Abstract
Introduction
Klippel–Feil Syndrome is the congenital fusion of at least two cervical vertebrae. Often asymptomatic, though in rare cases it may lead to severe cervical spine deformity and neurologic injury.
Case presentation
We report a case of a 48-year-old woman with a history of Klippel–Feil Syndrome and congenital scoliosis who developed progressive cervical myelopathy. She was surgically treated with anterior C5 corpectomy and arthrodesis. Pre-operative evaluation was facilitated by 3D printed models. The surgical decompression and spinal reconstruction was completed with the use of a patient-specific, custom-made cervical spine locking plate.
Discussion
Pre-operative evaluation with 3D printing technology was useful in understanding the patient’s complex curve pattern and in designing a patient specific implant. Custom designed implant is a reasonable option to treat cervical myelopathy associated with complex cervical deformity.
Similar content being viewed by others
Introduction
Klippel–Feil Syndrome (KFS), first described in 1912 by Klippel and Feil [1] is the congenital fusion of two or more cervical vertebrae and may be associated with additional congenital changes in the thoracic or lumbar spine [2]. Other clinical features include a short neck, limited cervical range of motion, and a low hairline [3]. However, most patients will not exhibit all of these clinical features [2].
The extent of the cervical anomalies is highly variable and has been classified into Type I (single fused segment), Type II (multiple noncontiguous fusions), and Type III (multiple contiguous fusions) [4]. The congenital cervical fusions may be asymptomatic and identified incidentally [5]. Other patients may present with significant cervical spine deformity and rarely patients may develop cervical myelopathy [3, 6, 7].
We present the case of a patient with severe cervical deformity and progressive cervical myelopathy related to KFS. Due to her markedly abnormal anatomy, surgical planning and decompression and fusion were facilitated by the use of a 3D printed model for bettering anatomic understanding and a patient-specific cervical spine locking plate (CSLP) was used to better match the patient’s complex deformity.
Case presentation
A 48-year-old woman was diagnosed with KFS and congenital scoliosis during infancy. She was initially braced between the ages of two and five, subsequently undergoing posterior fusion of the lower cervical and upper thoracic spine. In adulthood she noted intermittent and progressing neck pain, headaches, bilateral upper extremity paresthesia, and worsening difficulty with balance and gait concerning for cervical myelopathy. Her examination revealed 4/5 iliopsoas weakness bilaterally as well as 4/5 hand intrinsic weakness. Examination of the left hand is difficult due to congenital differences in the hand. The rest of her exam demonstrated full strength. She does have numbness and tingling in both hands in a nonspecific dermatomal distribution. Gait was analyzed with traditional inline gait assessment, tandem gait (one foot over the other), and heel and toe walking. She performed poorly with inline gait and heel and toe walking due to decreased balanced. She was able to perform heel and toe walking, though she had a significantly unsteady gait pattern. She was unable to perform tandem gait. She had a positive Romberg sign. She had a positive Hoffman and intrinsic wasting on the right. We were unable to fully assess the left hand due ot her congenital differences on that side. She had negative Babinski bilaterally. She did have hyperreflexia in the bilateral lower extremities bilaterally. The patient had intact bowel and bladder function and her spinal cord injury was rated as ASIA impairment scale D.
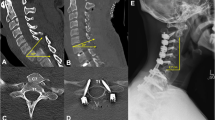
Radiographs of the cervical spine demonstrated congenital scoliosis of the cervical spine with left sided cervicothoracic curve measuring 67 degrees (Fig. 1). Congenital fusions were present at C2–C4 and C6–C7 (Fig. 2). MRI revealed severe stenosis with severe cord compression behind the C5 body resulting from her deformity and degenerative changes (Fig. 3). An EMG demonstrated bilateral C5-6 radiculopathies. MRI evaluation demonstrated significant cord compression with T2 hyperintense signal changes within the cord at the level of C5 body and C4–C5 disc consistent with her symptoms of cervical myelopathy.
