Abstract
Study design
Retrospective descriptive study.
Objectives
To determine the incidence and probable etiology of sperm DNA fragmentation (SDF) in a sample of males with spinal cord injury (SCI).
Setting
Hospital in Toledo, Spain; University-based Genetics laboratory in Madrid, Spain.
Methods
Semen collected by vibro-stimulation from 27 males with various levels of spinal cord injury. Classical semen parameters, SDF, leukocytospermia and pro-oxidant capacity were assessed and compared with a cohort of normozoospermic fertile donors (n = 10).
Results
Males with SCI presented with lower semen quality compared with normozoospermic donors with respect to progressive motility (p = 0.0002), SDF (p < 0.00005), pro-oxidant capacity (p = 0.0191) and leukocytospermia (p < 0.00005). Although there was no significant correlation between semen quality and time since the lesion occurred, the period of abstinence appeared to be positively correlated with SDF (r = 0.486; p = 0.041). When the semen parameters of males with SCI were categorized based on those with cervical and thoracic lesions, sperm concentration was higher for those with cervical damage (p = 0.0257). Males with complete lesions (AIS A) had ejaculates that were lower in progressive motility (p = 0.0040) than those with incomplete injuries (AIS B–D).
Conclusions
Ejaculates of males with SCI have excessively elevated SDF when compared with normozoospermic donors, which is likely to be associated with coincident high levels of leucocytospermia and pro-oxidant capacity. We propose that these phenomena are caused by the accumulation and degeneration of spermatozoa in the cauda epididymidis.
Similar content being viewed by others
Introduction
The semen of males with SCI is typically characterized by low sperm motility and viability, variable morphology, and alterations in semen plasma content [1,2,3]. The cause of poor sperm quality in males with SCI, although controversial, is likely to be multifactorial, potentially associated with anejaculation, reproductive tract infections, prostatic dysfunction and/or a failure to regulate scrotal temperature [2, 4, 5]. While the classical semen characteristics of males with SCI have been studied post injury, and these studies have demonstrated no chronic change in semen parameters [3], there are other factors that may be adversely affecting fertility, including changes to structural proteins, mitochondrial activity, and DNA damage [4, 6, 7].
There are numerous studies that relate the presence of sperm DNA damage to male infertility (Reviewed by Rex et al. [8]). These studies suggest that between 30 and 80% of cases of sub-fertility by male factor can be attributed to the damaging effects of oxidative stress. This stress occurs when reactive oxidative species (ROS) exceeds the natural antioxidant capacity of semen, causing cell membrane damage and increasing the rate of DNA fragmentation. The two largest contributors of ROS in semen are leukocytes and spermatozoa, although the production of ROS by leukocytes is up to 1000 times more potent than that attributed to spermatozoa. The superoxide anion is considered to be the principal ROS generated and the primary cause of this damage [9]. While the World Health Organisation [10] initially set a reference standard of leukocytes in the ejaculate at 1.5 × 106 mL−1, there are studies that have shown that even low levels of leukocytospermia can be associated with sperm DNA damage [11].
This retrospective study was conducted to investigate semen characteristics of males with SCI in a reference Hospital in Spain and to compare these data with a control sample of fertile semen donors in an attempt to establish the relationship between SDF, pro-oxidant capacity and leukocytospermia. Our principal aims were to explore the etiology of SDF in males with SCI and to contribute to the database of classical semen characteristics from SCI males, with the additional perspective of pro-oxidant capacity related with the presence of leukocytes. This database is likely to be useful in clinical service to provide improved fertility diagnosis and treatment recommendations.
Methods
Male cohort description
Semen samples were obtained from 27 males with SCI attending the Sexual and Fertility Unit, Hospital Nacional de Parapléjicos de Toledo, Spain (Table 1). For statistical analysis, males with SCI were grouped in two main groups, those presenting with cervical (C) injury (tetraplegia) and those presenting with thoracic (T) injury (paraplegia) and according to the American Spinal Injury Association Impairment Scale (AIS; [12]) with respect to occurrence of a complete lesion (A) or an incomplete lesions (B, C, and D). For a control comparison, fertile semen samples surplus to requirements, from ten normozoospermic donors without SCI, were provided by a clinic in Seville, Spain; SDF analysis of these semen samples was conducted at the Universidad Autónoma de Madrid. The mean (SD) age of males with SCI and the semen normozoospermic donors was 32.1 (9.0) and 30.1 (8.5), respectively. Males with SCI presented with a large variation in the time of abstinence with respect to their last ejaculation; this period ranged from 12 or more months of abstinence (11/27; Table 1) to less than 12 months of abstinence (7/27; Table 1) with a maximum of 68 months and a minimum of 1 month. In nine patients, only one ejaculate was obtained [denoted as not determined (ND) in Table 1]. Considering the large range of abstinence time as an unavoidable possible confounding factor, we conducted a correlation analysis with respect to months of abstinence and the different sperm quality parameters assessed.
