Abstract
Cancer is the leading cause of death worldwide, and its treatment and outcomes have been dramatically revolutionised by targeted therapies. As the most frequently mutated oncogene, Kirsten rat sarcoma viral oncogene homologue (KRAS) has attracted substantial attention. The understanding of KRAS is constantly being updated by numerous studies on KRAS in the initiation and progression of cancer diseases. However, KRAS has been deemed a challenging therapeutic target, even “undruggable”, after drug-targeting efforts over the past four decades. Recently, there have been surprising advances in directly targeted drugs for KRAS, especially in KRAS (G12C) inhibitors, such as AMG510 (sotorasib) and MRTX849 (adagrasib), which have obtained encouraging results in clinical trials. Excitingly, AMG510 was the first drug-targeting KRAS (G12C) to be approved for clinical use this year. This review summarises the most recent understanding of fundamental aspects of KRAS, the relationship between the KRAS mutations and tumour immune evasion, and new progress in targeting KRAS, particularly KRAS (G12C). Moreover, the possible mechanisms of resistance to KRAS (G12C) inhibitors and possible combination therapies are summarised, with a view to providing the best regimen for individualised treatment with KRAS (G12C) inhibitors and achieving truly precise treatment.
Similar content being viewed by others
Introduction
Kirsten rat sarcoma viral oncogene homologue (KRAS) is the best-known oncogene with the highest mutation rate among all cancers and is associated with a series of highly fatal cancers, including pancreatic ductal adenocarcinoma (PDAC), nonsmall-cell lung cancer (NSCLC), and colorectal cancer (CRC). The identification of tumour driver genes and the development of specific inhibitors have revolutionised cancer treatment strategies and clinical outcomes. Numerous clinical results have shown that targeted therapies significantly extend progression-free survival and are less toxic than standard chemotherapy1,2,3. For instance, targeted therapies in patients harbouring epidermal growth factor receptor (EGFR)-sensitive mutation or anaplastic lymphoma kinase (ALK) gene fusion have markedly enhanced survival time, with a median overall survival of 3 years or more4,5,6. Unfortunately, despite 40 years of proprietary drug efforts, there are still no effective strategies targeting KRAS mutations, except for sotorasib, which has just been approved to target the mutated KRAS subtype KRAS (G12C). Due to the intrinsic characteristics of KRAS proteins, targeting KRAS has been considered to be quite challenging. Therefore, many efforts have focused on indirectly targeting KRAS, including targeting its downstream signalling effectors7, epigenetic approaches such as telomerase inhibitors8 and RNA interference9 and synthetic lethality approaches, such as cyclin-dependent kinase inhibitors10. However, most of these strategies have failed due to a lack of activity or selectivity. In addition, patients with KRAS mutations usually have a poor response to current standard therapy11. There has been an urgent and unmet need to target KRAS mutations in KRAS-driven cancer. Recently, there has been light on the horizon for one specific mutation, KRAS (G12C). With the discovery of a new allosteric site of KRAS (G12C), several irreversible covalently binding inhibitors of KRAS (G12C) have emerged, raising the hope of drugging KRAS. In this review, the latest research progress on KRAS, the characteristics of KRAS mutations, the relationship between the KRAS mutations and tumour immunity, and the targeted research progress of KRAS, especially KRAS (G12C), are summarised. Furthermore, the potential mechanisms of resistance to KRAS (G12C) inhibitors and promising combination strategies to determine the best-individualised treatment regimen are outlined.
KRAS and signal transduction
Introduction to KRAS
The KRAS gene is a member of the rat sarcoma viral oncogene family (RAS), which includes two other isoforms in humans: the Harvey and neuroblastoma rat sarcoma viral oncogenes (HRAS, NRAS). In 1982, Weinberg and Barbacid isolated a gene from human bladder cancer cell lines. Subsequently, this gene was identified as a human homologue of the RAS gene, named HRAS, located on the short arm of chromosome 11 (11p15.1–11p15.3)12. In the same year, another homologue was found in human lung cancer cells, called KRAS, located on the short arm of chromosome 12 (12p11.1–12p12.1). The last gene, called NRAS, is found in human neuroblastoma and is located on the short arm of chromosome 1 (1p22–1p32)13,14,15. RAS genes are evolutionarily conserved with similar structures and are composed of four exons distributed on the full length of approximately 30 kb DNA. The KRAS gene encodes two highly related protein isoforms, KRAS-4B and KRAS-4A, which consist of 188 and 189 amino acids, respectively, due to different clipping of the fourth exon16. The other two RAS proteins all contain 189 amino acids. The term KRAS is generally referred to as KRAS-4B due to the high level of mRNA encoding KRAS-4B in cells17. The crystal structure of RAS reveals six beta strands and five alpha helices18. which form two major domains: a catalytic domain called the G domain and a hypervariable region (HVR)19. The G domain consists of three regions: switch I, switch II, and the P loop, which binds guanine nucleotides and activates signalling by interacting with effectors. The HVR comprises the CAAX motif related to membrane localisation20. From the perspective of function, RAS is a kind of membrane-bound regulatory protein (G protein) binding guanine nucleotide belonging to the family of guanosine triphosphatases (GTPases)21. RAS functions as a guanosine diphosphate (GDP)/triphosphate (GTP) binary switch, which controls important signal transduction from activated membrane receptors to intracellular molecules22 The binary switch is mainly determined by two kinds of regulatory proteins: guanine nucleotide exchange factors (GEFs) such as son of sevenless (SOS) and GTPase-activating proteins (GAPs) such as neurofibromin 1 (NF1)23. In the resting state, KRAS normally binds with GDP in an inactivated state due to the intrinsic GTPase activity of KRAS, which is able to hydrolyse GTP to GDP24. When the cells receive the relevant stimuli, such as the interaction of EGF and EGFR, the KRAS-GDP complex appears to have a decreased affinity of KRAS with GDP in the presence of GEFs, and then GDP is replaced by GTP, which has a higher affinity and an approximately 10-fold higher cellular concentration than GDP25. KRAS-GTP binding acquires an altered conformation in switches I and II of the G domain, and then KRAS is activated and binds to its downstream molecules as a monomer or dimer to mediate a series of signalling cascades. In contrast, GAPs promote the binding between GDP and KRAS by enhancing the GTPase activity of KRAS, thus maintaining the inactive state of KRAS (Fig. 1)26.
The structure and function of KRAS. a According to homology, KRAS, which consists of 188/189 amino acids can be divided into three parts. The first part consisting of the first 85 amino acid residues is a highly conserved region. The next 80 amino acid residues are defined as a second part where homology between any pair of human RAS genes is 85%. A third part is a highly variable region and homology is only 8%. KRAS forms two major domains: a catalytic domain called the G domain and a hypervariable region (HVR). The G domain consists of three regions: switch I, switch II and the P loop, which binds guanine nucleotides and activates signalling pathway by interacting with effectors. The HVR consists of a membrane-targeting domain containing the CAAX motif where C is a cysteine, A is any aliphatic amino acid and X is any amide acid, which acquires lipids by farnesyl or prenyl modification. b The normal function of KRAS depends on the membrane localisation of its post-transcriptional modification, which is mediated by a series of enzymes. KRAS functions as a guanosine diphosphate (GDP)/triphosphate (GTP) binary switch, which controls important signal transduction from activated membrane receptors to intracellular molecules. The binary switch is mainly determined by two kinds of regulatory proteins: guanine nucleotide exchange factors (GEFs) and GTPase-activating proteins (GAPs). FTase: farnesyltransferase; GGTase: geranyl geranyltransferase; RCE1: RAS-converting enzyme 1; ICMT: isoprenylcysteine carboxyl methyltransferase; PDEδ: phosphodiesterase δ
KRAS signalling
KRAS proteins function as a finely regulated molecular switch that controls multiple signalling cascades by cycling between activated and inactivated conformations. KRAS proteins can be activated by growth factors, chemokines, Ca2+ or receptor tyrosine kinase (RTK). Activated KRAS protein can activate multiple signalling pathways, including the rapidly accelerated fibrosarcoma (RAF)-mitogen-activated protein kinase (MEK)-extracellular regulated protein kinases (ERK) signalling pathway, phosphoinositide 3-kinase (PI3K)-protein kinase B (AKT)—mammalian target of rapamycin (mTOR) signalling pathway, and other signalling pathways, revealing a wide range of KRAS communications with multiple signalling pathways.
The upstream regulation of KRAS
GRB2-SOS1 complex
Many growth factors, such as epidermal growth factor (EGF), platelet-derived growth factor (PDGF) and fibroblast growth factors (FGFs), activate receptor tyrosine kinases (RTKs) and then activate KRAS proteins through intermediary molecules. A typical example is the interaction of EGF with EGFR, which results in the dimerisation and cross-phosphorylation of EGFR27. Phosphorylated EGFR is bound by growth factor receptor-bound protein 2 (GRB2), an adaptor molecule, through their respective SH2 domains. GRB2 consists of one SH2 domain and two SH3 domains, of which the SH3 domain is capable of binding SOS1, a kind of GEF. Activated SOS1 promotes the binding of GTP and KRAS and converts KRAS from an inactive state to an active state. As mentioned earlier, KRAS states are tuned primarily to GEFs/GAPs. Therefore, molecules upstream of KRAS mainly mediate the activation or inactivation of KRAS by regulating these two types of molecules. The signalling cascades mediated by the KRAS–GTP complex were thought to occur only within the cell membrane in the past. Recently, Tulpule and his colleagues revealed another novel, membrane-independent approach to activate KRAS28. Some fusion proteins containing RTKs, such as EML4-ALK, can combine with GRB2 and SOS to form membraneless cytoplasmic protein granules to actively activate KRAS and activate downstream signals. Notably, this approach is similar but not identical to phase separation.
RAS-GRF1
RAS protein-specific guanine nucleotide releasing factor 1 (RAS-GRF1) is another GEF and is expressed primarily in the brain. In mature neurons, RAS-GRF1 connects signals from glutamate receptors, such as N-methyl-D-aspartic acid receptor (NMDAR) to KRAS, thereby promoting the downstream MAPK/ERK cascade reaction29. The ability of RAS-GRF1 to activate KRAS was significantly enhanced with increasing Ca2+ concentration, revealing the internal communication and mutual regulation between KRAS signalling and Ca2+ signalling as well as metabolism30,31. In addition, chemokines can activate protein kinase A by producing cAMP after binding to chemokine receptors on the cell membrane. Activated protein kinase A can activate RAS-GRF1 through phosphorylation, thereby promoting the transformation of KRAS into an active state32.
SHP2
In addition to the GEFs mentioned above as a key molecule in KRAS activation, Src homology phosphatase 2 (SHP2) plays an integral role in KRAS activation33,34. SHP2 is a protein tyrosine phosphatase (PTP) containing Src homology 2 (SH2) domains and is encoded by PTPN1135. Unlike most PTPs, numerous studies indicate that SHP2 functions in the activation of intracellular signalling pathways, particularly the KRAS/ERK pathway34. SHP2 is a common signalling button that mediates numerous receptor tyrosine kinase signals to KRAS-ERK signalling36. On the one hand, SHP2 can promote the recruitment of the GRB2-SOS1 compound to the receptor. Studies have identified some phosphorylation sites of SHP2 as the major binding site for GRB2, such as tyrosine 542 and 580 of SHP237. SHP2 therefore acts as a scaffolding effect in GRB2 recruitment. On the other hand, SHP2 catalytic activity was required to promote the activation of KRAS through the dephosphorylation of the SHP2 substrate38. Some dephosphorylation substrates of SHP-2 have been shown to promote KRAS activation. There are also many negative regulatory molecules involved in KRAS activation, such as p120-RASGAP, a GTPase-activating protein. RASGAP has been shown to be a target for SHP2. SHP2 dephosphorylates the p120-Rasgap junction and regulates the recruitment of p120-RASGAP near KRAS, thus relieving the negative regulatory effect of p120-RASGAP39,40. In addition to p120-RASGAP, SHP2 plays a signalling role by dephosphorylating other molecules, including negative regulators of Sprouty and activators of Src, through the dephosphorylation of Src-regulatory proteins41,42.
The KRAS-mediated signalling pathway
The RAF-MEK-ERK pathway
The RAF-MEK-ERK pathway is the canonical downstream target of KRAS signalling43. Activated KRAS-GTP can recruit rapidly accelerating fibrosarcoma (RAF), a serine/threonine-specific protein kinase, from the cytoplasm to the plasma membrane, induce conformational changes in RAF and promote the activation of RAF by homologous or heterologous dimerization44. The C-terminal catalytic domain of RAF binds to MEK1/2 and activates it by phosphorylation. MEK1/2 phosphorylates and activates ERK1/2, and activated ERK phosphorylates ribosomal S6 kinase (RSK), serum response factor (SRF), E26 transformation-specific transcription factors (ETS) and ETS like-1 protein to regulate the transcription and translation of corresponding target genes, thus participating in the regulation of cell proliferation, differentiation, migration and other life activities44,45,46.
The PI3K-AKT-mTOR pathway
KRAS was also found to be involved in the PI3K-AKT-mTOR pathway, which is considered to play an important role in cell life activities such as cell proliferation, differentiation, apoptosis and glucose transport and has a great influence on the generation of tumour resistance47. Activated KRAS can activate PI3K by binding to its p110 subunit. Activated PI3K-catalysed phosphatidylinositol 4,5-bisphosphate (PIP2) is converted to phosphatidylinositol 3,4,5-trisphosphate (PIP3)48. PIP3 promotes phosphoinositide-dependent kinase 1 (PDK1) to phosphorylate AKT at Thr308. mTOR complex 2 further phosphorylates the serine phosphorylation site of AKT (Ser473), resulting in full AKT activation49. Activated AKT enters the nucleus, activates or inhibits many downstream pathways, and regulates cell proliferation, apoptosis and metabolic processes50. On the one hand, AKT can directly activate mTOR target proteins, which play an important role in cell proliferation, survival, metabolism, protein synthesis, and transcription51. On the other hand, AKT phosphorylates and activates Bcl-XL/Bcl-2-associated death promoters (BADs), facilitating the binding of BAD to the companion protein 14-3-3 instead of Bcl-2/Bcl-XL, thus inhibiting apoptosis52.
Other signalling pathways
RAL guanine nucleotide dissociation stimulator (RalGDS) is a downstream signalling protein of KRAS that functions as a GTP/GDP exchange factor to promote the GDP/GTP conversion of RAS-like protein (RAL)53,54,55. Downstream effector factors of RAL proteins include Rac/cell division cycle 42 (Cdc42) associated with cell migration, TANK binding kinase 1 (TBK1) associated with viral immunity, and phospholipase D (PLD) associated with endocytosis. KRAS also regulates TIAM1 and RAC1-specific guanine nucleotide exchange factors, to activate RAC1 signalling pathways that affect cell shape, migration, adhesion, actin cytoskeleton formation, endocytosis, and membrane trafficking56. In addition, KRAS can also regulate phosphatidylinositol signal pathway by activating PLCε57. In short, KRAS-mediated signal networks are complex and related to a variety of life activities (Fig. 2).