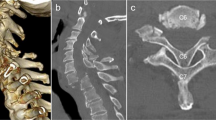
A 3D printed spine model was created from a high-resolution CT scan of the cervical spine for surgical planning. Due congenitally altered posterior elements and limited options for posterior fixation, the surgical plan was made for an anterior-only approach. Considering the degree of her myelopathic symptoms, as well as the rigidity of her deformity, the primary goal of surgery was decompression of the spinal cord rather than deformity correction. In order to facility fusion, a custom-made, patient-specific CSLP (Depuy Synthes, Warsaw, IN) was constructed based upon the CT and 3D printed model to match the patient’s unique anatomy (Fig. 4). The patient underwent C5 corpectomy and bilateral foraminotomies at C4–5 and C5–6. Arthrodesis was performed between C4 and C6 with the use of tricortical iliac crest allograft. The custom-made, patient-specific CSLP was then applied to the anterior of the spine and secured to the C3 and C7 vertebral bodies (Fig. 4). Throughout the case, the 3D model was used to assist in identifying appropriate anatomic landmarks. In particular, the patient’s cervical deformity led to aberrant paths of the vertebral and carotid arteries, which were more easily identified with reference to the 3D printed model.
A 3D printed model of the patient’s cervical spine. The custom-made, patient-specific cervical spine locking plate (CSLP) fits onto the model. A paperclip has been passed through the transverse foramen to visualize the course of the vertebral arteries. B Tricortical iliac crest allograft filling the C5 corpectomy defect. C The custom CSLP is implanted in appropriate alignment and fully conforms to the patient’s anatomy.
The patient was last seen at 9 months after the surgery (Figs. 1 and 5). At last follow-up she reported no neck pain denied issues with dysphagia or dysphonia. She had 5/5 strength in lower extremities to manual motor testing except for just some mild weakness of her hand intrinsics related to her congenital differences on the left side. No significant upper motor neuron findings on examination. She reported marked improvement in her extremity numbness and tingling as well as her balance and gait.
Discussion
In the current case, a patient with congenital scoliosis in the setting of KFS and prior spinal fusion presented with markedly abnormal anatomy resulting in progressive cervical myelopathy. The complex anatomy greatly increased the technical difficulty of performing cervical decompression and fusion. An anatomic 3D printed model was used to assist with detailed anatomic understanding of the patient’s specific pathology and to aid in surgical planning.
There has been growing interest in the application of both 3D models and custom implants for surgical management of patients with complex spinal pathology [8]. 3D models have been shown to improve surgeon understanding of spine deformity when compared to imaging alone [9]. Surgeons who were surveyed following the use of 3D models for complex deformity and spine tumor cases reported that the models allowed for better anatomic visualization and surgical planning [9]. The use of 3D models in the pre-operative planning stages can facilitate improved intra-operative workflow to reduce surgical time and help reduce potential intra-operative mistakes [10]. Better anatomic understanding of by reference 3D models, both pre-operatively and intra-operative, can help improve patient safety by reducing the probability of encountering unexpected anatomic variations [10]. In addition, the use of 3D printed models may also help improve the accuracy of placing instrumentation. A comparison of the accuracy of free-hand pedicle screw placement in patients with congenital scoliosis demonstrated improved accuracy of the pedicle screw placement [11].
Patient-specific implants have also shown increasing use in spine surgery, particularly in complex spine deformity or oncologic reconstructive procedures [12, 13]. Improved anatomic conformity of patient specific implants allows appropriate implant alignment and maximize bone-implant surface to minimize stress risers when off-the-shelf implants are otherwise unable to adequately match the anatomy [12, 14]. A systematic review of the literature demonstrated that the use of patient-specific implant may help reduce the risk of implant associated failures in the cases of significantly altered anatomy [12].
The current case shows a rare case of cervical myelopathy associated with severe cervical spine deformity in a patient with KFS. A patient-specific implant allowed for optimal fit of the cervical plate given the patient’s complex deformity, which otherwise precluded the use of a standard, off-the-shelf implant. The use of a 3D printed model allowed for understanding of the patient’s complex deformity to limit the chance of damaging nearby structures and improve patient safety. The case demonstrates how 3D printed models and patient-specific implants may be useful tools for the surgeon when dealing with the challenges associated with complex spine pathologies.
References
Klippel, Feil A. Un cas d’absence des vertébres cervicales cage thoracique remontant jusqu’a la base du crâne. Bull et Mémoires de la Société d’Anthropologie de Paris. 1912;3:101–2.
Hensinger RN, Lang JE, MacEwen GD. Klippel-Feil syndrome; a constellation of associated anomalies. J Bone Jt Surg Am. 1974;56:1246–53.
