Abstract
Purpose
Prostate-specific membrane antigen radioligand therapy (PSMA-RLT) is a novel treatment for castration-resistant prostate cancer (mCRPC). While the majority of patients responds to PSMA-RLT, with 10–15% having an exceptional response, approximately 30% of patients is unresponsive to PSMA-RLT. The molecular underpinning may in part explain these varying responses. This study investigated alterations in DNA damage repair (DDR) genes in tumour biopsies and their association with response to PSMA-RLT.
Methods
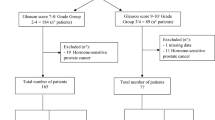
A predefined retrospective cohort study was performed in mCRPC patients of whom the tumours had undergone next-generation sequencing of 40 DDR genes and received Lu-177-PSMA and/or Ac-225-PSMA-RLT. The primary outcome of this study was to compare the progression free survival (PFS) after PSMA-RLT for patients with and without pathogenic DDR aberrations in their tumour. Secondary outcomes were prostate-specific antigen (PSA) response and overall survival (OS).
Results
A total of 40 patients were included of which seventeen had a tumour with a pathogenic DDR aberration (DDR+), of which eight had defects in BRCA1/2. DDR+ patients had an equal varying response to PSMA-RLT compared to those without pathological DDR anomalies (DDR-) in terms of PFS (5.9 vs. 6.4 months, respectively; HR 1.14; 95% CI 0.58-2.25; p = 0.71), ≥50% PSA response (59% vs. 65%, respectively; p = 0.75) or OS (11.1 vs. 10.7 months, respectively; HR 1.40; 95% CI: 0.68–2.91; p = 0.36).
Conclusion
In this study of a selected cohort, pathogenic DDR aberrations were not associated with exceptional responsiveness to PSMA-RLT. Translational studies in larger prospective cohorts are warranted to associate DDR gene defects with differential responses to PSMA-RLT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data accessibility
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA: Cancer J Clin. 2019;69:7–34.
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, et al. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur Urol. 2014;65:467–79.
Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res. 1997;3:81–85.
Ghosh A, Heston WD. Tumor target prostate specific membrane antigen (PSMA) and its regulation in prostate cancer. J Cell Biochem. 2004;91:528–39.
Bravaccini S, Puccetti M, Bocchini M, Ravaioli S, Celli M, Scarpi E, et al. PSMA expression: a potential ally for the pathologist in prostate cancer diagnosis. Sci Rep. 2018;8:4254.
Wright GL Jr., Grob BM, Haley C, Grossman K, Newhall K, Petrylak D, et al. Upregulation of prostate-specific membrane antigen after androgen-deprivation therapy. Urology. 1996;48:326–34.
Hofman MS, Violet J, Hicks RJ, Ferdinandus J, Thang SP, Akhurst T, et al. [(177)Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): a single-centre, single-arm, phase 2 study. Lancet Oncol. 2018;19:825–33.
Heck MM, Tauber R, Schwaiger S, Retz M, D’Alessandria C, Maurer T, et al. Treatment outcome, toxicity, and predictive factors for radioligand therapy with 177177177Lu-PSMA-I&T in metastatic castration-resistant prostate cancer. Eur Urol. 2019;75:920–6.
Sathekge M, Bruchertseifer F, Vorster M, Lawal IO, Knoesen O, Mahapane J, et al. Predictors of overall and disease-free survival in metastatic castration-resistant prostate cancer patients receiving (225)Ac-PSMA-617 radioligand therapy. J Nucl Med: Off Publ, Soc Nucl Med. 2020;61:62–69.
Rahbar K, Ahmadzadehfar H, Kratochwil C, Haberkorn U, Schafers M, Essler M, et al. German multicenter study investigating 177Lu-PSMA-617 radioligand therapy in advanced prostate cancer patients. J Nucl Med. 2017;58:85–90.
Hofman MS, Emmett L, Sandhu S, Iravani A, Joshua AM, Goh JC, et al. [177177177Lu] Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet. 2021;397:797–804.
Sathekge M, Bruchertseifer F, Knoesen O, Reyneke F, Lawal I, Lengana T, et al. 225Ac-PSMA-617 in chemotherapy-naive patients with advanced prostate cancer: a pilot study. Eur J Nucl Med Mol Imaging. 2019;46:129–38.
