Abstract
Study design
Randomized crossover.
Objectives
To test differences in the duration and magnitude of physiological response to isocaloric moderate intensity continuous (MICE) and high-intensity interval exercise (HIIE) sessions in persons with spinal cord injury (SCI).
Setting
Academic medical center in Miami, FL, USA.
Methods
Ten adult men (mean ± s.d.; 39 ± 10 year old) with chronic (13.2 ± 8.8 year) paraplegia (T2–T10) completed a graded exercise test. Then, in a randomized order, participants completed MICE and HIIE for a cost of 120 kcal. MICE was performed at 24.6% POpeak. During HIIE, exercise was completed in 2 min work and recovery phases at 70%:10% POpeak.
Results
MICE and HIIE were isocaloric (115.9 ± 21.8 and 116.6 ± 35.0 kcal, respectively; p = 0.903), but differed in duration (39.8 ± 4.6 vs 32.2 ± 6.2 min; p < 0.001) and average respiratory exchange ratio (RER; 0.90 ± 0.08 vs 1.01 ± 0.07; p = 0.002). During MICE, a workrate of 24.6 ± 6.7% POpeak elicited a V̇O2 of 53.1 ± 6.5% V̇O2peak (10.1 ± 2.2 ml kg−1 min−1). During HIIE, a workrate at 70% POpeak elicited 88.3 ± 6.7% V̇O2peak (16.9 ± 4.2 ml kg−1 min−1), and 29.4 ± 7.7% of the session was spent at or above 80% V̇O2peak. During HIIE working phase, RER declined from the first to last interval (1.08 ± 0.07 vs 0.98 ± 0.09; p < 0.001), reflecting an initially high but declining glycolytic rate.
Conclusions
Compared with MICE, HIIE imposed a greater physiological stimulus while requiring less time to achieve a target caloric expenditure. Thus, exercise intensity might be an important consideration in the tailoring of exercise prescription to address the cardiometabolic comorbidities of SCI.
Similar content being viewed by others
Introduction
Spinal cord injury (SCI) results in changes in bodily functions that accelerate risk for cardiometabolic disease (CMD) [1]. Specifically, SCI increases risk of cardiometabolic syndrome [2] with a clustering of component risk factors unique to this population [3]. Recently, the Consortium for Spinal Cord Medicine released the first Clinical Practice Guidelines for management of CMD in SCI which recommends ≥150 min of exercise per week [1]. Other recently published population-specific guidelines [4] recommend ≥30 min of moderate-to-vigorous intensity performed three times per week for cardiometabolic health benefits. However, current guidelines do not provide clear instruction regarding exercise intensity. Guidelines that specifically address the important role of exercise intensity would be extremely valuable, especially given the growing body of evidence demonstrating greater improvements in cardiometabolic health outcomes using high-intensity exercise compared with moderate intensity exercise [5].
High-intensity interval exercise (HIIE) is a method for structuring a session of physical activity that involves alternating the intensity of a task through routine work and recovery cycles [6]. A HIIE workout can be accomplished using any mode of rhythmic/endurance exercise. In the general population, HIIE is usually conducted with physical activities that involve large muscle groups, and heart rate (HR) is commonly used to monitor exercise intensity. However, when greater control is desired, a preferred practice is to use ergometry to prescribe HIIE relative to the peak power output (% POpeak) achieved during a prior graded exercise test (GXT). Precise methods of delivering HIIE are especially important in clinical populations where pathophysiology leads to unique responses to exercise and greater exercise risks [7].
Training with HIIE (i.e., HIIT) has been prescribed for some athletes, to enhance specific adaptations related to their physical performance requirements [8], while moderate intensity continuous exercise (MICE) is recommended for the general health benefits of exercise [9]. However, the benefits of HIIE have now been realized in the context of health [7, 10,11,12]. Notably, to achieve some specific physiological adaptations, less time is required when using HIIE than MICE [13]. Furthermore, adaptations to HIIE better target the component risks of CMD than MICE [10,11,12]. While the overwhelming majority of HIIT research involves lower extremity exercise, a recent study in persons without SCI demonstrated that arm cycling HIIT induced superior fitness and performance adaptations compared with training with MICE [14]. In persons with SCI, there is limited evidence suggesting superior adaptations to HIIT in SCI [5], and the few HIIT interventions in SCI are limited by small sample size [15, 16] and short training duration [17]. Furthermore, there is little evidence to guide the selection of HIIT protocol in this population. Knowledge about the acute physiological response to HIIE [18,19,20,21] in persons with SCI can inform the tailoring of HIIT protocols targeting specific components of physiological function.