The regulation of KRAS activation and signal transduction. The canonical and well-known pattern of activating KRAS is dependent on correct membrane localisation and adjacent activation of membrane receptors. In the resting state, KRAS normally binds with GDP in an inactivated state. When the extracellular growth factors such as EGF transmit signals to receptors, the SOS, a kind of GEF, interacts with the KRAS-GDP complex leading to the release of GDP and the replacement of GTP. The tether of GTP and KRAS induces structural changes of switch I and switch II, thereby activating KRAS. In contrast, GAPs enhance intrinsic GTPase activity in KRAS to accelerate the reaction in which GTP is hydrolysed to GDP. The KRAS cycle between the activated and inactivated conformations functions as a finely regulated molecular switch that controls multiple signalling cascades, including the canonical RAF-MEK-ERK pathway, which controls proliferation; PI3K-AKT-mTOR pathway, which promotes cell survival; and other signalling pathways, which are required for KRAS-dependent tumour growth and endocytosis, and cytoskeletal organisation
KRAS mutations and cancer
Mutation characteristics of the oncogene KRAS
KRAS is the most commonly mutated member of the RAS family and is considered to be the most common oncogenic gene driver in human cancers58,59. KRAS mutations are most common in PDAC, CRC, and NSCLC. The profile of KRAS mutations differs significantly among different cancer types (Table 1). KRAS mutations are dominated by single-base missense mutations, 98% of which are found at codon 12 (G12), codon 13 (G13), or codon 61 (Q61)60. Notably, KRAS mutations occur in many cancers with different mutation frequencies, but there is also a large variation in mutation subtypes (Fig. 3a, b). For example, in NSCLC, KRAS mutations account for 20.4% of KRAS, and the dominant substitution is G12C (glycine (GGT) to cysteine (TGT)), while KRAS mutation accounts for up to 67.6% of KRAS in pancreatic adenocarcinoma, and KRAS (G12D) is the dominant mutant subtype. It is important to note that the mutation rate of KRAS in the pancreas was 67.6% based on this acquired data analysis, which is lower than the commonly cited 90%. The incidence of KRAS mutations between 25 and 35% in smokers and 5% in nonsmokers has been reported, and smoking is usually considered to be a relevant factor61. Furthermore, the profiles of KRAS mutations are distinct in smokers and nonsmokers, and not all mutations in KRAS are driver mutations. For instance, KRAS (G12C) is usually found in heavily smoking patients, while KRAS (G12D) is more usually identified in tumours from nonsmoking patients62.
KRAS mutation in cancer. a The frequency of KRAS mutations across tumour types, including the mutation frequency of common sites and the subtype with the highest mutation rate in different tumour types. KRAS mutations are characterised by single-base missense mutations, 98% of which are found at codon 12, codon 13, or codon 61. Please refer to Table 1 for specific figures. b Specific mutant subtypes and percentages were represented in the top three cancers with the highest mutation rates of KRAS including pancreatic cancer, colorectal cancer, and nonsmall-cell lung cancer. c Frequency of co-occurring aberrations in KRAS mutant cells. Only a mutant prevalence of at least 3% is shown in addition to EGFR mutation, given the important effect of EGFR mutation on NSCLC. TP53: tumour protein p53 gene; DDR2: discoidin domain receptor tyrosine kinase 2 gene; MET: MNNG HOST Transforming gene; PIK3CA: phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha gene; STK11: serine/threonine kinase 11 gene; KEAP1: kelch-like ECH-associated protein 1 gene; ATM: ATM serine/threonine kinase gene, PIK3CG: phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit gamma gene; ERBB4: erb-b2 receptor tyrosine kinase 4 gene; KDR: kinase insert domain receptor gene; KIT: KIT proto-oncogene receptor tyrosine kinase gene; NFE2L2 nuclear factor erythroid 2, like 2 gene; PDGFR previous symbol of PDGFRB (platelet-derived growth factor receptor beta gene). Data acquired from The Cancer Genome Atlas (pan-Cancer) from cBioPortal
Biochemical heterogeneity of KRAS mutations
The conventional understanding of KRAS mutations is that alterations in KRAS proteins caused by mutations impede the interaction of KRAS with GAPs and the hydrolysis of GTP bound to KRAS, leaving KRAS in a constitutively active state63,64. Numerous studies have shown biochemical heterogeneity of KRAS mutations in various aspects, including intrinsic GTPase activity and the affinity of effectors and metastatic sites. For instance, alterations in codons 12, 13, and 61 usually lead to impaired intrinsic GTPase activity of KRAS, distinct from KRAS (G12D), KRAS (G12C), and KRAS (G13D)65. The type of KRAS mutation can also affect the interaction with its effectors. Moreover, KRAS (G12) and KRAS (Q61) are insensitive to neurofibromin 1-mediated hydrolysis, whereas KRAS (G13) is partially sensitive to neurofibromin 1, a kind of GAP66. In addition, cells containing KRAS (G12C) or KRAS (G12V) have increased levels of RAS-related protein (RAL) A/B signalling and decreased levels of phosphorylation of protein kinase B (AKT) compared with cells with other KRAS mutations or wild-type cells67. However, cell lines with KRAS (G12D) have higher levels of phosphorylated AKT68. According to the binding affinity of KRAS with RAF, an important effector, the mutation can be divided into two classes: high affinity (G12A, G12V, G12R, Q61H, and Q61L) and low affinity (G12R, G12D, and G12V). Different KRAS-mutant subtypes have different sensitivities to targeted therapy. For example, a study performing high-fidelity CRISPR-based engineering found that KRAS (G12D) is sensitive to EGFR inhibition in pancreatic cancer models, while KRAS (G12C) mutants selectively respond only to covalent G12C inhibitors when EGFR is inhibited69. In terms of metastasis, patients with KRAS mutations have a greater opportunity for lung or brain metastases70. Another study revealed that patients harbouring G12V mutations often have pleuropericardial metastases, while patients harbouring G12C and G12D mutations usually have bone metastases71,72. Notably, not all KRAS-mutant tumours are KRAS-dependent tumours. More relevant future studies are needed to expand the heterogeneity map of KRAS mutations and provide a theoretical basis for more accurate individualised treatments.
Comutations of KRAS mutations
In addition to the different mutation levels and mutation subtypes in different cancer tissues, KRAS mutations may have different comutations, which may influence the function of KRAS and the occurrence and development of tumours. Analysing 1078 NSCLCs harbouring KRAS mutations, 557 patients (53.5%) harboured comutations, and of 14 analysed genes, tumour protein p53 gene (TP53) mutation was the most common comutation, accounting for 39. 4%73. In addition, 101 patients in the above cohort were analysed for an additional 14 genes, such as the serine/threonine kinase 11 gene (STK11) and kelch-like ECH-associated protein 1 gene (Fig. 1c)73. Interestingly, the study indicated that EGFR mutations were detected in KRAS-mutated NSCLC, albeit with a very low frequency of less than 1%. This is controversial because it is widely believed that EGFR and KRAS mutations are mutually exclusive74,75. In the future, larger amounts of clinical data will be needed to draw more comprehensive and rational conclusions. There were distinct biological behaviours in different subgroups, altering outcomes in patients76. For example, tumours with KRAS/STK11 comutation commonly have a tumour microenvironment (TME) with poor immune response, lacking CD8 + tumour-infiltrating lymphocytes but including an abundance of T regulator cells. However, CD8 + tumour-infiltrating lymphocytes and activated dendritic cells are rich, and there are few T regulator cells in the TMEt with the KRAS/P53 comutation77. In addition, clinical evidence has shown that patients harbouring ALK or EGFR/KRAS commutations respond poorly to tyrosine kinase inhibitor therapy78. In 11,951 Chinese tumour samples, KRAS mutations accounted for 16.6%, of which KRAS (G12C) accounted for 14.5%. Almost all patients (99.6%) with G12C mutations were associated with genomic aberrations that were associated with the RAS/RTK pathway79. In general, when treating KRAS-driven tumours, it is necessary to pay attention to other gene changes and pay attention to individualised treatment (Fig. 3c).
KRAS mutations and TME
There is growing evidence that patterns of genetic changes influence the immune environment of cancer. Increasing evidence shows that KRAS mutations in tumours not only dedicate the intrinsic characteristics of tumours, such as survival and proliferation but also form a tumour microenvironment (TME), especially affecting immune cells in the TME and eventually resulting in tumour progression and immune escape80,81.
KRAS mutations and inflammatory TME
The TME in the presence of KRAS mutations often appears to be inflammatory and infiltrated with multiple immune cells. This inflammatory microenvironment is facilitated by high levels of a series of inflammatory cytokine and chemokine factors mediated by the KRAS signalling pathway81,82,83. The overactivation of KRAS signalling has been shown to enhance the secretion of interleukin-6 (IL-6), which is necessary for tumour initiation and progression and is important for the crosstalk between tumours and inflammation84,85. KRAS-induced IL-6 promoted the activation of Janus activated kinase 1 (JAK1) and phosphorylation of signal transducer and activator of transcription 3 (STAT3), contributing to tumorigenic cellular processes in a variety of tumour types86,87,88. In pancreatic cancer, IL-6 not only promotes the occurrence and progression of tumours through JAK1/STAT3 but also activates reactive oxygen species through the ERK pathway89. Studies have also shown that IL-6 secreted by myeloid cells can promote pancreatic intraepithelial neoplasia progression and PDAC with KRAS mutations90. IL-8 is also related to tumours and inflammation as a C-X-C motif chemokine receptor 2(CXCR2) ligand. IL-8 has been identified as a transcriptional target for KRAS-mediated ERK or PI3K signal transduction, affecting endothelial cell recruitment, tumour-associated inflammation formation, and tumour angiogenesis90,91. The role of inflammation in promoting lung cancer has been shown to be mediated in part by activation of the IL-8/CXCR2 pathway and subsequent neutrophil recruitment and release of neutrophil elastase91. KRAS/CXCR2 signalling in PDAC has been shown to induce the generation of cancer-associated fibroblasts with enhanced secretory function that mainly secrete protumorigenic cytokines92. In addition, KRAS has been reported to be involved in the formation of PDAC and the development of NSCLC by promoting the production of IL-1α and the activation of IKKβ/NF-κB93,94.
Several chemokines are involved in the proinflammatory TME. Although elevated levels of C-C chemokine ligand 5(CCL5) were detected in a variety of tumours and were associated with tumour progression, CCL5 also exhibited antitumour abilities by recruiting T cells and dendritic cells to the TME. In KRAS-driven lung cancer, the IKK-related kinases TBK1 and IKKε downstream of KRAS signalling increased CCL5 levels95. KRAS mutations in pancreatic acinar cells were able to upregulate intercellular adhesion molecule 1, resulting in chemical absorption by M1 macrophages96. M1 macrophages release stromal degrading enzymes, such as matrix metalloproteinase-9 and cytokines such as tumour necrosis factor (TNF), which contribute to the formation of an inflammatory environment. Several KRAS-mutant TMEs showed increased Th17 cells and significantly increased IL-17 levels. For example, in pancreatic KRAS mouse genetic models, increased numbers of Th17 cells were observed in PDAC and participated in tumour initiation and progression by the production of IL-1797. Recently, KRAS-mediated activation of NLRP3, which forms inflammasomes, was shown to affect the pathogenesis of KRAS-driven myeloproliferation98. In conclusion, this evidence supports that KRAS induces an inflammatory TME and facilitates the occurrence and development of tumours by inducing inflammation.
KRAS mutations and tumour immune escape
The TME of most solid tumours is infiltrated by multiple immune cells, including T cells and macrophages. However, many of these immune cells have been cultured in the TME and ultimately possess immunosuppressive properties, such as regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs), tumour-associated macrophages (TAMs), neutrophils, and mast cells. KRAS-mediated signalling plays an important role in the formation of an immunosuppressive TME and the modification of immune cells (Fig. 4)99.
KRAS-mediated immune escape in tumour microenvironment. KRAS mediates immune escape in the tumour macroenvironment by upregulating PD-L1expression, downregulating MHC1expression of tumour cells, and enhancing the secretion of a variety of cytokines and chemokines to recruit immunosuppressive immune cells. The black arrow represents facilitation, and the opposite red arrow represents inhibition. MDSCs: myeloid-derived suppressor cells; Treg cells: regulatory T cells; IL-10: interleukin-10; TGF-β: transforming growth factor-β; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL-23: interleukin-23
KRAS mutations mediate immune escape by regulating the intrinsic characteristics of tumour cells. In KRAS-driven tumours, mutant KRAS mediates tumour immune escape by upregulating PD-L1 expression. There is growing evidence that KRAS (G12C), KRAS (G12V), KRAS (G12D), and KRAS (G13D) mutations are associated with high PD-L1 expression in lung cancer100,101. The mutant KRAS signalling pathway upregulates the expression of PD-L1 in tumour cells by improving the stability of PD-L1 mRNA102. Although the AU-rich binding protein tristerrolin (TTP) reduced the expression of PD-L1 through AU-rich elements in the PD-L1 mRNA 3’UTR, the MEK signal downstream of KRAS phosphorylates and inhibits TTP, thus increasing the expression level of PD-L1102. Another study reported another mechanism of KRAS (G12V)-mediated upregulation of PD-L1 expression by promoting ROS production and inducing FGFR1 expression103. After antioxidant treatment, PD-L1 expression in KRAS-mutant cells was largely eliminated, and FGFR1 gene knockout also led to reduced PD-L1 expression and impaired tumour growth in vivo103. Notably, the PD-L1 expression levels in CRC patients were not significantly associated with KRAS mutations, unlike LAC. In addition to affecting PD-L1 expression, KRAS mutation impacted the immunogenicity of tumour cells by downregulating the expression of major histocompatibility complex (MHC) class I molecules in a CRC cell line with the KRAS (G13D) mutation104. MHC1 is essential for antigen presentation and antigen recognition by T cells. Downregulation of MHC 1 severely attenuated the tumour killing effect of T cells, especially CD8 + cytotoxic T cells. The knockdown of mutant KRAS (G12D) in a poorly immunogenic CRC model improved the immune response and caused tumour regression105.
In addition to affecting tumour cells, KRAS mutations play an immune evasion role by affecting immune cells in the TME, such as the acquisition and recruitment of inhibitory phenotypes of immune cells. KRAS mutations induce CD4+ T cells in the TME to transform into immunosuppressive Treg cells by promoting the secretion of related cytokines. The KRAS (G12C)-mediated phenotypic transformation of T cells has been shown to be a consequence of the secretion of IL-10 and TGF-β1 mediated by MEK/ERK/AP-1 signalling in CRC106. In the KRAS transgenic lung cancer model, the gene ablation of Treg cells resulted in inhibition of the occurrence and progression of lung cancer, indicating the necessity of Treg cells in the development of lung tumours107. In addition, the KRAS (G12V) and KRAS (G12D) mutations enhanced the infiltration of MDSCs in the TME by upregulating GM-CSF in PDAC and CRC, thereby leading to antitumour immune escape108,109. Another study has shown that KRAS (G12D) appears to inhibit the secretion of interferon regulatory factor 2 (IRF2), thereby promoting increased CXCL3 secretion, which acts on CXCR2 on MDSCs, leading to MDSC migration to the TME110.
