Abstract
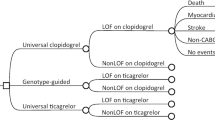
Current guidelines recommend dual antiplatelet therapy (DAPT) consisting of aspirin and a P2Y12 inhibitors following percutaneous coronary intervention (PCI). CYP2C19 genotype can guide DAPT selection, prescribing ticagrelor or prasugrel for loss-of-function (LOF) allele carriers (genotype-guided escalation). Cost-effectiveness analyses (CEA) are traditionally grounded in clinical trial data. We conduct a CEA using real-world data using a 1-year decision-analytic model comparing primary strategies: universal empiric clopidogrel (base case), universal ticagrelor, and genotype-guided escalation. We also explore secondary strategies commonly implemented in practice, wherein all patients are prescribed ticagrelor for 30 days post PCI. After 30 days, all patients are switched to clopidogrel irrespective of genotype (nonguided de-escalation) or to clopidogrel only if patients do not harbor an LOF allele (genotype-guided de-escalation). Compared with universal clopidogrel, both universal ticagrelor and genotype-guided escalation were superior with improvement in quality-adjusted life years (QALY’s). Only genotype-guided escalation was cost-effective ($42,365/QALY) and demonstrated the highest probability of being cost-effective across conventional willingness-to-pay thresholds. In the secondary analysis, compared with the nonguided de-escalation strategy, although genotype-guided de-escalation and universal ticagrelor were more effective, with ICER of $188,680/QALY and $678,215/QALY, respectively, they were not cost-effective. CYP2C19 genotype-guided antiplatelet prescribing is cost-effective compared with either universal clopidogrel or universal ticagrelor using real-world implementation data. The secondary analysis suggests genotype-guided and nonguided de-escalation may be viable strategies, needing further evaluation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wiviott SD. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2007;357:2001–15.
Wallentin L, Becker RC, Budaj A, Cannon CP, Emanuelsson H, Held C, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361:1045–57.
Benjamin EJ, Virani SS, Callaway CW, Chamberlain AM, Chang AR, Cheng S, et al. Heart disease and stroke statistics: 2018 update: a report from the American Heart Association. Circulation. 2018;137:e67–e492.
Levine GN, Bates ER, Blankenship JC, Bailey SR, Bittl JA, Cercek B, et al. 2015 ACC/AHA/SCAI focused update on primary percutaneous coronary intervention for patients with ST-elevation myocardial infarction: an update of the 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention and the 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines and the society for cardiovascular angiography and interventions. Circulation. 2016;133:1135–47.
Fan W, Plent S, Prats J, Deliargyris EN. Trends in P2Y12 inhibitor use in patients referred for invasive evaluation of coronary artery disease in contemporary US practice. Am J Cardiol. 2016;117:1439–43.
Mega JL, Simon T, Collet JP, Anderson JL, Antman EM, Bliden K, et al. Reduced-function CYP2C19 genotype and risk of adverse clinical outcomes among patients treated with clopidogrel predominantly for PCI: a meta-analysis. J Am Med Assoc. 2010;304:1821–30.
Mega JL, Close SL, Wiviott SD, Shen L, Hockett RD, Brandt JT, et al. Cytochrome P450 genetic polymorphisms and the response to prasugrel: relationship to pharmacokinetic, pharmacodynamic, and clinical outcomes. Circulation. 2009;119:2553–60.
Wallentin L, James S, Storey RF, Armstrong M, Barratt BJ, Horrow J, et al. Effect of CYP2C19 and ABCB1 single nucleotide polymorphisms on outcomes of treatment with ticagrelor versus clopidogrel for acute coronary syndromes: a genetic substudy of the PLATO trial. Lancet. 2010;376:1320–8.
Abdel-Qadir H, Roifman I, Wijeysundera HC. Cost-effectiveness of clopidogrel, prasugrel and ticagrelor for dual antiplatelet therapy after acute coronary syndrome: a decision-analytic model. CMAJ Open. 2015;3:E438–46.
