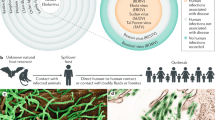
Abstract
Ebola disease is a severe disease with extremely high case-fatality rates ranging from 28–100%. Observations made during the 2013–2016 West African epidemic improved our understanding of the clinical course of Ebola disease and accelerated the study of therapeutic and preventative strategies. The epidemic also highlighted the unique challenges associated with providing optimal care for children during Ebola disease outbreaks. In this review, we outline current understanding of Ebola disease epidemiology, pathogenesis, management, and prevention, highlighting data pertinent to the care of children.
Impact
In this review, we summarize recent advancements in our understanding of Ebola disease epidemiology, clinical presentation, and therapeutic and preventative strategies. We highlight recent data pertinent to the care of children and pregnant women and identify research gaps for this important emerging viral infection in children.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
All data referenced in this review article are in publicly available published articles and repositories.
References
Kuhn, J. H. et al. New filovirus disease classification and nomenclature. Nat. Rev. Microbiol. 17, 261–263 (2019).
Kuhn, J. H. et al. ICTV virus taxonomy profile: filoviridae. J. Gen. Virol. 100, 911–912 (2019).
Jacob, S. T. et al. Ebola virus disease. Nat. Rev. Dis. Prim. 6, 13 (2020).
Towner, J. S. et al. Newly discovered ebola virus associated with hemorrhagic fever outbreak in Uganda. PLoS Pathog. 4, e1000212 (2008).
Coltart, C. E. M., Lindsey, B., Ghinai, I., Johnson, A. M. & Heymann, D. L. The Ebola outbreak, 2013–2016: old lessons for new epidemics. Philos. Trans. R Soc. Lond. B Biol. Sci. 372, 20160297 (2017).
Aruna, A. et al. Ebola virus disease outbreak - democratic republic of the Congo, August 2018-November 2019. MMWR Morb. Mortal. Wkly Rep. 68, 1162–1165 (2019).
Erb-Alvarez, J., Wendelboe, A. M. & Chertow, D. S. Ebola virus in the democratic republic of the Congo: advances and remaining obstacles in epidemic control, clinical care, and biomedical research. Chest 157, 42–46 (2020).
Report of an International Commission. Ebola haemorrhagic fever in Zaire, 1976. Bull. World Health Organ 56, 271–293 (1978).
Report of a WHO/International Study Team. Ebola haemorrhagic fever in Sudan, 1976. Report of a WHO/International Study Team. Bull. World Health Organ 56, 247–270 (1978).
Languon, S. & Quaye, O. Filovirus disease outbreaks: a chronological overview. Virology (Auckl.) 10, 1178122×1984992 (2019).
CDC. History of Ebola Disease Outbreaks. https://www.cdc.gov/vhf/ebola/history/chronology.html.
World Health Organization, Ebola Outbreak 2014–2016. https://www.who.int/emergencies/situations/ebola-outbreak-2014-2016-West-Africa.
Lado, M. & Howlett, P. Ebola virus disease in children: towards a better clinical picture and improved management. Lancet Glob. Health 4, e436–e437 (2016).
Long, S. S. Ebola virus disease in children presents special challenges. J. Pediatr. 182, 1 (2017).
Di Paola, N., Sanchez-Lockhart, M., Zeng, X., Kuhn, J. H. & Palacios, G. Viral genomics in Ebola virus research. Nat. Rev. Microbiol. 18, 365–378 (2020).
Leroy, E. M. et al. Fruit bats as reservoirs of Ebola virus. Nature 438, 575–576 (2005).
Judson, S. D., Fischer, R., Judson, A. & Munster, V. J. Ecological contexts of index cases and spillover events of different ebolaviruses. PLoS Pathog. 12, e1005780 (2016).
Vetter, P. et al. Ebola virus shedding and transmission: review of current evidence. J. Infect. Dis. 214, S177–S184 (2016).