Semen sample processing
All semen samples from males with SCI in this study were obtained using vibro-stimulation (Ferticare Personal®, https://medicalvibrator.com) and a subsample of each ejaculate cryopreserved using a slow freezing protocol (Cryosperm, Origio, Malov, Denmark) for later assessment of sperm DNA fragmentation. The remaining sperm parameters (motility, pro-oxidant capacity, and leukocyte concentration) were assessed directly from freshly collected ejaculates ~30 min following liquefaction. Given the large amount of cellular debris in the ejaculate, sperm motility and leukocyte concentration were visually assessed under phase contrast microscopy. To assess pro-oxidant capacity of the whole sperm sample, the Oxisperm II kit (Halotech DNA, Madrid, Spain) was employed. The colorimetric values of the pro-oxidant reaction were obtained after applying image analysis protocols (ImageJ-NIH; https://imagej.nih.gov/ij/index.html) and following mixing of 50 µL of semen with 50 µL of reactive gel containing nitro blue tetrazolium according with the manufacturer’s specifications. The color intensity of the reaction was evaluated using image analysis protocols as gray level units, which has been previously described by de la Casa et al. [13]. Sperm DNA fragmentation was assessed from frozen-thawed samples using Halosperm (Halotech DNA, Madrid, Spain) according with the manufacturer’s recommendations.
Statistical analysis
Descriptive and comparative statistics were performed using the Minitab®16 package (Minitab, LLC, USA). Seminal parameter data were mostly right-skewed so that conservative nonparametric statistical methods were employed to assess statistic difference of the medians. Seminal parameters of males with and without SCI were compared using the Mann–Whitney/Wilcoxon test. Seminal parameters of males with SCI were also compared with respect to the spinal level of their lesion (cervical or thoracic) and based on their AIS level (A verses B–D) using the Mann–Whitney/Wilcoxon test. Nonparametric confidence intervals for the difference in medians as an estimate of the differences in location are also reported. Correlations were analyzed using a Spearman Rho correlation using SPSSV25 (IBM® SPSS® Statistics, USA). Significance for all statistical tests was set at p value of 0.05 level and a two-tailed test used.
Results
Semen quality of males with SCI compared with normozoospermic donors without SCI
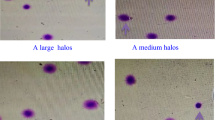
Figure 1 summarizes the descriptive statistics of the different semen parameters measured in males with and without SCI; Table 2 reports the p values and confidence intervals, when comparing the different parameter values obtained for each cohort. While sperm concentration in some males with SCI was very high, significant differences were not obtained when this sample was compared with the normozoospermic donor cohort. On the contrary, median progressive motility was higher in the normozoospermic donor cohort. The median sperm DNA Fragmentation (SDF) of males with SCI was estimated to be 5.4× that of the normozoospermic donor controls, with 70% of the SCI patients presenting with values higher than 70% SDF. Despite higher levels of sperm DNA damage in the ejaculates of males with SCI (compare red arrows with green arrows in Fig. 2a), some males with SCI still displayed haloes of dispersed chromatin representative of normal DNA quality. The median leukocyte concentration was estimated to be 6.5× higher in males with SCI compared with normozoospermic donors (Fig. 1 and Table 2). While the median pro-oxidant capacity of males with SCI was higher than that of normozoospermic donors (Fig. 1; Table 2), not all the leukocytes showed the same level of labeling response to the NBT reaction, as both high and low ROS activity were often visualized in the same field of view (Fig. 2b). In general, all semen parameters analyzed in males with SCI showed a higher degree of variability than the normozoospermic control donors (Fig. 1).
a Spermatozoa from a male with SCI showing fragmented and non-fragmented sperm DNA following the SCD (Halosperm®) assay—Red arrowheads represent sperm nuclei with fragmented sperm DNA and green arrowheads represent sperm with normal DNA; b NBT response of a male with SCI showing leukocytes with high and levels of activities after reaction for 15 min in the reactive gel.