There is a unique relationship between PO and oxygen consumption (V̇O2) during arm cycling [22], which contributes to unique physiological response to exercise in persons with SCI [23]. Quantification of exercise intensity based on V̇O2 has limited clinical utility and is hardly a comprehensive physiological parameter for understanding the benefits of HIIE. However, quantifying the V̇O2 response to HIIE allows for comparison of the physiological response between different exercise conditions and between populations. Our previous work shows that a relatively low % POpeak is required to elicit a target % V̇O2peak compared with persons without SCI performing leg cycling [23]. For example, we previously showed that during arm ergometry 22% and 49% POpeak elicited 46% and 68% V̇O2peak, respectively [23]. These data suggest that when prescribing HIIE for persons with SCI, ~ 70% POpeak working phases will elicit V̇O2 excursions in intensity to the ≥90% V̇O2peak zone. On the contrary, in persons without SCI performing lower body cycling, 95% POpeak elicited a maximal 90.7% V̇O2peak during HIIE with longer 2 min intervals [24], and 1 min intervals at 90% POpeak elicited responses as low as 77.3% V̇O2peak during the entire work duration depending on the work-to-recovery ratio [25]. Thus, the delivery of HIIE in SCI is best served by a modest alteration to HIIE whereby workrate is slightly reduced compared with “standard” practice. Indeed, of the two most recent studies of acute physiological response to HIIE in SCI, one study showed that a 1 min working phase at 70% POpeak elicited 86.9% V̇O2peak during the last 15 s of work [18], and the other study showed 1 min working phases at 85% POpeak, with longer recovery phases (60:120 s), elicited 86.7% V̇O2peak during the last 15 s of work [19]. In these studies, HIIE was compared with MICE. One study was not matched for total energy expenditure [18] and the other reported no differences in duration of isocaloric bouts of MICE or HIIE [19]. Therefore, the aim of this study was to examine differences in the duration and magnitude of physiological responses to isocaloric MICE and HIIE in persons with SCI.
Methods
This study was conducted as a component of a randomized repeated measures counter-balanced study that was registered with ClinicalTrials.gov (NCT03545867). The protocol has been published in full [26], with trial enrollment and eligibility testing all conducted in accordance with Standard Protocol Items: Recommendations for Interventional Trials guidelines [26]. All procedures were in accordance with, and approved by the Human Subjects Research Office, University of Miami Miller School of Medicine.
Participants
Ten adult males with chronic, neurologically stable thoracic (T1 or lower) non-ambulatory (AIS A-C) SCI participated in this study. Inclusion and exclusion criteria are detailed elsewhere [26]. Descriptive characteristics and basic injury characteristics of the ten men with chronic SCI who completed the trial are presented in Table 1. Participants were of “good” cardiorespiratory fitness (19.2 ± 5.2 ml kg−1 min−1) based on normative classification [27], but fitness varied within the group. Peak HR of 169 ± 16 min−1 suggests that injury did not result in disruption of sympathetic nervous system outflow to the heart.
Baseline assessments and HIIE familiarization
Participants attended two preliminary sessions including baseline assessments and a HIIE familiarization session before completing the two experimental conditions. Participants were instructed to refrain from exercise/alcohol/caffeine for 24 h prior to testing and to arrive at the laboratory normally hydrated (500 ml of water within 1 h of testing). During their first visit, participants’ cardiorespiratory fitness was assessed via a GXT as previously described [26]. All exercise was conducted on a wall-mounted electronically braked arm crank ergometer (Angio CPET, Lode B.V., Groningen, The Netherlands). The GXT was conducted with 3 min stages where PO increased 20 W stage−1 from a starting PO (10–40 W) estimated to elicit volitional exhaustion. During this and all subsequent arm cycling participants were advised to maintain a cadence of ~ 65 rpm but could vary cadence to their liking between 40 and 90 rpm. Data from the last minute of each stage of the GXT were used to generate a PO vs V̇O2 linear regression equation. The data from this individualized equation were used to calculate energy expenditure [28] and thus estimate a PO during MICE that would elicit 50% V̇O2peak for an exercise duration that would result in a total energy expenditure of 120 kcal.
