Abstract
Introduction
The etiologies of pediatric spinal cord infarction are commonly cardiovascular problems resulting from hypotensive events from trauma and abdominal aortic surgery. Non-traumatic spinal cord infarction in children is rare and remains difficult to diagnose. We report a case of non-traumatic spinal cord infarction of the conus medullaris in a child who recovered after receiving only rehabilitative treatment.
Case presentation
A 12-year-old female patient experienced sudden low back pain for 2 days, followed by weakness in the lower extremities and difficulties in micturition. On admission, magnetic resonance imaging indicated spinal cord infarction of the conus medullaris. After initial treatment with prednisone and mannitol, a few weeks of intensive rehabilitation was recommended. Physical therapy focused on improving lower limb strength. A plastic solid ankle-foot orthosis was used with Lofstrand crutches throughout the period of rehabilitation. After 2 months of rigorous therapy, she was able to walk independently.
Discussion
Non-traumatic spinal cord infarction of the conus medullaris in children is extremely rare. The current case is unique because it involves a patient who presented with pediatric spinal cord infarction of the conus medullaris and showed remarkable neurological recovery after rehabilitation. The case describes a rare spinal cord infarction in a pediatric patient and emphasizes the importance of providing an accurate diagnosis and treatment.
Similar content being viewed by others
Introduction
Spinal cord infarction in children is very rare. It has various etiologies, and its prognosis is usually poor [1]. In adults, spinal cord infarction tends to be due to aortic pathologies, such as aortic surgery or dissection, fibrocartilaginous embolism, systemic hypotension, vascular malformation, and vasculitis [2]. In children, it is mainly caused by traumatic injury to the spinal cord [3]. Therefore, non-traumatic pediatric spinal cord infarction is extremely rare, and hence, difficult to differentiate from some pathological conditions, such as transverse myelitis, necrotic myelitis, multiple sclerosis, intramedullary arteriovenous malformation, and acute multiple neuropathy [4,5,6].
Among the various levels of spinal cord injury, conus medullaris infarction is very rare for anatomical reasons. At the conus, ischemia is very unlikely due to abundant lateral branching and vascular anastomosis in the conus medullaris [7]. Therefore, few cases of spinal cord infarctions of the conus in children have been reported. We report a rare case of non-traumatic spinal cord infarction of the conus medullaris in a child and her expeditious neurological recovery after receiving rehabilitation therapy.
Case presentation
A 12-year-old female visited the emergency room with complaints of weakness in her lower extremities and voiding difficulties; which were decreased voiding sensation and difficulty in voiding initiation. Her history showed that a month earlier, she had an upper respiratory tract infection that resolved within days after been treated conservatively in a local clinic. Two days before she presented at the emergency room, she complained of weakness in both lower extremities with sudden low back pain.
The initial physical examination of the patient showed a motor grade of 2/5 in the hip flexors, knee extensors, and ankle dorsiflexors, was noted bilaterally; however, sensory function was normal. Bilateral knee jerks and ankle jerks were hypoactive, but there was no pathologic reflex in both legs.
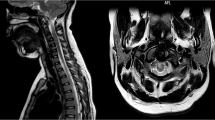
When she was first admitted into the pediatrics department, her pediatricians considered Guillain–Barre syndrome (GBS), acute transverse myelitis, and spinal cord infarction as differential diagnoses based on the patient’s history of sudden weakness in both lower extremities. Non-contrast enhanced magnetic resonance imaging (MRI) of the brain yielded normal results; therefore, they were able to rule out brain-related lesions or diseases that necessarily accompany brain lesions, such as multiple sclerosis and acute disseminated encephalomyelitis (ADEM). GBS was ruled out after a cerebrospinal fluid (CSF) study, which revealed no evidence of albuminocytological dissociation.
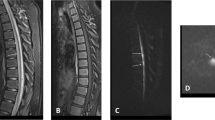
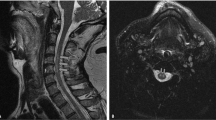
Additional tests including a nerve conduction study (NCS), somatosensory evoked potential study (SSEP), and contrast-enhanced MRI of the whole spine were performed. NCS showed no abnormality in peripheral nerve conduction, while SSEP revealed a central conduction disorder in the posterior column of the spinal cord. Based on these results, she was suspected of having a form of myelopathy, and she had symptoms that were similar to those of spinal cord infarction, such as severe back pain and loss of deep tendon reflexes. MRI revealed T2-weighted hyperintensity with diffusion restriction in the spinal cord from the 12th thoracic vertebra to the conus medullaris, indicating a high probability of spinal cord infarction (Fig. 1). On the MRI, marked diffusion restriction accompanied by swelling of the spinal cord was found in 1–2 vertebral segments, although the CSF analysis was normal. Her symptoms lasted for several seconds to several minutes, so it was possible to diagnose spinal cord infarction rather than acute transverse myelitis. A follow-up CSF study revealed that CSF pressure was >30 cmH2O. She was prescribed 15 mg of oral prednisolone thrice daily for 6 days and 55 mL of intravenous mannitol six times daily for 8 days for decompression, after which her lower limb weakness gradually improved.