Time between ejaculations
For males with SCI, the time between two serial ejaculations was highly variable, ranging from 1–68 months (Table 1), however, the majority of semen parameters showed no significant correlation with respect to the time period between two sequential ejaculations (Sperm concentration: r = 0.453; p = 0.059; Sperm motility: r = −0.059; p = 0.817; Pro-oxidant capacity: r = −0.218; p = 0.385 Leukocyte concentration: r = 0.296; p = 0.233), except for SDF which was positively correlated with time of abstinence (r = 0.486; p = 0.041). There was no significant correlation with respect to the time since the lesion had occurred for any of the semen parameters (Sperm concentration: r = 0.303; p = 0.125; Sperm motility: r = −0.194; p = 0.331; SDF: r = 0.159; p = 0.428; Pro-oxidant capacity: r = −0.027; p = 0.895; Leukocyte concentration: r = −0.175; p = 0.383). In addition, SDF was significantly positively correlated with sperm concentration (r = 0.488; p = 0.010) and not surprisingly, negatively correlated with progressive motility (r = −0.522; p = 0.003).
Level of injury
Two different analyses were performed. In the first, males with SCI were grouped into those with either cervical (C—tetraplegic) or thoracic (T—paraplegic) injuries; those individuals in AIS category D were excluded (n = 2; see Table 1). Those males with SCI at the cervical region showed no significant difference in semen parameters compared with those with thoracic injury (Table 3), except for sperm concentration, the median of which was estimated to be 2.1× higher in males with cervical lesions. In the second analysis, only progressive motility was significantly different between these two groups, with AIS A SCI males having a median estimated to be 2.3× lower than males with SCI that had incomplete injuries (AIS B–D) (Table 4).
Discussion
The results of this study have shown that apart from sperm concentration, the semen quality of males with SCI was remarkably poor compared with that of the normozoospermic “high fertility proven” donor samples provided by the clinic with respect to every parameter analysed. This is consistent with previous observations based on standard semen parameters [3, 6, 14, 15], although here, we have expanded the standard analysis to include SDF measured using the SCDt, pro-oxidant capacity detected in the ejaculate and the presence of active leukocytes. In addition, we have established comparative semen characteristics between males with cervical and thoracic level damage as well as between complete and incomplete damage as defined by the AIS scale.
Although sperm concentration in males with SCI evaluated in this study tended to be high in some individuals, it was not statistically different compared to the normozoospermic donor samples. While similar to the results of Rolf et al. [16], this finding needs to be treated with caution, as the variability of the sperm concentration in the males with SCI was high and the sample small. Within the samples from males with SCI, sperm concentration ranged from 1 × 106 mL−1 to 422 × 106 mL−1, whereas the donor’s sperm concentration only ranged from 33 × 106 mL−1 to 91 × 106 mL−1. Even if sperm concentration was higher in males with SCI, it is unlikely to have any beneficial relevance to the overall semen quality; it may actually be detrimental. In this study, we found a positive correlation between sperm concentration and SDF in the ejaculates of males with SCI. Interestingly, the adverse effect of high sperm concentration has also been reported in a range of animal species not affected by spinal injury (e.g., [17]).
The presence of semen reactive oxygen species in males with SCI has been iteratively reported (e.g., [18, 19]). We propose that amplified oxidative stress mediated by active leukocytes associated with sperm degeneration in the cauda epididymidis is likely contributing to an aggressive environment for the stored spermatozoa. Our results appear to support this hypothesis, as we found a higher level of pro-oxidant capacity associated with the presence of active leukocytes in the ejaculates of males with SCI compared with normozoospermic donors. In our study, there was also an increase in the level of pro-oxidant capacity and leukocyte concentration in the ejaculate of males with SCI. Therefore, we suggest that there is most likely to be a cooperative and degrading process, whereby the ROS activity present in the leukocytes is affecting normal spermatozoa and these damaged sperm could subsequently be further contributing to ROS generation as they breakdown, setting up a “vortex” of cellular degradation. Given this background, it is not surprising that we describe here a significant reduction in the progressive motility and sperm DNA quality in males with SCI.
In addition to ROS production, leukocytes can also affect sperm viability by the combined assembly of extracellular neutrophil traps, the so-called neutrophil extracellular traps (NETs). These structures are made up of DNA fibers ejected by neutrophils to capture and degrade bacteria and which are also able to trap spermatozoa [20]. Granules associated with forming part of these NETs contain microbicidal proteins, such as myeloperoxidase, calprotectin, or cathepsin G [21], all of which may negatively affect sperm quality.
