Abstract
Background
Survival rates are increasing in patients with prostate cancer, and second primary cancers (SPCs) are becoming more common in these patients. However, the etiology and clinical consequences of SPCs are not well-known. We define the impact of family history on SPC and causes of mortality in these patients.
Patients and methods
A nation-wide cohort study based on the Swedish Family-Cancer Database covering 4.4 million men and 80,449 prostate cancers diagnosed between 1990 and 2015. Relative risks (RRs) and cumulative incidence for SPCs and for familial SPC were calculated for prostate cancer patients.
Results
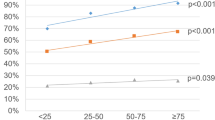
SPC was diagnosed in 6,396 men and more than a third of these patients had a first-degree family history of any cancer; the familial risk was 1.37 (95% CI: 1.27–1.40), compared to 1.10 (1.08–1.16), without a family history. Cumulative incidence by the age of 83 years reached 21% for prostate cancer alone, 28% in those with SPC, and 35% in patients with SPC and family history. Family history was associated with the risk of seven specific SPCs, including colorectal, lung, kidney, bladder and skin (both melanoma and squamous cell) cancers, and leukemia. Colorectal and lung cancers were common SPCs, and family history doubled the risk of these SPCs. In patients with SPC, half of all causes of death were due to SPC and only 12.77% were due to prostate cancer. Most deaths in SPC were caused by lung and colorectal cancers.
Conclusions
SPCs were an important cause of death in patients with prostate cancer and family history was an important risk factor for SPCs. Prevention of SPC should be essential when prostate cancer survival rates are being improved and this could start by conducting a thorough assessment of family history at the time of prostate cancer diagnosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Chen T, Fallah M, Jansen L, Castro FA, Krilavicuite A, Katalinic A et al. Distribution and risk of the second discordant primary cancers combined after a specific first primary cancer in German and Swedish cancer registries. Cancer Lett 2015;369:152–166.
CentreforEpidemiology. Cancer incidence in Sweden 2012. Stockholm: The National Board of Health and Welfare; 2013.
Travis LB, Demark Wahnefried W, Allan JM, Wood ME, Ng AK. Aetiology, genetics and prevention of secondary neoplasms in adult cancer survivors. Nat Rev Clin Oncol. 2013;10:289–301.
Wood ME, Vogel V, Ng A, Foxhall L, Goodwin P, Travis LB. Second malignant neoplasms: assessment and strategies for risk reduction. J Clin Oncol 2012;30:3734–3745.
Schaapveld M, Aleman BM, van Eggermond AM, Janus CP, Krol AD, van der Maazen RW et al. Second Cancer Risk Up to 40 Years after Treatment for Hodgkin's Lymphoma. N Engl J Med 2015;373:2499–2511.
Riihimaki M, Thomsen H, Brandt A, Sundquist J, Hemminki K. What do prostate cancer patients die of? Oncologist. 2011;16:175–81.
Donin N, Filson C, Drakaki A, Tan HJ, Castillo A, Kwan L et al. Risk of second primary malignancies among cancer survivors in the United States, 1992 through 2008. Cancer 2016;122: 3075–3086.
Zaorsky NG, Churilla TM, Egleston BL, Fisher SG, Ridge JA, Horwitz EM et al. Causes of death among cancer patients. Ann Oncol 2017;28:400-407.
Vogt A, et al. Multiple primary tumours: challenges and approaches, a review. ESMO Open. 2017;2:e000172.
Keehn A, Ludmir E, Taylor J, Rabbani F. Incidence of bladder cancer after radiation for prostate cancer as a function of time and radiation modality. World J Urol. 2017;35:713–20.
Jin T, Song T, Deng S, Wang K. Radiation-induced secondary malignancy in prostate cancer: a systematic review and meta-analysis. Urol Int. 2014;93:279–88.
Landgren O, Pfeiffer RM, Stewart L, Gridley G, Mellemkjaer L, Hemminki K et al. Risk of second malignant neoplasms among lymphoma patients with a family history of cancer. Int J Cancer 2007;120:1099–1102.
Sud A, Thomsen H, Sundquist K, Houlston RS, Hemminki K. Risk of second cancer in Hodgkin lymphoma survivors and the influence of family history. J Clin Oncol. 2017;35:1584–90.
Frank C, Sundquist J, Yu H, Hemminki A, Hemminki K. Concordant and discordant familial cancer: familial risks, proportions and population impact. Int J Cancer. 2017;140:1510–6.
Hemminki K, Ji J, Brandt A, Mousavi SM, Sundquist J. The Swedish Family-Cancer Database 2009: prospects for histology-specific and immigrant studies. Int J Cancer. 2010;126:2259–67.