Wang WE, Wang H, Wang X, Zeng C. A transition of P2Y12 antagonists for acute coronary syndrome: benefits, risks and costs. J Thromb Thrombolysis. 2014;37:102–6.
Liew D, De Abreu Lourenco R, Adena M, Chim L, Aylward P. Cost-effectiveness of 12-month treatment with ticagrelor compared with clopidogrel in the management of acute coronary syndromes. Clin Ther. 2013;35:1110–.e1119.
Crespin DJ, Federspiel JJ, Biddle AK, Jonas DE, Rossi JS. Ticagrelor versus genotype-driven antiplatelet therapy for secondary prevention after acute coronary syndrome: a cost-effectiveness analysis. Value Health. 2011;14:483–91.
Jiang M, You JH. Cost-effectiveness analysis of personalized antiplatelet therapy in patients with acute coronary syndrome. Pharmacogenomics. 2016;17:701–13.
Johnson SG, Gruntowicz D, Chua T, Morlock RJ. Financial analysis of CYP2C19 genotyping in patients receiving dual antiplatelet therapy following acute coronary syndrome and percutaneous coronary intervention. J Manag Care Spec Pharm. 2015;21:552–7.
Kazi DS, Garber AM, Shah RU, Dudley RA, Mell MW, Rhee C, et al. Cost-effectiveness of genotype-guided and dual antiplatelet therapies in acute coronary syndrome. Ann Intern Med. 2014;160:221–32.
Scott SA, Sangkuhl K, Stein CM, Hulot JS, Mega JL, Roden DM, et al. Clinical Pharmacogenetics Implementation Consortium guidelines for CYP2C19 genotype and clopidogrel therapy: 2013 update. Clin Pharm Ther. 2013;94:317–23.
Empey PE, Stevenson JM, Tuteja S, Weitzel KW, Angiolillo DJ, Beitelshees AL, et al. Multisite investigation of strategies for the implementation of CYP2C19 genotype-guided antiplatelet therapy. Clin Pharm Ther. 2018;104:664–74.
Cavallari LH, Beitelshees AL, Blake KV, Dressler LG, Duarte JD, Elsey A, et al. The IGNITE Pharmacogenetics Working Group: an opportunity for building evidence with pharmacogenetic implementation in a real-world setting. Clin Transl Sci. 2017;10:143–6.
Harada S, Zhou Y, Duncan S, Armstead AR, Coshatt GM, Dillon C, et al. Precision medicine at the University of Alabama at Birmingham: laying the foundational processes through implementation of genotype-guided antiplatelet therapy. Clin Pharm Ther. 2017;102:493–501.
Cavallari LH, Lee CR, Beitelshees AL, Cooper-DeHoff RM, Duarte JD, Voora D, et al. Multisite investigation of outcomes with implementation of CYP2C19 genotype-guided antiplatelet therapy after percutaneous coronary intervention. JACC Cardiovasc Inter. 2018;11:181–91.
Sonnenberg FA, Beck JR. Markov models in medical decision making: a practical guide. Med Decis Mak. 1993;13:322–38.
De Luca L, D’Ascenzo F, Musumeci G, Saia F, Parodi G, Varbella F, et al. Incidence and outcome of switching of oral platelet P2Y12 receptor inhibitors in patients with acute coronary syndromes undergoing percutaneous coronary intervention: the SCOPE registry. EuroIntervention. 2017;13:459–66.
Zettler ME, Peterson ED, McCoy LA, Effron MB, Anstrom KJ, Henry TD, et al. Switching of adenosine diphosphate receptor inhibitor after hospital discharge among myocardial infarction patients: insights from the treatment with adenosine diphosphate receptor inhibitors: longitudinal assessment of treatment patterns and events after acute coronary syndrome (TRANSLATE-ACS) observational study. Am Heart J. 2017;183:62–8.
Lee CR, Sriramoju VB, Cervantes A, Howell LA, Varunok N, Madan S, et al. Clinical outcomes and sustainability of using CYP2C19 genotype–guided antiplatelet therapy after percutaneous coronary intervention. Circulation. 2018;11:e002069.