Kreuels, B. et al. A case of severe Ebola virus infection complicated by gram-negative septicemia. N. Engl. J. Med. 371, 2394–2401 (2014).
Jacobs, M. et al. Late Ebola virus relapse causing meningoencephalitis: a case report. Lancet 388, 498–503 (2016).
Bausch, D. G. et al. Assessment of the risk of Ebola virus transmission from bodily fluids and fomites. J. Infect. Dis. 196, S142–S147 (2007).
Varkey, J. B. et al. Persistence of Ebola virus in ocular fluid during convalescence. N. Engl. J. Med. 372, 2423–2427 (2015).
Rodriguez, L. L. et al. Persistence and genetic stability of Ebola virus during the outbreak in Kikwit, Democratic Republic of the Congo, 1995. J. Infect. Dis. 179, S170–S176 (1999).
Caluwaerts, S. et al. Dilemmas in managing pregnant women with Ebola: 2 case reports: Table 1. Clin. Infect. Dis. 62, 903–905 (2016).
Reichler, M. R. et al. Household transmission of Ebola virus: risks and preventive factors, Freetown, Sierra Leone, 2015. J. Infect. Dis. 218, 757–767 (2018).
Dean, N. E., Halloran, M. E., Yang, Y. & Longini, I. M. Transmissibility and pathogenicity of Ebola virus: a systematic review and meta-analysis of household secondary attack rate and asymptomatic infection. Clin. Infect. Dis. 62, 1277–1286 (2016).
Victory, K. R. et al. Ebola transmission linked to a single traditional funeral ceremony—Kissidougou, Guinea, December, 2014-January 2015. MMWR Morb. Mortal. Wkly Rep. 64, 386–388 (2015).
Deen, G. F. et al. Ebola RNA persistence in semen of Ebola virus disease survivors—final report. N. Engl. J. Med. 377, 1428–1437 (2017).
PREVAIL III Study Group et al. A longitudinal study of Ebola sequelae in Liberia. N. Engl. J. Med. 380, 924–934 (2019).
Dyal, J. et al. Risk factors for Ebola virus persistence in semen of survivors in Liberia. Clin. Infect. Dis. 76, e849–e856 (2023).
Keita, A. K. et al. A 40 months follow-up of Ebola virus disease survivors in Guinea (Postebogui) reveals longterm detection of Ebola viral RNA in semen and breast milk. Open Forum Infect. Dis. ofz482. https://doi.org/10.1093/ofid/ofz482 (2019).
Christie, A. et al. Possible sexual transmission of Ebola virus—Liberia, 2015. MMWR Morb. Mortal. Wkly Rep. 64, 479–481 (2015).
Mate, S. E. et al. Molecular evidence of sexual transmission of Ebola virus. N. Engl. J. Med. 373, 2448–2454 (2015).
Mbala-Kingebeni, P. et al. Ebola virus transmission initiated by relapse of systemic Ebola virus disease. N. Engl. J. Med. 384, 1240–1247 (2021).
Keita, A. K. et al. Resurgence of Ebola virus in 2021 in Guinea suggests a new paradigm for outbreaks. Nature 597, 539–543 (2021).
Medina-Rivera, M. et al. Presence of Ebola virus in breast milk and risk of mother-to-child transmission: synthesis of evidence. Ann. N. Y. Acad. Sci. 1488, 33–43 (2021).
Foeller, M. E. et al. Pregnancy and breastfeeding in the context of Ebola: a systematic review. Lancet Infect. Dis. 20, e149–e158 (2020).
Kayem, N. D. et al. Ebola virus disease in pregnancy: a systematic review and meta-analysis. Trans. R. Soc. Trop. Med. Hyg. 116, 509–522 (2022).
World Health Organization. Guidelines for the management of pregnant and breastfeeding women in the context of Ebola virus disease. (World Health Organization, 2020).