On the 26th day of admission, she was transferred to the rehabilitation unit. Results of follow-up examination of motor grades of both legs were as follows: 3/3 for hip flexors, 3/3 for knee extensors, 4/4 for ankle dorsiflexors, 3/4 for long toe extensors, and 1/1 for ankle plantar flexors. Voluntary anal contraction was absent, but the perianal pin prick and light touch were intact. The anal reflex and clitocavernous reflex were absent. The neurological status of the spinal cord lesion was L2 ASIA Impairment Scale D. Urodynamic study revealed acontractile detrusor, and the patient used intermittent catheterization (IC) for voiding, which was performed every 4 h a day. She also had a neurogenic bowel that was controlled with 10 g, once daily, of oral macrogel. She reported neuropathic pain in both legs, which was controlled with 200 mg, once daily, of oral gabapentin.
The calcaneal gait pattern was evident, which was characterized as weakness of the ankle plantar flexors for all gait phases, especially during mid-stance. Therefore, physical therapy focused on gait training with strengthening of the lower extremities. The rehabilitation programs included physical therapy, occupational therapy and function electrical stimulation for 2 h 5 days a week. Intensive gait training and aquatic therapy were performed in physical therapy.
In addition, dorsiflexion-stopped ankle-foot orthoses were donned bilaterally to improve the gait pattern of the patient. As she regained strength, the patient gradually became able to walk using Lofstrand crutches. After 2 months of intensive gait training, she was able to walk independently.
The patient was followed up on an outpatient basis for 6 months post-discharge, and remarkable changes were observed during this period. The motor grades of both leg muscles, especially the ankle plantar flexors, improved to grade 4. Gait performance was also improved accordingly. Voiding and defecation difficulties remained somewhat improved. Post-void residuals have been reduced and voiding frequencies have been improved. The patient was educated to catheterize less intermittently every 6–8 h. Most importantly, she gained self-confidence in all activities of daily living, including attending school.
The study was approved by the institution review board (IRB) of Pusan National University Yangsan Hospital (IRB no. 05-2020-059).
Discussion
The spinal cord is supplied by three longitudinal arteries, namely an anterior spinal artery (ASA) and two posterior spinal arteries (PSAs), that anastomose to form the arterial vasocorona [8]. Originating from the vertebral arteries and medullary segmental arteries that branch out from the aorta, the single ASA supplies the anterior third of the spinal cord, including the central gray matter and anterolateral white matter, while the paired PSAs supplies the posterior third, including the posterior columns [9, 10]. The vasculature of the spinal cord at the level of the conus medullaris has different features compared to the vasculature of the upper thoracic segment [7]. First, it is known that the ASA and PSAs anastomose at this level forming the anastomotic ansa of the conus [11]. Second, the largest segmental medullary artery that branches out into the ASA, also known as artery of Adamkiewicz, branches from an intercostal artery or the internal iliac artery and extends from the T8 to the conus [10]. With this kind of vasculature, the probability that infarction will occur at the conus medullaris is much lower than that at other levels of the spinal cord [7].
Since it is a rare condition, spinal cord infarction in children remains poorly described. Postmortem studies of infantile deaths within 4 weeks of birth revealed ischemic spinal cord damage in 21 out of 900 children [12]. Poor autoregulation of spinal blood flow in premature infants is believed to be a cause of damage [12]. Other studies suggest various causes of pediatric spinal cord infarction, such as achondroplasia, systemic hypotension, arteriovenous malformation, vascular compromise because of aortic dissection, hyperflexion position, cerebellar herniation following lumbar puncture, and fibrocartilaginous embolism [13,14,15,16,17,18]. Imaging technologies, such as diffusion-weighted MRI, have enabled early diagnosis of spinal cord infarction in children, but the causes of spinal infarction in children remains less well-known compared to cases in adults [19].