Unbalanced levels of ROS appear in part to be responsible for the presence of high levels of SDF (e.g., [22]). Consequently, an uncontrolled REDOX system may also be responsible for the extremely high levels of SDF described in males with SCI in our study. The level of SDF in males with SCI in our study appears to be higher than that reported in generalized male factor infertility, such as leukocytospermia, Chlamydia infection, testicular cancer or varicocele [23]. In fact, the level of SDF in some males with SCI we analysed was as high as 100% and the median estimated to be 5.4× higher than that observed in the control donor cohort. If these males with SCI are ultimately going to achieve pregnancy, then it will be necessary that these high levels of SDF are reduced to around 15–25% so that procedures such as ICSI might be employed.
When the semen quality of males with SCI in this study was analysed based on whether they had cervical or thoracic spinal cord lesions, no significant differences were observed in any semen parameter, except that of sperm concentration. It is possible that males with SCI at the cervical level are less likely to ejaculate spontaneously, thereby increasing cauda epididymidal reserves. Neither the length of time after the injury the semen sample was collected, nor the period of anejaculation appeared to have any effect on ejaculate quality, except that SDF was positively correlated with abstinence time. As the impact of anejaculation has been challenged by Brackett et al. [6] and we only had small number of observations in our current study, this phenomenon will require further investigation.
When the semen parameter data for the SCI patients were analysed according to AIS categories [complete (A) verses incomplete (B–D)], the only significant difference in semen parameters in our study was found for progressive motility, which was higher in males with SCI with incomplete lesions. The neurophysiology of this observation is difficult to explain but is possibly related to the varying higher degrees of spinal cord integrity in this broad category of males with SCI; given the relatively low numbers of males with SCI in each sub-category injury, their interpretation needs to be regarded with caution.
In general, semen samples from the males with SCI were highly abnormal, so it is unlikely that these samples would result in pregnancy using standard intra-uterine artificial insemination techniques. Rather, it is anticipated that males with SCI would need some form of sperm selection to create an enriched subpopulation of suitable spermatozoa, closer to the donor standard in terms of sperm motility and acceptable levels of SDF, to in order to obtain a pregnancy in their partners. If classical sperm selection protocols for improving human sperm quality such as “swim up” or density gradient centrifugation (DGC) were not sufficient to improve the proportion of fertile spermatozoa, then a synergistic combination of DGC and magnetic activated cell sorting (MACS) may need to be employed. DGC is likely to be particularly useful for males with SCI, as it can eliminate a large proportion of somatic cells (e.g., leukocytes) and thereby decrease the effect of post-ejaculation sperm DNA fragmentation [24]. In addition, the MACS procedure may remove spermatozoa containing DNA damaged undergoing apoptosis by targeting the cell surface of these spermatozoa for capture with annexin-V antibodies [25].
Increased ejaculation frequency has been shown to be effective for improving semen parameters in males with SCI (e.g., [26]), even affecting the level of SDF of the selected sperm subpopulation following DGC [27]. While such a strategy appears intuitive, there are also obvious practical challenges in obtaining repeated ejaculations, including problems associated with the overuse of procedures such as vibro-ejaculation or electro-ejaculation that could potentially be harmful to males with SCI. In addition, the current evidence that repeated ejaculation in males with SCI might be helpful in improving semen quality, still appears equivocal (e.g., [6, 28]).
Another possibility to improve the sperm quality of males with SCI is harvesting spermatozoa directly from the testis for use in intracytoplasmic sperm injection procedures. Spermatozoa recovered from the testis, typically have a lower level of sperm DNA fragmentation [29] and in addition, they have not been exposed to oxidative damage associated with sperm accumulation and deterioration in the cauda epididymidis or from being adversely affected by leukocyte presence. In addition, is it unnecessary to undergo aggressive artificial induction of ejaculation, since sperm recovery can be performed using TESA/TESE or micro-TESE. When testicular sperm recovery is used in severe oligospermic males, it may reduce the level of SDF from ~40% of SDF within the ejaculate to 8%, resulting in a reduction in the probability of a miscarriage [30].
In reality, males with SCI are likely to require a combination of customized sperm selection and assisted fertilization techniques depending on their specific pathology and circumstances but these procedures will need to be able to reduce the proportion of SDF in the semen sample in order to achieve a successful pregnancy. For example, this may involve serial ejaculations with a period of abstinence for at least two days, followed by a protocol of DGC for sperm selection to reduce the proportion of leukocyte and/or immature spermatozoa causing oxidative damage so as to increase the final proportion of spermatozoa free of DNA damage. If the sperm concentration recruited is optimal for this treatment, then additional possibilities also exist to use procedures such as MACS for a second tier of sperm selection.