Fitzgerald, F. et al. Ebola virus disease in children, Sierra Leone, 2014–2015. Emerg. Infect. Dis. 22, 1769–1777 (2016).
Shah, T. et al. Inpatient signs and symptoms and factors associated with death in children aged 5 years and younger admitted to two Ebola management centres in Sierra Leone, 2014: a retrospective cohort study. Lancet Glob. Health 4, e495–e501 (2016).
Organization, W. H. Target product profile for Zaire Ebolavirus rapid, simple test to be used in the control of the Ebola outbreak in West Africa. World Health Organization, Geneva, Switzerland. http://www.who.int/medicines/publications/target-product-profile.pdf (2014).
Mukadi-Bamuleka, D. et al. Field performance of three Ebola rapid diagnostic tests used during the 2018–20 outbreak in the eastern Democratic Republic of the Congo: a retrospective, multicentre observational study. Lancet Infect. Dis. 22, 891–900 (2022).
Chippaux, J.-P. Outbreaks of Ebola virus disease in Africa: the beginnings of a tragic saga. J. Venom. Anim. Toxins Incl. Trop. Dis. 20, 44 (2014).
Kiiza, P., Adhikari, N. K. J., Mullin, S., Teo, K. & Fowler, R. A. Principles and practices of establishing a hospital-based Ebola treatment unit. Crit. Care Clin. 35, 697–710 (2019).
WHO Ebola Response Team et al. Ebola virus disease among children in West Africa. N. Engl. J. Med. 372, 1274–1277 (2015).
Centers for Disease Control and Prevention (CDC). Outbreak of Ebola haemorrhagic fever, Uganda, August 2000-January 2001. Wkly. Epidemiol. Rec. 76, 41–46 (2001).
Mupere, E., Kaducu, O. F. & Yoti, Z. Ebola haemorrhagic fever among hospitalised children and adolescents in northern Uganda: epidemiologic and clinical observations. Afr. Health Sci. 1, 60–65 (2001).
Chérif, M. S. et al. Ebola virus disease in children during the 2014-2015 epidemic in Guinea: a nationwide cohort study. Eur. J. Pediatr. 176, 791–796 (2017).
Smit, M. A., Michelow, I. C., Glavis-Bloom, J., Wolfman, V. & Levine, A. C. Characteristics and outcomes of pediatric patients with Ebola virus disease admitted to treatment units in Liberia and Sierra Leone: a retrospective cohort study. Clin. Infect. Dis. 64, 243–249 (2017).
Shears, P. & Garavan, C. The 2018/19 Ebola epidemic the Democratic Republic of the Congo (DRC): epidemiology, outbreak control, and conflict. Infect. Prev. Pract. 2, 100038 (2020).
Henao-Restrepo, A. M. et al. Efficacy and effectiveness of an rVSV-vectored vaccine in preventing Ebola virus disease: final results from the Guinea ring vaccination, open-label, cluster-randomised trial (Ebola Ça Suffit!). Lancet 389, 505–518 (2017).
Dyer, O. Two Ebola treatments halve deaths in trial in DRC outbreak. BMJ l5140 https://doi.org/10.1136/bmj.l5140 (2019).
WHO. Disease Outbreak News: Ebola virus disease—Democratic Republic of the Congo. https://www.who.int/emergencies/disease-outbreak-news/item/2019-DON226 (2019).
WHO. Disease Outbreak News: Ebola virus disease—Democratic Republic of the Congo. https://www.who.int/emergencies/disease-outbreak-news/item/ebola-virus-disease-democratic-republic-of-the-congo-draft (2020).
Centers for Disease Control and Prevention (CDC). Outbreak of Ebola hemorrhagic fever Uganda, August 2000-January 2001. MMWR Morb. Mortal. Wkly Rep. 50, 73–77 (2001).
Okware, S. et al. An outbreak of Ebola in Uganda. Trop. Med. Int. Health 7, 1068–1075 (2002).