Kratochwil C, Bruchertseifer F, Rathke H, Hohenfellner M, Giesel FL, Haberkorn U, et al. Targeted alpha-therapy of metastatic castration-resistant prostate cancer with (225)ac-psma-617: swimmer-plot analysis suggests efficacy regarding duration of tumor control. J Nucl Med. 2018;59:795–802.
Yadav MP, Ballal S, Sahoo RK, Tripathi M, Seth A, Bal C. Efficacy and safety of 225225225Ac-PSMA-617 targeted alpha therapy in metastatic castration-resistant prostate cancer patients. Theranostics. 2020;10:9364–77.
Jackson SP, Bartek J. The DNA-damage response in human biology and disease. Nature. 2009;461:1071–8.
Kan C, Zhang J. BRCA1 mutation: a predictive marker for radiation therapy? Int J Radiat Oncol, Biol, Phys. 2015;93:281–93.
Isaacsson Velho P, Qazi F, Hassan S, Carducci MA, Denmeade SR, Markowski MC, et al. Efficacy of Radium-223 in bone-metastatic castration-resistant prostate cancer with and without homologous repair gene defects. Eur Urol. 2019;76:170–6.
Lord CJ, Ashworth A. The DNA damage response and cancer therapy. Nature. 2012;481:287–94.
de Bono JS, Fizazi K, Saad F, Shore N, Sandhu SK, Mehra N, et al. 847PD Central, prospective detection of homologous recombination repair gene mutations (HRRm) in tumour tissue from >4000 men with metastatic castration-resistant prostate cancer (mCRPC) screened for the PROfound study. Ann Oncol. 2019;30:v325–55.
Robinson D, Van Allen EM, Wu YM, Schultz N, Lonigro RJ, Mosquera JM, et al. Integrative clinical genomics of advanced prostate cancer. Cell. 2015;161:1215–28.
Mateo J, Carreira S, Sandhu S, Miranda S, Mossop H, Perez-Lopez R, et al. DNA-repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697–708.
Castro E, Mejorada RL, Saez M, De Giorgi UFF, Aragón I, Romero Laorden N, et al. Impact of germline mutations in Homologous Recombination (HR) genes on the response to Radium-223 for metastatic castration prostate cancer. Ann Oncol. 109; 30 v325–55.
Kirova YM, Savignoni A, Sigal-Zafrani B, de La Rochefordiere A, Salmon RJ, This P, et al. Is the breast-conserving treatment with radiotherapy appropriate in BRCA1/2 mutation carriers? Long-term results and review of the literature. Breast Cancer Res Treat. 2010;120:119–26.
Castro E, Goh C, Leongamornlert D, Saunders E, Tymrakiewicz M, Dadaev T, et al. Effect of BRCA mutations on metastatic relapse and cause-specific survival after radical treatment for localised prostate cancer. Eur Urol. 2015;68:186–93.
Cornford P, Bellmunt J, Bolla M, Briers E, De Santis M, Gross T, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur Urol. 2017;71:630–42.
Weren RD, Mensenkamp AR, Simons M, Eijkelenboom A, Sie AS, Ouchene H, et al. Novel BRCA1 and BRCA2 tumor test as basis for treatment decisions and referral for genetic counselling of patients with ovarian carcinomas. Hum Mutat. 2017;38:226–35.
Kroeze LI, de Voer RM, Kamping EJ, von Rhein D, Jansen EAM, Hermsen MJW, et al. Evaluation of a hybrid capture-based pan-cancer panel for analysis of treatment stratifying oncogenic aberrations and processes. J Mol Diagn. 2020;22:757–69.
Cardinale J, Schafer M, Benesova M, Bauder-Wust U, Leotta K, Eder M, et al. Preclinical evaluation of (18)F-PSMA-1007, a new prostate-specific membrane antigen ligand for prostate cancer imaging. J Nucl Med. 2017;58:425–31.