Although the findings of our study are limited to some degree by its retrospective design, the small sample size of ejaculates collected from males with SCI, the convenience of using our own reference data for estimating the semen parameters of donors and the use of nonparametric statistics, we have clearly demonstrated that semen quality of males with SCI is significantly poorer than that of donors. Given this phenomenon, most males with SCI will most likely require some form of assisted reproductive technology to increase their chances of successful conception. While this study provides preliminary insights into the aeitology of poor semen quality in males with SCI, there is still much to learn, particularly with respect to the relationship between anejaculation, sperm concentration, and SDF.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Sønksen J. Assisted ejaculation and semen characteristics in spinal cord injured males. Scand J Urol Nephrol Suppl. 2003;213:1–31.
Patki P, Woodhouse J, Hamid R, Craggs M, Shah J. Effects of spinal cord injury on semen parameters. J Spinal Cord Med. 2008;31:27–32.
Iremashvili V, Brackett NL, Ibrahim E, Aballa TC, Lynne CM. Semen quality remains stable during the chronic phase of spinal cord injury: a longitudinal study. J Urol. 2010;184:2073–7.
Qiu Y, Wang LG, Zhang LH, Li J, Zhang AD, Zhang MH. Sperm chromosomal aneuploidy and DNA integrity of infertile men with anejaculation. J Assist Reprod Genet. 2012;29:185–94.
Dimitriadis F, Karakitsios K, Tsounapi P, Tsambalas S, Loutradis D, Kanakas N, et al. Erectile function and male reproduction in men with spinal cord injury: a review. Andrologia. 2010;42:139–65.
Brackett NL, Ibrahim E, Grotas JA, Aballa TC, Lynne CM. Higher sperm DNA damage in semen from men with spinal cord injuries compared with controls. J Androl. 2008;29:93–99.
Barbonetti A, Vassallo MRC, Di Rosa A, Leombruni Y, Felnazi G, Gandini L, et al. Involvement of mitochondrial dysfunction in the adverse effect exerted by seminal plasma from men with spinal cord injury on sperm motility. Andrology. 2013;1:456–63.
Rex AS, Aagaard J, Fedder J. DNA fragmentation in spermatozoa: a historical review. Andrology. 2017;5:622–30.
Iommiello VM, Albani E, Di Rosa A, Marras A, Menduni F, Morreale G, et al. Ejaculate oxidative stress is related with sperm DNA fragmentation and round cells. Int J Endocrinol. 2015; 321901.
World Health Organisation. WHO laboratory manual for the examination and processing of human semen. 5th ed. Switzerland: WHO Press; 2010.
Agarwal A, Mulgund A, Alshahrani S, Assidi M, Abuzenadah AM, Sharma R, et al. Reactive oxygen species and sperm DNA damage in infertile men presenting with low level leukocytospermia. Reprod Biol Endrocrin. 2014;12:126.
Kirshblum S, Waring W 3rd. Updates for the International Standards for Neurological Classification of spinal cord injury. Phys Med Rehabil Clin N Am. 2014;25:505–17.
de la Casa M, Fernández JL, Badajoz V, López-Fernández C, Johnston SD, Gosálvez J. Semi-quantitative assessment of superoxide in the human neat ejaculate using a nitro blue tetrazolium based assay: a comparison of visual, spectrophotometric and image analysis approaches. J Fertil In Vitro. 2014; 03. https://doi.org/10.4172/2375-4508.1000146
Brackett NL, Santa-Cruz C, Lynne CM. Sperm from spinal cord injured men lose motility faster than sperm from normal men: the effect is exacerbated at body compared to room temperature. J Urol. 1997;157:2150–3.
Brackett NL, Ferrell SM, Aballa TC, Amador MJ, Lynne CM. Semen quality in spinal cord injured men: does it progressively decline postinjury? Arch Phys Med Rehab. 1998;79:625–8.
Rolf C, Cooper TG, Yeung CH, Nieschlag E. Antioxidant treatment of patients with asthenozoospermia or moderate oligoasthenozoospermia with high-dose vitamin C and vitamin E: a randomized, placebo-controlled, double-blind study. Hum Reprod. 1999;14:1028–33.