Lorenzo Bermejo J, Hemminki K. Familial risk of cancer shortly after diagnosis of the first familial tumor. J Natl Cancer Inst. 2005;97:1575–9.
Hemminki K, Ankerst DP, Sundquist J, Mousavi SM. Prostate cancer incidence and survival in immigrants to Sweden. World J Urol. 2013;31:1483–8.
Centre for Epidemiology. Cancer Incidence in Sweden 1990. The National Board of Health and Welfare; 1993. p. 139.
Trama A, Foschi R, Larranaga N, Sant M, Fuentes-Raspall R, Serraino D et al. Survival of male genital cancers (prostate, testis and penis) in Europe 1999-2007: Results from the EUROCARE-5 study. Eur J Cancer 2015;51:2206–2216.
Ji J, Sundquist K, Sundquist J, Hemminki K. Comparability of cancer identification among Death Registry, Cancer Registry and Hospital Discharge Registry. Int J Cancer. 2012;131:2085–93.
Rahman N. Realizing the promise of cancer predisposition genes. Nature. 2014;505:302–8.
Giri VN, Knudsen KE, Kelly WK, Abida W, Andriole GL, Bangma CH et al. Role of Genetic Testing for Inherited Prostate Cancer Risk: Philadelphia Prostate Cancer Consensus Conference 2017. J Clin Oncol. 2018;36:4, 414–424
Moller P, Seppala TT, Bernstein I, Holinski-Feder E, Sala P, Gareth Evans D et al. Cancer risk and survival in path_MMR carriers by gene and gender up to 75 years of age: a report from the Prospective Lynch Syndrome Database. Gut 2018;67:1306–1316.
Frank C, Sundquist J, Hemminki A, Hemminki K. Familial associations between prostate cancer and other cancers. Eur Urol. 2017;71:162–5.
Yu H, Frank C, Hemminki A, Sundquist K, Hemminki K. Other cancers in lung cancer families are overwhelmingly smoking-related cancers. ERJ Open Research. 2017;3:00006–02017.
Yu H, Hemminki A, Sundquist K, Hemminki K. Familial associations of colorectal cancer with other cancers. Sci Rep. 2017;7:5243.
Frank C, Sundquist J, Hemminki A, Hemminki K. Risk of other cancers in families with melanoma: novel familial links. Sci Rep. 2017;7:42601.
Hemminki K, Hemminki O, Försti A, Sundquist J, Sundquist K, Li X. Familial Risks Between Urolithiasis and Cancer. Scientific Reports 2018;8:3083.
Gu J, Wu X. Genetic susceptibility to bladder cancer risk and outcome. Pers Med. 2011;8:365–74.
Wen-Qing Li, Abrar A. Qureshi, Jing Ma, Alisa M. Goldstein, Edward L. Giovannucci, Meir J. Stampfer, and Jiali Han. J Clin Oncol. 2013;31:35,4394–99
Hemminki K, Bevier M, Hemminki A, Sundquist J. Survival in cancer of unknown primary site: population-based analysis by site and histology. Ann Oncol. 2012;23:1854–63.
Hemminki K, Riihimaki M, Sundquist K, Hemminki A. Site-specific survival rates for cancer of unknown primary according to location of metastases. Int J Cancer. 2013;133:182–9.
Hemminki K, Bevier M, Sundquist J, Hemminki A. Cancer of unknown primary (CUP): does cause of death and family history implicate hidden phenotypically changed primaries? Ann Oncol. 2012;23:2720–24.
Paller CJ, Cole AP, Partin AW, Carducci MA, Kanarek NF. Risk factors for metastatic prostate cancer: a sentinel event case series. Prostate. 2017;77:1366–72.
IARC. Personal habits and indoor combustion. Lyon: International Agency for Research on Cancer; 2012. p. 575.
Acknowledgements
We are grateful for Patrick Reilly for language editing. This project was supported by grants from the German Cancer Aid, the Swedish Research Council (2014-2517, 2014-10134, and 2016-01176), and ALF funding from Region Skane, Sweden.
Author contributions
The study was designed by KH, OH, SC. KS, and JS were responsible for data acquisition. Data analysis was done by SC, KH, and AF. All authors interpreted the data. The first draft of the report was written by KH. The decision to submit the report for publication was made by all the authors. All authors contributed to the review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chattopadhyay, S., Hemminki, O., Försti, A. et al. Impact of family history of cancer on risk and mortality of second cancers in patients with prostate cancer. Prostate Cancer Prostatic Dis 22, 143–149 (2019). https://doi.org/10.1038/s41391-018-0089-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0089-y