Fendler WP, Calais J, Eiber M, Flavell RR, Mishoe A, Feng FY, et al. Assessment of 68Ga-PSMA-11 PET accuracy in localizing recurrent prostate cancer: a prospective single-arm clinical trial. JAMA Oncol. 2019;5:856–63.
Kratochwil C, Bruchertseifer F, Giesel FL, Weis M, Verburg FA, Mottaghy F, et al. 225Ac-PSMA-617 for PSMA-targeted alpha-radiation therapy of metastatic castration-resistant prostate cancer. J Nucl Med. 2016;57:1941–4.
Kratochwil C, Giesel FL, Stefanova M, Benesova M, Bronzel M, Afshar-Oromieh A, et al. PSMA-targeted radionuclide therapy of metastatic castration-resistant prostate cancer with 177Lu-Labeled PSMA-617. J Nucl Med. 2016;57:1170–6.
Scher HI, Morris MJ, Stadler WM, Higano C, Basch E, Fizazi K, et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the prostate cancer clinical trials working group 3. J Clin Oncol. 2016;34:1402–18.
van der Velden DL, Hoes LR, van der Wijngaart H, van Berge Henegouwen JM, van Werkhoven E, Roepman P, et al. The Drug Rediscovery protocol facilitates the expanded use of existing anticancer drugs. Nature. 2019;574:127–31.
Morris MJ, Bono JSD, Chi KN, Fizazi K, Herrmann K, Rahbar K, et al. Phase III study of lutetium-177-PSMA-617 in patients with metastatic castration-resistant prostate cancer (VISION). J Clin Oncol. 2021;39:LBA4-LBA4.
Kratochwil C, Fendler WP, Eiber M, Baum R, Bozkurt MF, Czernin J, et al. EANM procedure guidelines for radionuclide therapy with (177)Lu-labelled PSMA-ligands ((177)Lu-PSMA-RLT). Eur J Nuclear Med Mol Imaging. 2019; 46:2536–44.
Violet J, Jackson P, Ferdinandus J, Sandhu S, Akhurst T, Iravani A, et al. Dosimetry of (177)Lu-PSMA-617 in metastatic castration-resistant prostate cancer: correlations between pretherapeutic imaging and whole-body tumor dosimetry with treatment outcomes. J Nucl Med. 2019;60:517–23.
Edler von Eyben F, Singh A, Zhang J, Nipsch K, Meyrick D, Lenzo N, et al. (177)Lu-PSMA radioligand therapy of predominant lymph node metastatic prostate cancer. Oncotarget. 2019;10:2451–61.
van der Doelen MJ, Mehra N, van Oort IM, Looijen-Salamon MG, Janssen MJR, Custers JAE, et al. Clinical outcomes and molecular profiling of advanced metastatic castration-resistant prostate cancer patients treated with (225)Ac-PSMA-617 targeted alpha-radiation therapy. Urol Oncol. 2021. (in press).
Schulz A, Meyer F, Dubrovska A, Borgmann K. Cancer stem cells and radioresistance: DNA repair and beyond. Cancers. 2019;11:862.
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature. 2006;444:756–60.
Nikjoo S, Uehara WE, Wilson M, Hoshi DT, Goodhead H. Track structure in radiation biology: theory and applications. Int J Radiat Biol. 1998;73:355–64.
Ward JF. DNA damage and repair. Basic Life Sci. 1991;58:403–15. discussion 415-21.
Crumbaker M, Emmett L, Horvath LG, Joshua AM. Exceptional response to 177 lutetium prostate-specific membrane antigen in prostate cancer harboring DNA repair defects. JCO Precis Oncol. 2019;3:1–5.
Kratochwil C, Giesel FL, Heussel CP, Kazdal D, Endris V, Nientiedt C, et al. Patients resistant against PSMA-targeting alpha-radiation therapy often harbor mutations in DNA-repair associated genes. J Nuclear Med. 2020;61:683–8.
Stover EH, Konstantinopoulos PA, Matulonis UA, Swisher EM. Biomarkers of response and resistance to DNA repair targeted therapies. Clin Cancer Res. 2016;22:5651–60.
Pouget JP, Lozza C, Deshayes E, Boudousq V, Navarro-Teulon I. Introduction to radiobiology of targeted radionuclide therapy. Front Med. 2015;2:12.