The differential diagnosis of spinal cord infarction is very diverse and has a common point of acute non-traumatic myelopathy. Diseases to be distinguished mainly include GBS, idiopathic transverse myelitis (ITM), and ADEM [20]. All ADEM patients with spinal cord involvement have cerebral involvement, so simultaneous cerebral imaging can be used to differentiate ADEM from spinal cord infarction [21]. GBS can be diagnosed by CSF analysis when there is an increased CSF total protein level without cellular reaction [22]. ITM is a disease that is especially difficult to differentiate from spinal cord infarction because both disorders have very similar symptoms. ITM is mainly differentiated via MRI findings and the patient’s history. MRI findings of ITM patients commonly show centrally located lesions occupying >2/3 of the cord’s cross-sectional area, which extends to more than three to four vertebral segments in length [23]. The time between the onset of symptoms to the maximum intensity of symptoms in ITM is usually between a few hours to 21 days and CSF findings of pleocytosis or elevated IgG levels are often observed [24]. On the other hand, in cases of spinal cord infarction, pencil-like hyperintensity and cord swelling are observed on MRI, and marked restriction is observed via diffusion-weighted imaging [25]. The time between the onset of symptoms to the peak of spinal cord infarction is commonly several minutes to 12 h [26].
A unique aspect of this case was the occurrence of non-traumatic spinal infarction in a child, and more importantly, conus medullaris infarctions are rare, even in adults. Since there was no evidence of trauma or surgery in this case, the cause of the spinal cord infarction in this case was classified as idiopathic. Fortunately, in this case, an accurate diagnosis was made using SEP, NCS, and contrast-enhanced MRI, and both acute-phase neurological and rehabilitation treatment could be performed. The patient visited the hospital in a bedridden state and was able to walk independently after 6 months of follow-up.
Conus medullaris syndrome in children is rare, even without any history of trauma. In general, the clinical manifestations of conus medullaris syndrome are sudden non-specific low back pain, bilateral pain and weakness in the leg, and accompanying bladder and bowel dysfunction [27]. It also shows signs of lower motor neuron syndrome, including decreased limb reflexes, and flaccid muscles with atrophic changes [28]. The patient showed overall decreased deep tendon reflexes in the lower extremities but had no pathologic reflexes. Comparing to the poor prognosis of conus medullaris infarction in adults, our patient showed good recovery even without antiplatelet or anticoagulant treatment. We focused on improving her walking performance, especially using ankle plantar flexor strengthening.
In some studies, researchers have compared the outcomes of spinal cord infarction in adults with those of children. The percentage of adults that regain the ability to walk independently varies from 11% to 41%; in children, 32% walked independently and an additional 52% were able to walk using aids [29]. Although age has not been definitively shown to affect recovery from spinal cord infarction, the prognoses in pediatric cases appear to be better than those in adults. Similarly, pediatric stroke patients are reported to have better outcomes than adult stroke patients, and this could be attributed to cerebral plasticity. In general, the outcomes of spinal cord infarction are known to be closely related to initial neurological deficits, especially motor deficits or severity of injury [29]. A retrospective study of spinal cord infarction revealed that predictors of prognosis include initial neurological assessment following ASIA/International Medical Society of Paraplegia classification and the patient’s age [30].
Non-traumatic spinal infarction in the conus medullaris is extremely rare. In particular, it is unique that this disease occurs in children who experience traumatic spinal cord infarction much more frequently than non-traumatic spinal cord infarction. To date, there have been few studies on this rare disease, and further studies on its prognosis and risk factors are needed. This case underscores the importance of considering the possibility of conus medullaris infarction in such cases, which may improve outcomes.
References
Robertson CE, Brown RD, Wijdicks EF, Rabinstein AA. Recovery after spinal cord infarcts: long-term outcome in 115 patients. Neurology. 2012;78:114–21.
Mullen M, McGarvey M. Spinal cord infarction: vascular anatomy and etiologies. Waltham, MA: UpToDate; 2015.
Vargas M, Gariani J, Sztajzel R, Barnaure-Nachbar I, Delattre BM, Lovblad KO, et al. Spinal cord ischemia: practical imaging tips, pearls, and pitfalls. Am J Neuroradiol 2015;36:825–30.
Küker W, Weller M, Klose U, Krapf H, Dichgans J, Nägele T. Diffusion-weighted MRI of spinal cord infarction. J Neurol. 2004;251:818–24.
Thron AK. Vascular anatomy of the spinal cord: neuroradiological investigations and clinical syndromes. Berlin: Springer Science & Business Media; 1988.
Masson C, Pruvo J, Meder J, Cordonnier C, Touzé E, De La Sayette V, et al. Spinal cord infarction: clinical and magnetic resonance imaging findings and short term outcome. J Neurol Psychiatry. 2004;75:1431–5.
Wong JJ, Dufton J, Mior SA. Spontaneous conus medullaris infarction in a 79 year-old female with cardiovascular risk factors: a case report. J Can Chiropr Assoc. 2012;56:58.
Hendelman W. Atlas of functional neuroanatomy. 2nd edn. Florida: CRC Press; 2005. p. 296.
