Abstract
Background
To investigate the efficacy of transperineal targeted microwave ablation (TMA) in treating localized prostate cancer (PCa).
Methods
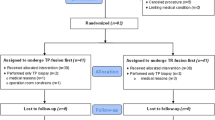
This is a single-centre prospective phase 2 trial recruiting men with low to intermediate-risk localized PCa to undergo transperineal TMA. TMA was performed with MRI-Ultrasound fusion guidance and organ-based tracking. A per-protocol 6-month MRI and biopsy were performed for all patients. The primary outcome was any cancer detected on biopsy of each ablated area. Secondary outcomes included per-patient analysis of positive biopsy, complications, urinary symptom score, erectile function and quality of life (QOL) scores.
Results
In the first 15 men, 23 areas were being treated. The median age was 70 years, number of TMA ablations were 5 (range 2–8), and the total ablation time and operating time was 22 (IQR 14–28) and 75 (IQR 65-85) minutes, respectively. PSA level dropped from a median of 7.7 to 2.4 ng/mL. For the primary outcome, 91.3% (21/23) ablated area had no cancer in 6-month biopsy. In per-patient analysis, 33.3% (5/15) had in or out-of-field positive biopsy at 6 months. Among these five cases, four of them were amenable to active surveillance and 1 (6.7%) case with out-of-field ISUP grade group 2 cancer received radiotherapy. The urinary symptoms, uroflowmetry, erectile function, and QOL scores had no significant difference at 6 months. One patient (out of five patients with normal erection) in the cohort complained of significant worsening of erectile function after TMA. Grade 1 complications including hematuria (33.3%), dysuria (6.7%), and perineal discomfort (13.4%) were observed.
Conclusions
In this first pilot study, transperineal TMA guided by MRI-Ultrasound fusion guidance and organ-based tracking was shown to be effective, safe, and easily applicable in men with localized PCa.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mottet N, van den Bergh RCN, Briers E, Van den Broeck T, Cumberbatch MG, De Santis M, et al. EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate cancer-2020 update. part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2021;79:243–62.
Bokhorst LP, Valdagni R, Rannikko A, Kakehi Y, Pickles T, Bangma CH, et al. A decade of active surveillance in the PRIAS study: an update and evaluation of the criteria used to recommend a switch to active treatment. Eur Urol. 2016;70:954–60.
Hamdy FC, Donovan JL, Lane JA, Mason M, Metcalfe C, Holding P, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415–24.
Stabile A, Orczyk C, Hosking-Jervis F, Giganti F, Arya M, Hindley RG, et al. Medium-term oncological outcomes in a large cohort of men treated with either focal or hemi-ablation using high-intensity focused ultrasonography for primary localized prostate cancer. BJU Int. 2019;124:431–40.
van Son MJ, Peters M, Reddy D, Shah TT, Hosking-Jervis F, Robinson S, et al. Conventional radical versus focal treatment for localised prostate cancer: a propensity score weighted comparison of 6-year tumour control. Prostate Cancer Prostatic Dis. 2021;24:1120–8.
Shah TT, Reddy D, Peters M, Ball D, Kim NH, Gomez EG, et al. Focal therapy compared to radical prostatectomy for non-metastatic prostate cancer: a propensity score-matched study. Prostate Cancer Prostatic Dis. 2021;24:567–74.
Rakauskas A, Marra G, Heidegger I, Kasivisvanathan V, Kretschmer A, Zattoni F, et al. Focal therapy for prostate cancer: complications and their treatment. Front Surg. 2021;8:696242.
Barry Delongchamps N, Schull A, Anract J, Abecassis JP, Zerbib M, Sibony M, et al. Feasibility and safety of targeted focal microwave ablation of the index tumor in patients with low to intermediate risk prostate cancer: results of the FOSTINE trial. PLoS One. 2021;16:e0252040.
Chiu PK, Teoh JY, Lau SY, Yee CH, Lo KL, Li KM, et al. Transperineal microwave ablation of prostate cancer guided by MRI ultrasound fusion and organ based tracking technology. Eur Urol Open Sci 2020;19(Suppl 2):e2344.
Fainberg JS, Al Awamlh BAH, DeRosa AP, Chesnut GT, Coleman JA, Lee T, et al. A systematic review of outcomes after thermal and nonthermal partial prostate ablation. Prostate Int. 2021;9:169–75.
Guillaumier S, Peters M, Arya M, Afzal N, Charman S, Dudderidge T, et al. A multicentre study of 5-year outcomes following focal therapy in treating clinically significant nonmetastatic prostate cancer. Eur Urol. 2018;74:422–9.
Durand M, Barret E, Galiano M, Rozet F, Sanchez-Salas R, Ahallal Y, et al. Focal cryoablation: a treatment option for unilateral low-risk prostate cancer. BJU Int. 2014;113:56–64.
van den Bos W, Scheltema MJ, Siriwardana AR, Kalsbeek AMF, Thompson JE, Ting F, et al. Focal irreversible electroporation as primary treatment for localized prostate cancer. BJU Int. 2018;121:716–24.
Taneja SS, Bennett J, Coleman J, Grubb R, Andriole G, Reiter RE, et al. Final results of a phase I/II multicenter trial of WST11 vascular targeted photodynamic therapy for hemi-ablation of the prostate in men with unilateral low risk prostate cancer performed in the United States. J Urol. 2016;196:1096–104.
Ukimura O, Desai MM, Palmer S, Valencerina S, Gross M, Abreu AL, et al. 3-Dimensional elastic registration system of prostate biopsy location by real-time 3-dimensional transrectal ultrasound guidance with magnetic resonance/transrectal ultrasound image fusion. J Urol. 2012;187:1080–6.
Acknowledgements
There was no financial support or grant funding for this project. The microwave needle ablation system was provided by Koelis company for study purpose only. Besides equipment support, Koelis has no role in protocol development, trial conductance, and manuscript preparation.
Author information
Authors and Affiliations
Contributions
PK-FC: Design of trial, Patient recruitment, Perform treatment, Data analysis, Drafting of manuscript. C-HC: Data analysis, Drafting of manuscript. C-HY: Perform treatment, critical review of manuscript. S-YL: Patient recruitment, Data analysis. Patient follow-up. JY-CT: Design of trial, critical review of manuscript. H-FW: Perform treatment, critical review of manuscript. K-LL: Perform treatment, supervision. T-YY: MRI imaging and reporting, Treatment planning. H-YH: MRI imaging and reporting, Treatment planning. CC-MC: MRI imaging and reporting, Treatment planning, critical review of manuscript. C-FN: Critical review of manuscript, Supervision.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chiu, P.KF., Chan, CH., Yee, CH. et al. Transperineal Targeted Microwave Ablation (TMA) of localized prostate cancer guided by MRI-Ultrasound fusion and organ-based tracking: a pilot study. Prostate Cancer Prostatic Dis 26, 736–742 (2023). https://doi.org/10.1038/s41391-022-00577-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-022-00577-8
This article is cited by
-
“Targeted microwave ablation: another way to kick the can(cer) down the road?”
Prostate Cancer and Prostatic Diseases (2023)
-
Oncological results and cancer control definition in focal therapy for Prostate Cancer: a systematic review
Prostate Cancer and Prostatic Diseases (2023)
-
Functional outcomes and safety of focal therapy for prostate cancer: a systematic review on results and patient-reported outcome measures (PROMs)
Prostate Cancer and Prostatic Diseases (2023)