Abstract
Background
During the past two decades, new antitumor drugs (Abiraterone, Enzalutamide, Radium-223, Cabazitaxel, and Docetaxel) have been introduced for treatment of prostate cancer with distant metastases (mPCa). Each drug have demonstrated a survival gain in studies, but little is known about the impact in a general population of mPCa patients. In this study we assessed survival before and after introduction of the new drugs for Norwegian mPCa patients.
Methods
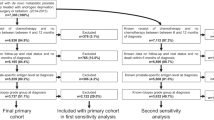
Survival was assessed in 5542 patients with primary mPCa. The patients were diagnosed between 2004 and 2018, identified in the Norwegian Cancer Registry. We also analyzed a subgroup of 2738 patients possibly eligible for treatment with new drugs (age <80 years, WHO performance status ≤2 and not dead within 3 months from diagnosis). We estimated overall (OS) and cause-specific survival (CSS) across three diagnostic time periods reflecting to the drugs’ introduction in Norway: Before (2004–2009), “in between” (2010–2014) and after the introduction (2015–2018). We used Kaplan-Meier survival analysis and multivariable Cox regression.
Results
Median OS increased from 2.3 years in 2004–2009 to 3.3 years in 2015–2018. 3-year OS improved from 41% in 2004–2009 to 51% in 2015–2018. An earlier diagnostic period, a more advanced T stage, higher ISUP grade group, lower WHO status and higher PSA levels were associated with a lower CSS. Similar results was found for the subgroup.
Conclusions
Median OS of mPCa has increased by one year since 2004 for mPCa patients in Norway. Survival improvement persisted after adjustment for recognized prognostic factors and may be related to the introduction of new drugs in Norway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Norway Cro. Cancer in Norway 2018—Cancer incidence, mortality, survival and prevalence in Norway. 2019.
Pagliarulo V, Bracarda S, Eisenberger MA, Mottet N, Schröder FH, Sternberg CN, et al. Contemporary role of androgen deprivation therapy for prostate cancer. Eur Urol. 2012;61:11–25.
Fossa SD, Jacobsen AB, Ginman C, Jacobsen IN, Overn S, Iversen JR, et al. Weekly docetaxel and prednisolone versus prednisolone alone in androgen-independent prostate cancer: a randomized phase II study. Eur Urol. 2007;52:1691–8.
Petrylak DP, Tangen CM, Hussain MH, Lara PN Jr., Jones JA, Taplin ME, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–20.
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12.
de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995–2005.
de Bono JS, Oudard S, Ozguroglu M, Hansen S, Machiels J-P, Kocak I, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376:1147–54.
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fosså SD, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–23.
Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–97.
James ND, Spears MR, Clarke NW, Dearnaley DP, De Bono JS, Gale J, et al. Survival with newly diagnosed metastatic prostate cancer in the “docetaxel era”: data from 917 patients in the control arm of the STAMPEDE Trial (MRC PR08, CRUK/06/019). Eur Urol. 2015;67:1028–38.
Westerberg M, Franck Lissbrant I, Damber JE, Robinson D, Garmo H, Stattin P. Temporal changes in survival in men with de novo metastatic prostate cancer: nationwide population-based study. Acta Oncol. 2019;59:1–6.
Bandini M, Pompe RS, Marchioni M, Zaffuto E, Gandaglia G, Fossati N, et al. Improved cancer-specific free survival and overall free survival in contemporary metastatic prostate cancer patients: a population-based study. Int Urol Nephrol. 2018;50:71–8.
Helgstrand JT, Roder MA, Klemann N, Toft BG, Lichtensztajn DY, Brooks JD, et al. Trends in incidence and 5-year mortality in men with newly diagnosed, metastatic prostate cancer—a population-based analysis of 2 national cohorts. Cancer. 2018;124:2931–8.
Hernes E, Kyrdalen A, Kvåle R, Hem E, Klepp O, Axcrona K, et al. Initial management of prostate cancer: first year experience with the Norwegian National Prostate Cancer Registry. BJU Int. 2010;105:805–11. discussion 11.
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–56.
James ND, Sydes MR, Clarke NW, Mason MD, Dearnaley DP, Spears MR, et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet. 2016;387:1163–77.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA. The 2014 International Society of Urological Pathology (ISUP) consensus conference on gleason grading of prostatic carcinoma: definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40:244–52.
http://www.norpd.no/: The Norwegian Prescription Database; 2021.
Author information
Authors and Affiliations
Contributions
Anne Holck Storås—original draft preparation, statistical analysis. Sophie D. Fosså—Conceptualization, writing-reviewing. Giske Ursin—Conceptualization, writing-reviewing. Bettina Kulle Andreassen—Conceptualization, writing-reviewing, statistical analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Storås, A.H., Fosså, S.D., Ursin, G. et al. Survival trends for patients with primary metastatic prostate cancer before and after the introduction of new antitumor drugs. Prostate Cancer Prostatic Dis 26, 53–58 (2023). https://doi.org/10.1038/s41391-021-00445-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-021-00445-x
This article is cited by
-
Resource use in the last year of life of prostate cancer patients—A register-based analysis
Prostate Cancer and Prostatic Diseases (2023)