In KRAS-mutated tumours, other oncogenes or tumour suppressor genes may also be abnormal and may participate in immune escape together with KRAS mutation. For example, in lung cancer, coactivation of KRAS (G12D) and MYC drove the aggregation of anti-inflammatory macrophages, but the absence of T, B, and NK cells was due to the effect of CCL9 and IL-23111. In PDAC, studies have shown that KRAS (G12D) and TP53 jointly activate the ARF6/AMAP1 pathway, affect the level and presentation of PD-L1, and promote tumour development and immune invasion112. Through the analysis of clinical data of LAC patients, the expression of PD-L1 in tumours in the TP53/KRAS comutation group was increased, and the proportion of CD8 + T cells in TME was higher, which was consistent with the clinical benefit of TP53, KRAS or TP53/KRAS-mutated cancer patients after treatment with PD-1 inhibitors113. In addition, patients with STK11/LKB1 mutations were resistant to PD-1 inhibitors in KRAS-mutant LAC, suggesting a new mechanism of resistance103. In general, KRAS comutations affect the immune regulation of the KRAS-driven TME by recruiting immunosuppressive cells and increasing PD-L1 expression.
Successfully targeting KRAS (G12C)
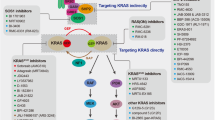
KRAS plays a central role in signal transduction, and KRAS mutations are closely related to tumour initiation and development. Successful targeting of mutant KRAS will lead to a new platform for targeted oncology therapy. However, after nearly 40 years of effort, KRAS remains an unsolved puzzle. Due to the difficulty of direct targeting, researchers switched to other important molecules in the KRAS signalling pathway, such as RAF, ERK, and MEK. However, there has been no significant success in KRAS-driven tumours. KRAS-mutant tumours have obvious heterogeneity, which partly explains the poor efficacy of nonspecifically targeting KRAS. Selective inhibitors targeting specific KRAS mutations are urgently needed to effectively inhibit different mutant KRAS functions in line with the requirements of precision oncology.
The first signs of the dawn appear on the horizon for one specific mutation, KRAS (G12C). Unlike KRAS (G12D) and KRAS (G12V), KRAS (G12C) can maintain alternative interactions with its downstream effectors through an active cycle between the GDP-bound and GTP-bound states114. This difference enables KRAS (G12C) to be locked into an inactive conformation by reacting with cysteine residues. For instance, SML-8-73-1, a GDP analogue in which beta phosphate binds an electrophilic chloroacetamide, can covalently react with cysteine 12 (Cys12) of KRAS (G12C). However, SML-8-73-1 appears to be cell impermeable due to possessing two negative charges115. SML-10-70-1, a caged version of SML-8-73-1, can penetrate cells but lacks selectivity and antitumour activity116. Fortunately, another attractive target has been identified: the thiol group in the cysteine residue forms a disulphide bridge with specific inhibitors with prolonged target engagement. Notably, this strategy exhibits not only remarkable selectivity but also greater activity because Cys12 is located near switch regions involved in the interaction with effectors and the nucleotide. Novel inhibitors targeting KRAS (G12C) have displayed promising results in preclinical and clinical trials, showing promise for targeting KRAS, which was considered to be undruggable in the past (Fig. 5).
Current targeted strategies for KRAS (G12C). a Despite the mutation occurring in KRAS, KRAS (G12C) still continues to perform the KRAS-GDP/GTP cycle. Covalent inhibitors such as AMG510 and MRTX849 lock KRAS (G12C) in inactivated GDP-bound state, thus decreasing functional KRAS. Another strategy is to increase the degradation of mutant KRAS (G12C) proteins. Based on the previously described KRAS (G12C) covalent inhibitors, LC-2, an endogenous KRAS (G12C) degrader, has been developed to promote KRAS (G12C) degradation. b The development and chemical structures of KRAS (G12C) inhibitors
The discovery of a new allosteric regulatory site in KRAS (G12C)
Initially, Shokat and colleagues designed a sequence of small molecule compounds based on the specific nucleophilic properties of the mutant half cystine thiols for targeting KRAS (G12C)117. The crystal structure of compound 6 combined with the KRAS (G12C)-GDP complex clearly indicates that compound six extends from Cys12 into an adjacent pocket named the switch-II pocket (S-IIP), rather than binding to the nucleotide pocket117. Notably, S-IIP is not obvious in other published structures of other KRAS or isoforms, although a groove is visible in some cases118,119. Shokat and colleagues changed the drug-design strategy to focus on acrylamides, vinyl sulfonamides and carbon-based electrophiles rather than continuing with disulfide-based compounds. This strategy was not only chemically selective but irreversible. Finally, they obtained the most potent acrylamide 12. Cells containing the KRAS (G12C) mutation exhibited reduced viability and increased cell apoptosis when treated with compound 12 compared with cells without the KRAS (G12C) mutation117. Overall, a new allosteric pocket in KRAS (G12C) and relatively specific inhibitors were identified, providing structure-based validation that KRAS is targetable58.
Preclinical exploration of KRAS (G12C) inhibitors
The discovery of a new allosteric site has reignited researchers’ enthusiasm for targeting KRAS (G12C). Based on previous compounds reported by Shokat and colleagues, a series of compounds were constantly developed to seek the path to success, including ARS-853 and ARS-1620 (Fig. 4b). ARS-853 was developed from first-generation compounds 6 and 12 by optimising electrophilic localisation and modifying the scaffold that interacts with a hydrophobic portion of the S-IIP120,121. Studies have shown that ARS-853 inhibits the KRAS (G12C) protein by locking it in the inactive state of the GDP-GTP binding state cycle. Increasing evidence suggests that KRAS mutations are unequal and that different KRAS mutations reflect biological heterogeneity122. Furthermore, by using ARS-853 as a molecular tool, a study found that KRAS (G12C) is rapidly cycling rather than in a statically active state, that KRAS (G12C)-mediated signalling can be regulated by upstream effectors, and that cell viability and growth depend on KRAS mutations to varying degrees even within KRASG12 cancer cell lines120,123.
ARS-1620 is the first proof-of-concept molecule to demonstrate the feasibility of targeting KRAS (G12C) in vivo. According to the cocrystal structure, ARS-1620 possesses an additional covalent interaction with His95 of KRAS (G12C), which provides a more stable and preferred conformation than ARS-853124. In brief, ARS-1620 is an orally selective and potent agent in a variety of models. An apparent paradox is why a target without conventional binding pockets can nevertheless be inhibited by small molecules with powerful efficiency from the nanomolar to the low micromolar range. The study of Rasmus Hansen and colleagues demonstrated that inhibitors exhibit an only weak and reversible binding affinity for KRAS (G12C), but the chemical reaction between the targeted cysteine and inhibitors is powerful and is accelerated by KRAS (G12C) as an enzyme that specifically catalyses covalent bond formation between groups with low intrinsic reactivity125. Therefore, the main driver of potency is KRAS-mediated acceleration of the chemical reactions of specific KRAS (G12C) inhibitors.
Clinical exploration of KRAS (G12C) inhibitors
AMG510 (sotorasib)
AMG510 is the first small molecule inhibitor specifically targeting KRAS (G12C) to enter clinical trials (NCT03600883), which specifically and irreversibly bind to Cys12 in the inducible S-IIP and lock KRAS (G12C) protein in an inactive status126,127. AMG510 is additionally combined with a novel surface groove formed by an alternative orientation of His95 on KRAS, resulting in a tenfold improvement in efficiency compared to ARS-1620128,129. Excitingly, AMG510 was approved by the U.S. Food and Drug Administration, on May 28, 2021, as the first treatment for adult patients with NSCLC whose tumours harbour KRAS (G12C) mutations and who have received at least one prior systemic therapy130. This is a milestone because it is the first drug to directly target mutated KRAS. The full Phase I cohort (n = 129) receiving daily AMG510 monotherapy in the clinical trial (NCT03600883) was completed131. This monotherapy trial showed responses across all dose levels tested, and no dose-limiting toxicity or treatment-related deaths were observed131. A total of 56.6% patients (n = 76) reported treatment-related adverse events, of which 11.6% of patients (n = 15) reported grade 3 or 4 events, including reversible elevations in alanine aminotransferase levels, diarrhoea, vomiting, and anaemia. In the subgroup with NSCLC (n = 59), the ORR and DCR were 32.2% (n = 19) and 88.1%, respectively. Simultaneously, a median progression-free survival of 6.3 months and the recommended phase II dose of 960 mg daily were identified131. On January 28, 2021, Amgen revealed the results of a phase II clinical trial (NCT03600883) evaluating 126 patients with KRAS (G12C)-mutant NSCLC. Surprisingly, an ORR of 37.1%, including 3 complete responses and 43 partial responses, and a DCR of 80.6% were reported. A median duration of response of 10 months and a median progression-free survival of 6.8 months were also confirmed, in line with earlier Phase I results. The security of AMG510 was further confirmed. Grade 3 adverse events were reported in 19.8% of patients, and grade 4 adverse events were reported in only 1 patient (0.8%), with no treatment-related deaths. These attractive results make AMG510 the first approved drug to treat KRAS (G12C)-mutant NSCLC. There are other ongoing clinical trials to further assess AMG510, such as NCT04185883, which will assess the safety and feasibility of various combinational therapies; NCT04303780, which compares AMG510 with docetaxel; and NCT04625647, which will evaluate the response rate of AMG510 in participants with KRAS (G12C)-mutated stage IV or recurrent nonsquamous NSCLC (Table 2).
MRTX849 (adagrasib)
The ongoing clinical study of MRTX849 also offers hope for the successful targeting of KRAS (G12C) proteins132. The preliminary results of a multiple expansion study evaluating MRTX849 (NCT03785249) were reported at the 2019 EORTC-NCI-AACR annual symposium. In the cohort of 12 patients, including six NSCLC patients, most patients received a 600 mg dose twice daily, of whom three NSCLC patients reported unconfirmed partial responses and other patients experienced stable disease. MRTX849 was well tolerated in most patients with only grade 1 adverse events, of which diarrhoea and nausea were the most common. Another phase I/II study (NCT04330664) is also ongoing to assess MRTX849 in combination with NO155, an SHP2 inhibitor, in patients with the KRAS (G12C) mutation (Table 3).
Other specific KRAS (G12C) inhibitors from other companies are also under investigation in clinical trials, including LY3499446, D-1553, and ARS-3248/JNJ-74699157, which are next-generation ARS-1620 inhibitors (NCT04006301, NCT04165031, and NCT04585035) (Table 2). Notably, the first KRAS (G12C) inhibitor, LY3499446, from Eli Lilly, has been discontinued due to safety concerns, but at the 2021 AACR meeting, they presented preclinical results for another inhibitor, LY3537982. In KRAS (G12C) mutant H358 cells, the inhibitory activity of LY3537982 was at least 10 times higher than that of AMG510 and MRTX849. We look forward to the further results of LY3537982 in preclinical and clinical trials.
Proteolysis-targeting chimaeras (PROTACs) targeting KRAS (G12C) proteins
Compared with direct inhibition, degradation could be a more potent strategy that affects cell proliferation and downstream signalling responses133,134. Based on covalent inhibitors targeting KRAS (G12C), PROTACs, as small molecule degraders, are designed to target KRAS (G12C). The E3 ligase is able to ubiquitinate proximal lysine residues of KRAS (G12C) by PROTACs that simultaneously engage KRAS (G12C) and E3 ligase substrate receptor proteins, such as the cerebellum (CRBN). A covalently degrading molecule based on a thalidomide scaffold and ARS-1620 has been reported to degrade the artificial GFP-KRAS (G12C) fusion protein in reporter cells135. Subsequently, LC-2, the first endogenous KRAS (G12C) degrader, was reported to rapidly engage and continuously degrade KRAS (G12C), which consisted of MRTX849 and the VHL E3 ligase ligand136. LC-2-mediated KRAS (G12C) degradation requires an intact proteasome system and VHL E3 ligase complex assembly, of which neddylation is important for the formation and function of the VHL E3 ligase complex; nevertheless, the lysosomal pathway is nonessential137. After treatment with LC-2, multiple cancer cells demonstrated ERK phosphorylation inhibition136. Moreover, there has been an unmet need for reversible degraders attacking KRAS (G12C) because LC-2 cannot be involved in multiple catalytic cycles of degradation, limiting its potency due to its covalent nature138. The development of PROPACS is considered to be an important direction for antitumour therapy and has broad prospects, although in vivo data have yet to be obtained (Fig. 5a).
Targeting other KRAS mutations and indirect strategies
Targeting other KRAS mutations
The successful development of KRAS (G12C) inhibitors is dependent on the covalent inhibition of cysteine and high GTPase activity of KRAS (G12C), which is not fully present in other KRAS-mutant types114. New strategies are therefore needed to target other common KRAS mutations, such as KRAS (G12D) and KRAS (G12V). By screening a random peptide library for purified recombinant KRAS (G12D) exhibited on the T7 phage, researchers obtained a novel and selective inhibitory peptide (KRpep-2d) to KRAS (G12D), as in the first report of a KRAS (G12D)-selective inhibitor139. Then, KS-58 based on KRpep-2d was reported as the first KRAS (G12D)-selective inhibitory peptide presenting in vivo anticancer activity140. Another study suggested that CRISPR/Cas9 could be delivered via exosomes to target KRAS (G12D) in models of pancreatic cancer141. Koide and his colleagues successfully developed a noncovalent inhibitor, termed 12VC1, that can highly selectively bind to the active states of KRAS (G12V) and KRAS (G12C) in vivo and in vitro142. Based on this monomer, PROTAC-like fusion proteins were developed, which could electively and effectively increase the degradation of KRAS (G12V). Using a structure-based drug design, Kessler and colleagues discovered that Bi-2852, a nonspecific KRAS inhibitor, binds with nanomolar affinity to a pocket between switches I and II on active and inactive KRAS in vitro143. Overall, the development of specific inhibitors for other KRAS mutations is still in its infancy and is a long way from reaching clinical trials. However, the emergence of new approaches also offers hope for successfully targeting different KRAS-mutated subtypes.