During their second visit, participants completed an HIIE familiarization trial. The aim of our HIIE protocol was to elicit a physiological intensity of >80% V̇O2peak during the working phase, with a peak intensity of ~ 90% V̇O2peak. The cycle ergometer was programmed to vary power output so that a warm-up and cool-down (2 min each) and the recovery phases were completed at 10% POpeak, and the working phases completed at 70% POpeak. The warm and cool-down duration were purposefully short to reduce the contribution of these components of HIIE to the total exercise energy expenditure during HIIE, thus improving accuracy of using HIIE familiarization to estimate HIIE energy expenditure. The work and recovery phases were 2 min each. Our HIIE protocol (70:10% POpeak at 2:2 min) was chosen based on previous literature [18, 19] with the intention of maximizing the time spent at/above 80% V̇O2peak (a more detailed rational for choosing this duration is provided in the discussion). During the HIIE familiarization participants completed a warm-up, three work and recovery cycles, and a cool-down. Expired gas data from this trial were used to compute energy expenditure in order to estimate the duration of HIIE required to match the energy cost of MICE.
Experimental exercise trials
A web tool (http://www.randomization.com) was used to ensure trials were performed in a randomized order. Trails were separated by 2–10 days. Before exercise trials, participants were asked to abstain from strenuous exercise, caffeine, and alcohol for 24 h. On the morning of the trials, participants were instructed to consume ~ 10 ml kg−1 of water and report to the laboratory following an overnight fast (≥10 h). Based on the PO–V̇O2 regression equation, participants conducted 39.8 ± 4.6 min of MICE at 26.1 ± 7.3% POpeak. Expired gas was analyzed breath-by-breath continuous during MICE and HIIE trials. HIIE was conducted in the same manner as in the HIIE familiarization trial for a duration that would elicit a total energy expenditure of 120 kcal. Calculations from the HIIE familiarization trial determined that 32.2 ± 6.2 min of HIIE (5–9 intervals) would be required to expend 120 kcal.
Data analysis
Expired gas data were recorded breath-by-breath and then averaged offline into 20 s windows. For HIIE, data are an average of the entire session or are an average of the last minute of the work and recovery cycles (e.g., Table 2). For the calculation of energy expenditure, the appropriate stoichiometric equations [28] were applied to indirect calorimetry data. These updated equations were calibrated for high-intensity exercise where an estimated 80% of carbohydrate oxidation is attributed to intramuscular glycogen stores [28].
Statistical analysis
Statistical analysis was conducted using IBM’s SPSS (v25, Chicago, IL, USA). To assess reliability of the physiological response to HIIE, intraclass correlation coefficients (ICCs; two-way rand effect, absolute agreement [29]) and Pearson correlation coefficients were computed comparing the HIIE familiarization and the first three intervals of HIIE. Because participants completed HIIE to a calorie target based, the number of intervals each participant completed was different and based on their HIIE familiarization. The differential number of intervals completed by each participant confounded the use of a repeated measures analysis of variance, and thus paired t-tests were used to compare differences in the means between exercise conditions. Normality of distribution was checked via Shapiro–Wilks test, and data were normally distributed (average p = 0.505 for all comparisons reported in Table 2). For HIIE, a paired t-test was also used to compare the first interval to the last interval. Statistical significance was set at an alpha level of p ≤ 0.05.
Results
All participants completed all assessment and exercise sessions as required. No sessions were aborted due to exhaustion, and no adverse events were reported.
The PO–V̇O2 relationship calculated from the GXT was:
Correlation for the PO–V̇O2 and %PO–%V̇O2 relationships were strong (R2 = 0.899 and 0.901, respectively). When comparing the HIIE familiarization session to the beginning of the HIIE session, the test–retest reliability of V̇O2 was acceptable based on ICC (mean = 0.797, range = 0.556–0.942) and Pearson correlation (R = 0.864).