WHO. Ebola disease caused by Sudan ebolavirus—Uganda. https://www.who.int/emergencies/disease-outbreak-news/item/2023-DON433 (2023).
UNICEF. Uganda Country Office Sudan Virus Disease (SVD) Final Humanitarian Situation Report. https://www.unicef.org/media/135146/file/Uganda-Humanitarian-Situation-Report-No.8-(Sudan-Virus-Disease)-24-February-2023.pdf.
Kratz, T. et al. Ebola virus disease outbreak in Isiro, Democratic Republic of the Congo, 2012: signs and symptoms, management and outcomes. PLoS One 10, e0129333 (2015).
Le Guenno, B. et al. Isolation and partial characterisation of a new strain of Ebola virus. Lancet 345, 1271–1274 (1995).
Baseler, L., Chertow, D. S., Johnson, K. M., Feldmann, H. & Morens, D. M. The pathogenesis of Ebola virus disease. Annu. Rev. Pathol. 12, 387–418 (2017).
Geisbert, T. W. et al. Pathogenesis of Ebola hemorrhagic fever in cynomolgus macaques. Am. J. Pathol. 163, 2347–2370 (2003).
Yen, B., Mulder, L. C. F., Martinez, O. & Basler, C. F. Molecular basis for ebolavirus VP35 suppression of human dendritic cell maturation. J. Virol. 88, 12500–12510 (2014).
Basler, C. F. et al. The Ebola virus VP35 protein functions as a type I IFN antagonist. Proc. Natl Acad. Sci. USA 97, 12289–12294 (2000).
Reid, P. et al. Ebola virus VP24 binds karyopherin α1 and blocks STAT1 nuclear accumulation. J. Virol. 80, 5156–5167 (2006).
Zhang, A. P. P. et al. The ebolavirus VP24 interferon antagonist: know your enemy. Virulence 3, 440–445 (2012).
Ito, H., Watanabe, S., Takada, A. & Kawaoka, Y. Ebola virus glycoprotein: proteolytic processing, acylation, cell tropism, and detection of neutralizing antibodies. J. Virol. 75, 1576–1580 (2001).
Breman, J. G. & Johnson, K. M. Ebola then and now. N. Engl. J. Med. 371, 1663–1666 (2014).
Chertow, D. S. et al. Ebola virus disease in West Africa-clinical manifestations and management. N. Engl. J. Med. 371, 2054–2057 (2014).
Kjaldgaard, L. et al. Virus kinetics and biochemical derangements among children with Ebolavirus disease. eClinicalMedicine 53, 101638 (2022).
Cournac, J. M. et al. Rhabdomyolysis in Ebola virus disease. Results of an observational study in a treatment center in Guinea. Clin. Infect. Dis. 62, 19–23 (2016).
Lado, M. et al. Clinical features of patients isolated for suspected Ebola virus disease at Connaught Hospital, Freetown, Sierra Leone: a retrospective cohort study. Lancet Infect. Dis. 15, 1024–1033 (2015).
Rojek, A., Horby, P. & Dunning, J. Insights from clinical research completed during the West Africa Ebola virus disease epidemic. Lancet Infect. Dis. 17, e280–e292 (2017).
Jamieson, D. J., Uyeki, T. M., Callaghan, W. M., Meaney-Delman, D. & Rasmussen, S. A. What obstetrician-gynecologists should know about Ebola: a perspective from the Centers for Disease Control and Prevention. Obstet. Gynecol. 124, 1005–1010 (2014).
McElroy, A. K. et al. Ebola hemorrhagic fever: novel biomarker correlates of clinical outcome. J. Infect. Dis. 210, 558–566 (2014).
Chertow, D. S. et al. Severe meningoencephalitis in a case of Ebola virus disease: a case report. Ann. Intern Med 165, 301–304 (2016).
de Greslan, T. et al. Ebola virus-related encephalitis. Clin. Infect. Dis. 63, 1076–1078 (2016).