Indirect strategies of targeting KRAS
For more than 30 years after its discovery, KRAS was considered undruggable target due to the intrinsic characteristics of KRAS proteins. The KRAS is small and has a considerably smooth and shallow surface, resulting in difficulty of small molecule binding to the KRAS. There is no other pocket on the surface of KRAS that can bind to small molecules except the GTP binding pocket, but targeting the GTP binding pocket is quite difficult144. Because there is a quite high concentration of GTP in cells under physiological conditions and the affinity between the GTP and KRAS reaches the picomolar level, GTP almost monopolises the sole pocket on the surface of KRAS145. These inherent conditions make it nearly impossible to develop competitive KRAS inhibitors. Therefore, various explorations for indirectly targeting KRAS have been performed over the past 40 years, of which most strategies are nonspecific and inefficient, including reducing the expression of KRAS, interrupting the membrane location of KRAS, interfering with the interaction between the KRAS and its effectors, inhibiting upstream and downstream signalling, and synthetic lethality approaches such as cyclin-dependent kinase inhibitors. However, most of these strategies have failed due to a lack of activity or selectivity.
Reducing the expression of KRAS
RNA-based approaches to degrade overexpressed mRNAs have been shown to be effective and feasible in vivo, as exosomes can act as effective vectors for short interfering RNAs to mediate entry into tumour cells146,147. Among them, progress has been made in the development of antisense oligonucleotides (ASOs) targeting KRAS in lung cancer. AZD4785, developed by Macleod and colleagues, is an ASO targeting KRAS with a novel 2’-4’-constrained ethyl modification148. KRAS expression was significantly reduced in subcutaneous tumours after treatment with AZD4785, but the reduction was not obvious in clinical trials (NCT03101839).
Interrupting membranal location of KRAS
Only membrane-bound KRAS can be activated and activate downstream signalling pathways. Therefore, preventing membrane localisation after KRAS translation should be an effective strategy. Farnesyltransferase was first identified as a potential therapeutic target at an early stage due to its key role in the localisation of KRAS149. Of several powerful farnesyltransferase inhibitors (FTIs), tiffany and lonafarnib have entered phase III clinical trials150. However, the treatment of KRAS-driven cancers ultimately failed due to the lack of antitumour effects151. Subsequent findings indictaed that KRAS was successfully localised by modification of prenylation by geranylgeranyl transferase type 1-mediated substitution pathways in the absence of farnesyltransferase152. However, cotargeting of farnesyltransferase and geranyl transferase also did not produce significant antitumour effects153. Salirasib, as a second-generation FTI, was developed but terminated in phase II trials due to lack of efficacy154. Other post-translational modification enzymes, such as RAS-converting enzyme 1 or isoprenyl carboxyl methyltransferase may also be potential therapeutic targets to block KRAS membrane localisation, especially phosphodiesterase-δ, which assists the transport of KRAS from the Golgi apparatus to the plasma membrane155. However, relevant studies are still in the preclinical stage and different studies have presented puzzling and different results that need to be further investigated in the future156,157.
Inhibiting KRAS-mediated signal transmission
Protein-protein interactions are an important form of signalling. Therefore, interference in the interaction between the KRAS and other proteins may be a potential therapeutic strategy to block KRAS-mediated signal transmission. GEFs are a class of key proteins of KRAS interaction, such as SOS1. SOS1 inhibitors have also been designed and synthesised to block KRAS-SOS1 interaction158. Of them, BI1701963, an orally bioavailable pan-KRAS inhibitor, has entered a phase I clinical trial (NCT04111458), which has an obvious effect in inducing tumour senescence. In addition, stapled peptides, which can block the interaction of KRAS with SOS1 based on a stabilised α-helical structure, are also considered promising as an effective strategy to interfere with the interaction between KRAS and SOS1159. In addition, small molecules that directly block the interaction of KRAS and downstream effectors, primarily RAF kinases, have not yet been identified, because the interface is part of an antiparallel β-sheet where no appropriate pockets are offered for binding a small molecule at high affinity160. Through continuous modification and optimisation, effective cyclic peptides have been designed to prevent the binding of KRAS and RAF, such as cyclorasin 9 A and cyclic peptide kD2161,162. Inhibition of key KRAS upstream and downstream molecules can also effectively inhibit KRAS-mediated signal transduction, such as inhibiting upstream signalling, SHP2, and inhibiting downstream signalling, the RAF-MEK-ERK pathway and the PI3K-AKT-mTOR pathway. However, due to the extensive cross-talk of KRAS-mediated signalling pathways, it is difficult to achieve effective inhibition of signalling pathways only by inhibiting a single key molecule, and joint inhibition of multiple key molecules is often required.
Combination therapy with KRAS (G12C) inhibitors
KRAS (G12C) inhibitors have achieved surprising results in clinical trials, and AMG510 has been successfully approved for application in the clinic, achieving a breakthrough from scratch in the development of KRAS inhibitors. However, the subsequent acquired resistance greatly limits the application of KRAS (G12C) inhibitors. Combination therapy is an important and prioritised strategy to increase efficacy, reduce toxicity and side effects, and delay the acquisition of drug resistance. Either preclinical or preliminary clinical data indicate that combinational strategies can improve the antitumour efficacy of KRAS (G12C) inhibitors. A concerning problem with combination therapy is that patients are often unable to tolerate it due to severe side effects when two or more inhibitors are applied to patients at the same time163. However, clinical results of KRAS (G12C) inhibitors showed that most patients tolerated treatment well without serious adverse reactions. Therefore, KRAS (G12C) inhibitors could be used as a promising class of combinational agents to achieve better efficacy (Fig. 6).
Combined with chemoradiotherapy
Chemotherapy remains an important tumour treatment strategy for advanced cancer. However, the benefit to patients is usually limited due to severe side effects and the rapid emergence of drug resistance. The combination of conventional chemotherapy drugs and novel drugs is often considered to increase the benefit to patients. Covalent inhibitors targeting KRAS (G12C) combined with conventional chemotherapy drugs may be a potential strategy to improve efficacy. The study demonstrated that NSCLC models with KRAS mutations displayed a greater sensitivity to pemetrexed, and the expression of KRAS RNA changed after treatment with pemetrexed, leading to decreased angiogenesis, possibly because KRAS-mutant cells were more dependent on the folate metabolic pathway164. It is worth noting that different KRAS mutations differ in sensitivity to chemotherapy. For instance, tumour models with KRAS (G12C) responded well to taxanes and pemetrexed but responded poorly to cisplatin164. The combination of AMG510 and carboplatin significantly enhanced antitumour efficacy compared with AMG510 or carboplatin alone126. The above evidence shows that improved efficacy offers a sufficient theoretical basis for the application of a combination of KRAS (G12C) inhibitors and chemotherapeutic drugs in clinical trials. In addition, the clinical results of AMG510 and MTRX849 showed fewer adverse reactions and better tolerance, and the two resulted in more benefits and less toxicity when used in combination131.
In addition to the effect on chemotherapy, patients with KRAS-activated mutations often have radiation resistance and poor prognosis165. It has been suggested that radiation resistance is realised by EGFR-mediated chromatin condensation in KRAS-mutated lung cancers166,167. Radiotherapy resistance of CRC with KRAS mutations was realised by rapid upregulation of heterogeneous nuclear ribonucleoprotein KhnRNP K, and MEK inhibition effectively increased radiotherapy sensitivity of CRC168. Radiotherapy, as a means of cancer treatment, is widely used in many types of cancer. In radiation-naive tumours with KRAS mutations, it has been confirmed that radiotherapy combined with PD-1 antibody has an obvious synergistic effect169. Therefore, KRAS inhibitors in combination with radiotherapy may provide unexpected benefits to patients harbouring KRAS mutations.
Combined with targeted therapy
Combined with upstream molecules
RTKs inhibitors
The reactivation of KRAS and its downstream effectors induced by adaptive receptor tyrosine kinases (RTKs) is one of the mechanisms of drug resistance, so the combination of KRAS (G12C) inhibitors and drug-targeting RTKs may be a promising therapeutic strategy. The combined efficacy of KRAS (G12C) inhibitors and RTK inhibitors was explored in two studies120,121. The combined action of RTK inhibitors and ARS-853 significantly enhanced the antitumour activity in different cells, and the effects of different RTK inhibitors were different in KRAS (G12C) mutant cells, indicating the heterogeneity of the RTK action. Subsequent studies further indicated that the phosphorylation levels of several RTKs in different KRAS (G12C) models were increased and showed high heterogeneity. The synergistic effect of several RTK inhibitors and ARS-1620 showed strong antitumour activity170. Unfortunately, the synergistic results of these combinations are not generally effective across different KRAS (G12C) tumour models. Therefore, the efficacy of a single RTK inhibitor combined with KRAS (G12C) inhibitors may not be consistent in cancer treatment.
SHP2 inhibitors
Given the inconsistent effects of single RTK inhibitors, inhibiting common downstream nodes of multiple RTK signalling pathways may be broadly effective in overcoming the adaptive reactivation of KRAS170. Src homology region 2 domain-containing phosphatase 2 (SHP2) functions as a convergent node downstream of multiple RTKs to regulate RAS activation, which is encoded by tyrosine-protein phosphatase nonreceptor type11171. RTKs normally recruit the GRB2-SOS1 complex to activate RAS and do not depend on SHP2172. However, SHP2 is necessary for the proliferation of KRAS (G12C) mutant cancer in vivo, and adaptive reactivation of KRAS is heavily dependent on SHP2173,174,175. SHP2 inhibition has the potential to increase KRAS-GDP occupancy and suppress RAS-mediated signalling as well as adaptive signalling driving resistance to therapy176. For instance, the combination of ARS-1620 with SHP-099, an SHP2 inhibitor, significantly reduced tumour volume in tumour models with KRAS (G12C)170,177. The combination of MRTX849 with RMC-4550, another SHP2 inhibitor that also exhibited higher antitumour activity in both the sensitive and refractory MRTX849 models by further inhibiting the KRAS-mediated signalling pathway129. The combination of TNO155 with EGFR inhibitors, RAF inhibitors, and KRAS (G12C) inhibitors has shown synergistic effects in preclinical studies, suggesting that TNO155 effectively blocks feedback activation178. On the basis of these compelling preclinical data, multiple SHP2 inhibitors, such as JAB-3068, RLY-1971, and TNO155, have entered early clinical trials (NCT03518554, NCT04252339, and NCT03114319, respectively), and the combination of KRAS (G12C) and the SHP2 inhibitor is also testing in early-phase clinical trials (NCT04185883, NCT04330664).
SOS1 inhibitors
SOS1 is another common downstream effector of multiple RTK signalling pathways. There is increasing evidence that KRAS (G12C) rapidly cycles in the GDP or GTP binding status rather than being continuously active in the GTP-KRAS status, resulting in abnormal activation of downstream signals. This indicates that KRAS (G12C) is still sensitive to GEF (such as SOS1) regulation. A synergistic effect of BAY293, a potent SOS1 inhibitor, with ARS-853 has been reported because BAY293 is capable of disrupting the KRAS-SOS1 interaction to inhibit the activation of KRAS179.
Combined with downstream molecules
KRAS is cross-linked with various signalling pathways and plays a pivotal role in cell survival and proliferation. In turn, downstream effectors influence KRAS function to different degrees. For instance, ERK3 activity is required for KRAS-driven tumorigenesis in vitro, and ERK3 deficiency inhibits the oncogenic growth of KRAS (G12C)-mutant NSCLC in vivo, indicating the application of ERK3 inhibitors in KRAS (G12C)-driven tumours180. Recent studies have found that BRAF/ERK inhibitors can release the feedback inhibition of upstream RTK and RAS signals, and the liberation of RTK and RAS signals can facilitate other signalling pathways, such as the PI3K/AKT/mTORC1 pathway, which improves cell growth and proliferation181. The combination of a PI3K inhibitor and a KRAS (G12C) inhibitor showed obvious synergistic effects in various models. In addition, targeting ATK or mTOR, important PI3K downstream effectors, has been proven to be effective when used in combination with KRAS (G12C) inhibitors126,129,182. For instance, the combined use of mTOR inhibitors, IGF1R inhibitors, and ARS-1620 significantly enhanced tumour regression with low toxicity compared to combined MEK inhibitors or ARS-1620 alone in a series of KRAS-driven mouse lung cancer models183.
Combined with cell cycle checkpoint
Excessive proliferation is a common feature of tumour cells, and CDK4/6 is a key regulator of the cell cycle, playing a key role in the G1-to-S-phase transition. CDK4/6 can be affected by KRAS through multiple signalling pathways, such as the RAF-MEk-ERK and PI3K-Akt pathways. Studies have shown that the CDK4/6 inhibitor palbociclib enhances the overall effect of a KRAS inhibitor (ARS-1620). Similarly, another study indicated that MRTX849 and palbociclib, in several MRTX849-refractory models, exhibited significant general tumour regression, which may be due to increased inhibition of the retinoblastoma protein/E2F transcription factor pathway129. In addition to CDK, aurora kinase A (AURKA) plays an important role in the cell cycle by ensuring the proper separation of chromosomes and the smooth completion of cytokinesis during mitosis. ARS-1620 was observed to have a significant synergistic effect with alisertib, a specific AURKA inhibitor, for the treatment of ARS-1620-refractory KRAS (G12C) mutant cancer. In terms of mechanism, targeting AURKA not only affects the normal proliferation division of cells but also disrupts the stable interaction between KRAS and c-RAF, resulting in difficulty in RAF activation177. Several clinical trials are currently underway in which combinations of CDK4/6 inhibitors have been added (Table 3).
Combined with synthetic lethality screen
Compared with wild-type KRAS cells, mutant KRAS-driven tumour cells may be more dependent on certain genes that are necessary for the maintenance of the KRAS-driven cellular state. Targeting these vulnerable genes in synthetic lethality screens could serve as an alternative approach, called synthetic lethality screens. Some large-scale genetic screening has been performed to find vulnerable genes that are uniquely essential to KRAS-mutant tumours184,185,186. The study found that in addition to KRAS itself, the critical genes for Kras mutation were RAF1 (encoding CRAF) and SHOC2187. Genetic deletion of CRAF significantly reduced tumour size in a KRAS (G12V) mouse model, which was independent of the function of CRAF kinase188. SHOC2 enhances RAF dimerization and is necessary for maximum ERK activity. Ablation of SHOC2 reduced the growth of KRAS-mutant NSCLC cells and sensitised the cells to MEK inhibition189. Another experiment showed strong enrichment of genes with mitotic functions in a genome-wide RNAi screen of KRAS-mutant cells186. In addition, genome-wide CRISPR/Cas9-based genetic screening has been widely used to find ideal synthetic lethal targets in KRAS-mutated tumours190. Unfortunately, the degree of overlap between these screen results is low, except for the proteasome system, and has not yet led to successful clinical approaches. However, the successful development of KRAS (G12C) inhibitors provides a new direction for KRAS mutation-related synthetic lethality. One study proposed that the bypass pathways (defined as collateral dependencies (CDs)) that maintain cancer cell survival may be different from those dependent on the excessive activation of KRAS signalling when acute deprivation drives oncogene activity with KRAS (G12C) inhibitors and target CDs will promote the response to KRASG12C inhibitors191. At the same time, two classes of combination strategies targeting CDs are proposed to either strengthen KRAS (G12C) target engagement (namely, EGFR, FGFR, or SHP2 inhibitors) or independently restrain persistent survival pathways (namely, PI3K, or CDK4/6 inhibitors)191.