Metabolic and cardiovascular response to exercise
Physiological responses to exercise are presented in Table 2. The total caloric cost of exercise was similar between MICE and HIIE, (115.9 ± 21.8 vs 116.6 ± 35.0 kcal; p = 0.90), although MICE required more time than HIIE to reach this target (39.8 ± 4.6 vs 32.2 ± 6.2 min; p < 0.001). When averaging over the entire MICE or HIIE sessions, the relative intensity for HR (62.3 ± 7.0% vs 73.3 ± 7.7% HRpeak; p = 0.009) and V̇O2 (53.0 ± 6.6% vs 66.1 ± 5.2% V̇O2peak; p < 0.001), respiratory exchange ratio (RER; 0.90 ± 0.08 vs 1.01 ± 0.07; p = 0.002), and rate of energy expenditure (2.90 ± 0.44 vs 3.60 ± 0.66 kcal min−1; p = 0.001) were all lower in MICE than HIIE. During MICE, a 24.6 ± 6.7% POpeak elicited a V̇O2 of 53.1 ± 6.5% V̇O2peak (10.1 ± 2.2 ml kg−1 min−1).
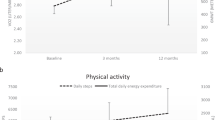
Figure 1 shows the time course of V̇O2, HR, and RER during MICE and HIIE in a representative individual. This participant’s response demonstrates the steady-state physiological response during MICE. Furthermore, Fig. 1 demonstrates the peaks and valleys during HIIE that correspond with working and recovery phases. The fluctuations in this representative individual were typical of the group (Fig. 2 and Table 2). As a group, 70% POpeak work cycle elicited a V̇O2 of 88.3 ± 6.7% V̇O2peak (16.9 ± 4.2 ml kg−1 min−1) during the last 1 min of each interval. V̇O2 recovered to 49.2 ± 6.8% V̇O2peak (9.3 ± 2.2 ml kg−1 min−1), also as an average of the final minute of all recovery cycles. A total of 29.4 ± 7.7% and 33.4 ± 25.9% of the session time was spent at or above 80% V̇O2peak and HRpeak, respectively.
A representative individual’s physiological response throughout the time course of both exercise conditions. HIIE high-intensity interval exercise, MICE moderate intensity continuous exercise, V̇O2 rate of oxygen consumption, HR heart rate, RER respiratory exchange ratio. For the HIIE condition, the gray vertical bars in the plot area represent 2 min work phases (70% POpeak) and the white spaces between denote 2 min recovery phases (70% POpeak). For this individual MICE was conducted at 24.2% POpeak.
Figure 2 shows the change in V̇O2, HR, and RER from the first to last interval. The peak working phase V̇O2 and HR observed in any 20 s time window during HIIE occurred during the last interval. The last interval elicited higher V̇O2 than the first interval (Fig. 2a; 18.7 ± 4.9 vs 16.2 ± 4.1 ml kg−1 min−1; p ≤ 0.001). Furthermore, RER during work and recovery phases was lower in the last interval compared with the first (Fig. 2c).
Discussion
This study provides first evidence that when structured as described, HIIE requires less time than MICE to achieve a target energy cost in persons with SCI. In order to achieve a time-efficient and attainable session, we delivered HIIE with 2 min work and recovery phases and modestly reduced workrate (70%:10% POpeak) compared with “standard” practice with leg cycling in persons without SCI (e.g. [6, 24, 25]).
The results from our GXT showed that when persons with paraplegia are conducting arm cycle exercise, an increase in power by 1 W causes an increase in V̇O2 of 9.59 ± 1.53 ml min−1. Smith et al. showed that the PO–V̇O2 relationship during arm cycling in persons without SCI was 16.2 ml min−1 W−1 [22]. The difference in these findings can be partially accounted for by differences in body mass, with participants in the current study (75.0 kg) being substantially lighter than in Smith (84.7 kg). However, after normalization of the PO–V̇O2 relationship to mass, there is still a 39.7% difference between our findings and those of Smith et al. [22]. This difference indirectly suggests that persons with SCI are more efficient during arm cycling; corroborating our previous findings [23]. If this is true, the increased ACE efficiency is likely due to differences in arm cycling technique and/or adaptations to upper body musculature that occurs due to habitual use of upper extremities in ways uncommon in the general population.