Chen SY, Wang PY. Cervical myelopathy in Klippel-Feil syndrome. Acta Neurol Taiwan. 2008;17:217–8.
Samartzis DD, Herman J, Lubicky JP, Shen FH. Classification of congenitally fused cervical patterns in Klippel-Feil patients: epidemiology and role in the development of cervical spine-related symptoms. Spine (Philos Pa 1976). 2006;31:E798–804.
Gruber J, Saleh A, Bakhsh W, Rubery PT, Mesfin A. The Prevalence of Klippel-Feil Syndrome: A Computed Tomography-Based Analysis of 2,917 Patients. Spine Deform. 2018;6:448–53.
Ducker TB. Cervical myeloradiculopathy: Klippel-Feil deformity. J Spinal Disord. 1990;3:439–40. discussion 441-4
Hsieh M-H, Yeh K-T, Chen H, Yu T-C, Peng C-H, Liu K-L, et al. Cervical Klippel-Feil syndrome progressing to myelopathy following minor trauma. Tzu Chi Med J. 2014;26:47–50.
Mobbs RJ, Coughlan M, Thompson R, Sutterlin CE, Phan K. The utility of 3D printing for surgical planning and patient-specific implant design for complex spinal pathologies: case report. J Neurosurg: Spine. 2017;26:513–8.
Izatt MT, Thorpe PL, Thompson RG, D’Urso PS, Adam CJ, Earwaker JW, et al. The use of physical biomodelling in complex spinal surgery. Eur Spine J. 2007;16:1507–18.
Parr WCH, Burnard JL, Wilson PJ, Mobbs RJ. 3D printed anatomical (bio)models in spine surgery: clinical benefits and value to health care providers. J Spine Surg. 2019;5:549–60.
Wu ZX, Huang LY, Sang HX, Ma ZS, Wan SY, Cui G, et al. Accuracy and safety assessment of pedicle screw placement using the rapid prototyping technique in severe congenital scoliosis. J Spinal Disord Tech. 2011;24:444–50.
Burnard JL, Parr WC, Choy WJ, Walsh WR, Mobbs RJ. 3D-printed spine surgery implants: a systematic review of the efficacy and clinical safety profile of patient-specific and off-the-shelf devices. Eur Spine J. 2020;29:1248–60.
Senkoylu A, Daldal I, Cetinkaya M. 3D printing and spine surgery. J Orthop Surg. 2020;28:2309499020927081.
Wallace N, Schaffer NE, Aleem IS and Patel R. 3D-printed patient-specific spine implants: a systematic review. Clin Spine Surg. 2020;33:400–7.
Author information
Authors and Affiliations
Contributions
(1) TJJ was responsible for literature review, primary drafting of the paper, and creating Figures. (2) BAF assisted with paper drafting and review and project direction. (3) JMM assisted with paper drafting and review as well as 3D model design. (4) BLC assisted with paper drafting and review and was assisting attending surgeon. (5) ANN assisted with paper drafting and review was the primary surgeon and senior author.
Corresponding author
Ethics declarations
Competing interests
(1) TJJ, MD declares no competing interests. (2) BAF, MD reports equity in Clear Choice Therapeutics, Inc, outside the submitted work. (3) JMM, MD declares no competing interests. (4) BLC, MD reports royalties from DePuy Synthes, other from Zimmer Biomet, outside the submitted work; In addition, Dr BLC has a patent with DePuy Synthes with royalties paid, and a patent with Zimmer Biomet with royalties paid. (5) AN, MD reports research support to the institution from pfizer, research support to the institution from premia spine, research and fellowship support to the institution from AO Spine North America, outside the submitted work.
Ethics approval
The Mayo Clinic Institutional Review Board (IRB) acknowledges that in accordance with the Code of Federal Regulations, 45 CFR 46.102, the above noted activity does not require IRB review. IRB Exemption: As a case report of a single patient, in accordance with the Mayo Clinic IRBe and the Code of Federal Regulations, 45 CFR 46.102, the above noted project does not require IRB review. The paper is compliant with HIPAA regulations and no patient identifiers are included.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Jackson, T.J., Freedman, B.A., Morris, J.M. et al. Cervical myelopathy in a patient with Klippel–Feil syndrome treated with a patient-specific custom cervical spine locking plate. Spinal Cord Ser Cases 8, 6 (2022). https://doi.org/10.1038/s41394-022-00478-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41394-022-00478-x