In general, synergistically targeting upstream molecules of KRAS is considered to offer advantages over synergistically targeting downstream molecules of KRAS. Synergistic targeting of upstream molecules can effectively inhibit multiple downstream pathways, avoiding the activation of other parallel signalling pathways that can induce tumour growth. However, the identification of appropriate upstream targets for KRAS is challenging and risky because cotargeting upstream targets may bring more side effects192. Synthetic lethal approaches using genome-wide screening are also promising, but more detailed screening conditions are needed to address complex tumour types. In addition, effective synthetic lethal targets may be defined as subsets of tumours. For instance, KRAS-mutated tumours with KEAP1 mutations and activation of NRF2 antioxidant programmes make them more susceptible to interference with the glutathione pathway193.
Combined with immune therapy
In recent decades, great progress has been made in immunotherapy for advanced cancer194. There has been substantial clinical evidence that the survival of patients with advanced cancer is improved by immune checkpoint inhibitors (ICIs), especially by blocking the immune checkpoint axis involved in programmed cell death 1 (PD-1) and programmed death ligand 1 (PD-L1)195,196. PD-1/PD-L1 inhibitors such as durvalumab, nivolumab, pembrolizumab, and atezolizumab have entered the clinic as first-line or second-line therapy for advanced NSCLC196,197,198. However, clinical data from anti-PD therapy indicated that only approximately 15–25% of patients with NSCLC responded to ICIs199. Among various drug resistance factors, the TME plays a significant role in the host response to immunotherapy200. Targeted therapy usually has a profound immunomodulatory effect on the TME, so targeted therapy combined with immunotherapy should be a strong combinational therapeutic strategy201,202.
Novel KRAS (G12C) inhibitors induce a shift of the TME from immunosuppressive to immunoreactive. Increasing evidence suggests that KRAS mutation preferentially induces an immunosuppressive TME by promoting the expression of immunomodulatory factors of tumour cells, such as transforming growth factor-β, interleukin-6, and interleukin-10203. KRAS inhibition might be able to block the effect. A relative study has shown that the infiltration of CD8 + T cells, macrophages and dendritic cells in the TME is significantly increased after AMG510 treatment, including CD103 + cross-presenting dendritic cells, which are necessary for T cell priming, activation, and recruitment126. In general, AMG510 therapy contributes to the formation of a proinflammatory microenvironment and enhances immunosurveillance. Similarly, MRTX849 treatment decreased intratumoural immunosuppressive myeloid-derived suppressor cells and M2-polarised macrophages and increased immune-promoting M1-polarised macrophages, dendritic cells, and CD4 + and NKT cells in KRAS (G12C) tumours by altering the expression of tumour RNA and protein implicated in the presentation of tumour antigens or mediating an immunosuppressive TME204. These results agree with a previous study showing that patients with KRAS mutations usually have an inflammatory TME and higher tumour immunogenicity, leading to a better response to PD-1/PD-L1 inhibitors205. Notably, the durable antitumour response to KRAS (G12C) inhibitors depends on the engagement of the immune system. Treatment with high doses of AMG510 displayed sustained tumour regression in immunocompetent mouse models, whereas tumours recovered rapidly after a short response in immunocompromised mouse models126. These studies suggest that specific inhibitors targeting KRAS (G12C) in tumour cells profitably result in a turn from an immunosuppressive to an immunocompetent TME; thus, the combination of KRAS (G12C) inhibitors and immune checkpoint inhibitors is a promising reciprocal strategy for patients with KRAS (G12C) mutant NSCLC.
Combined with tumour metabolism therapy
One of the hallmarks of cancer is the reprogramming of cellular metabolism, in which the tumour efficiently uses substances in the environment to provide more energy and biomacromolecules to meet the nutritional needs of the tumour’s uncontrolled proliferation206. Many studies have shown that KRAS mutations can lead to tumour-specific metabolic changes, thereby regulating oncogenic signalling networks and promoting tumour progression207,208. Mutant KRAS upregulates the expression of the GLUT1 glucose transporter to promote glucose uptake and glycolytic rate-limiting enzymes hexokinase 1 and 2 to increase glycolytic activity209,210. Mutant KRAS also promotes the generation of precursors of many biomacromolecules, such as the precursor for lipid and protein glycosylation through the hexosamine biosynthesis pathway211 and the backbone for nucleic acid production212. In addition to affecting glycolysis, mutant KRAS promotes tumorigenesis and development by regulating glutamine decomposition, lipid metabolism and fatty acid biosynthesis213,214,215,216. In addition, mutated KRAS meets its own needs of high nutrition and rapid metabolism through high levels of autophagy and macropinocytosis217,218,219.
The close association between the KRAS signalling and cellular metabolic remodelling makes it possible to exploit KRAS-associated metabolic fragility to treat KRAS-driven cancers220. As expected, targeted metabolic enzymes have been shown to be effective in some KRAS-mutant cancer cell lines and in mouse models221,222. Some clinical trials are underway targeting the metabolic pathways of KRAS mutant cancers, including strategies to combat glutamine breakdown and autophagy. Indeed, synergistic effects of several metabolic pathway inhibitors in combination with KRAS (G12C) inhibitors, such as the mTOR inhibitors and MRTX849, have been tested in preclinical settings with encouraging results223. However, much work remains to be done to explore the therapeutic potential of targeting metabolic changes in KRAS-driven cancers and synergistic effects with KRAS inhibitors224.
The acquired resistance mechanism of covalent KRAS (G12C) inhibitors
Despite acquiring promising preliminary clinical results from KRAS (G12C) inhibitors, it should be cautioned that complete responses are rare in clinical trials225,226. The diversity of tumour status and the sensitivity to drugs in different populations reasonably influence the drug effect because of various intrinsic factors, including dependency on KRAS-mediated signalling and concurrent genetic alterations227. Simultaneously, rapidly acquired drug resistance after promising initial responses is a very common challenge in cancer monotherapy targeting oncogenic driving enzymes. It is conceivable that there is a high possibility of acquired resistance after treatment with KRAS (G12C) inhibitors228. Several adaptive resistance mechanisms have been proposed, including the release of ERK-mediated feedback inhibition, activation of other bypasses, secondary KRAS mutations, and multiple resistance mechanisms that occur simultaneously (Fig. 7).
Acquired resistance mechanism of covalent KRAS (G12C) inhibitors. The figure on the left blue background represents cells that are sensitive to KRAS (G12C) inhibitors, while the figure on right red background represents cells that are resistant to inhibitors. The black dotted line represents inhibition of KRAS signalling in the presence of KRAS (G12C) inhibitors. The solid red lines represent the resistance mechanisms identified after the use of KRAS (G12C) inhibitors, including the relief of ERK-mediated feedback inhibition, which reactivates the MAPK pathway by wild-type RAS (NRAS and HRAS) or new synthetic KRAS (G12C); the activation of other bypasses, such as PI3K activation by the IGFR–IRS1 pathway and secondary or additional mutations in KRAS. This figure generated from BioRender
Experience gained from RAF and MEK inhibitors that have been applied in the clinic and extensively studied in their resistance mechanisms could provide important insights into the resistance mechanisms to KRAS (G12C) inhibitors229. For instance, an increased level of ERK phosphorylation after long-term treatment with RAF or MEK inhibitors was reported, which may be caused by the amplification of upstream drivers such as RTKs230. Similarly, after ARS-1620 treatment for a period of time, the recovery of ERK pathway flux was identified in vivo124. These results strongly imply that treatment with KRAS (G12C) inhibitors triggers the release of ERK-mediated feedback inhibition, which reactivates KRAS signalling in turn to confer therapeutic resistance. The work of Ryan and colleagues supported this view and proposed the mechanism of drug resistance in which wild-type RAS, which could not be specifically inhibited by KRAS (G12C) inhibitors, rapidly activated the adaptive KRAS feedback pathway mediated by PTK after ARS-1620 and AMG510 treatment in vitro170. Furthermore, no prominent RTK was found to mediate this process in all KRAS (G12C) tumour models, suggesting that this feedback may be comodulated by multiple RTKs. In an in vivo study, KRAS (G12C) colorectal cancer models had higher basal receptor tyrosine kinase (RTK) activation, especially EGFR signalling, than NSCLC cell lines. Therefore, KRAS (G12C) inhibition induced a higher rebound of phosphorylated ERK than NSCLC cells, which resulted in a generally poorer response to KRAS (G12C) inhibitors in CRC patients than in NSCLC231. Another in vitro study also supported this view, but a diverse mechanistic interpretation suggested that after treatment with KRAS (G12C) inhibitors, some cancer cells were in a stationary state with low KRAS activity, while others were equipped to resume proliferation due to the synthesis of new KRAS (G12C)177,232. In addition, it was reported that GEFR promoted the activation of newly synthesised KRAS, and aurora kinase A (AURKA) avoided drug-induced tumour cells by interacting with KRAS (G12C) and the downstream protein c-RAF177.
Epithelial-to-mesenchymal transition (EMT), another reason for resistance, was proposed in the presence of KRAS (G12C) inhibitors, which activated the PI3K pathway to lead to endogenous and acquired drug resistance. In EMT-induced cells, Adachi and colleagues demonstrated that PI3K remained activated in the presence of KRAS (G12C) inhibitors and was dominantly regulated by the IGFR–IRS1 pathway233. Another in vitro study supported this view and proposed that epithelial cells compensated through the ERBB2/3 signalling pathway, while mesenchymal cells exhibited high basal and feedback FGFR activation following inhibition of ERK and AKT signalling by ARS-1620 treatment234. In addition, considering that KRAS (G12C) covalent inhibitors inactivate KRAS (G12C) by locking them in GDP binding status, KRAS (G12C) could have a secondary mutation that led to the loss of GTPase activity or decreased affinity with GDP235. A clinical report described a patient with KRAS (G12C) NSCLC who developed polyclonal acquired resistance to MRTX849 and showed 10 heterogeneous resistance changes in 4 genes, including KRAS, NRAS, BRAF, and MAP2K1. Of note, the researchers identified an important mutation in KRAS (Y96D) that affected the Switch-II pocket, resulting in resistance to all current KRAS (G12C) inhibitors236. At the same time, researchers also reported a novel, functionally distinct tricomplex KRAS (G12C) active-state inhibitor RM-018, which can inhibit KRAS (G12C/Y96D) in vitro237. Evidence from in vitro studies confirms this conclusion, and it is clear that either AMG510 or MRTX849 results in a high proportion of KRAS secondary mutations, and 12 different secondary KRAS mutations are found. Different KRAS secondary mutations have different resistances to different drugs. For example, G13D, R68M, A59S and A59T mutations were resistant to AMG510 but sensitive to MRTX849, while the Q99L mutation was resistant to MRTX849 and remained sensitive to AMG510238. The results of a clinical cohort study (NCT03785249) further indicate that the mechanisms of acquired resistance to KRAS (G12C) inhibitors are quite complex, with multiple mechanisms occurring simultaneously in a single patient. Required KRAS alterations included G12D/R/V/W, G13D, Q61H, R68S, H95D/Q/R, Y96C and high-level amplification of the KRASG12C allele. Acquired bypass mechanisms of resistance included MET amplification, activating mutations in NRAS, BRAF, MAP2K1, and RET, oncogenic fusions involving ALK, RET, BRAF, RAF1, and FGFR3, and loss-of-function mutations in NF1 and PTEN239.
These studies partly explained why patients responded partially but not completely to treatment with KRAS (G12C) inhibitors. Furthermore, resistance could potentially occur via other undiscovered mechanisms, even multiple mechanisms, emphasizing the need to further investigate resistance mechanisms to select optimal treatment regimens for best benefits.
Concluding remarks and future perspectives
Targeting KRAS is an attractive strategy because of the high prevalence of KRAS mutations and its importance in initiating and sustaining tumour growth. Because of its own characteristics, targeting KRAS directly was once thought impossible. With continuous active exploration, there have been several novel insights to better understand KRAS mutations, promoting the development of drugs targeting KRAS. Progress has been made in targeting KRAS, especially targeting KRAS (G12C). New techniques, such as NMR-based fragment screening, tethering, and in silico drug design, have been used to discover novel agents that bind directly to KRAS, although there is a long way off to achieve targeted KRAS. Unconventional approaches of peptides and proteins are also promising but face delivery-related challenges. A class of covalently specific small molecules that bind KRAS (G12C), such as AMG510 and MRTX849, have been identified and have shown promising results in clinical trials. However, there are also some problems to be solved for its subsequent development, including the evaluation of clinical safety in a larger cohort, the optimisation of clinical efficacy and the overcoming of drug resistance. The response to KRAS (G12C) inhibitors in patients is diverse, implicating the existence of intrinsic resistance. Further exploration of intrinsic resistance should be conducted to identify biomarkers that indicate the appropriate population and tumour type in the clinic. Meanwhile, the common challenge for targeted drugs is the emergence of acquired resistance. Further investigation is clearly warranted to comprehensively elucidate the mechanisms of acquired resistance to obtain optimal treatment options.
References
Sequist, L. V. et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J. Clin. Oncol. 31, 3327–3334 (2013).
Katayama, R., Lovly, C. M. & Shaw, A. T. Therapeutic targeting of anaplastic lymphoma kinase in lung cancer: a paradigm for precision cancer medicine. Clin. Cancer Res 21, 2227–2235 (2015).
Mok, T. S. et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med 361, 947–957 (2009).
Soria, J.-C. et al. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N. Engl. J. Med 378, 113–125 (2018).
Kwak, E. L. et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N. Engl. J. Med 363, 1693–1703 (2010).
Shaw, A. T. et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N. Engl. J. Med 371, 1963–1971 (2014).
Schram, A. M. et al. A phase Ib dose-escalation and expansion study of the oral MEK inhibitor pimasertib and PI3K/MTOR inhibitor voxtalisib in patients with advanced solid tumours. Br. J. cancer 119, 1471–1476 (2018).
Liu, W. et al. Kras mutations increase telomerase activity and targeting telomerase is a promising therapeutic strategy for Kras-mutant NSCLC. Oncotarget 8, 179–190 (2017).
Srikar, R. et al. Targeted nanoconjugate co-delivering siRNA and tyrosine kinase inhibitor to KRAS mutant NSCLC dissociates GAB1-SHP2 post oncogene knockdown. Sci. Rep. 6, 30245 (2016).
Puyol, M. et al. A synthetic lethal interaction between K-Ras oncogenes and Cdk4 unveils a therapeutic strategy for non-small cell lung carcinoma. Cancer Cell 18, 63–73 (2010).
Gao, W. et al. KRAS and TP53 mutations in bronchoscopy samples from former lung cancer patients. Mol. Carcinog. 56, 381–388 (2017).