Authoritative exercise guidelines for persons without disability state that HIIE work phases should elicit intensities between 64 and >100% V̇O2peak [10], with health adaptation optimized by intensities >90% V̇O2peak [6]. The HIIE protocol employed in this study achieved V̇O2 excursions into this target intensity zone. However, the clinical utility of V̇O2 measurement is limited [7], and exercise intensity during HIIE is commonly expressed as a percent of peak HR or percent heart rate reserve (%HRR) [11]. The HR response in the current study showed dynamic response to HIIE, with 10.5 ± 8.6 min of the HIIE session spent above 80% HRpeak. Figure 1 allows for comparison of the V̇O2 and HR responses during HIIE from a representative individual (Participant 03 in Table 1). This participant was chosen as the representative because their characteristics are representative of the SCI community at large: they are an obese (BMI = 28.2 kg m−2) [1] middle-aged man of “average” fitness [27]. Figure 1a, b shows a tight coupling between V̇O2 and HR, with a greater dynamic fluctuation in V̇O2, corresponded with HIIE work and recovery phases. MICE elicited a steady-state response without evidence of V̇O2 drift, as observed by others [30]. Figure 1a, b also demonstrates the gradual increase in the highest physiological response during consecutive HIIE working phases. This “treppe” phenomenon was a common feature during HIIE (Fig. 2). Notable in the representative participant’s response is that V̇O2 and HR both exceeded peak values achieved during GXT. This phenomenon was also common, with the highest V̇O2 and HR (observed in a 20 s window) throughout the HIIE sessions being 97.3 ± 8.8% V̇O2peak and 91.4 ± 9.0% HRpeak, respectively (Fig. 2a). It should be noted that the variability in the HR response was greater than that of the V̇O2 response. In certain clinical populations an atypical HR response to exercise can confound the use of HR as a proxy to quantify exercise intensity [7]. Changes in left ventricular global function [31] and the unique V̇O2–HR relationship during arm cycling [32] could have contributed to the greater variability of HR response to HIIE in SCI.
One of the primary benefits of HIIE is that a reduced time commitment is required to achieve a given physiological response [13]. Of the HIIE [18, 19] and sprint interval exercise (105–115% POpeak) [18,19,20,21] studies in SCI, ours is the first to demonstrate a reduced exercise duration required to achieve a target energy expenditure. But, it is important to remember that high-intensity exercise training elicits superior adaptations to moderate intensity despite substantial differences in the total work, and thus energy expenditure, completed in individual exercise sessions [13]. Thus, the comparison of energy expenditure from HIIE and MICE likely overlooks the totality of the potential benefits of HIIE. Indeed, glycogen cycling and disruptions in cellular homeostasis are important considerations for the benefits of high-intensity exercise [33]. These metabolic responses contribute to the energetic requirements of recovery from exercise, measured as excess postexercise oxygen consumption, in a manner dependent on exercise intensity [34], which might be increased in HIIE in SCI. Thus, energy cost, like V̇O2, should be considered a useful but incomplete measurement of the physiological intensity of exercise, and other metabolic parameters should be taken into account when considering HIIE programming. Accordingly, our HIIE protocol was guided by knowledge of the heavy reliance on carbohydrates during exercise in persons with SCI [23]. Due to this heavy reliance on carbohydrates, we anticipated that a “long” [6] recovery phase would facilitate the clearance of metabolic byproducts produced during the working phase; mitigating accumulation throughout each successive interval. Examination of Fig. 1c shows the coupling of RER with working and recovery phase. Furthermore, the highest RER seen during a 20 s window decreased from the first to last bout (Fig. 2c). This dynamic fluctuation during HIIE is common in lower extremity HIIE [35], and reflects a metabolic shift likely reflective of some degree of relative muscle glycogen depletion that is characteristic of HIIE [33]. The total energy expenditure in our HIIE protocol is below what would likely result in relative glycogen depletion during leg exercise in persons without disability. However, the arms are substantially more reliant than the legs on anaerobic metabolism during exercise [36], and relatively “short” (30 min) high-intensity arm exercise has been shown to decrease glycogen concentrations of the triceps and deltoid muscles by 83.4% and 28.0%, respectively [37]. Furthermore, training status has been shown to have little effect on the high reliance on anaerobic metabolism during arm exercise [38], thus the participants in our study likely experienced some degree of relative glycogen depletion. This metabolic challenge, and the accompanying disruption to cellular homeostasis within skeletal muscle that comes with high-intensity exercise, likely has persistent metabolic effects long into the postexercise recovery period that emphasizes glucose uptake and storage and fat oxidation. Thus, shifts in RER seen in HIIE but not MICE are reflective of physiological responses to HIIE that likely confer benefits beyond the mere caloric time-efficiency of HIIE. In persons without SCI adaptations to chronic HIIE training have been shown to improve the ability to use fat during exercise in a variety of context [35], and if similar adaptations to HIIT interventions are shown in SCI then this exercise strategy could be a promising strategy for targeting cardiometabolic risks in this population [3].