Khreish F, Ebert N, Ries M, Maus S, Rosar F, Bohnenberger H, et al. (225)Ac-PSMA-617/(177)Lu-PSMA-617 tandem therapy of metastatic castration-resistant prostate cancer: pilot experience. Eur J Nuclear Med Mol Imaging. 2020;47:721–8.
Feuerecker B, Tauber R, Knorr K, Heck M, Beheshti A, Seidl C, et al. Activity and adverse events of actinium-225-PSMA-617 in advanced metastatic castration-resistant prostate cancer after failure of lutetium-177-PSMA. Eur Urol. 2021;79:343–50.
Paschalis A, Sheehan B, Riisnaes R, Rodrigues DN, Gurel B, Bertan C, et al. Prostate-specific membrane antigen heterogeneity and DNA repair defects in prostate cancer. Eur Urol. 2019;76:469–78.
Acknowledgements
We thank all the investigators of the study, the patients and their families.
Author information
Authors and Affiliations
Contributions
Conceptualisation: Privé, Slootbeek, Laarhuis, van der Doelen, Gerritsen, Heskamp, Janssen, Nagarajah, Mehra Methodology: Privé, Slootbeek, van der Doelen, Heskamp, Janssen, Gerritsen, Nagarajah, Mehra Software: Privé, Slootbeek, Pamidimarri Naga; Validation: Privé, Slootbeek, Laarhuis, van der Doelen, Gerritsen, Nagarajah, Mehra Formal Analysis: Privé, Slootbeek, Laarhuis, van der Doelen, Gerritsen, Nagarajah, Mehra Investigation: Privé, van Kalmthout, Gotthardt, Janssen, Gerritsen, Nagarajah, Mehra Resources: de Keizer, Ezziddin, Kratochwil, Morgenstern, Bruchertseifer, Ligtenberg, Witjes, van Oort, Gotthardt, Janssen, Gerritsen, Nagarajah, Mehra Data Curation: Privé, Slootbeek, Laarhuis, Janssen, Gerritsen, Nagarajah, Mehra Writing – Original Draft: Privé, Laarhuis, Nagarajah, Mehra. Writing – Review & Editing: Slootbeek, van der Doelen, van Kalmthout, de Keizer, Ezziddin, Ligtenberg, Witjes, van Oort, Gotthardt, Heskamp, Janssen, Gerritsen, Nagarajah, Mehra Visualisation: Privé, Pamidimarri Naga Supervision: Gotthardt, Heskamp, Gerritsen, Nagarajah, Mehra Project Administration: Privé, Laarhuis, van der Doelen, Mehra.
Corresponding author
Ethics declarations
Competing interests
C. Kratochwil is co-inventor of some patents in the field of PSMA-targeting radio-ligand therapy. The other authors have declared that no competing interest exists for this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Privé, B.M., Slootbeek, P.H.J., Laarhuis, B.I. et al. Impact of DNA damage repair defects on response to PSMA radioligand therapy in metastatic castration-resistant prostate cancer. Prostate Cancer Prostatic Dis 25, 71–78 (2022). https://doi.org/10.1038/s41391-021-00424-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-021-00424-2
This article is cited by
-
Implementing Ac-225 labelled radiopharmaceuticals: practical considerations and (pre-)clinical perspectives
EJNMMI Radiopharmacy and Chemistry (2024)
-
Radiotheranostics in advanced prostate cancer: Current and future directions
Prostate Cancer and Prostatic Diseases (2024)
-
[177Lu]Lu-PSMA-617 as first-line systemic therapy in patients with metastatic castration-resistant prostate cancer: a real-world study
European Journal of Nuclear Medicine and Molecular Imaging (2024)
-
Therapeutic sensitivity to standard treatments in BRCA positive metastatic castration-resistant prostate cancer patients—a systematic review and meta-analysis
Prostate Cancer and Prostatic Diseases (2023)
-
89Zr-labeled PSMA ligands for pharmacokinetic PET imaging and dosimetry of PSMA-617 and PSMA-I&T: a preclinical evaluation and first in man
European Journal of Nuclear Medicine and Molecular Imaging (2022)