Uyeki, T. M. et al. Clinical management of Ebola virus disease in the United States and Europe. N. Engl. J. Med. 374, 636–646 (2016).
Bwaka, M. A. et al. Ebola hemorrhagic fever in Kikwit, Democratic Republic of the Congo: clinical observations in 103 patients. J. Infect. Dis. 179, S1–S7 (1999).
Chertow, D. S., Childs, R. W., Arai, A. E. & Davey, R. T. Cardiac MRI findings suggest myocarditis in severe Ebola virus disease. JACC Cardiovasc Imaging 10, 711–713 (2017).
WHO. Ebola haemorrhagic fever in Zaire, 1976. Bull. World Health Organ. 56, 271–293 (1978).
Damkjær, M., Rudolf, F., Mishra, S., Young, A. & Storgaard, M. Clinical features and outcome of Ebola virus disease in pediatric patients: a retrospective case series. J. Pediatr. 182, 378–381.e1 (2017).
Hunt, L. et al. Clinical presentation, biochemical, and haematological parameters and their association with outcome in patients with Ebola virus disease: an observational cohort study. Lancet Infect. Dis. 15, 1292–1299 (2015).
Fitzgerald, F., Awonuga, W., Shah, T. & Youkee, D. Ebola response in Sierra Leone: the impact on children. J. Infect. 72, S6–S12 (2016).
The Lancet Child Adolescent Health, null. Children’s needs in an Ebola virus disease outbreak. Lancet Child Adolesc. Health 3, 55 (2019).
Chowdhury, M. et al. Strengthening the maternal and child health responses to Ebola outbreaks in Uganda. Lancet Infect. Dis. 23, 650–651 (2023).
UNICEF. UNICEF Uganda Ebola Virus Disease (EVD) update. https://www.unicef.org/uganda/reports/unicef-uganda-ebola-virus-disease-evd-update (2022).
WHO. Case definition recommendations for Ebola or Marburg virus diseases. https://www.who.int/publications/i/item/WHO-EVD-CaseDef-14.1 (2014).
Bettini, A., Lapa, D. & Garbuglia, A. R. Diagnostics of Ebola virus. Front. Public Health 11, 1123024 (2023).
WHO. Optimized supportive care for Ebola virus disease clinical management standard operating procedures. https://apps.who.int/iris/handle/10665/325000 (2019).
Lamontagne, F. et al. Evidence-based guidelines for supportive care of patients with Ebola virus disease. Lancet 391, 700–708 (2018).
Misasi, J. & Sullivan, N. J. Immunotherapeutic strategies to target vulnerabilities in the Ebolavirus glycoprotein. Immunity 54, 412–436 (2021).
Rayaprolu, V. et al. Structure of the Inmazeb cocktail and resistance to Ebola virus escape. Cell Host Microbe 31, 260–272.e7 (2023).
Mulangu, S. et al. A randomized, controlled trial of Ebola virus disease therapeutics. N. Engl. J. Med. 381, 2293–2303 (2019).
Liu, G. et al. A Pan-ebolavirus Monoclonal Antibody Cocktail Provides Protection against Ebola and Sudan Viruses. J. Infect. Dis. jiad205. https://doi.org/10.1093/infdis/jiad205 (2023).
Cross, R. W. et al. Combination therapy with remdesivir and monoclonal antibodies protects nonhuman primates against advanced Sudan virus disease. JCI Insight 7, e159090 (2022).
Wohl, D. A. et al. Post-Ebola symptoms 7 years after infection: the natural history of long Ebola. Clin. Infect. Dis. 76, e835–e840 (2023).
Clark, D. V. et al. Long-term sequelae after Ebola virus disease in Bundibugyo, Uganda: a retrospective cohort study. Lancet Infect. Dis. 15, 905–912 (2015).
Scott, J. T. et al. Post-Ebola syndrome, Sierra Leone. Emerg. Infect. Dis. 22, 641–646 (2016).