Our study is subject to a number of limitations. Most importantly, we did not directly compare different HIIE protocols in order to determine differences in the physiological response to different HIIE paradigms. Thus, this study does not allow for conclusions to be drawn about the optimal HIIE protocol for a target physiological response. There are limitations to using indirect calorimetry to calculate energy expenditure during exercise dominated by anaerobic metabolism [39], and some of the assumptions of the stoichiometry equations [28] were violated during certain parts of HIIE. Furthermore, matching the calorie cost of HIIE and MICE placed artificial constraints on the potential benefits of MICE. It can be argued that MICE has a greater potential capacity for energy expenditure because a greater exercise duration is possible due to the steady-state nature whereas fatigue during HIIE likely limits the capacity for total calorie cost due to exhaustion. However, it should be noted that the exercise intensity used in our study (53% V̇O2peak) was similar to other HIIE publications in SCI [19] making our data comparable to existing literature. Furthermore, long duration MICE could be considered undesirable due to the time commitment and mundane nature of the task. With respect to our population, while the participants in this study had a wide range of physical characteristics and fitness levels (Table 1), 50% of our sample had above-average cardiorespiratory fitness. Thus, the results of our study may be less applicable to persons with SCI who are at the lower end of the cardiorespiratory fitness spectrum [27]. Finally, while autonomic function was not directly tested, our data (Table 1) showed that our participants had retained cardioacceleratory capacity and thus are not likely experiencing the full extent of autonomic impairment that occurs with higher level SCI. Therefore the results of this study cannot necessarily be applied to persons with higher level injuries that result in paralysis of muscles involved in arm cycling along with stark autonomic impairments that predispose an early onset of fatigue due to cardiovascular and neuroendocrine limitations.
Our study is the first to demonstrate in SCI that, when appropriately adjusted, HIIE requires less time to elicit a target calorie expenditure compared with MICE. Furthermore, fluctuations in RER during HIIE, but not MICE, demonstrate differences in substrate partitioning between the two exercise protocols. In order to deliver this sufficiently intense and time-efficient HIIE session in SCI, we used 2 min work and recovery phases prescribed at a workrate (70%:10% POpeak) relatively lower than would be used in persons without disability completing leg cycling. Future studies should determine if differences in the acute physiological response to MICE and HIIE lead to differential adaptations to training interventions using these exercises to target health and fitness.
Data availability
The dataset generated from the current study is available from the corresponding author on reasonable request.
References
Nash MS, Groah SL, Gater DR Jr, Dyson-Hudson TA, Lieberman JA, Myers J, et al. Identification and management of cardiometabolic risk after spinal cord injury: clinical practice guideline for health care providers. Top Spinal Cord Inj Rehabil. 2018;24:379–423.
de Groot S, Adriaansen JJ, Tepper M, Snoek GJ, van der Woude LHV, Post MWM. Metabolic syndrome in people with a long-standing spinal cord injury: associations with physical activity and capacity. Appl Physiol Nutr Me. 2016;41:1190–6.
Libin A, Tinsley EA, Nash MS, Mendez AJ, Burns P, Elrod M, et al. Cardiometabolic risk clustering in spinal cord injury: results of exploratory factor analysis. Top Spinal Cord Inj Rehabil. 2013;19:183–94.
Martin Ginis KA, van der Scheer JW, Latimer-Cheung AE, Barrow A, Bourne C, Carruthers P, et al. Evidence-based scientific exercise guidelines for adults with spinal cord injury: an update and a new guideline. Spinal Cord. 2018;56:308–21.
Nightingale TE, Metcalfe RS, Vollaard NB, Bilzon JL. Exercise guidelines to promote cardiometabolic health in spinal cord injured humans: time to raise the intensity? Arch Phys Med Rehabil. 2017;98:1693–704.