Cannon CP, Harrington RA, James S, Ardissino D, Becker RC, Emanuelsson H, et al. Comparison of ticagrelor with clopidogrel in patients with a planned invasive strategy for acute coronary syndromes (PLATO): a randomised double-blind study. Lancet. 2010;375:283–93.
Yusuf S, Zhao F, Mehta SR, Chrolavicius S, Tognoni G, Fox KK. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med. 2001;345:494–502.
Held C, Asenblad N, Bassand JP, Becker RC, Cannon CP, Claeys MJ, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes undergoing coronary artery bypass surgery: results from the PLATO (Platelet Inhibition and Patient Outcomes) trial. J Am Coll Cardiol. 2011;57:672–84.
Mega JL, Close SL, Wiviott SD, Shen L, Walker JR, Simon T, et al. Genetic variants in ABCB1 and CYP2C19 and cardiovascular outcomes after treatment with clopidogrel and prasugrel in the TRITON-TIMI 38 trial: a pharmacogenetic analysis. Lancet. 2010;376:1312–9.
Yeh RW, Sidney S, Chandra M, Sorel M, Selby JV, Go AS. Population trends in the incidence and outcomes of acute myocardial infarction. N Engl J Med. 2010;362:2155–65.
Fox KA, Mehta SR, Peters R, Zhao F, Lakkis N, Gersh BJ, et al. Benefits and risks of the combination of clopidogrel and aspirin in patients undergoing surgical revascularization for non-ST-elevation acute coronary syndrome: the Clopidogrel in Unstable angina to prevent Recurrent ischemic Events (CURE) Trial. Circulation. 2004;110:1202–8.
Fryback DG, Dasbach EJ, Klein R, Klein BE, Dorn N, Peterson K, et al. The Beaver Dam Health Outcomes Study: initial catalog of health-state quality factors. Med Decis Mak. 1993;13:89–102.
Tsevat J, Goldman L, Soukup JR, Lamas GA, Connors KF, Chapin CC, et al. Stability of time-tradeoff utilities in survivors of myocardial infarction. Med Decis Mak. 1993;13:161–5.
Bamezai A, Melnick G, Nawathe A. The cost of an emergency department visit and its relationship to emergency department volume. Ann Emerg Med. 2005;45:483–90.
Healthcare Cost and Utilization Project. Overview of the nationwide inpatient sample. www.hcup-us.ahrq.gov/nisoverview.jsp. Accessed 5 Jan 2019.
Neumann PJ, Cohen JT, Weinstein MC. Updating cost-effectiveness-the curious resilience of the $50,000-per-QALY threshold. N Engl J Med. 2014;371:796–7.
Saltelli A. Making best use of model evaluations to compute sensitivity indices. Computer Phys Commun. 2002;145:280–97.
Keeling NJ, Rosenthal MM, West-Strum D, Patel AS, Haidar CE, Hoffman JM. Preemptive pharmacogenetic testing: exploring the knowledge and perspectives of US payers. Genet Med. 2019;21:1224–32.
Hicks JK, Stowe D, Willner MA, Wai M, Daly T, Gordon SM, et al. Implementation of clinical pharmacogenomics within a large health system: from electronic health record decision support to consultation services. Pharmacotherapy. 2016;36:940–8.
Guzauskas GF, Hughes DA, Bradley SM, Veenstra DL. A risk-benefit assessment of prasugrel, clopidogrel, and genotype-guided therapy in patients undergoing percutaneous coronary intervention. Clin Pharm Ther. 2012;91:829–37.
Reese ES, Daniel Mullins C, Beitelshees AL, Onukwugha E. Cost-effectiveness of cytochrome P450 2C19 genotype screening for selection of antiplatelet therapy with clopidogrel or prasugrel. Pharmacotherapy. 2012;32:323–32.
Borse MS, Dong OM, Polasek MJ, Farley JF, Stouffer GA, Lee CR. CYP2C19-guided antiplatelet therapy: a cost-effectiveness analysis of 30-day and 1-year outcomes following percutaneous coronary intervention. Pharmacogenomics. 2017;18:1155–66.