Burns S, Mior S, O’Connor S. Cervical spondylotic myelopathy: Part I: anatomical and pathomechanical considerations. J Can Chiropr Assoc 1991;35:75.
Sladky JT, Rorke LB. Perinatal hypoxic/ischemic spinal cord injury. Pediatr Pathol 1986;6:87–101.
Monteiro L, Leite I, Pinto JA, Stocker A. Spontaneous thoracolumbar spinal cord infarction: report of six cases. Acta Neurol Scand 1992;86:563–6.
Sheikh A, Warren D, Childs AM, Russell J, Liddington M, Guruswamy V, et al. Paediatric spinal cord infarction—a review of the literature and two case reports. Child Nerv Syst 2017;33:671–6.
Norman MG. Respiratory arrest and cervical spinal cord infarction following lumbar puncture in meningitis. J Can Chiropr Assoc 1982;9:443–7.
Pang D, Wilberger JE. Spinal cord injury without radiographic abnormalities in children. J Neurosurg 1982;57:114–29.
Riche M, Modenesi-Freitas J, Djindjian M, Merland J. Arteriovenous malformations (AVM) of the spinal cord in children. Neuroradiology. 1982;22:171–80.
Hasegawa M, Yamashita J, Yamashima T, Ikeda K, Fujishima Y, Yamazaki M. Spinal cord infarction associated with primary antiphospholipid syndrome in a young child: case report. J Neurosurg 1993;79:446–50.
Wieting JM, Krach LE. Spinal cord injury rehabilitation in a pediatric achondroplastic patient: case report. Arch Phys Med Rehabil 1994;75:106–8.
Davis GA, Klug GL. Acute-onset nontraumatic paraplegia in childhood: fibrocartilaginous embolism or acute myelitis? Child Nerv Syst 2000;16:551–4.
Beslow LA, Ichord RN, Zimmerman RA, Smith S, Licht D. Role of diffusion MRI in diagnosis of spinal cord infarction in children. Neuropediatrics. 2008;39:188–91.
Boddu SR, Cianfoni A, Kim KW, Banihashemi MA, Pravatà E, Gobin YP, et al. Spinal cord infarction and differential diagnosis. Neurovascular imaging: from basics to advanced concepts. New York: Springer New York; 2014. p. 1–64.
Khong P-L, Ho H-K, Cheng P-W, Wong VC, Goh W, Chan F-LJPr. Childhood acute disseminated encephalomyelitis: the role of brain and spinal cord MRI. Pediatr Radiol 2002;32:59–66.
van Doorn PAJLPM. Diagnosis, treatment and prognosis of Guillain–Barré syndrome (GBS). Presse Méd. 2013;42:e193–201.
Choi KH, Lee KS, Chung SO, Park JM, Kim YJ, Kim HS, et al. Idiopathic transverse myelitis: MR characteristics. Am J Neuroradiol 1996;17:1151–60.
Transverse Myelitis Consortium Working Group. Proposed diagnostic criteria and nosology of acute transverse myelitis. Neurology. 2002;59:499–505.
Weidauer S, Nichtweiss M, Lanfermann H, Zanella FEJN. Spinal cord infarction: MR imaging and clinical features in 16 cases. Neuroradiology. 2002;44:851–7.
Geldmacher DS. Vascular diseases of the nervous system: spinal cord vascular disease. Bradley’s neurology in clinical practice. 6th ed. Philadelphia: Elsevier; 2012.
Ebner FH, Roser F, Acioly MA, Schoeber W, Tatagiba M. Intramedullary lesions of the conus medullaris: differential diagnosis and surgical management. Neurosurg Rev 2009;32:287.
Kingwell SP, Curt A, Dvorak MF. Factors affecting neurological outcome in traumatic conus medullaris and cauda equina injuries. Neurosurg Focus. 2008;25:E7.
Stettler S, El-Koussy M, Ritter B, Boltshauser E, Jeannet PY, Kolditz P, et al. Non-traumatic spinal cord ischaemia in childhood—clinical manifestation, neuroimaging and outcome. Eur J Paediatr Neurol 2013;17:176–84.
de la Barrera SS, Barca-Buyo A, Montoto-Marques A, Ferreiro-Velasco M, Cidoncha-Dans M, Rodriguez-Sotillo A. Spinal cord infarction: prognosis and recovery in a series of 36 patients. Spinal Cord 2001;39:520.
Acknowledgements
This work was supported by a 2-year Research Grant of Pusan National University. We acknowledge Editage (www.editage.com), an English editing service, for editing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Seo, Z.W., Huh, S. & Ko, HY. Non-traumatic spinal cord infarction of the conus medullaris in a child: a case report. Spinal Cord Ser Cases 7, 59 (2021). https://doi.org/10.1038/s41394-021-00425-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41394-021-00425-2