Barbacid, M. ras genes. Annu Rev. Biochem 56, 779–827 (1987).
McBride, O. W. et al. Regional chromosomal localization of N-ras, K-ras-1, K-ras-2 and myb oncogenes in human cells. Nucleic Acids Res 11, 8221–8236 (1983).
Kirsten, W. H., Schauf, V. & McCoy, J. Properties of a murine sarcoma virus. Bibl. Haematol. 246–249, (1970).
Chang, E. H. et al. Human genome contains four genes homologous to transforming genes of Harvey and Kirsten murine sarcoma viruses. Proc. Natl Acad. Sci. USA 79, 4848–4852 (1982).
Karnoub, A. E. & Weinberg, R. A. Ras oncogenes: split personalities. Nat. Rev. Mol. Cell Biol. 9, 517–531 (2008).
Plowman, S. J. et al. K-ras 4A and 4B are co-expressed widely in human tissues, and their ratio is altered in sporadic colorectal cancer. J. Exp. Clin. Cancer Res 25, 259–267 (2006).
Santos, E. & Nebreda, A. R. Structural and functional properties of ras proteins. FASEB J. 3, 2151–2163 (1989).
Vetter, I. R. & Wittinghofer, A. The guanine nucleotide-binding switch in three dimensions. Science 294, 1299–1304 (2001).
Bourne, H. R., Sanders, D. A. & McCormick, F. The GTPase superfamily: conserved structure and molecular mechanism. Nature 349, 117–127 (1991).
Takai, Y., Sasaki, T. & Matozaki, T. Small GTP-binding proteins. Physiol. Rev. 81, 153–208 (2001).
Roman, M. et al. KRAS oncogene in non-small cell lung cancer: clinical perspectives on the treatment of an old target. Mol. Cancer 17, 33 (2018).
Drugan, J. K. et al. The Ras/p120 GTPase-activating protein (GAP) interaction is regulated by the p120 GAP pleckstrin homology domain. J. Biol. Chem. 275, 35021–35027 (2000).
Bos, J. L., Rehmann, H. & Wittinghofer, A. GEFs and GAPs: critical elements in the control of small G proteins. Cell 129, 865–877 (2007).
Iversen, L. et al. Molecular kinetics. Ras activation by SOS: allosteric regulation by altered fluctuation dynamics. Science 345, 50–54 (2014).
Pamonsinlapatham, P. et al. p120-Ras GTPase activating protein (RasGAP): a multi-interacting protein in downstream signaling. Biochimie 91, 320–328 (2009).
Voldborg, B. R., Damstrup, L., Spang-Thomsen, M. & Poulsen, H. S. Epidermal growth factor receptor (EGFR) and EGFR mutations, function and possible role in clinical trials. Ann. Oncol. 8, 1197–1206 (1997).
Tulpule, A. et al. Kinase-mediated RAS signaling via membraneless cytoplasmic protein granules. Cell. 184, 2649–2664.e18 (2021).
Brambilla, R. et al. A role for the Ras signalling pathway in synaptic transmission and long-term memory. Nature 390, 281–286 (1997).
Farnsworth, C. L. et al. Calcium activation of Ras mediated by neuronal exchange factor Ras-GRF. Nature 376, 524–527 (1995).
Cullen, P. J. & Lockyer, P. J. Integration of calcium and Ras signalling. Nat. Rev. Mol. Cell Biol. 3, 339–348 (2002).
Smith, F. D. et al. AKAP-Lbc enhances cyclic AMP control of the ERK1/2 cascade. Nat. Cell Biol. 12, 1242–1249 (2010).
Zhu, G. et al. Phase Separation of Disease-Associated SHP2 Mutants Underlies MAPK Hyperactivation. Cell. 183, 490–502 (2020).
Ruess, D. A. et al. Mutant KRAS-driven cancers depend on PTPN11/SHP2 phosphatase. Nat. Med 24, 954–960 (2018).
Chan, R. J. & Feng, G.-S. PTPN11 is the first identified proto-oncogene that encodes a tyrosine phosphatase. Blood 109, 862–867 (2007).
Chen, Y.-N. P. et al. Allosteric inhibition of SHP2 phosphatase inhibits cancers driven by receptor tyrosine kinases. Nature 535, 148–152 (2016).
Eulenfeld, R. & Schaper, F. A new mechanism for the regulation of Gab1 recruitment to the plasma membrane. J. Cell Sci. 122, 55–64 (2009).
Bunda, S. et al. Inhibition of SHP2-mediated dephosphorylation of Ras suppresses oncogenesis. Nat. Commun. 6, 8859 (2015).
Agazie, Y. M. & Hayman, M. J. Molecular mechanism for a role of SHP2 in epidermal growth factor receptor signaling. Mol. Cell Biol. 23, 7875–7886 (2003).
Ekman, S. et al. SHP-2 is involved in heterodimer specific loss of phosphorylation of Tyr771 in the PDGF beta-receptor. Oncogene 21, 1870–1875 (2002).
Dance, M. et al. The molecular functions of Shp2 in the Ras/Mitogen-activated protein kinase (ERK1/2) pathway. Cell Signal 20, 453–459 (2008).
Hanafusa, H., Torii, S., Yasunaga, T. & Nishida, E. Sprouty1 and Sprouty2 provide a control mechanism for the Ras/MAPK signalling pathway. Nat. Cell Biol. 4, 850–858 (2002).
Prior, I. A., Hood, F. E. & Hartley, J. L. The Frequency of Ras Mutations in Cancer. Cancer Res 80, 2969–2974 (2020).
Martinelli, E., Morgillo, F., Troiani, T. & Ciardiello, F. Cancer resistance to therapies against the EGFR-RAS-RAF pathway: The role of MEK. Cancer Treat. Rev. 53, 61–69 (2017).
Gualdrini, F. et al. SRF Co-factors Control the Balance between Cell Proliferation and Contractility. Mol. Cell 64, 1048–1061 (2016).
Liu, R.-Y. et al. Role of p90 ribosomal S6 kinase in long-term synaptic facilitation and enhanced neuronal excitability. Sci. Rep. 10, 608 (2020).
Fruman, D. A. et al. The PI3K Pathway in Human Disease. Cell 170, 605–635 (2017).
Fresno Vara, J. A. et al. PI3K/Akt signalling pathway and cancer. Cancer Treat. Rev. 30, 193–204 (2004).
Kilic, U. et al. Particular phosphorylation of PI3K/Akt on Thr308 via PDK-1 and PTEN mediates melatonin’s neuroprotective activity after focal cerebral ischemia in mice. Redox Biol. 12, 657–665 (2017).
Hoxhaj, G. & Manning, B. D. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism. Nat. Rev. Cancer 20, 74–88 (2020).
Liu, G. Y. & Sabatini, D. M. mTOR at the nexus of nutrition, growth, ageing and disease. Nat. Rev. Mol. Cell Biol. 21, 183–203 (2020).
She, Q.-B. et al. The BAD protein integrates survival signaling by EGFR/MAPK and PI3K/Akt kinase pathways in PTEN-deficient tumor cells. Cancer Cell 8, 287–297 (2005).
Seger, R. & Krebs, E. G. The MAPK signaling cascade. FASEB J. 9, 726–735 (1995).
Tomasini, P. et al. Targeting the KRAS Pathway in Non-Small Cell Lung Cancer. oncologist 21, 1450–1460 (2016).
Schubbert, S., Shannon, K. & Bollag, G. Hyperactive Ras in developmental disorders and cancer. Nat. Rev. Cancer 7, 295–308 (2007).
Vigil, D., Cherfils, J., Rossman, K. L. & Der, C. J. Ras superfamily GEFs and GAPs: validated and tractable targets for cancer therapy? Nat. Rev. Cancer 10, 842–857 (2010).
Rhee, S. G. Regulation of phosphoinositide-specific phospholipase C. Annu Rev. Biochem 70, 281–312 (2001).
Cox, A. D. et al. Drugging the undruggable RAS: Mission possible? Nat. Rev. Drug Disco. 13, 828–851 (2014).
Prior, I. A., Lewis, P. D. & Mattos, C. A comprehensive survey of Ras mutations in cancer. Cancer Res 72, 2457–2467 (2012).
Wood, K., Hensing, T., Malik, R. & Salgia, R. Prognostic and Predictive Value in KRAS in Non-Small-Cell Lung Cancer: A Review. JAMA Oncol. 2, 805–812 (2016).
Dearden, S., Stevens, J., Wu, Y. L. & Blowers, D. Mutation incidence and coincidence in non small-cell lung cancer: meta-analyses by ethnicity and histology (mutMap). Ann. Oncol. 24, 2371–2376 (2013).
Dogan, S. et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin. Cancer Res 18, 6169–6177 (2012).
Hunter, J. C. et al. Biochemical and Structural Analysis of Common Cancer-Associated KRAS Mutations. Mol. Cancer Res 13, 1325–1335 (2015).
Ostrem, J. M. & Shokat, K. M. Direct small-molecule inhibitors of KRAS: from structural insights to mechanism-based design. Nat. Rev. Drug Disco. 15, 771–785 (2016).
Hunter, J. C. et al. Biochemical and Structural Analysis of Common Cancer-Associated KRAS Mutations. Mol. Cancer Res. 13, 1325–1335 (2015).
Rabara, D. et al. KRAS G13D sensitivity to neurofibromin-mediated GTP hydrolysis. Proc. Natl Acad. Sci. USA 116, 22122–22131 (2019).
Nadal, E. et al. KRAS-G12C mutation is associated with poor outcome in surgically resected lung adenocarcinoma. J. Thorac. Oncol.: Off. Publ. Int. Assoc. Study Lung Cancer 9, 1513–1522 (2014).
Ihle, N. T. et al. Effect of KRAS oncogene substitutions on protein behavior: implications for signaling and clinical outcome. J. Natl Cancer Inst. 104, 228–239 (2012).
Zafra, M. P. et al. An In Vivo Kras Allelic Series Reveals Distinct Phenotypes of Common Oncogenic Variants. Cancer Discov. 10, 1654–1671 (2020).
Zhao, N. et al. Alterations of LKB1 and KRAS and risk of brain metastasis: comprehensive characterization by mutation analysis, copy number, and gene expression in non-small-cell lung carcinoma. Lung cancer (Amst., Neth.) 86, 255–261 (2014).
Jia, Y. et al. Characterization of distinct types of mutation and its impact on first-line platinum-based chemotherapy in Chinese patients with advanced non-small cell lung cancer. Oncol. Lett. 14, 6525–6532 (2017).
Arriaga, J. M. et al. A MYC and RAS co-activation signature in localized prostate cancer drives bone metastasis and castration resistance. Nat. Cancer 1, 1082–1096 (2020).
Scheffler, M. et al. K-ras Mutation Subtypes in NSCLC and Associated Co-occuring Mutations in Other Oncogenic Pathways. J. Thorac. Oncol.: Off. Publ. Int. Assoc. Study Lung Cancer 14, 606–616 (2019).
Zheng, S. et al. Targeted next-generation sequencing for cancer-associated gene mutation and copy number detection in 206 patients with non-small-cell lung cancer. Bioengineered 12, 791–802 (2021).
Skoulidis, F. & Heymach, J. V. Co-occurring genomic alterations in non-small-cell lung cancer biology and therapy. Nat. Rev. Cancer 19, 495–509 (2019).
Arbour, K. C. et al. Effects of Co-occurring Genomic Alterations on Outcomes in Patients with -Mutant Non-Small Cell Lung Cancer. Clin. Cancer Res 24, 334–340 (2018).
Bange, E. et al. Impact of and Co-Mutations on Outcomes After First-Line Systemic Therapy Among Patients With -Mutated Advanced Non-Small-Cell Lung Cancer. JCO Precis Oncol. 3, e2037120 (2019).
Schmid, S. et al. Clinical Outcome of ALK-Positive Non-Small Cell Lung Cancer (NSCLC) Patients with De Novo EGFR or KRAS Co-Mutations Receiving Tyrosine Kinase Inhibitors (TKIs). J. Thorac. Oncol.: Off. Publ. Int. Assoc. Study Lung Cancer 12, 681–688 (2017).
Loong, H. H. et al. KRAS G12C mutations in Asia: a landscape analysis of 11,951 Chinese tumor samples. Transl. Lung Cancer Res. 9, 1759–1769 (2020).
Pylayeva-Gupta, Y., Grabocka, E. & Bar-Sagi, D. RAS oncogenes: weaving a tumorigenic web. Nat. Rev. Cancer 11, 761–774 (2011).
Dias Carvalho, P. et al. KRAS Oncogenic Signaling Extends beyond Cancer Cells to Orchestrate the Microenvironment. Cancer Res. 78, 7–14 (2018).
Kitajima, S., Thummalapalli, R. & Barbie, D. A. Inflammation as a driver and vulnerability of KRAS mediated oncogenesis. Semin. cell developmental Biol. 58, 127–135 (2016).
Golay, H. G. & Barbie, D. A. Targeting cytokine networks in KRAS-driven tumorigenesis. Expert Rev. Anticancer Ther. 14, 869–871 (2014).
Taniguchi, K. & Karin, M. IL-6 and related cytokines as the critical lynchpins between inflammation and cancer. Semin. Immunol. 26, 54–74 (2014).
Lesina, M. et al. Stat3/Socs3 activation by IL-6 transsignaling promotes progression of pancreatic intraepithelial neoplasia and development of pancreatic cancer. Cancer Cell 19, 456–469 (2011).
Ancrile, B., Lim, K. H. & Counter, C. M. Oncogenic Ras-induced secretion of IL6 is required for tumorigenesis. Genes Dev. 21, 1714–1719 (2007).
Brooks, G. D. et al. IL6 Trans-signaling Promotes KRAS-Driven Lung Carcinogenesis. Cancer Res 76, 866–876 (2016).
Corcoran, R. B. et al. STAT3 plays a critical role in KRAS-induced pancreatic tumorigenesis. Cancer Res 71, 5020–5029 (2011).
Zhang, Y. et al. Interleukin-6 is required for pancreatic cancer progression by promoting MAPK signaling activation and oxidative stress resistance. Cancer Res 73, 6359–6374 (2013).
Sunaga, N. et al. Oncogenic KRAS-induced interleukin-8 overexpression promotes cell growth and migration and contributes to aggressive phenotypes of non-small cell lung cancer. Int J. Cancer 130, 1733–1744 (2012).
Sparmann, A. & Bar-Sagi, D. Ras-induced interleukin-8 expression plays a critical role in tumor growth and angiogenesis. Cancer Cell 6, 447–458 (2004).
Awaji, M. et al. CXCR2 signaling promotes secretory cancer-associated fibroblasts in pancreatic ductal adenocarcinoma. FASEB J. 34, 9405–9418 (2020).
Ling, J. et al. KrasG12D-induced IKK2/β/NF-κB activation by IL-1α and p62 feedforward loops is required for development of pancreatic ductal adenocarcinoma. Cancer Cell 21, 105–120 (2012).