López-Fernández C, Johnston SD, Fernández JL, Wilson RJ, Gosálvez J. Fragmentation dynamics of frozen-thawed ram sperm DNA is modulated by sperm concentration. Theriogenology. 2010;74:1362–70.
De Lamirande E, Leduc BE, Iwasaki A, Hassouna M, Gagnon C. Increased reactive oxygen species formation in semen of patients with spinal cord injury. Fertil Steril. 1995;63:637–42.
Padron OF, Lynne CM, Brackett NL, Tomas AJ, Sharma RK, Agarwa A. Seminal reactive oxygen species and sperm motility and morphology in men with spinal cord injury. Fertil Steril. 1997;67:1115–20.
Alghamdi AS, Foster DN. Seminal DNase frees spermatozoa entangled in neutrophil extracellular traps. Biol Reprod. 2005;73:1174–81.
Brinkmann V, Zychlinsky A. Neutrophil extracellular traps: Is immunity the second function of chromatin? J Cell Biol. 2012;198:773–83.
Dorostghoal M, Kazeminejad SR, Shahbazian N, Pourmehdi M, Jabbari A. Oxidative stress status and sperm DNA fragmentation in fertile and infertile men. Andrologia. 2017;49:e12762.
Esteves SC, Gosálvez J, Lopez-Fernandez C, Nunez-Calonge R, Caballero P, Ashok A, et al. Diagnostic accuracy of sperm DNA degradation index (DDSi) as a potential noninvasive biomarker to identify men with varicocele‑associated infertility. Int Urol Nephrol. 2015;47:1471–7. https://doi.org/10.1007/s11255-015-1053-6
Muratori M, Tarozzi N, Carpentiero F, Danti S, Perrone FM, Cambi M, et al. Sperm selection with density gradient centrifugation and swim up: effect on DNA fragmentation in viable spermatozoa. Sci Rep. 2019;9:7492.
Lee TH, Liu CH, Shih YT, Tsao HM, Huang CC, Chen HH, et al. Magnetic-activated cell sorting for sperm preparation reduces spermatozoa with apoptotic markers and improves the acrosome reaction in couples with unexplained infertility. Hum Reprod. 2010;25:839–46.
Levitas E, Lunenfeld E, Weiss N, Friger M, Har-Vardi I, Koifman A, et al. Relationship between the duration of sexual abstinence and semen quality: analysis of 9,489 semen samples. Fertil Steril. 2005;83:1680–6.
Gosálvez J, González-Martınez M, López-Fernández C, Fernández JL, Sánchez-Martín P. Shorter abstinence decreases sperm deoxyribonucleic acid fragmentation in ejaculate. Fertil Steril. 2011;96:1083–6.
Hamid R, Patki P, Bywater H, Shah PJ, Craggs MD. Effects of repeated ejaculations on semen characteristics following spinal cord injury. Spinal Cord. 2006;44:369–73.
Greco E, Scarselli F, Iacobelli M, Rienzi L, Ubaldi F, Ferrero S, et al. Efficient treatment of infertility due to sperm DNA damage by ICSI with testicular spermatozoa. Hum Reprod. 2005;20:226–30.
Esteves SC, Sanchez-Martín F, Sanchez-Martín P, Schneider DT, Gosálvez J. Comparison of reproductive outcome in oligozoospermic men with high sperm DNA fragmentation undergoing intracytoplasmic sperm injection with ejaculated and testicular sperm. Fertil Steril. 2015;104:1398–405.
Acknowledgements
We are grateful for the technical expertize of Prof Carmen López-Fernández for help with sperm DNA analysis.
Funding
This work was partially supported with funds from the Spanish Ministry of Economy, Industry and Competitiveness: RTC-2016-4733-1.
Author information
Authors and Affiliations
Contributions
EVB, ASR, and AAM were in charge of patient involvement; they contributed in the experimental design and provided cases for inclusion of the study and editing of initial drafts of the manuscript. SDJ, RW, and JG were instrumental in the experimental design, statistical analysis, composition and draft, and final writing and editing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was supervised and approved by the clinical ethic committee of the National Hospital of Paraplegics (Toledo, Spain); we further certify that all applicable institutional and governmental regulations concerning the ethical use of human were followed during the course of this research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Vargas-Baquero, E., Johnston, S., Sánchez-Ramos, A. et al. The incidence and etiology of sperm DNA fragmentation in the ejaculates of males with spinal cord injuries. Spinal Cord 58, 803–810 (2020). https://doi.org/10.1038/s41393-020-0426-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-020-0426-6