Flory, David Forbes, Carol E. Franz, Sandro Galea, Melanie E. Garrett, Bizu Gelaye, Joel Gelernter, Elbert Geuze, Charles F. Gillespie, Aferdita Goci, Slavina B. Goleva, Scott D. Gordon, Lana Ruvolo Grasser, Camila Guindalini, Magali Haas, Saskia Hagenaars, Michael A. Hauser, Andrew C. Heath, Sian M.J. Hemmings, Victor Hesselbrock, Ian B. Hickie, Kelleigh Hogan, David Michael Hougaard, Hailiang Huang, Laura M. Huckins, Kristian Hveem, Miro Jakovljevic, Arash Javanbakht, Gregory D. Jenkins, Jessica Johnson, Ian Jones, Tanja Jovanovic, Karen-Inge Karstoft, Milissa L. Kaufman, James L. Kennedy, Ronald C. Kessler, Alaptagin Khan, Nathan A. Kimbrel, Anthony P. King, Nastassja Koen, Roman Kotov, Henry R. Kranzler, Kristi Krebs, William S. Kremen, Pei-Fen Kuan, Bruce R. Lawford, Lauren A. M. Lebois, Kelli Lehto, Daniel F. Levey, Catrin Lewis, Israel Liberzon, Sarah D. Linnstaedt, Mark W. Logue, Adriana Lori, Yi Lu, Benjamin J. Luft, Michelle K. Lupton, Jurjen J. Luykx, Iouri Makotkine, Jessica L. Maples-Keller, Shelby Marchese, Charles Marmar, Nicholas G. Martin, Gabriela A. MartÍnez-Levy, Kerrie McAloney, Alexander McFarlane, Katie A. McLaughlin, Samuel A. McLean, Sarah E. Medland, Divya Mehta, Jacquelyn Meyers, Vasiliki Michopoulos, Elizabeth A. Mikita, Lili Milani, William Milberg, Mark W. Miller, Rajendra A. Morey, Charles Phillip Morris, Ole Mors, Preben Bo Mortensen, Mary S. Mufford, Elliot C. Nelson, Merete Nordentoft, Sonya B. Norman, Nicole R. Nugent, Meaghan O’Donnell, Holly K. Orcutt, Pedro M. Pan, Matthew S. Panizzon, Gita A. Pathak, Edward S. Peters, Alan L. Peterson, Matthew Peverill, Robert H. Pietrzak, Melissa A. Polusny, Bernice Porjesz, Abigail Powers, Xue-Jun Qin, Andrew Ratanatharathorn, Victoria B. Risbrough, Andrea L. Roberts, Barbara O. Rothbaum, Alex O. Rothbaum, Peter Roy-Byrne, Kenneth J. Ruggiero, Ariane Rung, Heiko Runz, Bart P. F. Rutten, Stacey Saenz de Viteri, Giovanni Abrahão Salum, Laura Sampson, Sixto E. Sanchez, Marcos Santoro, Carina Seah, Soraya Seedat, Julia S. Seng, Andrey Shabalin, Christina M. Sheerin, Derrick Silove, Alicia K. Smith, Jordan W. Smoller, Scott R. Sponheim, Dan J. Stein, Synne Stensland, Jennifer S. Stevens, Jennifer A. Sumner, Martin H. Teicher, Wesley K. Thompson, Arun K. Tiwari, Edward Trapido, Monica Uddin, Robert J. Ursano, Unnur Valdimarsdóttir, Leigh Luella van den Heuvel, Miranda Van Hooff, Sanne J. H. van Rooij, Eric Vermetten, Christiaan H. Vinkers, Joanne Voisey, Zhewu Wang, Yunpeng Wang, Monika Waszczuk, Heike Weber, Frank R. Wendt, Thomas Werge, Michelle A. Williams, Douglas E. Williamson, Bendik S. Winsvold, Sherry Winternitz, Erika J. Wolf, Christiane Wolf, Yan Xia, Ying Xiong, Rachel Yehuda, Ross McD Young, Keith A. Young, Clement C. Zai, Gwyneth C. Zai, Mark Zervas, Hongyu Zhao, Lori A. Zoellner, John-Anker Zwart, Murray B. Stein, Kerry J. Ressler, and Karestan C. Koenen.
Author information
Authors and Affiliations
Consortia
Contributions
CMN, EJW, JAS, and SMJH supervised the project. SMJH, AR, and AXM designed the study. The PGC-PTSD and CHARGE consortia provided data used in analyses. AXM performed statistical analyses. AXM prepared the first draft of the manuscript. AKS, AOR, AR, CMN, EAM, EJW, JAS, JS, KCK, KHC, KJR, MBS, RAS, RMS, RP, SMJH, SS, TW, and VM critically revised and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
MBS has in the past 3 years received consulting income from Actelion, Acadia Pharmaceuticals, Aptinyx, Bionomics, BioXcel Therapeutics, Clexio, EmpowerPharm, GW Pharmaceuticals, Janssen, Jazz Pharmaceuticals, and Roche/Genentech and has stock options in Oxeia Biopharmaceuticals and Epivario. RP received a research grant and is paid for his editorial work on the journal Complex Psychiatry. All other authors report no biomedical financial interests or potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maihofer, A.X., Ratanatharathorn, A., Hemmings, S.M.J. et al. Effects of genetically predicted posttraumatic stress disorder on autoimmune phenotypes. Transl Psychiatry 14, 172 (2024). https://doi.org/10.1038/s41398-024-02869-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-024-02869-0