Meylan, E. et al. Requirement for NF-kappaB signalling in a mouse model of lung adenocarcinoma. Nature 462, 104–107 (2009).
Zhu, Z. et al. Inhibition of KRAS-driven tumorigenicity by interruption of an autocrine cytokine circuit. Cancer Disco. 4, 452–465 (2014).
Liou, G. Y. et al. Mutant KRAS-induced expression of ICAM-1 in pancreatic acinar cells causes attraction of macrophages to expedite the formation of precancerous lesions. Cancer Disco. 5, 52–63 (2015).
McAllister, F. et al. Oncogenic Kras activates a hematopoietic-to-epithelial IL-17 signaling axis in preinvasive pancreatic neoplasia. Cancer Cell 25, 621–637 (2014).
Hamarsheh, S. et al. Oncogenic Kras(G12D) causes myeloproliferation via NLRP3 inflammasome activation. Nat. Commun. 11, 1659 (2020).
van Maldegem, F. & Downward, J. Mutant KRAS at the Heart of Tumor Immune Evasion. Immunity 52, 14–16 (2020).
Sumimoto, H., Takano, A., Teramoto, K. & Daigo, Y. RAS-Mitogen-Activated Protein Kinase Signal Is Required for Enhanced PD-L1 Expression in Human Lung Cancers. PLoS One 11, e0166626 (2016).
Chen, N. et al. KRAS mutation-induced upregulation of PD-L1 mediates immune escape in human lung adenocarcinoma. Cancer Immunol., immunotherapy: CII 66, 1175–1187 (2017).
Coelho, M. A. et al. Oncogenic RAS Signaling Promotes Tumor Immunoresistance by Stabilizing PD-L1 mRNA. Immunity 47, 1083–1099.e1086 (2017).
Glorieux, C. et al. Regulation of PD-L1 expression in K-ras-driven cancers through ROS-mediated FGFR1 signaling. Redox Biol. 38, 101780 (2021).
Testorelli, C. et al. Dacarbazine-induced immunogenicity of a murine leukemia is attenuated in cells transfected with mutated K-ras gene. J. Exp. Clin. Cancer Res 16, 15–22 (1997).
Smakman, N. et al. Dual effect of Kras(D12) knockdown on tumorigenesis: increased immune-mediated tumor clearance and abrogation of tumor malignancy. Oncogene 24, 8338–8342 (2005).
Zdanov, S. et al. Mutant KRAS Conversion of Conventional T Cells into Regulatory T Cells. Cancer Immunol. Res. 4, 354–365 (2016).
Granville, C. A. et al. A central role for Foxp3+ regulatory T cells in K-Ras-driven lung tumorigenesis. PLoS One 4, e5061 (2009).
Petanidis, S. et al. Differential expression of IL-17, 22 and 23 in the progression of colorectal cancer in patients with K-ras mutation: Ras signal inhibition and crosstalk with GM-CSF and IFN-γ. PLoS One 8, e73616 (2013).
Bayne, L. J. et al. Tumor-derived granulocyte-macrophage colony-stimulating factor regulates myeloid inflammation and T cell immunity in pancreatic cancer. Cancer Cell 21, 822–835 (2012).
Liao, W. et al. KRAS-IRF2 Axis Drives Immune Suppression and Immune Therapy Resistance in Colorectal Cancer. Cancer Cell 35, 559–572.e557 (2019).
Kortlever, R. M. et al. Myc Cooperates with Ras by Programming Inflammation and Immune Suppression. Cell 171, 1301–1315.e1314 (2017).
Hashimoto, S. et al. ARF6 and AMAP1 are major targets of KRAS and TP53 mutations to promote invasion, PD-L1 dynamics, and immune evasion of pancreatic cancer. Proc. Natl Acad. Sci. USA 116, 17450–17459 (2019).
Dong, Z. Y. et al. Potential Predictive Value of TP53 and KRAS Mutation Status for Response to PD-1 Blockade Immunotherapy in Lung Adenocarcinoma. Clin. Cancer Res 23, 3012–3024 (2017).
Hunter, J. C. et al. Biochemical and Structural Analysis of Common Cancer-Associated KRAS Mutations. Mol. Cancer Res 13, 1325–1335 (2015).
Lim, S. M. et al. Therapeutic targeting of oncogenic K-Ras by a covalent catalytic site inhibitor. Angew. Chem. Int Ed. Engl. 53, 199–204 (2014).
Hunter, J. C. et al. In situ selectivity profiling and crystal structure of SML-8-73-1, an active site inhibitor of oncogenic K-Ras G12C. Proc. Natl Acad. Sci. USA 111, 8895–8900 (2014).
Ostrem, J. M. et al. K-Ras(G12C) inhibitors allosterically control GTP affinity and effector interactions. Nature 503, 548–551 (2013).
Milburn, M. V. et al. Molecular switch for signal transduction: structural differences between active and inactive forms of protooncogenic ras proteins. Science 247, 939–945 (1990).
Taveras, A. G. et al. Ras oncoprotein inhibitors: the discovery of potent, ras nucleotide exchange inhibitors and the structural determination of a drug-protein complex. Bioorg. Med Chem. 5, 125–133 (1997).
Lito, P. et al. Allele-specific inhibitors inactivate mutant KRAS G12C by a trapping mechanism. Science 351, 604–608 (2016).
Patricelli, M. P. et al. Selective Inhibition of Oncogenic KRAS Output with Small Molecules Targeting the Inactive State. Cancer Discov. 6, 316–329 (2016).
Shipman, L. Signalling: Putting the brakes on KRAS-G12C nucleotide cycling. Nat. Rev. Cancer 16, 127 (2016).
Hobbs, G. A., Wittinghofer, A. & Der, C. J. Selective Targeting of the KRAS G12C Mutant: Kicking KRAS When It’s Down. Cancer Cell 29, 251–253 (2016).
Janes, M. R. et al. Targeting KRAS Mutant Cancers with a Covalent G12C-Specific Inhibitor. Cell. 172, 578–589.e17 (2018).
Hansen, R. et al. The reactivity-driven biochemical mechanism of covalent KRAS inhibitors. Nat. Struct. Mol. Biol. 25, 454–462 (2018).
Canon, J. et al. The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature 575, 217–223 (2019).
Papadopoulos, K. P. et al. A phase I/II multiple expansion cohort trial of MRTX849 in patients with advanced solid tumors with KRAS G12C mutation. J. Clin. Oncol. 37, TPS3161–TPS3161 (2019).
Gentile, D. R. et al. Ras Binder Induces a Modified Switch-II Pocket in GTP and GDP States. Cell Chem. Biol. 24, 1455–1466 (2017).
Hallin, J. et al. The KRAS(G12C) Inhibitor MRTX849 Provides Insight toward Therapeutic Susceptibility of KRAS-Mutant Cancers in Mouse Models and Patients. Cancer Disco. 10, 54–71 (2020).
FDA Approves First KRAS Inhibitor: Sotorasib. Cancer Discov. 11, OF4, (2021).
Hong, D. S. et al. KRAS Inhibition with Sotorasib in Advanced Solid Tumors. N. Engl. J. Med 383, 1207–1217 (2020).
Fell, J. B. et al. Identification of the Clinical Development Candidate MRTX849, a Covalent KRAS(G12C) Inhibitor for the Treatment of Cancer. J. Med Chem. 63, 6679–6693 (2020).
Gu, S. et al. PROTACs: An Emerging Targeting Technique for Protein Degradation in Drug Discovery. Bioessays 40, e1700247 (2018).
Burslem, G. M. et al. The Advantages of Targeted Protein Degradation Over Inhibition: An RTK Case Study. Cell Chem. Biol. 25, 67–77 (2018).
Zeng, M. et al. Exploring Targeted Degradation Strategy for Oncogenic KRAS. Cell Chem. Biol. 27, 19–31 (2020).
Bond, M. J. et al. Targeted Degradation of Oncogenic KRAS by VHL-Recruiting PROTACs. ACS Cent. Sci. 6, 1367–1375 (2020).
Merlet, J., Burger, J., Gomes, J. E. & Pintard, L. Regulation of cullin-RING E3 ubiquitin-ligases by neddylation and dimerization. Cell Mol. Life Sci. 66, 1924–1938 (2009).
Lim, S. et al. Exquisitely Specific anti-KRAS Biodegraders Inform on the Cellular Prevalence of Nucleotide-Loaded States. ACS Cent. Sci. 7, 274–291 (2021).
Sakamoto, K. et al. K-Ras(G12D)-selective inhibitory peptides generated by random peptide T7 phage display technology. Biochem Biophys. Res Commun. 484, 605–611 (2017).
Sakamoto, K., Masutani, T. & Hirokawa, T. Generation of KS-58 as the first K-Ras(G12D)-inhibitory peptide presenting anti-cancer activity in vivo. Sci. Rep. 10, 21671 (2020).
McAndrews, K. M. et al. Exosome-mediated delivery of CRISPR/Cas9 for targeting of oncogenic Kras in pancreatic cancer. Life Sci Alliance. 4, e202000875 (2021).
Teng, K. W. et al. Selective and noncovalent targeting of RAS mutants for inhibition and degradation. Nat. Commun. 12, 2656 (2021).
Kessler, D. et al. Drugging an undruggable pocket on KRAS. Proc. Natl Acad. Sci. USA 116, 15823–15829 (2019).
Dang, C. V., Reddy, E. P., Shokat, K. M. & Soucek, L. Drugging the ‘undruggable’ cancer targets. Nat. Rev. Cancer 17, 502–508 (2017).
Goody, R. S., Frech, M. & Wittinghofer, A. Affinity of guanine nucleotide binding proteins for their ligands: facts and artefacts. Trends Biochem Sci. 16, 327–328 (1991).
Kamerkar, S. et al. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature 546, 498–503 (2017).
Kole, R., Krainer, A. R. & Altman, S. RNA therapeutics: beyond RNA interference and antisense oligonucleotides. Nat. Rev. Drug Discov. 11, 125–140 (2012).
Ross, S. J. et al. Targeting KRAS-dependent tumors with AZD4785, a high-affinity therapeutic antisense oligonucleotide inhibitor of KRAS. Sci Transl Med. 9, eaal5253 (2017).
Cox, A. D., Der, C. J. & Philips, M. R. Targeting RAS Membrane Association: Back to the Future for Anti-RAS Drug Discovery? Clin. Cancer Res 21, 1819–1827 (2015).
Basso, A. D., Kirschmeier, P. & Bishop, W. R. Lipid posttranslational modifications. Farnesyl transferase inhibitors. J. Lipid Res 47, 15–31 (2006).
Berndt, N., Hamilton, A. D. & Sebti, S. M. Targeting protein prenylation for cancer therapy. Nat. Rev. Cancer 11, 775–791 (2011).
Whyte, D. B. et al. K- and N-Ras are geranylgeranylated in cells treated with farnesyl protein transferase inhibitors. J. Biol. Chem. 272, 14459–14464 (1997).
Lobell, R. B. et al. Preclinical and clinical pharmacodynamic assessment of L-778,123, a dual inhibitor of farnesyl:protein transferase and geranylgeranyl:protein transferase type-I. Mol. Cancer Ther. 1, 747–758 (2002).
Riely, G. J. et al. A phase II trial of Salirasib in patients with lung adenocarcinomas with KRAS mutations. J. Thorac. Oncol.: Off. Publ. Int. Assoc. Study Lung Cancer 6, 1435–1437 (2011).
Winter-Vann, A. M. & Casey, P. J. Post-prenylation-processing enzymes as new targets in oncogenesis. Nat. Rev. Cancer 5, 405–412 (2005).
Majmudar, J. D. et al. Amide-modified prenylcysteine based Icmt inhibitors: Structure-activity relationships, kinetic analysis and cellular characterization. Bioorg. Med Chem. 20, 283–295 (2012).
Zimmermann, G. et al. Small molecule inhibition of the KRAS-PDEδ interaction impairs oncogenic KRAS signalling. Nature 497, 638–642 (2013).
Patgiri, A., Yadav, K. K., Arora, P. S. & Bar-Sagi, D. An orthosteric inhibitor of the Ras-Sos interaction. Nat. Chem. Biol. 7, 585–587 (2011).
Leshchiner, E. S. et al. Direct inhibition of oncogenic KRAS by hydrocarbon-stapled SOS1 helices. Proc. Natl Acad. Sci. USA 112, 1761–1766 (2015).
Mott, H. R. & Owen, D. Structures of Ras superfamily effector complexes: What have we learnt in two decades? Crit Rev Biochem Mol Biol. 50, 85–133 (2015).
Upadhyaya, P. et al. Inhibition of Ras signaling by blocking Ras-effector interactions with cyclic peptides. Angew. Chem. Int Ed. Engl. 54, 7602–7606 (2015).
Zhang, Z. et al. GTP-State-Selective Cyclic Peptide Ligands of K-Ras(G12D) Block Its Interaction with Raf. ACS Cent. Sci. 6, 1753–1761 (2020).
Bayat Mokhtari, R. et al. Combination therapy in combating cancer. Oncotarget 8, 38022–38043 (2017).
Sun, J.-M. et al. Prognostic and predictive value of KRAS mutations in advanced non-small cell lung cancer. PloS one 8, e64816 (2013).
Mak, R. H. et al. Outcomes by tumor histology and KRAS mutation status after lung stereotactic body radiation therapy for early-stage non-small-cell lung cancer. Clin. Lung Cancer 16, 24–32 (2015).
Wang, M. et al. EGFR-mediated chromatin condensation protects KRAS-mutant cancer cells against ionizing radiation. Cancer Res 74, 2825–2834 (2014).
Wang, M. et al. Radiation Resistance in KRAS-Mutated Lung Cancer Is Enabled by Stem-like Properties Mediated by an Osteopontin-EGFR Pathway. Cancer Res 77, 2018–2028 (2017).
Gurtner, K. et al. Radioresistance of KRAS/TP53-mutated lung cancer can be overcome by radiation dose escalation or EGFR tyrosine kinase inhibition in vivo. Int J. Cancer 147, 472–477 (2020).
Herter-Sprie, G. S. et al. Synergy of radiotherapy and PD-1 blockade in Kras-mutant lung cancer. JCI Insight 1, e87415 (2016).
Ryan, M. B. et al. Vertical Pathway Inhibition Overcomes Adaptive Feedback Resistance to KRAS Inhibition. Clin. Cancer Res. 26, 1633–1643 (2020).
Zhang, J., Zhang, F. & Niu, R. Functions of Shp2 in cancer. J. Cell Mol. Med 19, 2075–2083 (2015).
Fedele, C. et al. SHP2 Inhibition Prevents Adaptive Resistance to MEK Inhibitors in Multiple Cancer Models. Cancer Discov. 8, 1237–1249 (2018).
Nichols, R. J. et al. RAS nucleotide cycling underlies the SHP2 phosphatase dependence of mutant BRAF-, NF1- and RAS-driven cancers. Nat. Cell Biol. 20, 1064–1073 (2018).