Claassens DMF, Vos GJA, Bergmeijer TO, Hermanides RS, van’t Hof AWJ, van der Harst P, et al. A genotype-guided strategy for oral P2Y12 inhibitors in primary PCI. N Engl J Med. 2019;381:1621–31.
Deiman BA, Tonino PA, Kouhestani K, Schrover CE, Scharnhorst V, Dekker LR, et al. Reduced number of cardiovascular events and increased cost-effectiveness by genotype-guided antiplatelet therapy in patients undergoing percutaneous coronary interventions in the Netherlands. Neth Heart J. 2016;24:589–99.
Antman EM, Wiviott SD, Murphy SA, Voitk J, Hasin Y, Widimsky P, et al. Early and late benefits of prasugrel in patients with acute coronary syndromes undergoing percutaneous coronary intervention: a TRITON-TIMI 38 (TRial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet InhibitioN with Prasugrel-Thrombolysis In Myocardial Infarction) analysis. J Am Coll Cardiol. 2008;51:2028–33.
Becker RC, Bassand JP, Budaj A, Wojdyla DM, James SK, Cornel JH, et al. Bleeding complications with the P2Y12 receptor antagonists clopidogrel and ticagrelor in the PLATelet inhibition and patient Outcomes (PLATO) trial. Eur Heart J. 2011;32:2933–44.
Sibbing D, Aradi D, Jacobshagen C, Gross L, Trenk D, Geisler T, et al. Guided de-escalation of antiplatelet treatment in patients with acute coronary syndrome undergoing percutaneous coronary intervention (TROPICAL-ACS): a randomised, open-label, multicentre trial. Lancet. 2017;390:1747–57.
Martin J, Williams AK, Klein MD, Sriramoju VB, Madan S, Rossi JS, et al. Frequency and clinical outcomes of CYP2C19 genotype-guided escalation and de-escalation of antiplatelet therapy in a real-world clinical setting. Genet Med. 2020;2:160–9.
Cuisset T, Deharo P, Quilici J, Johnson TW, Deffarges S, Bassez C, et al. Benefit of switching dual antiplatelet therapy after acute coronary syndrome: the TOPIC (timing of platelet inhibition after acute coronary syndrome) randomized study. Eur Heart J. 2017;38:3070–8.
Holmes MV, Perel P, Shah T, Hingorani AD, Casas JP. CYP2C19 genotype, clopidogrel metabolism, platelet function, and cardiovascular events: a systematic review and meta-analysis. J Am Med Assoc. 2011;306:2704–14.
Doll JA, Neely ML, Roe MT, Armstrong PW, White HD, Prabhakaran D, et al. Impact of CYP2C19 metabolizer status on patients with ACS treated with prasugrel versus clopidogrel. J Am Coll Cardiol. 2016;67:936–47.
Pare G, Mehta SR, Yusuf S, Anand SS, Connolly SJ, Hirsh J, et al. Effects of CYP2C19 genotype on outcomes of clopidogrel treatment. N Engl J Med. 2010;363:1704–14.
Sorich MJ, Vitry A, Ward MB, Horowitz JD, McKinnon RA. Prasugrel vs. clopidogrel for cytochrome P450 2C19-genotyped subgroups: integration of the TRITON-TIMI 38 trial data. J Thromb Haemost. 2010;8:1678–84.
Esteve-Pastor MA, Ruiz-Nodar JM, Orenes-Pinero E, Rivera-Caravaca JM, Quintana-Giner M, Veliz-Martinez A, et al. Temporal trends in the use of antiplatelet therapy in patients with acute coronary syndromes. J Cardiovasc Pharm Ther. 2018;23:57–65.
Bonaca MP, Bhatt DL, Oude Ophuis T, Steg PG, Storey R, Cohen M, et al. Long-term tolerability of ticagrelor for the secondary prevention of major adverse cardiovascular events: a secondary analysis of the PEGASUS-TIMI 54 trial. JAMA Cardiol. 2016;1:425–32.