Diallo, M. S. K. et al. Understanding long-term evolution and predictors of sequelae of Ebola virus disease survivors in Guinea: a 48-month prospective, longitudinal cohort study (PostEboGui). Clin. Infect. Dis. 73, 2166–2174 (2021).
Amuzu, C., James, P. B., Bah, A. J., Bayoh, A. V. S. & Singer, S. R. Post-Ebola sequelae among Ebola child survivors in Sierra Leone. BMC Pediatr. 21, 482 (2021).
McElroy, A. K. et al. Human Ebola virus infection results in substantial immune activation. Proc. Natl Acad. Sci. USA 112, 4719–4724 (2015).
Rowe, A. K. et al. Clinical, virologic, and immunologic follow‐up of convalescent Ebola hemorrhagic fever patients and their household contacts, Kikwit, Democratic Republic of the Congo. J. Infect. Dis. 179, S28–S35 (1999).
Mattia, J. G. et al. Early clinical sequelae of Ebola virus disease in Sierra Leone: a cross-sectional study. Lancet Infect. Dis. 16, 331–338 (2016).
Paquin-Proulx, D. et al. Associations between antibody fc-mediated effector functions and long-term Sequelae in Ebola Virus survivors. Front. Immunol. 12, 682120 (2021).
UNICEF. Thousands of Children Still Need Care and Support in Wake of Ebola Epidemic. https://www.unicefusa.org/press/thousands-children-still-need-care-and-support-wake-ebola-epidemic (2015).
Ainsworth, M. & Filmer, D. Inequalities in children’s schooling: AIDS, orphanhood, poverty, and gender. World Dev. 34, 1099–1128 (2006).
WHO. How to conduct safe and dignified burial of a patient who has died from suspected or confirmed Ebola or Marburg virus disease. https://www.who.int/publications/i/item/WHO-EVD-Guidance-Burials-14.2 (2014).
WHO, Prequalified Vaccines. https://extranet.who.int/pqweb/vaccines/prequalified-vaccines.
Woolsey, C. & Geisbert, T. W. Current state of Ebola virus vaccines: a snapshot. PLoS Pathog. 17, e1010078 (2021).
Bockstal, V. et al. Non-human primate to human immunobridging demonstrates a protective effect of Ad26.ZEBOV, MVA-BN-Filo vaccine against Ebola. NPJ Vaccines 7, 1–8 (2022).
PREVAC Study Team. Randomized trial of vaccines for Zaire Ebola virus disease. N. Engl. J. Med. 387, 2411–2424 (2022).
WHO. ICG for Ebola virus disease vaccine. https://www.who.int/groups/icg/ebola-virus-disease.
Anywaine, Z. et al. Safety and immunogenicity of 2-dose heterologous Ad26.ZEBOV, MVA-BN-Filo Ebola vaccination in children and adolescents in Africa: A randomised, placebo-controlled, multicentre Phase II clinical trial. PLoS Med. 19, e1003865 (2022).
Afolabi, M. O. et al. Safety and immunogenicity of the two-dose heterologous Ad26.ZEBOV and MVA-BN-Filo Ebola vaccine regimen in children in Sierra Leone: a randomised, double-blind, controlled trial. Lancet Infect. Dis. 22, 110–122 (2022).
Agnandji, S. T. et al. Safety and immunogenicity of rVSVΔG-ZEBOV-GP Ebola vaccine in adults and children in Lambaréné, Gabon: a phase I randomised trial. PLoS Med. 14, e1002402 (2017).
Manno, D. et al. Safety and immunogenicity of an Ad26.ZEBOV booster dose in children previously vaccinated with the two-dose heterologous Ad26.ZEBOV and MVA-BN-Filo Ebola vaccine regimen: an open-label, non-randomised, phase 2 trial. Lancet Infect. Dis. 23, 352–360 (2023).