Buchheit M, Laursen PB. High-intensity interval training, solutions to the programming puzzle. Sports Med. 2013;43:927–54.
Nash MS, Groah SL, Gater DR, Dyson-Hudson TA, Lieberman JA, Myers J, et al. Identification and management of cardiometabolic risk after spinal cord injury: clinical practice guideline for health care providers. J Spinal Cord Med. 2019;24:1–35.
Billat LV. Interval training for performance: a scientific and empirical practice—special recommendations for middle- and long-distance running, part I: aerobic interval training. Sports Med. 2001;31:13–31.
Pate RR, Pratt M, Blair SN, Haskell WL, Macera CA, Bouchard C, et al. Physical-activity and public-health—a recommendation from the Centers-for-Disease-Control-and-Prevention and the American-College-of-Sports-Medicine. J Am Med Assoc. 1995;273:402–7.
Campbell WW, Kraus WE, Powell KE, Haskell WL, Janz KF, Jakicic JM, et al. High-intensity interval training for cardiometabolic disease prevention. Med Sci Sports Exerc. 2019;51:1220–6.
Batacan RB Jr., Duncan MJ, Dalbo VJ, Tucker PS, Fenning AS. Effects of high-intensity interval training on cardiometabolic health: a systematic review and meta-analysis of intervention studies. Br J Sports Med. 2017;51:494–503.
Weston KS, Wisloff U, Coombes JS. High-intensity interval training in patients with lifestyle-induced cardiometabolic disease: a systematic review and meta-analysis. Brit J Sport Med. 2014;48:1227–52.
Burgomaster KA, Howarth KR, Phillips SM, Rakobowchuk M, MacDonald MJ, Mcgee SL, et al. Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J Physiol. 2008;586:151–60.
Schoenmakers P, Reed K, Van Der Woude L, Hettinga FJ. High intensity interval training in handcycling: the effects of a 7 week training intervention in able-bodied men. Front Physiol. 2016;7:638.
Harnish CR, Daniels JA, Caruso D. Training response to high-intensity interval training in a 42-year-old man with chronic spinal cord injury. J Spinal Cord Med. 2017;40:246–9.
de Groot PC, Hjeltnes N, Heijboer AC, Stal W, Birkeland K. Effect of training intensity on physical capacity, lipid profile and insulin sensitivity in early rehabilitation of spinal cord injured individuals. Spinal Cord. 2003;41:673–9.
Harnish C, Sabo R, Daniels J, Caruso D. The effects of two weeks of arm crank sprint interval training in men with chronic spinal cord injury. Int J Sports Exerc Med. 2017;3:56–9.
Astorino TA, Thum JS. Within-session responses to high-intensity interval training in spinal cord injury. Disabil Rehabil. 2018;40:444–9.
Astorino TA. Hemodynamic and cardiorespiratory responses to various arm cycling regimens in men with spinal cord injury. Spinal Cord Ser Cases. 2019;5:8.
Graham K, Yarar-Fisher C, Li J, McCully KM, Rimmer JH, Powell D, et al. Effects of high-intensity interval training versus moderate-intensity training on cardiometabolic health markers in individuals with spinal cord injury: a pilot study. Top Spinal Cord Inj Rehabil. 2019;25:248–59.
McLeod JC, Diana H, Hicks AL. Sprint interval training versus moderate-intensity continuous training during inpatient rehabilitation after spinal cord injury: a randomized trial. Spinal Cord. 2020;58:106–15.
Smith PM, Amaral I, Doherty M, Price M, Jones A. The influence of ramp rate on V· O2 peak and “Excess” V· O2 during arm crank ergometry. Int J sports Med. 2006;27:610–6.
Jacobs KA, Burns P, Kressler J, Nash MS. Heavy reliance on carbohydrate across a wide range of exercise intensities during voluntary arm ergometry in persons with paraplegia. J Spinal Cord Med. 2013;36:427–35.
Zafeiridis A, Kounoupis A, Dipla K, Kyparos A, Nikolaidis MG, Smilios I, et al. Oxygen delivery and muscle deoxygenation during continuous, long- and short-interval exercise. Int J Sports Med. 2015;36:872–80.
Gosselin LE, Kozlowski KF, DeVinney-Boymel L, Hambridge C. Metabolic response of different high-intensity aerobic interval exercise protocols. J Strength Cond Res. 2012;26:2866–71.