Wiviott SD, Braunwald E, McCabe CH, Horvath I, Keltai M, Herrman JP, et al. Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON-TIMI 38 trial: a subanalysis of a randomised trial. Lancet. 2008;371:1353–63.
Garg P, Cohen DJ, Gaziano T, Mauri L. Balancing the risks of restenosis and stent thrombosis in bare-metal versus drug-eluting stents: results of a decision analytic model. J Am Coll Cardiol. 2008;51:1844–53.
Centers for Medicare & Medicaid Services. Medicare provider analysis and review (MEDPAR) file. www.cms.gov/Research-Statistics-Data-and-Systems/Files-for-Order/IdentifiableDataFiles/. Accessed 5 Jan 2019.
Patrick AR, Avorn J, Choudhry NK. Cost-effectiveness of genotype-guided warfarin dosing for patients with atrial fibrillation. Circ Cardiovasc Qual Outcomes. 2009;2:429–36.
Schleinitz MD, Weiss JP, Owens DK. Clopidogrel versus aspirin for secondary prophylaxis of vascular events: a cost-effectiveness analysis. Am J Med. 2004;116:797–806.
Cohen DJ, Breall JA, Ho KK, Kuntz RE, Goldman L, Baim DS, et al. Evaluating the potential cost-effectiveness of stenting as a treatment for symptomatic single-vessel coronary disease. Use of a decision-analytic model. Circulation. 1994;89:1859–74.
Freeman JV, Zhu RP, Owens DK, Garber AM, Hutton DW, Go AS, et al. Cost-effectiveness of dabigatran compared with warfarin for stroke prevention in atrial fibrillation. Ann Intern Med. 2011;154:1–11.
Acknowledgements
This work was supported by grants from the National Institutes of Health: RO1HL092173 and K24HL133373, Clinical and Translational Science Award UL1TR000165, University of Alabama Birmingham’s Health Service Foundations’ General Endowment Fund and Hugh Kaul Personalized Medicine Institute (NAL, CD); U01 HG007269, U01 GM074492 and U01 HL105198, UL1 TR000064 and UL1 TR001427 (LHC, YG, JAJ); U01 HG007762 (AMH, TCS), K23 GM112014 (JDD), UL1 TR001857, an Anonymous Donor, and internal funds from UPMC, the University of Pittsburgh Clinical and Translational Science Institute, and the Institute for Precision Medicine (PEE), U01HG007775 (ALB) NIH Common Fund Program in Health Economics; NHGRI 1R01HG009694–01and NHLBI U01HL122904 (JFP).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
JFP is a consultant for Color Genomics Inc. No other authors have any conflicts of interest to declare.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Limdi, N.A., Cavallari, L.H., Lee, C.R. et al. Cost-effectiveness of CYP2C19-guided antiplatelet therapy in patients with acute coronary syndrome and percutaneous coronary intervention informed by real-world data. Pharmacogenomics J 20, 724–735 (2020). https://doi.org/10.1038/s41397-020-0162-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41397-020-0162-5
This article is cited by
-
Clinical impact of preemptive pharmacogenomic testing on antiplatelet therapy in a real-world setting
European Journal of Human Genetics (2024)
-
Economic evaluation of pharmacogenomic-guided antiplatelet treatment in Spanish patients suffering from acute coronary syndrome participating in the U-PGx PREPARE study
Human Genomics (2023)
-
Safety and Efficacy of CYP2C19 Genotype-Guided Escalation of P2Y12 Inhibitor Therapy After Percutaneous Coronary Intervention in Chronic Kidney Disease: a Post Hoc Analysis of the TAILOR-PCI Study
Cardiovascular Drugs and Therapy (2022)
-
Cost Effectiveness of a CYP2C19 Genotype-Guided Strategy in Patients with Acute Myocardial Infarction: Results from the POPular Genetics Trial
American Journal of Cardiovascular Drugs (2022)
-
Cost-effectiveness of CYP2C19-guided antiplatelet therapy for acute coronary syndromes in Singapore
The Pharmacogenomics Journal (2021)