Wong, G. S. et al. Targeting wild-type KRAS-amplified gastroesophageal cancer through combined MEK and SHP2 inhibition. Nat. Med 24, 968–977 (2018).
Mainardi, S. et al. SHP2 is required for growth of KRAS-mutant non-small-cell lung cancer in vivo. Nat. Med 24, 961–967 (2018).
Fedele, C. et al. SHP2 inhibition diminishes KRASG12C cycling and promotes tumor microenvironment remodeling. J Exp Med. 218, e20201414 (2021).
Xue, J. Y. et al. Rapid non-uniform adaptation to conformation-specific KRAS(G12C) inhibition. Nature 577, 421–425 (2020).
Liu, C. et al. Combinations with Allosteric SHP2 Inhibitor TNO155 to Block Receptor Tyrosine Kinase Signaling. Clin. Cancer Res 27, 342–354 (2021).
Hillig, R. C. et al. Discovery of potent SOS1 inhibitors that block RAS activation via disruption of the RAS-SOS1 interaction. Proc. Natl Acad. Sci. USA 116, 2551–2560 (2019).
Bogucka, K. et al. ERK3/MAPK6 is required for KRAS-mediated NSCLC tumorigenesis. Cancer Gene Ther. 28, 359–374 (2020).
Lito, P. et al. Relief of profound feedback inhibition of mitogenic signaling by RAF inhibitors attenuates their activity in BRAFV600E melanomas. Cancer Cell 22, 668–682 (2012).
Misale, S. et al. KRAS G12C NSCLC Models Are Sensitive to Direct Targeting of KRAS in Combination with PI3K Inhibition. Clin. Cancer Res 25, 796–807 (2019).
Molina-Arcas, M. et al. Development of combination therapies to maximize the impact of KRAS-G12C inhibitors in lung cancer. Sci. Transl. Med. 11, 359–374 (2019).
Barbie, D. A. et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature 462, 108–112 (2009).
Kumar, M. S. et al. The GATA2 transcriptional network is requisite for RAS oncogene-driven non-small cell lung cancer. Cell 149, 642–655 (2012).
Luo, J. et al. A genome-wide RNAi screen identifies multiple synthetic lethal interactions with the Ras oncogene. Cell 137, 835–848 (2009).
Wang, T. et al. Gene Essentiality Profiling Reveals Gene Networks and Synthetic Lethal Interactions with Oncogenic Ras. Cell. 168, 890–903.e15. (2017).
Sanclemente, M. et al. c-RAF Ablation Induces Regression of Advanced Kras/Trp53 Mutant Lung Adenocarcinomas by a Mechanism Independent of MAPK Signaling. Cancer Cell. 33, 217–228.e4 (2018).
Sulahian, R. et al. Synthetic Lethal Interaction of SHOC2 Depletion with MEK Inhibition in RAS-Driven Cancers. Cell Rep. 29, 118–134.e8(2019).
Michels, B. E. et al. Pooled In Vitro and In Vivo CRISPR-Cas9 Screening Identifies Tumor Suppressors in Human Colon Organoids. Cell Stem Cell. 26, 782–792.e7 (2020).
Lou, K. et al. KRAS inhibition produces a driver-limited state revealing collateral dependencies. Sci Signal. 12, eaaw9450 (2019).
Yaeger, R. & Solit, D. B. Overcoming Adaptive Resistance to KRAS Inhibitors Through Vertical Pathway Targeting. Clin. Cancer Res 26, 1538–1540 (2020).
Romero, R. et al. Keap1 loss promotes Kras-driven lung cancer and results in dependence on glutaminolysis. Nat. Med 23, 1362–1368 (2017).
Kruger, S. et al. Advances in cancer immunotherapy 2019 - latest trends. J. Exp. Clin. Cancer Res 38, 268 (2019).
Addeo, A., Banna, G. L. & Metro, G. & Di Maio, M. Chemotherapy in Combination With Immune Checkpoint Inhibitors for the First-Line Treatment of Patients With Advanced Non-small Cell Lung Cancer: A Systematic Review and Literature-Based Meta-Analysis. Front. Oncol. 9, 264 (2019).
Brahmer, J. et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N. Engl. J. Med 373, 123–135 (2015).
Mok, T. S. K. et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 393, 1819–1830 (2019).
Antonia, S. J. et al. Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N. Engl. J. Med 379, 2342–2350 (2018).
Mazieres, J. et al. Immune checkpoint inhibitors for patients with advanced lung cancer and oncogenic driver alterations: results from the IMMUNOTARGET registry. Ann. Oncol. 30, 1321–1328 (2019).
Binnewies, M. et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med 24, 541–550 (2018).
Hughes, P. E., Caenepeel, S. & Wu, L. C. Targeted Therapy and Checkpoint Immunotherapy Combinations for the Treatment of Cancer. Trends Immunol. 37, 462–476 (2016).
Mathew, M., Enzler, T., Shu, C. A. & Rizvi, N. A. Combining chemotherapy with PD-1 blockade in NSCLC. Pharm. Ther. 186, 130–137 (2018).
Cullis, J., Das, S. & Bar-Sagi, D. Kras and Tumor Immunity: Friend or Foe? Cold Spring Harb Perspect Med. 8, a031849 (2018).
Briere, D. M. et al. The KRASG12C Inhibitor MRTX849 Reconditions the Tumor Immune Microenvironment and Sensitizes Tumors to Checkpoint Inhibitor Therapy. Mol. Cancer Ther. 20, 975–985 (2021).
Liu, C. et al. The superior efficacy of anti-PD-1/PD-L1 immunotherapy in KRAS-mutant non-small cell lung cancer that correlates with an inflammatory phenotype and increased immunogenicity. Cancer Lett. 470, 95–105 (2020).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Mayers, J. R. et al. Tissue of origin dictates branched-chain amino acid metabolism in mutant Kras-driven cancers. Science 353, 1161–1165 (2016).
Yuneva, M. O. et al. The metabolic profile of tumors depends on both the responsible genetic lesion and tissue type. Cell Metab. 15, 157–170 (2012).
Amendola, C. R. et al. KRAS4A directly regulates hexokinase 1. Nature 576, 482–486 (2019).
Yun, J. et al. Glucose deprivation contributes to the development of KRAS pathway mutations in tumor cells. Science 325, 1555–1559 (2009).
Slawson, C., Copeland, R. J. & Hart, G. W. O-GlcNAc signaling: a metabolic link between diabetes and cancer? Trends Biochem Sci. 35, 547–555 (2010).
Santana-Codina, N. et al. Oncogenic KRAS supports pancreatic cancer through regulation of nucleotide synthesis. Nat. Commun. 9, 4945 (2018).
Gaglio, D. et al. Oncogenic K-Ras decouples glucose and glutamine metabolism to support cancer cell growth. Mol. Syst. Biol. 7, 523 (2011).
Son, J. et al. Glutamine supports pancreatic cancer growth through a KRAS-regulated metabolic pathway. Nature 496, 101–105 (2013).
Gouw, A. M. et al. Oncogene KRAS activates fatty acid synthase, resulting in specific ERK and lipid signatures associated with lung adenocarcinoma. Proc. Natl Acad. Sci. USA 114, 4300–4305 (2017).
Salloum, D. et al. Mutant ras elevates dependence on serum lipids and creates a synthetic lethality for rapamycin. Mol. Cancer Ther. 13, 733–741 (2014).
Amaravadi, R. K., Kimmelman, A. C. & Debnath, J. Targeting Autophagy in Cancer: Recent Advances and Future Directions. Cancer Disco. 9, 1167–1181 (2019).
Guo, J. Y. et al. Activated Ras requires autophagy to maintain oxidative metabolism and tumorigenesis. Genes Dev. 25, 460–470 (2011).
Humpton, T. J. et al. Oncogenic KRAS Induces NIX-Mediated Mitophagy to Promote Pancreatic Cancer. Cancer Disco. 9, 1268–1287 (2019).
Kelly, M. R. et al. Combined Proteomic and Genetic Interaction Mapping Reveals New RAS Effector Pathways and Susceptibilities. Cancer Discov. 10, 1950–1967 (2020).
Romero, R. et al. Keap1 loss promotes Kras-driven lung cancer and results in dependence on glutaminolysis. Nat. Med 23, 1362–1368 (2017).
Saqcena, M. et al. Blocking anaplerotic entry of glutamine into the TCA cycle sensitizes K-Ras mutant cancer cells to cytotoxic drugs. Oncogene 34, 2672–2680 (2015).
Hallin, J. et al. The KRAS(G12C) Inhibitor MRTX849 Provides Insight toward Therapeutic Susceptibility of KRAS-Mutant Cancers in Mouse Models and Patients. Cancer Disco. 10, 54–71 (2020).
Kerk, S. A., Papagiannakopoulos, T., Shah, Y. M. & Lyssiotis, C. A. Metabolic networks in mutant KRAS-driven tumours: tissue specificities and the microenvironment. Nat. Rev. Cancer 21, 510–525 (2021).
Singh, A. et al. A gene expression signature associated with “K-Ras addiction” reveals regulators of EMT and tumor cell survival. Cancer Cell 15, 489–500 (2009).
Muzumdar, M. D. et al. Survival of pancreatic cancer cells lacking KRAS function. Nat. Commun. 8, 1090 (2017).
Holohan, C., Van Schaeybroeck, S., Longley, D. B. & Johnston, P. G. Cancer drug resistance: an evolving paradigm. Nat. Rev. Cancer 13, 714–726 (2013).
Garraway, L. A. & Jänne, P. A. Circumventing cancer drug resistance in the era of personalized medicine. Cancer Discov. 2, 214–226 (2012).
Poulikakos, P. I. & Solit, D. B. Resistance to MEK inhibitors: should we co-target upstream. Sci. Signal 4, pe16 (2011).
Luebker, S. A. & Koepsell, S. A. Diverse Mechanisms of BRAF Inhibitor Resistance in Melanoma Identified in Clinical and Preclinical Studies. Front. Oncol. 9, 268 (2019).
Amodio, V. et al. EGFR Blockade Reverts Resistance to KRAS Inhibition in Colorectal Cancer. Cancer Disco. 10, 1129–1139 (2020).
Another KRAS Inhibitor Holds Its Own. Cancer Discov. 10, OF2, (2020).
Adachi, Y. et al. Epithelial-to-Mesenchymal Transition is a Cause of Both Intrinsic and Acquired Resistance to KRAS G12C Inhibitor in KRAS G12C-Mutant Non-Small Cell Lung Cancer. Clin. Cancer Res 26, 5962–5973 (2020).
Solanki, H. S. et al. Cell Type-specific Adaptive Signaling Responses to KRAS(G12C) Inhibition. Clin. Cancer Res. 27, 2533–2548 (2021).
Hallin, J. et al. The KRAS Inhibitor MRTX849 Provides Insight toward Therapeutic Susceptibility of KRAS-Mutant Cancers in Mouse Models and Patients. Cancer Discov. 10, 54–71 (2020).
Tanaka, N. et al. Clinical acquired resistance to KRASG12C inhibition through a novel KRAS switch-II pocket mutation and polyclonal alterations converging on RAS-MAPK reactivation. Cancer Discov. 11, 1913–1922 (2021).
Tanaka, N. et al. Clinical Acquired Resistance to KRAS Inhibition through a Novel KRAS Switch-II Pocket Mutation and Polyclonal Alterations Converging on RAS-MAPK Reactivation. Cancer Discov. 11, 1913–1922 (2021).
Koga, T. et al. KRAS secondary mutations that confer acquired resistance to KRAS G12C inhibitors, sotorasib and adagrasib, and overcoming strategies: insights from the in vitro experiments. J. Thorac. Oncol. 16, 1321–1332 (2021).
Awad, M. M. et al. Acquired Resistance to KRAS Inhibition in Cancer. N. Engl. J. Med 384, 2382–2393 (2021).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (NO. 82073882) and Natural Science Research Team Foundation of Guangdong Province, China (NO. 2016A030312014).
Author information
Authors and Affiliations
Contributions
L.F. conceived the review. L.H. conducted the literature search and draughted the manuscript. F.W. and Z.G. revised the manuscript. All authors have read and approved the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Abbreviation:
KRAS: Kirsten rat sarcoma viral oncogene homologue; NSCLC: small-cell and nonsmall-cell lung cancer; EGFR: epithelial growth factor receptor; ALK: anaplastic lymphoma kinase; CDK: cyclin-dependent kinase; HRAS: Harvey rat sarcoma virus oncogene; NRAS: neuroblastoma rat sarcoma virus oncogene; HVR: hypervariable region; GEFs: guanine nucleotide exchange factors; SOS: son-of-sevenless homologue; GAPs: GTPase-activating proteins; RAF: rapidly accelerated fibrosarcoma; MAPK: mitogen-activated protein kinase; ERK: extracellular signal regulated kinase; PI3K: phosphatidylinositol 3-kinase; GRB2: growth factor receptor-bound protein 2; AKT: protein kinase B; mTOR: mammalian target of rapamycin; RALGDS: RAS-related protein guanine nucleotide dissociation stimulator; RAL: RAS -related protein; TIAM1: tumour invasion and metastasis-inducing protein 1; FTIs: farnesyltransferase inhibitors; RCE: RAS-converting enzyme; ICMT1: isoprenyl carboxyl methyltransferase 1; PDE δ: phosphodiesterase-δ; Cys12: Cysteine 12; ORR: objective response rate; DCR: disease control rate; GFP: green fluorescent proteins; RTK: receptor tyrosine kinase; SHP2: Src homology region 2 domain-containing phosphatase-2; PD-1: programmed cell death 1; PD-L1: programmed death-ligand 1; TME: tumour microenvironment; IGFR: insulin-like growth factor receptor; IRS1: insulin receptor substrate 1; EMT: epithelial-to-mesenchymal transition; PROTACs: Proteolysis-targeting chimeras.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, L., Guo, Z., Wang, F. et al. KRAS mutation: from undruggable to druggable in cancer. Sig Transduct Target Ther 6, 386 (2021). https://doi.org/10.1038/s41392-021-00780-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41392-021-00780-4
This article is cited by
-
BI-2865, a pan-KRAS inhibitor, reverses the P-glycoprotein induced multidrug resistance in vitro and in vivo
Cell Communication and Signaling (2024)
-
IκBα kinase inhibitor BAY 11-7082 promotes anti-tumor effect in RAS-driven cancers
Journal of Translational Medicine (2024)
-
CRISPR/Cas9 gene editing: a novel strategy for fighting drug resistance in respiratory disorders
Cell Communication and Signaling (2024)
-
Deciphering the molecular interplay and tumorigenesis in hepatocellular carcinoma through insights into FBXL6 and KRASG12D
Military Medical Research (2024)
-
Targeting AGTPBP1 inhibits pancreatic cancer progression via regulating microtubules and ERK signaling pathway
Molecular Medicine (2024)