Nyombayire, J. et al. Monitoring of adverse events in recipients of the 2-dose Ebola vaccine regimen of Ad26.ZEBOV followed by MVA-BN-Filo in the UMURINZI Ebola Vaccination Campaign. J. Infect. Dis. 227, 268–277 (2023).
Sullivan, N. J., Martin, J. E., Graham, B. S. & Nabel, G. J. Correlates of protective immunity for Ebola vaccines: implications for regulatory approval by the animal rule. Nat. Rev. Microbiol. 7, 393–400 (2009).
Plotkin, S. A. Recent updates on correlates of vaccine-induced protection. Front. Immunol. 13, 1081107 (2023).
Medaglini, D., Santoro, F. & Siegrist, C.-A. Correlates of vaccine-induced protective immunity against Ebola virus disease. Semin. Immunol. 39, 65–72 (2018).
Hoff, N. A. et al. Immunogenicity of rVSVΔG-ZEBOV-GP Ebola vaccination in exposed and potentially exposed persons in the Democratic Republic of the Congo. Proc. Natl Acad. Sci. USA 119, e2118895119 (2022).
Shultz, J. M., Espinel, Z., Espinola, M. & Rechkemmer, A. Distinguishing epidemiological features of the 2013–2016 West Africa Ebola virus disease outbreak. Disaster Health 3, 78–88 (2016).
Maganga, G. D. et al. Ebola virus disease in the Democratic Republic of Congo. N. Engl. J. Med 371, 2083–2091 (2014).
Nsio, J. et al. 2017 outbreak of Ebola virus disease in Northern Democratic Republic of Congo. J. Infect. Dis. https://doi.org/10.1093/infdis/jiz107 (2019).
Gryseels, S. et al. Role of wildlife in emergence of Ebola virus in Kaigbono (Likati), Democratic Republic of the Congo, 2017. Emerg. Infect. Dis. 26, 2205–2209 (2020).
Burki, T. DRC getting ready to introduce a second Ebola vaccine. Lancet Infect. Dis. 19, 1174–1175 (2019).
Adepoju, P. Ebola returns to Guinea and DR Congo. Lancet 397, 781 (2021).
WHO. Disease Outbreak News: Ebola virus disease—Democratic Republic of the Congo. https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON398 (2022).
WHO. Disease Outbreak News: Ebola virus disease—Democratic Republic of the Congo. https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON411 (2022).
WHO/International Study Team. Ebola haemorrhagic fever in Sudan, 1976. Bull. World Health Organ. 56, 247–270 (1978).
Baron, R. C., McCormick, J. B. & Zubeir, O. A. Ebola virus disease in southern Sudan: hospital dissemination and intrafamilial spread. Bull. World Health Organ. 61, 997–100 (1983).
World Health Organization. Outbreak of Ebola haemorrhagic fever in Yambio, south Sudan, April – June 2004. Wkly. Epidemiol. Rec. 80, 369–376 (2005).
Shoemaker, T. et al. Reemerging Sudan Ebola virus disease in Uganda, 2011. Emerg. Infect. Dis. 18, 1480 (2012).
Albariño, C. G. et al. Genomic analysis of filoviruses associated with four viral hemorrhagic fever outbreaks in Uganda and the Democratic Republic of the Congo in 2012. Virology 442, 97–100 (2013).
Author information
Authors and Affiliations
Contributions
K.R.D. and A.L.: substantial contributions to conception and design; interpretation of literature; drafting the article or revising it critically for important intellectual content; and final approval of the version to be published. A.E.D.: drafting the article or revising it critically for important intellectual content; and final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dobbs, K.R., Lobb, A. & Dent, A.E. Ebola virus disease in children: epidemiology, pathogenesis, management, and prevention. Pediatr Res 95, 488–495 (2024). https://doi.org/10.1038/s41390-023-02873-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02873-y
This article is cited by
-
Trends in prenatal and pediatric viral infections, and the impact of climate change
Pediatric Research (2024)