McMillan DW, Maher JL, Jacobs KA, Mendez AJ, Nash MS, Bilzon JLJ. Influence of upper-body continuous, resistance or high-intensity interval training (CRIT) on postprandial responses in persons with spinal cord injury: study protocol for a randomised controlled trial. Trials. 2019;20:497–50.
Simmons OL, Kressler J, Nash MS. Reference fitness values in the untrained spinal cord injury population. Arch Phys Med Rehab. 2014;95:2272–8.
Jeukendrup AE, Wallis GA. Measurement of substrate oxidation during exercise by means of gas exchange measurements. Int J Sports Med 2005;26(Suppl 1):S28–37.
Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016;15:155–63.
Horiuchi M, Fukuoka Y. Absence of cardiovascular drift during prolonged arm-crank exercise in individuals with spinal cord injury. Spinal Cord. 2019;57:942–52.
Currie KD, West CR, Krassioukov AV. Differences in left ventricular global function and mechanics in paralympic athletes with cervical and thoracic spinal cord injuries. Front Physiol. 2016;7:110.
Vokac Z, Bell H, Bautz-Holter E, Rodahl K. Oxygen uptake/heart rate relationship in leg and arm exercise, sitting and standing. J Appl Physiol. 1975;39:54–9.
Scribbans TD, Edgett BA, Vorobej K, Mitchell AS, Joanisse SD, Matusiak JB, et al. Fibre-specific responses to endurance and low volume high intensity interval training: striking similarities in acute and chronic adaptation. PLoS ONE. 2014;9:e98119.
Børsheim E, Bah R. Effect of exercise intensity, duration and mode on post-exercise oxygen consumption. Sports Med. 2003;33:1037–60.
Hetlelid KJ, Plews DJ, Herold E, Laursen PB, Seiler S. Rethinking the role of fat oxidation: substrate utilisation during high-intensity interval training in well-trained and recreationally trained runners. BMJ Open Sport Exerc Med. 2015;1:e000047.
Ahlborg G, Jensen-Urstad M. Metabolism in exercising arm vs. leg muscle. Clin Physiol. 1991;11:459–68.
Kiilerich K, Birk JB, Damsgaard R, Wojtaszewski JF, Pilegaard H. Regulation of PDH in human arm and leg muscles at rest and during intense exercise. Am J Physiol-Endocrinol Metab. 2008;294:E36–42.
Jensen-Urstad M, Ahlborg G. Is the high lactate release during arm exercise due to a low training status? Clin Physiol. 1992;12:487–96.
Scott CB, Leighton BH, Ahearn KJ, McManus JJ. Aerobic, anaerobic, and excess postexercise oxygen consumption energy expenditure of muscular endurance and strength: 1-set of bench press to muscular fatigue. J Strength Cond Res. 2011;25:903–8.
Acknowledgements
The authors thank Gregory Bigford, Rachel Tan, and Richie Goulding for their contributions to the manuscript.
Funding
This study was funded by the Miami Project to Cure Paralysis.
Author information
Authors and Affiliations
Contributions
DWM contributed to study design, data collection, data organization/analysis, and writing of the manuscript; JLM contributed to study design, data collection, and writing of the manuscript; KAJ contributed to data analysis and writing of the manuscript; MSN contributed to study design and writing of the manuscript; JLJB contributed to study design and writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
We certify that all applicable institutional and governmental regulation concerning the ethical use of human volunteers were following during the course of this research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
McMillan, D.W., Maher, J.L., Jacobs, K.A. et al. Physiological responses to moderate intensity continuous and high-intensity interval exercise in persons with paraplegia. Spinal Cord 59, 26–33 (2021). https://doi.org/10.1038/s41393-020-0520-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-020-0520-9
This article is cited by
-
Exercise intensity during exergaming in wheelchair-dependent persons with SCI
Spinal Cord (2023)
-
Hybrid high-intensity interval training using functional electrical stimulation leg cycling and arm ski ergometer for people with spinal cord injuries: a feasibility study
Pilot and Feasibility Studies (2022)
-
Recent Updates in Nutrition After Spinal Cord Injury: 2015 Through 2021
Current Physical Medicine and Rehabilitation Reports (2022)
-
Exercise for people with SCI: so important but difficult to achieve
Spinal Cord (2021)